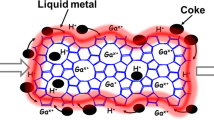
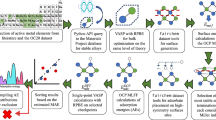
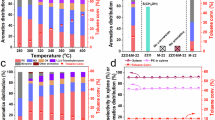
Abstract
Methanol-to-hydrocarbons (MTH) processes offer the potential for transitioning the production of base chemicals and liquid fuels to alternative fossil and renewable resources. The commercialization of ZSM-5 (MFI) and SAPO-34 (CHA) catalysts, coupled with the complexity of the reaction mechanism, has spurred extensive research to increase the selectivity for desired hydrocarbons and also mitigate coke-induced deactivation. Here we present a quantitative performance assessment of the zeolites and zeotypes studied as MTH catalysts over the past three decades. We leverage a comprehensive dataset to evaluate selectivity–stability profiles and examine how structural descriptors and operating conditions govern performance, with additional insights imparted through machine-learning-based analytics. These structure–conditions–performance relationships are discussed in the context of current mechanistic understanding, providing a perspective on MTH catalysis. The analysis shows that product selectivity and coking sensitivity are strongly determined by catalyst structure, particularly the micropore topology, but are also influenced by conditions such as temperature and the addition of co-feeds, which substantially influence competing pathways.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The data that support the findings of this study are provided in Supplementary Information. Data are also available from the corresponding authors upon request.
References
Olsbye, U. et al. Conversion of methanol to hydrocarbons: how zeolite cavity and pore size controls product selectivity. Angew. Chem. Int. Ed. 51, 5810–5831 (2012).
Tian, P., Wei, Y., Ye, M. & Liu, Z. Methanol to olefins (MTO): from fundamentals to commercialization. ACS Catal. 5, 1922–1938 (2015).
Yarulina, I., Chowdhury, A. D., Meirer, F., Weckhuysen, B. M. & Gascon, J. Recent trends and fundamental insights in the methanol-to-hydrocarbons process. Nat. Catal. 1, 398–411 (2018).
Sattler, J. J. H. B., Ruiz-Martinez, J., Santillan-Jimenez, E. & Weckhuysen, B. M. Catalytic dehydrogenation of light alkanes on metals and metal oxides. Chem. Rev. 114, 10613–10653 (2014).
Li, T., Shoinkhorova, T. & Gascon, J. Aromatics production via methanol‑mediated transformation routes. ACS Catal. 11, 7780–7819 (2021).
Hirunsit, P., Senocrate, A., Gómez-Camacho, C. E. & Kiefer, F. From CO2 to sustainable aviation fuel: navigating the technology landscape. ACS Sustain. Chem. Eng. 12, 12143–12160 (2024).
Ilias, S. & Bhan, A. Mechanism of the catalytic conversion of methanol to hydrocarbons. ACS Catal. 3, 18–31 (2013).
Van Speybroeck, V. et al. First principle chemical kinetics in zeolites: the methanol-to-olefin process as a case study. Chem. Soc. Rev. 43, 7326–7357 (2014).
Bertau, M., Wernicke, H.-J. & Schmidt, F. in Methanol: The Basic Chemical and Energy Feedstock of the Future (eds Bertau, M. et al.) 423–499 (Springer, 2014).
Chen, N. Y. & Reagan, W. J. Evidence of autocatalysis in methanol to hydrocarbon reactions over zeolite catalysts. J. Catal. 59, 123–129 (1979).
Dessau, R. M. & LaPierre, R. B. On the mechanism of methanol conversion to hydrocarbons over HZSM-5. J. Catal. 78, 136–141 (1982).
Mole, T., Bett, G. & Seddon, D. Conversion of methanol to hydrocarbons over ZSM-5 zeolite: an examination of the role of aromatic hydrocarbons using 13carbon- and deuterium-labeled feeds. J. Catal. 84, 435–445 (1983).
Dahl, I. M. & Kolboe, S. On the reaction mechanism for hydrocarbon formation from methanol over SAPO‑34. 2. Isotopic labeling studies of the co‑reaction of propene and methanol. J. Catal. 161, 304–309 (1994).
