Abstract
The emergence of highly pathogenic avian influenza viruses (HPAIV) has caused widespread mortality wildlife globally. In 2023, mass mortalities of southern elephant seals Mirounga leonina were observed in South America, and the virus subsequently reached the sub-Antarctic, affecting multiple species. The remoteness of these islands has limited assessment of its true impact. Here we present evidence of HPAIV’s effect on the number of breeding females at the world’s largest southern elephant seal population at South Georgia. Following the virus’ arrival in 2023, we recorded a 47% (SD = 14.2%) decline in the number of breeding females at the three largest breeding colony beaches in 2024 compared to 2022. The apparent loss of nearly half the breeding female population has serious implications for recruitment and future stability of the population. These findings highlight the urgent need for continued, intensive monitoring to track the long-term effects on this species.
Similar content being viewed by others
Introduction
Southern elephant seals Mirounga leonina are the largest of the pinnipeds, and are a major predator with a circumpolar distribution in the Southern Hemisphere1. Southern elephant seals breed annually and come ashore on sub-Antarctic islands2 at the end of the Austral winter, where they form dense colonies comprised of competitive harems on beaches3,4,5. Large males haul out first, towards the end of September and early October, followed by pregnant females, which give birth ~3–5 days post-arrival6,7. Pups are then weaned approximately 22–23 days post-partum, with females coming into oestrus several days prior to weaning6,8,9. The number of female arrivals and departures follows a Gaussian (normal) distribution, where numbers are typically 50% of the maximum two to three weeks either side of the breeding peak3,6,7,10, which, at South Georgia, has historically fallen during the final week of October3,11,12.
Four genetically distinct populations have been identified within the Southern Hemisphere: the Peninsula Valdés population in Argentina; (ii) the South Georgia population, which includes the South Sandwich and Falkland Islands, in the South Atlantic; the Macquarie population in the South Pacific; and finally the Heard and Kerguelen populations, which includes the Crozet and Prince Edward archipelagos, in the south Indian Ocean13,14. These four broad areas comprise the principal breeding locations for this species in the Southern Ocean.
Population trajectories are varied throughout the Southern Ocean. The Peninsula Valdés population in Argentina has been growing at between 1 and 3.4% annually for the last five decades15. At its last census in 1995, the South Georgia population was deemed to be stable and accounted for ~54% of the global breeding population3. At Macquarie Island, the population declined through much of the last century, before growing slightly and once again declining more recently, with the overall population being negatively correlated with sea ice concentration16. In the Indian Ocean sector, populations on the Prince Edward archipelago, namely Marion Island, have declined by ~83% since the 1950s, with a more recent attrition rate of 5.8% annually17,18. At Îles Crozet, populations have decreased by 5.4% annually between 1970 and 199019,20,21. At Îles Kerguelen, the population almost halved in size from 70,000 females in 1952 to 37,400 in 198720. Following this, the population began to increase at almost 1% annually between 1987 and 200920,22. However, recent evidence suggests that both Îles Crozet and Îles Kerguelen populations have entered a growth phase, increasing annually at 5.1% and 1.6%, respectively23. Available population data from Heard Island is over three decades old, but trends showed a decrease of ~50% from 1949 to 1985, which was linked to changes in sea-ice dynamics, followed by a period of relative stability from 1985 to 199224.
The expansion of highly pathogenic avian influenza viruses (HPAIV) across the globe, notably from clade 2.3.4.4b in 2020, has impacted mortality in wildlife populations globally25. Initially detected in Europe26, this clade gained traction and crossed into North America27,28, before spreading down into South America29,30, culminating in mass mortalities of seabirds and marine mammals in 202229,31,32,33,34. Background sampling of sites through the sub-Antarctic and Antarctic region showed that, as of March 2023, HPAIV had not been carried into this region35. However, in September 2023, the first suspected avian case was reported in brown skuas, Stercorarius antarcticus, on Bird Island, South Georgia36, with confirmed mammalian occurrence of HPAIV several months later in both Antarctic fur seals Arctocephalus gazella and southern elephant seals31,36. As the season progressed, HPAIV was also confirmed in multiple other species island-wide36. In 2024, HPAIV was documented in the Indian Ocean, having been transferred from South Georgia to Îles Crozet and Îles Kerguelen37.
