Abstract
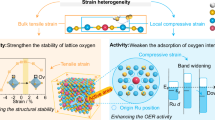
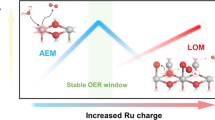
The durability of proton-exchange-membrane water electrolysers (PEMWE) is strongly influenced by the purity of the feedwater. Reverse osmosis (RO) is a cost-effective purification method, but the residual ions usually cause rapid degradation. Here we show that a standard PEMWE equipped with a cobalt-doped ruthenium dioxide (Co–RuO2) anode catalyst can operate stably for 2,000 h at 1.0 A cm−2 using RO-level impure water, with a degradation rate of 10.2 μV h−1. The catalyst provides two complementary protections: Co sites selectively and reversibly capture chloride ions (Cl−), forming a shielding layer against anions corrosion, and strain-activated Ru sites create a proton-rich interface that blocks impurity cations. Together, these effects maintain electrode activity and membrane conductivity. As a result, RO water electrolysis achieves a durability comparable to pure water operation while retaining the cost benefits of seawater-derived purification, offering a practical route towards efficient and affordable hydrogen production.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the paper and its Supplementary Information. Additional data supporting the findings of this study are available from the corresponding author upon reasonable request. Source data are provided with this paper.
References
Lagadec, M. F. & Grimaud, A. Water electrolysers with closed and open electrochemical systems. Nat. Mater. 19, 1140–1150 (2020).
Chong, L. et al. La- and Mn-doped cobalt spinel oxygen evolution catalyst for proton exchange membrane electrolysis. Science 380, 609–616 (2023).
Wu, Z.-Y. et al. Non-iridium-based electrocatalyst for durable acidic oxygen evolution reaction in proton exchange membrane water electrolysis. Nat. Mater. 22, 100–108 (2023).
Ram, R. et al. Water-hydroxide trapping in cobalt tungstate for proton exchange membrane water electrolysis. Science 384, 1373–1380 (2024).
Lin, C. et al. In-situ reconstructed Ru atom array on α-MnO2 with enhanced performance for acidic water oxidation. Nat. Catal. 4, 1012–1023 (2021).
Dresp, S. et al. Efficient direct seawater electrolysers using selective alkaline NiFe-LDH as OER catalyst in asymmetric electrolyte feeds. Energy Environ. Sci. 13, 1725–1729 (2020).
Dresp, S. et al. Molecular understanding of the impact of saline contaminants and alkaline pH on NiFe layered double hydroxide oxygen evolution catalysts. ACS Catal. 11, 6800–6809 (2021).
Hausmann, J. N. et al. Hyping direct seawater electrolysis hinders electrolyzer development. Joule 8, 2436–2442 (2024).
Hausmann, J. N., Schlögl, R., Menezes, P. W. & Driess, M. Is direct seawater splitting economically meaningful? Energy Environ. Sci. 14, 3679–3685 (2021).
Frisch, M. L. et al. Seawater electrolysis using all-PGM-free catalysts and cell components in an asymmetric feed. ACS Energy Lett. 8, 2387–2394 (2023).
Liu, Y. et al. Long-term durability of seawater electrolysis for hydrogen: from catalysts to systems. Angew. Chem. Int. Ed. 63, e202412087 (2024).
Tong, W. et al. Electrolysis of low-grade and saline surface water. Nat. Energy 5, 367–377 (2020).
Shi, W. et al. Ultrastable supported oxygen evolution electrocatalyst formed by ripening-induced embedding. Science 387, 791–796 (2025).
Lin, H. Y. et al. Leaching-induced Ti trapping stabilizes amorphous IrOx for proton exchange membrane water electrolysis. Angew. Chem. Int. Ed. 64, e202504212 (2025).
Venkatesan, S. et al. Rapid scalable one-step production of catalysts for low-iridium content proton exchange membrane water electrolyzers. Adv. Energy Mater. 15, 2401659 (2025).
Lindquist, G. A., Xu, Q., Oener, S. Z. & Boettcher, S. W. Membrane electrolyzers for impure-water splitting. Joule 4, 2549–2561 (2020).
Vos, J. G., Wezendonk, T. A., Jeremiasse, A. W. & Koper, M. T. M. MnOx/IrOx as selective oxygen evolution electrocatalyst in acidic chloride solution. J. Am. Chem. Soc. 140, 10270–10281 (2018).
Vos, J. G. et al. Selectivity trends between oxygen evolution and chlorine evolution on iridium-based double perovskites in acidic media. ACS Catal. 9, 8561–8574 (2019).
Kim, J., Usama, M., Exner, K. S. & Joo, S. H. Renaissance of chlorine evolution reaction: emerging theory and catalytic materials. Angew. Chem. Int. Ed. 64, e202417293 (2025).
Cui, R. et al. Effect of cations (Na+, Co2+, Fe3+) contamination in Nafion membrane: a molecular simulations study. Int. J. Hydrog. Energy 50, 635–649 (2024).