Haw, J. F. et al. Roles for cyclopentenyl cations in the synthesis of hydrocarbons from methanol on zeolite catalyst HZSM-5. J. Am. Chem. Soc. 122, 4763–4775 (2000).
Arstad, B. & Kolboe, S. Methanol-to-hydrocarbons reaction over SAPO-34. Molecules confined in the catalyst cavities at short time on stream. Catal. Lett. 71, 209–212 (2001).
Svelle, S. et al. Conversion of methanol into hydrocarbons over zeolite H-ZSM-5: ethene formation is mechanistically separated from the formation of higher alkenes. J. Am. Chem. Soc. 128, 14770–14771 (2006).
Brogaard, R. Y. et al. Methanol-to-hydrocarbons conversion: the alkene methylation pathway. J. Catal. 314, 159–169 (2014).
Sun, X. et al. On reaction pathways in the conversion of methanol to hydrocarbons on HZSM-5. J. Catal. 317, 185–197 (2014).
Zhang, W. et al. Influences of the confinement effect and acid strength of zeolite on the mechanisms of methanol-to-olefins conversion over H-ZSM-5: a theoretical study of alkenes-based cycle. Microporous Mesoporous Mater. 231, 216–229 (2016).
Xue, Y. et al. Co-reaction of methanol with butene over a high-silica H-ZSM-5 catalyst. J. Catal. 367, 315–325 (2018).
Borodina, E. et al. Influence of the reaction temperature on the nature of the active and deactivating species during methanol‑to‑olefins conversion over H‑SAPO‑34. ACS Catal. 7, 5268–5281 (2017).
Wang, S. et al. Polymethylbenzene or alkene cycle? Theoretical study on their contribution to the process of methanol to olefins over H‑ZSM‑5 zeolite. J. Phys. Chem. C 119, 28482–28498 (2015).
Jang, H. G., Min, H. K., Hong, S. B. & Seo, G. Tetramethylbenzenium radical cations as major active intermediates of methanol-to-olefin conversions over phosphorous-modified HZSM-5 zeolites. J. Catal. 299, 240–248 (2013).
Dai, W. et al. Intermediates and dominating reaction mechanism during the early period of the methanol-to-olefin conversion on SAPO-41. J. Phys. Chem. C 119, 2637–2645 (2015).
Zhang, W. et al. Methanol‑to‑olefins reaction over cavity‑type zeolite: cavity controls the critical intermediates and product selectivity. ACS Catal. 8, 10950–10963 (2018).
Hernandez, E. D. & Jentoft, F. C. Spectroscopic signatures reveal cyclopentenyl cation contributions in methanol‑to‑olefins catalysis. ACS Catal. 10, 5764–5782 (2020).
Zhang, W. et al. Methanol‑to‑olefins reaction route based on methylcyclopentadienes as critical intermediates. ACS Catal. 9, 7373–7379 (2019).
Dai, W. et al. Understanding the early stages of the methanol‑to‑olefin conversion on H‑SAPO‑34. ACS Catal. 5, 317–326 (2015).
Minova, I. B. et al. Elementary steps in the formation of hydrocarbons from surface methoxy groups in HZSM‑5 seen by synchrotron infrared microspectroscopy. ACS Catal. 9, 6564–6570 (2019).
Zhou, J. et al. Directed transforming of coke to active intermediates in methanol-to-olefins catalyst to boost light olefins selectivity. Nat. Commun. 12, 17 (2021).
Ilias, S. & Bhan, A. The mechanism of aromatic dealkylation in methanol-to-hydrocarbons conversion on H-ZSM-5: what are the aromatic precursors to light olefins? J. Catal. 311, 6–16 (2014).
Wang, C. et al. New insight into the hydrocarbon‑pool chemistry of the methanol‑to‑olefins conversion over zeolite H‑ZSM‑5 from GC‑MS, solid‑state NMR spectroscopy, and DFT calculations. Chem. A Eur. J. 20, 12432–12443 (2014).