Monitoring for HPAIV was conducted at South Georgia throughout the 2023/24 season, with opportunistic samples taken, where possible, at landing sites island-wide36. Transient observations, however, likely do not capture the true extent of the impact, as this virus affected southern elephant seals whilst ashore. Reports submitted from cruise ships38 and from research activities (JC pers. comms.) suggest that the impact of this outbreak is likely comparable to the mass mortalities observed in the Argentinian Valdés population, which saw pup mortality of 97%39. Here, we present dedicated aerial survey data that indicates population decreases of comparable magnitudes at South Georgia, with surveys of the three largest breeding colonies in 2024 indicating an average reduction in female seal attendance of 47% (SD = 14.2%).
Results
In 2024, seal breeding peaks were observed at St Andrews Bay on the 22nd October (4373 females ashore) and at Hound Bay on the 21st October (1154 females ashore), with subsequent counts showing declines. For the purpose of the HPAIV comparison, the number of females ashore on the comparison dates (Fig. 1) were 5.6% lower than the observed peak at St Andrews Bay (27th October), and 7.6% lower than the observed peak at Hound Bay (26th October). Peak dates were not available for Gold Harbour as, due to access issues, a time series could not be collected. Over both years, counts between the two observers were on average within 0.87% (SD = 1.1%) of each other.
“Survey dates for flights of the three largest southern elephant seal colonies on South Georgia”. “Dates of flights from the 2024 (black) and 2022 (red) seasons at the three largest southern elephant seal (Mirounga leonina) breeding colony beaches on South Georgia”.
In 2022, counts at St Andrews Bay, Gold Harbour and Hound Bay derived from ortho-rectified mosaics revealed that 6305 (95% CI: 6087.9–6528.1), 1599 (95% CI: 1538–1758) and 1901 (95% CI: 1784.2–2025.8) females were ashore at each of these respective beaches. Comparative counts from 2024 revealed an average reduction of 47% (SD = 14.2%) in the number of female seals present between 2022 and 2024, with only 4128 (95% CI: −3968.1 to 4323.9), 601 (95% CI: 538.2–673.8) and 1066 (95% CI: 982.6–1163.4) females present on each of the three colonies, respectively. When compared to long-term average counts (1958–2022), the observed number of females in 2024 constitutes an average decrease of 33.7% (SD = 2.3%) in the relative number of females present in 2024 (Table 1). If scaled to the entire island population at its last census3, not accounting for population change over the past three decades, we predict about 53,000 females missed breeding in the 2024 season.
Discussion
The last estimate of the South Georgia population, performed 30 years ago (1995), suggested that it represented over 50% of the global population of the species, with breeding sites located around the entirety of the island3. Here, we collected UAV aerial imagery at the three largest breeding beaches in both 2022 and 2024. Inter-observer variation was minimal, suggesting a high level of consistency, with variation stemming from minor adjustments in detection rather than systematic misclassification. Given this, we consider overhead counts to be a highly robust method for detecting population-level trends.
Arrival patterns of female southern elephant seals on breeding colony beaches are known to follow a distinct curve3,6,7,10, which gradually increases to a peak with numbers tailing off afterwards. For a given date, counts revealed an average reduction of 47% between the absolute counts of female seals observed in 2024 compared to 2022. The three counted beaches represent 15.6% of the total population based on the 19953, which, if scaled in line with the last island-wide population estimate in 19953, a reduction of 47% equates to an estimated 53,000 female southern elephant seals failing to return for breeding following the confirmed arrival of HPAIV to South Georgia. Determining the return rate of these animals in future years remains crucial to understanding the dynamics and trends of this population. These findings suggest a substantial reduction in the number of adult females ashore in response to HPAIV.
