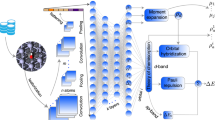
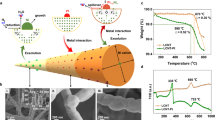
Abstract
The metal–support interaction (MSI) critically influences the performance of supported nanocatalysts and their long-term stability, yet the factors governing MSIs are multifaceted and challenging to sort out. Here we combine first-principles neural network molecular dynamics (NN-MD) simulations with interpretable machine learning (iML) to shed light on the factors determining MSIs for Pt nanoparticles on diverse metal–oxide supports. Our approach reveals the atomic-scale dynamics of sintering mechanisms and identifies key features of oxide supports governing MSI. We find that the surface energy, surface oxygen bond order, surface dipole and work function of the support are dominant in Pt–oxide interactions. Leveraging these insights, we screened promising sinter-resistant supports for Pt nanoparticles from over 10,000 metal–oxide surfaces and validated some cases by Monte Carlo simulations and experiments. This work integrates iML with NN-MD to accelerate the understanding and discovery of stable supported nanocatalysts, and should be broadly applicable to numerous catalytic applications.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Data supporting this study’s findings are provided in the article and Supplementary Information. The complete NN-MD trajectory files, associated statistical analysis data (including contact angle measurements and adhesion energy calculations), all statistical results from high-throughput screening, promising sintering-resistant supports, and their corresponding atomic structure files are publicly accessible via the Zenodo repository at https://doi.org/10.5281/zenodo.16878887 (ref. 41). All other relevant raw data are available from the corresponding authors upon reasonable request. Source data are provided with this paper.
Code availability
The software code for LASP and NN potentials used within the article are available on the website http://www.lasphub.com. The source code of iGAM (a.k.a. EBM) can be found at https://github.com/interpretml/interpret/. All well-established iGAM models are available in the GitHub repository (https://github.com/chenggoj/iGAM-MSI).
References
Liu, W. et al. Single-atom dispersed Co–N–C catalyst: structure identification and performance for hydrogenative coupling of nitroarenes. Chem. Sci. 7, 5758–5764 (2016).
Gholami, R., Alyani, M. & Smith, K. Deactivation of Pd catalysts by water during low temperature methane oxidation relevant to natural gas vehicle converters. Catalysts 5, 561–594 (2015).
Yati, I., et al. Effects of sintering-resistance and large metal-support interface of alumina nanorod-stabilized Pt nanoparticle catalysts on the improved high temperature water gas shift reaction activity. Catal. Commun. 56, 11–16 (2014).
Dai, Y., Lu, P., Cao, Z., Campbell, C. T. & Xia, Y. The physical chemistry and materials science behind sinter-resistant catalysts. Chem. Soc. Rev. 47, 4314–4331 (2018).
van Deelen, T. W., Hernández Mejía, C. & de Jong, K. P. Control of metal–support interactions in heterogeneous catalysts to enhance activity and selectivity. Nat. Catal. 2, 955–970 (2019).
Chen, J. et al. Metal–support interactions for heterogeneous catalysis: mechanisms, characterization techniques and applications. J. Mater. Chem. A 11, 8540–8572 (2023).
Campbell, C. T. & Sellers, J. R. V. Enthalpies and entropies of adsorption on well-defined oxide surfaces: experimental measurements. Chem. Rev. 113, 4106–4135 (2013).
Ro, I., Resasco, J. & Christopher, P. Approaches for understanding and controlling interfacial effects in oxide-supported metal catalysts. ACS Catal. 8, 7368–7387 (2018).
Campbell, C. T. The energetics of supported metal nanoparticles: relationships to sintering rates and catalytic activity. Acc. Chem. Res. 46, 1712–1719 (2013).
Campbell, C. T. & Sellers, J. R. V. Anchored metal nanoparticles: effects of support and size on their energy, sintering resistance and reactivity. Faraday Discuss. 162, 9–20 (2013).
Bartholomew, C. H. Mechanisms of catalyst deactivation. Appl. Catal. A 212, 17–60 (2001).
O’Connor, N. J., Jonayat, A. S. M., Janik, M. J. & Senftle, T. P. Interaction trends between single metal atoms and oxide supports identified with density functional theory and statistical learning. Nat. Catal. 1, 531–539 (2018).
Leybo, D. et al. Metal-support interactions in metal oxide-supported atomic, cluster and nanoparticle catalysis. Chem. Soc. Rev. 28, 10450–10490 (2024).
Hu, S. & Li, W.-X. Sabatier principle of metal-support interaction for design of ultrastable metal nanocatalysts. Science 374, 1360–1365 (2021).
Liu, J.-C. et al. Metal affinity of support dictates sintering of gold catalysts. J. Am. Chem. Soc. 144, 20601–20609 (2022).