VanSpeybroeck, V. et al. Mechanistic studies on chabazite‑type methanol‑to‑olefin catalysts: insights from time‑resolved UV/Vis microspectroscopy combined with theoretical simulations. ChemCatChem 5, 173–184 (2013).
Ferri, P. Chemical and structural parameters connecting cavity architecture, confined hydrocarbon-pool species, and MTO product selectivity in small-pore cage-based zeolites. ACS Catal. 9, 11542–11551 (2019).
Vandichel, M., Lesthaeghe, D., van der Mynsbrugge, J., Waroquier, M. & van Speybroeck, V. Assembly of cyclic hydrocarbons from ethene and propene in acid zeolite catalysis to produce active catalytic sites for MTO conversion. J. Catal. 271, 67–78 (2010).
Müller, S. et al. Hydrogen transfer pathways during zeolite‑catalyzed methanol conversion to hydrocarbons. J. Am. Chem. Soc. 138, 15994–16003 (2016).
Martínez-Espín, J. S. et al. Hydrogen transfer versus methylation: on the genesis of aromatics formation in the methanol‑to‑hydrocarbons reaction over H‑ZSM‑5. ACS Catal. 7, 5773–5780 (2017).
Müller, S. et al. Coke formation and deactivation pathways on H-ZSM-5 in the conversion of methanol to olefins. J. Catal. 325, 48–59 (2015).
Lee, S. & Choi, M. Unveiling coke formation mechanism in MFI zeolites during methanol-to-hydrocarbons conversion. J. Catal. 375, 183–192 (2019).
Wennmacher, J. T. C. et al. Electron diffraction enables the mapping of coke in ZSM‑5 micropores formed during methanol‑to‑hydrocarbons conversion. Angew. Chem. Int. Ed. 61, e202205413 (2022).
Rzepka, P., Sheptyakov, D., Wang, C., van Bokhoven, J. A. & Paunović, V. How micropore topology influences the structure and location of coke in zeolite catalysts. ACS Catal. 14, 5593–5604 (2024).
Wei, Y. et al. Coke formation and carbon atom economy of methanol-to-olefins reaction. ChemSusChem 5, 906–912 (2012).
Wang, C. et al. Structural changes of ZSM‑5 catalysts during methanol‑to‑hydrocarbons conversion processes. ACS Catal. 14, 12410–12424 (2024).
Wang, N. et al. Molecular elucidating of an unusual growth mechanism for polycyclic aromatic hydrocarbons in confined space. Nat. Commun. 11, 1079 (2020).
Martinez-Espin, J. S. et al. New insights into catalyst deactivation and product distribution of zeolites in the methanol-to-hydrocarbons (MTH) reaction with methanol and dimethyl ether feeds. Catal. Sci. Technol. 7, 2700–2716 (2017).
Hwang, A., Kumar, M., Rimer, J. D. & Bhan, A. Implications of methanol disproportionation on catalyst lifetime for methanol-to-olefins conversion by HSSZ-13. J. Catal. 346, 154–160 (2017).
Ni, Y., Zhu, W. & Liu, Z. H‑ZSM‑5‑catalyzed hydroacylation involved in the coupling of methanol and formaldehyde to aromatics. ACS Catal. 9, 11398–11403 (2019).
Zhao, X. et al. Achieving a superlong lifetime in the zeolite‑catalyzed MTO reaction under high pressure: synergistic effect of hydrogen and water. ACS Catal. 9, 3017–3025 (2019).
Paunović, V. et al. The formation, reactivity and transformation pathways of formaldehyde in the methanol-to-hydrocarbon conversion. Catal. Sci. Technol. 14, 1216–1228 (2024).
Pare, C. W. P. et al. Formaldehyde‑induced deactivation of ZSM‑5 catalysts during the methanol‑to‑hydrocarbons conversion. ACS Catal. 14, 463–474 (2024).
Liu, Y. et al. Critical role of formaldehyde during methanol conversion to hydrocarbons. Nat. Commun. 10, 1462 (2019).