At South Georgia, the severity of HPAIV’s impact was not spatially uniform36, similar to observations from the sub-Antarctic islands of the Indian Ocean37, yet differing from the apparent uniform impact at Peninsula Valdés39. Smaller or more isolated breeding beaches experienced different apparent infection rates, with factors such as colony size, density, localised transmission rates and species composition influencing outcomes36,37. Therefore, assuming homogeneity may overestimate mortality at South Georgia. Further site-specific assessments are needed to refine population-wide estimates and capture potential regional variation in disease impact.
Regular long-term monitoring of this species has not been carried out at South Georgia; although stints of work have been conducted at Husvik during the 1980s/1990s40,41,42,43 and work has been carried out intermittently in a potentially outlier population (due to its smaller size and location on an inner beach) at King Edward Cove over the past decade. The absence of regular monitoring on South Georgia’s accessible breeding colony beaches complicates our understanding of the observed decline. However, where records exist, in the absence of HPAIV, interannual variation in the number of breeding females present at Gold Harbour indicates a deviation of ±3% between 1959 and 1964 (Table 1). Comparatively, where published, data from sites free of HPAIV and further afield also reflect interannual fluctuations within the same order of magnitude, for instance, between 1990 and 1997 at l’îles de la Possession, Îles Crozet, interannual variation averaged ±7.9%, and on the Courbet Peninsula, Îles Kerguelen, females present fluctuated within ±6.9% between adjacent years21. Similarly, between 1995 and 1997, the population at Peninsula Valdés reported interannual fluctuations of ±5.93%44. These comparative records support our assertion that the observed 47% decrease in females between 2022 and 2024 on South Georgia is atypical.
Due to accessibility, weather and slight differences in breeding phenology, it was not possible to fly all sites on the same day in consecutive years. In 2024, counts were taken either on the same date (St Andrews Bay) or one day later (Hound Bay and Gold Harbour) than in 2022 (Fig. 1). Whilst the later count in 2024 at Hound Bay will have slightly exacerbated the observed decrease, with the count being 7.6% lower than the observed peak (1154 females on the 21st October), this does not offset the magnitude of the observed reduction from 2022. These findings reinforce the atypical low signal in 2024, suggesting the one-day lag is unlikely to significantly affect the observed decline.
Several plausible hypotheses exist that could explain the observed decline between 2022 and 2024 at South Georgia. One explanation for the observed decline could be related to the strenuous conditions of the HPAIV-impacted 2023 breeding season, which resulted in numerous pup mortalities and/or abandonments alongside mortalities in both adult males and females (JC, Pers. Comm.). After losing their pup or abandoning them due to their own HPAIV-induced stress, females may have left the breeding colony beaches prematurely before oestrus, resulting in reduced copulation rates. Subsequently, this would lead to fewer pregnancies and, ultimately, fewer females returning to give birth in the following (monitored) season. Limited observations from Marion Island45 suggest that at-sea copulation may occur, but its rarity implies that it is unlikely to offset reduced terrestrial breeding and low observed attendance in 2024. Alternatively, the emergence of HPAIV in the population may have triggered a shift in previous strong philopatry46,47, with females returning to outlier colonies in 2024, dispersing the population more widely and lowering counts at the historically preferred beaches. If mating took place at sea, a rapid return to normal breeding behaviour and output might be expected. Further long-term investigation into breeding dynamics is needed to understand phenology and recovery from this epidemic.
Finally, an unusual sea-ice anomaly in the South Atlantic during the 2023/2024 austral winter (National Snow and Ice Data Centre—https://nsidc.org/data/seaice_index) may have influenced elephant seal distribution and foraging, affecting their post-breeding/HPAIV recovery. However, given their wide-ranging movements48 and known sea ice-association49, the localised anomalous conditions in the South Atlantic are unlikely to have significantly impacted recovery or altered phenology.
While these hypotheses provide plausible explanations for some of the variation in population numbers, they alone, or in combination, are unlikely to fully account for the dramatic decline observed between 2022 and 2024. The temporal overlap of the arrival and prevalence of HPAIV in the elephant seal population during this period, coupled with the observed reductions, suggests a correlation that is too pronounced to be coincidental.