Chen, J. et al. Chloride residues in RuO2 catalysts enhance its stability and efficiency for acidic oxygen evolution reaction. Angew. Chem. Int. Ed. 64, e202420860 (2025).
Ryan, J. V. et al. Electronic connection to the interior of a mesoporous insulator with nanowires of crystalline RuO2. Nature 406, 169–172 (2000).
Wang, Y. et al. Unraveling oxygen vacancy site mechanism of Rh-doped RuO2 catalyst for long-lasting acidic water oxidation. Nat. Commun. 14, 1412 (2023).
Bajdich, M., García-Mota, M., Vojvodic, A., Nørskov, J. K. & Bell, A. T. Theoretical investigation of the activity of cobalt oxides for the electrochemical oxidation of water. J. Am. Chem. Soc. 135, 13521–13530 (2013).
Shan, J. et al. Short-range ordered iridium single atoms integrated into cobalt oxide spinel structure for highly efficient electrocatalytic water oxidation. J. Am. Chem. Soc. 143, 5201–5211 (2021).
Lee, S., Moysiadou, A., Chu, Y.-C., Chen, H. M. & Hu, X. Tracking high-valent surface iron species in the oxygen evolution reaction on cobalt iron (oxy) hydroxides. Energy Environ. Sci. 15, 206–214 (2022).
Kang, W. et al. Unraveling sequential oxidation kinetics and determining roles of multi-cobalt active sites on Co3O4 catalyst for water oxidation. J. Am. Chem. Soc. 145, 3470–3477 (2023).
Dong, H., Yu, W. & Hoffmann, M. R. Mixed metal oxide electrodes and the chlorine evolution reaction. J. Phys. Chem. C 125, 20745–20761 (2021).
Li, L. et al. Lanthanide-regulating Ru–O covalency optimizes acidic oxygen evolution electrocatalysis. Nat. Commun. 15, 4974 (2024).
Luo, Z. et al. Water-hydroxide trapping in hollandite-type iridium oxide enables efficient proton exchange membrane water electrolysis. Adv. Funct. Mater. 35, 2500044 (2025).
Kang, J. et al. Synthesis, molecular structure, and water electrolysis performance of TiO2-supported Raney-IrOx nanoparticles for the acidic oxygen evolution reaction. ACS Catal. 15, 5435–5446 (2025).
Kang, J. et al. Highly active IrRuOx/MnOx electrocatalysts with ultralow anode PGM demand in proton exchange membrane electrolyzers. Adv. Energy Mater. https://doi.org/10.1002/aenm.202405758 (2025).
Zlatar, M. et al. Evaluating the stability of Ir single atom and Ru atomic cluster oxygen evolution reaction electrocatalysts. Electrochim. Acta 444, 141982 (2023).
Li, A. et al. Atomically dispersed hexavalent iridium oxide from MnO2 reduction for oxygen evolution catalysis. Science 384, 666–670 (2024).
Acknowledgements
S.-Z.Q. acknowledges financial support provided by the Australian Research Council through the Discovery and Linkage Project Programs (DP230102027, IL230100039 and CE230100032). Y.Z. acknowledges financial support provided by the Australian Research Council through the Discovery and Linkage Project Programs (DP240102575 and CE230100032). We acknowledge Y. Hu at Lanzhou University for support with microscopy analysis. We acknowledge the Australian Synchrotron for access to the beamline facilities (mode 3).
Author information
Authors and Affiliations
Contributions
Y.Z. and S.-Z.Q. conceived and supervised the work. H.L. designed and carried out experiments. X.S. and H.L. conducted the technology economic analysis. H.L. and F.-Y.G. conducted the PEMWE tests. H.L., Y.Z. and S.-Z.Q. wrote and corrected the paper. All authors approved the final version of the paper.
Corresponding authors
Ethics declarations
Competing interests
H.L., Y.Z. and S.-Z.Q. are inventors on a patent application related to the technology described in this work.
Peer review
Peer review information
Nature Catalysis thanks Mark Mba-Wright, Ryuhei Nakamura and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Notes 1 and 2, Figs. 1–34, Table 1 and References.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data for Fig. 1.
Source Data Fig. 2 (download XLSX )
Statistical source data for Fig. 2.
Source Data Fig. 3 (download XLSX )
Statistical source data for Fig. 3.
Source Data Fig. 4 (download XLSX )
Statistical source data for Fig. 4.
Source Data Fig. 5 (download XLSX )
Statistical source data for Fig. 5.
Source Data Fig. 6 (download XLSX )
Statistical source data for Fig. 6.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, H., Sun, X., Gao, FY. et al. Cost-efficient and stable electrolysis of reverse osmosis water using a Co-RuO2-enabled PEM electrolyser. Nat Catal 9, 9–17 (2026). https://doi.org/10.1038/s41929-025-01456-w
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41929-025-01456-w
This article is cited by
-
Shielding PEM electrolysers from real-world water
Nature Catalysis (2026)
-
Systematic design of photovoltaic–electrolyzer coupled systems for energy storage
Research on Chemical Intermediates (2026)