Liu, Y. et al. Formation mechanism of the first carbon-carbon bond and the first olefin in the methanol conversion into hydrocarbons. Angew. Chem. Int. Ed. 55, 5723–5726 (2016).
Kilburn, L., DeLuca, M., Hoffman, A. J., Patel, S. & Hibbitts, D. Comparing alkene-mediated and formaldehyde-mediated diene formation routes in methanol-to-olefins catalysis in MFI and CHA. J. Catal. 400, 124–139 (2021).
Wei, Z. et al. Methane formation mechanism in the initial methanol-to-olefins process catalyzed by SAPO-34. Catal. Sci. Technol. 6, 5526–5533 (2016).
Paunović, V., Hemberger, P., Bodi, A., Hauert, R. & van Bokhoven, J. A. Impact of nonzeolite-catalyzed formation of formaldehyde on the methanol-to-hydrocarbons conversion. ACS Catal. 12, 13426–13434 (2022).
Bleken, F. et al. Conversion of methanol over 10-ring zeolites with differing volumes at channel intersections: comparison of TNU-9, IM-5, ZSM-11 and ZSM-5. Phys. Chem. Chem. Phys. 13, 2539–2549 (2011).
Teketel, S. et al. Shape selectivity in the conversion of methanol to hydrocarbons: the catalytic performance of one-dimensional 10‑ring zeolites: ZSM-22, ZSM-23, ZSM-48 and EU-1. ACS Catal. 2, 26–37 (2012).
Dyballa, M. et al. Parameters influencing the selectivity to propene in the MTO conversion on 10-ring zeolites: directly synthesized zeolites ZSM-5, ZSM-11 and ZSM-22. Appl. Catal. A Gen. 510, 233–243 (2016).
Kang, J. H. et al. Further studies on how the nature of zeolite cavities that are bounded by small pores influences the conversion of methanol to light olefins. ChemPhysChem 19, 412–419 (2018).
Kang, J. H., Alshafei, F. H., Zones, S. I. & Davis, M. E. Cage-defining ring: a molecular sieve structural indicator for light olefin product distribution from the methanol-to-olefins reaction. ACS Catal. 9, 6012–6019 (2019).
Ferri, P. et al. Impact of zeolite framework composition and flexibility on methanol-to-olefins selectivity: confinement or diffusion?. Angew. Chem. Int. Ed. 59, 19708–19715 (2020).
Li, C. et al. Synthesis of reaction-adapted zeolites as methanol-to-olefins catalysts with mimics of reaction intermediates as organic structure-directing agents. Nat. Catal. 1, 547–554 (2018).
Janssens, T. V. W. A new approach to the modeling of deactivation in the conversion of methanol on zeolite catalysts. J. Catal. 264, 130–137 (2009).
Janssens, T. V. W., Svelle, S. & Olsbye, U. Kinetic modeling of deactivation profiles in the methanol-to-hydrocarbons (MTH) reaction: a combined autocatalytic-hydrocarbon pool approach. J. Catal. 308, 122–130 (2013).
Shi, Z. & Bhan, A. Metrics of performance relevant in methanol-to-hydrocarbons catalysis. J. Catal. 421, 198–209 (2023).
Rojo-Gama, D. et al. Structure-deactivation relationships in zeolites during the methanol-to-hydrocarbons reaction: complementary assessments of the coke content. J. Catal. 351, 33–48 (2017).
Wang, S. et al. Relation of catalytic performance to the aluminum siting of acidic zeolites in the conversion of methanol to olefins, viewed via a comparison between ZSM‑5 and ZSM‑11. ACS Catal. 8, 5485–5505 (2018).
Shoinkhorova, T. et al. Highly selective and stable production of aromatics via high-pressure methanol conversion. ACS Catal. 11, 3602–3613 (2021).
Li, C. et al. Selective introduction of acid sites in different confined positions in ZSM‑5 and its catalytic implications. ACS Catal. 8, 7688–7697 (2018).
Liang, T. et al. Conversion of methanol to olefins over H‑ZSM‑5 zeolite: reaction pathway is related to the framework aluminum siting. ACS Catal. 6, 7311–7325 (2016).