The long-term impact of the observed decline in the elephant seal population at South Georgia is yet to be determined. Research on adjacent populations has shown that female survival is a critical determinant of population growth50,51. Although we cannot be certain that all female absences are due to mortality, it is probable that a significant portion of these absent seals have perished. At Peninsula Valdés, HPAIV has had devastating effects, with abnormal mortality observed in 202339 and subsequent low attendance in 202452. Demographic projection models for this population suggest that recovery could take decades52, particularly because adult females, normally long-lived with low mortality, have a disproportionate effect on long-term population trends51,53,54. Under severe scenarios, full rebound is unlikely before the next century52.
Due to the remoteness and relative inaccessibility of breeding colony beaches around much of South Georgia, accurately assessing mortality rates remains challenging. The average rate of female absence at South Georgia in 2024, whilst lower than the 67% observed at Peninsula Valdés, still remains significant. If the South Georgia population responds similarly to the modelled outlook at Peninsula Valdés, the future is bleak. This underscores the need for further research to understand the long-term impact on the South Georgia population, which, unlike Peninsula Valdés, lacks regular monitoring.
The average 47% decline observed between 2022 and 2024 across South Georgia’s three largest breeding colony beaches is attributed to the direct impact of HPAIV. This dramatic drop contrasts sharply with historical interannual variations, which typically remain within 10%, both locally and in comparable Southern Ocean populations. While external environmental factors and behavioural shifts may have contributed, they alone cannot explain the severity of this decline.
The scale of this event may impact ongoing recruitment for the world’s largest breeding population of southern elephant seals at South Georgia. To assess its short-term impact and determine whether the missing females represent true mortalities, follow-up monitoring, in line with recommendations55, in 2025 and 2026, is imperative. To assess the long-term impacts on recruitment, sustained monitoring at the major breeding colony beaches is essential. Over recent years, the increased availability of high-resolution satellite imagery has provided an opportunity to examine recent trajectories. Integrating remotely sensed counts with ground data would allow an assessment of the consequences of HPAIV, enabling researchers to distinguish short-term fluctuations from enduring population-level impacts.
Methods
Aerial surveys
Aerial imagery from an uncrewed aerial vehicle (UAV) was collected in the 2022 and 2024 breeding seasons from the three largest breeding colony beaches on South Georgia. Both fieldwork seasons underwent review by the animal ethical approvals board at the British Antarctic Survey and were permitted under AWREB:1071 & 1109 in 2022 and 2024, respectively. These sampled years straddled the emergence of HPAIV on South Georgia. Flights in 2022 were part of an extended field campaign, which targeted multiple locations and species around the South Georgia islands, meaning that sites were often only flown once. Conversely, in 2024, the field campaign specifically focused on southern elephant seals, which enabled sites to be flown multiple times successively (Figs. 1 and 2).
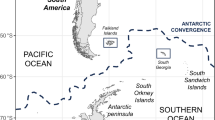
St Andrews Bay, Hound Bay and Gold Harbour (Fig. 1) were selected as they represent 15.6% of the island’s southern elephant seal population at the last census3. St Andrews Bay, representing 7.5%, was flown on the 27th October in both 2022 and 2024; Gold Harbour, representing 4.4%, was flown on the 10th November in both 2022 and 2024; and Hound Bay, representing 3.7%, was flown on the 26th October 2022 and 27th October 2024 (Fig. 2).
“Locations of the largest breeding colonies of southern elephant seal (Mirounga leonina) on South Georgia”. “Sites of the three largest breeding colony beaches of Southern elephant seals (Mirounga leonina) on South Georgia (by total number of breeding females from the 1995 census3) where aerial imagery was collected in 2022 and 2024”.