Wang, T. et al. Nature of metal-support interaction for metal catalysts on oxide supports. Science 386, 915–920 (2024).
Esterhuizen, J. A., Goldsmith, B. R. & Linic, S. Interpretable machine learning for knowledge generation in heterogeneous catalysis. Nat. Catal. 5, 175–184 (2022).
Omidvar, N. et al. Interpretable machine learning of chemical bonding at solid surfaces. J. Phys. Chem. Lett. 12, 11476–11487 (2021).
Xin, H., Mou, T., Pillai, H. S., Wang, S.-H. & Huang, Y. Interpretable machine learning for catalytic materials design toward sustainability. Acc. Mater. Res. 5, 22–34 (2024).
Wang, H. et al. Scientific discovery in the age of artificial intelligence. Nature 620, 47–60 (2023).
Esterhuizen, J. A., Goldsmith, B. R. & Linic, S. Uncovering electronic and geometric descriptors of chemical activity for metal alloys and oxides using unsupervised machine learning. Chem. Catal. 1, 923–940 (2021).
Goldsmith, B. R., Esterhuizen, J., Liu, J. X., Bartel, C. J. & Sutton, C. Machine learning for heterogeneous catalyst design and discovery. AlChE J. 64, 2311–2323 (2018).
Kang, P.-L., Shang, C. & Liu, Z.-P. Large-scale atomic simulation via machine learning potentials constructed by global potential energy surface exploration. Acc. Chem. Res. 53, 2119–2129 (2020).
Tran, R. et al. The open catalyst 2022 (OC22) dataset and challenges for oxide electrocatalysts. ACS Catal. 13, 3066–3084 (2023).
Jain, A. et al. Commentary: the materials project: a materials genome approach to accelerating materials innovation. APL Mater. 1, 011002 (2013).
Hansen, T. W., DeLaRiva, A. T., Challa, S. R. & Datye, A. K. Sintering of catalytic nanoparticles: particle migration or Ostwald ripening? Acc. Chem. Res. 46, 1720–1730 (2013).
Tran, R. et al. Surface energies of elemental crystals. Sci. Data 3, 160080 (2016).
Yu, J. et al. Highly stable Pt/CeO2 catalyst with embedding structure toward water-gas shift reaction. J. Am. Chem. Soc. 146, 1071–1080 (2023).
Esterhuizen, J. A., Goldsmith, B. R. & Linic, S. Theory-guided machine learning finds geometric structure-property relationships for chemisorption on subsurface alloys. Chem 6, 3100–3117 (2020).
Lou, Y., Caruana, R., Gehrke, J. & Hooker, G. Accurate intelligible models with pairwise interactions. In Proc. 19th ACM SIGKDD International Conference on Knowledge Discovery and Data Mining (ed. Dhillon, I. S.) 623–631 (ACM, 2013).
Rudin, C. Stop explaining black box machine learning models for high stakes decisions and use interpretable models instead. Nat. Mach. Intell. 1, 206–215 (2019).
Reshef, D. N. et al. Detecting novel associations in large datasets. Science 334, 1518–1524 (2011).
Hemmingson, S. L. & Campbell, C. T. Trends in adhesion energies of metal nanoparticles on oxide surfaces: understanding support effects in catalysis and nanotechnology. ACS Nano 11, 1196–1203 (2017).
Lou, Y., Caruana, R. & Gehrke, J. Intelligible models for classification and regression. In Proc. 18th ACM SIGKDD International Conference on Knowledge Discovery and Data Mining (ed. Yang, Q.) 150–158 (ACM, 2012).
Hu, S. & Li, W.-X. Metal–support interaction controlled migration and coalescence of supported particles. Sci. China Technol. Sci. 62, 762–772 (2019).
Limas, N. G. & Manz, T. A. Introducing DDEC6 atomic population analysis: part 4. Efficient parallel computation of net atomic charges, atomic spin moments, bond orders and more. RSC Adv. 8, 2678–2707 (2018).
Manz, T. A. Introducing DDEC6 atomic population analysis: part 3. Comprehensive method to compute bond orders. RSC Adv. 7, 45552–45581 (2017).
Smith, J. et al. Sintering mechanism of Pt/Al2O3 in complex emission gases elucidated via in situ environmental STEM. ACS Mater. Lett. 6, 3301–3311 (2024).
Huang, S.-D., Shang, C., Kang, P.-L., Zhang, X.-J. & Liu, Z.-P. LASP: fast global potential energy surface exploration. WIREs Comput. Mol. Sci. 9, e1415 (2019).
Kang, P.-L., Shang, C. & Liu, Z.-P. Recent implementations in LASP 3.0: global neural network potential with multiple elements and better long-range description. Chin. J. Chem. Phys. 34, 583–590 (2021).
Jiang, C. Predictive model for the discovery of sinter-resistant supports for metallic nanoparticle catalysts by interpretable machine learning. Zenodo https://doi.org/10.5281/zenodo.16878887 (2025).