Le, T. T. et al. Elemental zoning enhances mass transport in zeolite catalysts for methanol to hydrocarbons. Nat. Catal. 6, 254–265 (2023).
Bleken, F. et al. The effect of acid strength on the conversion of methanol to olefins over acidic microporous catalysts with the CHA topology. Top. Catal. 52, 218–228 (2009).
Zhu, X. et al. Trimodal porous hierarchical SSZ‑13 zeolite with improved catalytic performance in the methanol‑to‑olefins reaction. ACS Catal. 6, 2163–2177 (2016).
Dai, W., Wu, G., Li, L., Guan, N. & Hunger, M. Mechanisms of the deactivation of SAPO‑34 materials with different crystal sizes applied as MTO catalysts. ACS Catal. 3, 588–596 (2013).
Yarulina, I. et al. Structure–performance descriptors and the role of Lewis acidity in the methanol-to-propylene process. Nat. Chem. 10, 804–812 (2018).
Liutkova, A. et al. Role of strontium cations in ZSM‑5 zeolite in the methanol‑to‑hydrocarbons reaction. J. Phys. Chem. Lett. 14, 6506–6512 (2023).
Arora, S. S., Nieskens, D. L. S., Malek, A. & Bhan, A. Lifetime improvement in methanol-to-olefins catalysis over chabazite materials by high-pressure H2 co-feeds. Nat. Catal. 1, 666–672 (2018).
Altman, N. & Krzywinski, M. Points of significance: association, correlation and causation. Nat. Methods 12, 899–900 (2015).
Esterhuizen, J. A., Goldsmith, B. R. & Linic, S. Interpretable machine learning for knowledge generation in heterogeneous catalysis. Nat. Catal. 5, 175–184 (2022).
Database of Zeolite Structures (IZA, 2017); https://www.iza-structure.org/databases/
Lundberg, S. M. & Lee, S. I. A unified approach to interpreting model predictions. In Proc. Advanced Neural Information Processing Systems (eds Guyon, I. et al.), 4766–4775 (Curran Associates, 2017).
Apley, D. W. & Zhu, J. Visualizing the effects of predictor variables in black box supervised learning models. J. R. Stat. Soc. Ser. B Stat. Methodol. 82, 1059–1086 (2020).
Hwang, A. & Bhan, A. Bifunctional strategy coupling Y2O3‑catalyzed alkanal decomposition with methanol‑to‑olefins catalysis for enhanced lifetime. ACS Catal. 7, 4417–4422 (2017).
Barbera, K., Bonino, F., Bordiga, S., Janssens, T. V. W. & Beato, P. Structure-deactivation relationship for ZSM-5 catalysts governed by framework defects. J. Catal. 280, 196–205 (2011).
Dai, H. et al. Finned zeolite catalysts. Nat. Mater. 19, 1074–1080 (2020).
ScanIT software. https://www.amsterchem.com/scanit.html (Amsterchem, 2024).
Toyao, T. et al. Machine learning for catalysis informatics: recent applications and prospects. ACS Catal. 10, 2260–2297 (2020).
Acknowledgements
We acknowledge financial support from the Paul Scherrer Institut and ETH Zurich.
Author information
Authors and Affiliations
Contributions
V.P. conceived and designed the study, analysed the data, contributed analysis tools and wrote the paper. G.L. and K.S. contributed to analysis tools. J.A.v.B. contributed to data analysis and paper writing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks Abhishek Dutta Chowdhury and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Table of contents. Supplementary Table 1: structural information for different frameworks. Supplementary Table 2: catalysts, their properties, operating regime, and performance metrics in the MTH conversion. Supplementary Fig. 1: correlation matrix based on Spearman’s correlation coefficients (ρ) for different structural and reaction descriptors described in Fig. 7 of the main textt. Supplementary references.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Paunović, V., Liesche, G., Sundmacher, K. et al. Exploring the landscape of methanol-to-hydrocarbons conversion catalysts. Nat Catal 9, 348–362 (2026). https://doi.org/10.1038/s41929-026-01506-x
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41929-026-01506-x