In both seasons, flights were undertaken using a hand-launched, fixed-wing AgEagle eBee X UAV in fair weather with wind speeds <10 m/s. This UAV has a maximum flight time of ~90 min and was permitted for flights up to 182 m above surface level, although flights typically took ~15 min with a flight altitude of 90 m to achieve a suitable image resolution. The UAV carried a 24 megapixel (6000 \(\times\) 4000 pixel) Aeria X RGB camera designed specifically for photogrammetry and mapping applications, along with a dual-band global navigation satellite system (GNNS) receiver used to determine the position of the image centres accurately to 1.5 cm.
Image processing
Simultaneously with each flight, a Trimble R9 GNSS base station collected precise point positioning (PPP) data following the methodology set out in ref. 56. Their data were used to maximise the quality of the exterior orientation of the downstream image processing using the online Canadian Spatial Reference System Precise Point Positioning (CSRS-PPP, v3) service, applying the International GNSS Service’s (IGS) realisation of the International Terrestrial Reference Frame 2020 (ITRF2020). The precise location of the base station was then used to reprocess the eBee X’s onboard GNSS data using a post-processed kinematics (PPK) workflow in eMotion (v 3.23). This yielded an updated latitude, longitude and height (above mean sea level, MSL) for each image.
Images were then processed following a Structure-from-Motion (SfM) photogrammetry workflow in Pix4D (v.4.9.0). Exterior orientations (x, y, z, \(\Omega\), \(\Phi\) and \({{{\rm{{ K}}}}}\)—yaw, pitch and roll) of the camera during image capture were determined using both PPK solutions and common points within the images. These were used to derive a dense point cloud of the surface within the target area, with processing time being managed by downscaling images by a factor of 16. The dense point cloud was then orthorectified to produce a digital surface model (DSM) onto which the full-resolution original images were mapped; the effect of this was to remove distortion stemming from camera perspective and terrain shape, yielding a single orthorectified image mosaic for each flight.
In each of the ortho-rectified mosaics, adult females were counted, because they provide a reliable indicator of population trends in densely aggregated seal colonies3,57 and are easily distinguishable from other demographics in overhead drone imagery11. Experienced observers, familiar with both the study species and the interpretation of overhead imagery, conducted the count. Using QGIS (v.3.22.16) and ESRI’s ArcMap (v10.8), observers placed a point shapefile at the centre of each visible female along the three beaches. In 2022, each beach was counted twice by different observers, and, due to a high level of agreement between observers in 2022, in 2024 the counting process was streamlined, involving a first pass by one reviewer, followed by a second pass by an independent observer who reviewed the initial count, amending the shapefile for any false positives or negatives.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All UAV survey data are available on request from the NERC EDS UK Polar Data Centre. Data from the DPLUS109 2022/23 season for St Andrews Bay58,59; for Hound Bay60; and Gold Harbour61; these are under embargo until 31st December 2025. Data for the DPLUS214 2024/2025 season are available for St Andrews Bay62; Hound Bay63; and Gold Harbour64; these are under embargo until the 30th June 2026. Prior to these dates, please can interested parties contact the corresponding author to arrange access.
References
Ling, J. K. & Bryden, M. M. in Handbook of Marine Mammals Vol. 2 (eds S.H. Ridgway & R.J. Harrison) 297–328 (Academic Press, 1981).
Hindell, M. A. & Perrin, W. F. in Encyclopedia of Marine Mammals (eds W. F. Perrin, B. Wursig & J. G. M. Thewissen) (Academic Press, Elsevier, 2009).
Boyd, I. L., Walker, T. R. & Poncet, J. Status of southern elephant seals at South Georgia. Antarct. Sci. 8, 237–244 (1996).
van Aarde, R. J. Fluctuations in the population of southern elephant seals Mirounga leonina at Kerguelen Island. Afr. Zool. 15, 99–106 (1980).
Campagna, C., Lewis, M. & Baldi, R. Breeding biology of southern elephant seals in Patagonia. Mar. Mamm. Sci. 9, 34–47 (1993).
Le Boeuf, B. J. & Laws, R. M. Elephant Seals: An Introduction to the Genus. (University of California Press: Berkeley/Los Angeles, CA, 1994).