Shang, C. & Liu, Z.-P. in Quantum Chemistry in the Age of Machine Learning (ed. Dral, P. O.) 313–327 (Elsevier, 2023).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for liquid metals. Phys. Rev. B 47, 558–561 (1993).
Kresse, G. & Hafner, J. Ab initio molecular-dynamics simulation of the liquid-metal-amorphous-semiconductor transition in germanium. Phys. Rev. B 49, 14251–14269 (1994).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Wang, Y., Kiziltas, A., Blanchard, P. & Walsh, T. R. ContactAngleCalculator: an automated, parametrized and flexible code for contact angle estimation in visual molecular dynamics. J. Chem. Inf. Model. 62, 6302–6308 (2022).
Humphrey, W., Dalke, A. & Schulten, K. VMD: Visual Molecular Dynamics. J. Mol. Graph. 14, 33–38 (1996).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Sun, W. et al. The thermodynamic scale of inorganic crystalline metastability. Sci. Adv. https://doi.org/10.1126/sciadv.1600225 (2016).
Young, S. D., Chen, J., Sun, W., Goldsmith, B. R. & Pilania, G. Thermodynamic stability and anion ordering of perovskite oxynitrides. Chem. Mater. 35, 5975–5987 (2023).
Wang, V., Xu, N., Liu, J.-C., Tang, G. & Geng, W.-T. VASPKIT: a user-friendly interface facilitating high-throughput computing and analysis using VASP code. Comput. Phys. Commun. 267, 108033 (2021).
Ong, S. P. et al. The materials application programming interface (API): a simple, flexible and efficient API for materials data based on representational state transfer (REST) principles. Comput. Mater. Sci. 97, 209–215 (2015).
Hong, Q.-J., Ushakov, S. V., van de Walle, A. & Navrotsky, A. Melting temperature prediction using a graph neural network model: from ancient minerals to new materials. Proc. Natl Acad. Sci. USA 119, e2209630119 (2022).
Zandkarimi, B., Poths, P. & Alexandrova, A. N. When fluxionality beats size selection: acceleration of Ostwald ripening of sub-nano clusters. Angew. Chem. 133, 12080–12089 (2021).
Zhai, H. & Alexandrova, A. N. Ensemble-average representation of Pt clusters in conditions of catalysis accessed through GPU accelerated deep neural network fitting global optimization. J. Chem. Theory Comput. 12, 6213–6226 (2016).
Lide, D. R. (ed.) in CRC Handbook of Chemistry and Physics 88th edn 12–124 (CRC Press, 2008).
Acknowledgements
This material is based on work supported by NSF DMREF #2116646 (B.R.G. and S.L.). Experimental work was supported by the US DOE Office of Basic Energy Sciences, Division of Chemical Sciences (DE-SC0021008; S.L.).
Author information
Authors and Affiliations
Contributions
C.J. wrote the original draft, and performed all calculations and analysis. B.Y. performed all experiments. B.R.G. is a PhD co-adviser to C.J., and he supervised the project, analysed the data and contributed to writing. S.L. is a PhD co-adviser to C.J. and PhD adviser to B.Y., and he supervised the project, analysed the data and contributed to writing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Methods, Figs. 1–37 and Tables 1–5.
Supplementary Video 1 (download MP4 )
NN-MD simulation for 3 nm Pt NP on Ce2O3 (100)-Ce terminated surface.
Supplementary Video 2 (download MP4 )
NN-MD simulation for 3 nm Pt NP on CeO2 (110) surface.
Supplementary Video 3 (download MP4 )
NN-MD simulation for 3 nm Pt NP on anatase-TiO2 (101) surface.
Supplementary Video 4 (download MP4 )
NN-MD simulation for one 3 nm Pt NP surrounded by four 1 nm Pt NPs on Ce2O3 (100)-Ce terminated surface.
Supplementary Video 5 (download MP4 )
NN-MD simulation for one 3 nm Pt NP surrounded by four 1 nm Pt NPs on CeO2 (110) surface.
Supplementary Video 6 (download MP4 )
NN-MD simulation for one 3 nm Pt NP surrounded by four 1 nm Pt NPs on anatase-TiO2 (101) surface.
Supplementary Data 1 (download XLSX )
High-throughput screening results summary.
Supplementary Data 2 (download XLSX )
Promising sinter-resistant supports materials summary.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jiang, C., Yan, B., Goldsmith, B.R. et al. Predictive model for the discovery of sinter-resistant supports for metallic nanoparticle catalysts by interpretable machine learning. Nat Catal 8, 1038–1050 (2025). https://doi.org/10.1038/s41929-025-01417-3
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41929-025-01417-3
This article is cited by
-
A data-driven leap towards stable catalysts
Nature Catalysis (2025)