McCann, T. S. Population structure and social organization of Southern Elephant Seals, Mirounga leonina (L.). Biol. J. Linn. Soc. 14, 133–150 (1980).
Laws, R. The Elephant Seal (Mirounga leonina, Linn.): II. General, social and reproductive behaviour. F. I. D. S. Sci. Rep. 13, 88 (1956).
Carrick, R., Csordas, S. & Ingham, S. E. Studies on the southern elephant seal, Mirounga leonina (L.). IV. Breeding and development. CSIRO Wildl. Res. 7, 161–197 (1962).
Hindell, M. A. & Burton, H. R. Seasonal Haul-Out Patterns of the Southern Elephant Seal (Mirounga leonina L.), at Macquarie Island. J. Mammal. 69, 81–88 (1988).
Dickens, J. et al. Developing UAV Monitoring of South Georgia and the South Sandwich Islands’ iconic land-based marine predators. Front. Mar. Sci. https://doi.org/10.3389/fmars.2021.654215 (2021).
Rothery, P. & McCann, T. in Mammal Population Studies. (Clarendon Press).
Slade, R. W., Moritz, C., Hoelzel, A. R. & Burton, H. R. Molecular Population Genetics of the Southern Elephant Seal Mirounga leonina. Genetics 149, 1945–1957 (1998).
Rus Hoelzel, A., Campagna, C. & Arnbom, T. Genetic and morphometric differentiation between island and mainland southern elephant seal populations. Proc. R. Soc. Lond. Ser. B Biol. Sci. 268, 325–332 (2001).
Ferrari, M. A., Campagna, C., Condit, R. & Lewis, M. N. The founding of a southern elephant seal colony. Mar. Mamm. Sci. 29, 407–423 (2013).
van den Hoff, J. et al. Bottom-up regulation of a pole-ward migratory predator population. Proc. R. Soc. Lond. B Biol. Sci. 281, 20132842 (2014).
McMahon, C. R., Bester, M. N., Hindell, M. A., Brook, B. W. & Bradshaw, C. J. A. Shifting trends: detecting environmentally mediated regulation in long-lived marine vertebrates using time-series data. Oecologia 159, 69–82 (2009).
Oosthuizen, W. C., Bester, M. N., Altwegg, R., McIntyre, T. & de Bruyn, P. J. N. Decomposing the variance in southern elephant seal weaning mass: partitioning environmental signals and maternal effects. Ecosphere 6, art139 (2015).
Barrat, A. & Mougin, J. L’elephant de mer Mirounga leonina de l’ile de la Possession, archipel Crozet (46°25’S, 51°45’E). Mammalia 42, 143–174 (1978).
Guinet, C., Jouventin, P. & Weimerskirch, H. Population changes, movements of southern elephant seals on Crozet and Kerguelen Archipelagos in the last decades. Polar Biol. 12, 349–356 (1992).
Guinet, C., Jouventin, P. & Weimerskirch, H. Recent population change of the southern elephant seal at Îles Crozet and Îles Kerguelen: the end of the decrease?. Antarct. Sci. 11, 193–197 (1999).
Authier, M., Delord, K. & Guinet, C. Population trends of female Elephant Seals breeding on the Courbet Peninsula, îles Kerguelen. Polar Biol. 34, 319–328 (2011).
Laborie, J. et al. Estimation of total population size of southern elephant seals (Mirounga leonina) on Kerguelen and Crozet Archipelagos using very high-resolution satellite imagery. Front. Mar. Sci. https://doi.org/10.3389/fmars.2023.1149100 (2023).
Slip, D. J. & Burton, H. R. Population status and seasonal haulout patterns of the southern elephant seal (Mirounga leonina) at Heard Island. Antarct. Sci. 11, 38–47 (1999).
Wille, M. & Waldenström, J. Weathering the storm of high pathogenicity avian influenza in waterbirds. Waterbirds 46, 100–109, 110 (2023).
Adlhoch, C. et al. Avian influenza overview February—May 2021. EFSA J. 19, e06951 (2021).
Caliendo, V. et al. Transatlantic spread of highly pathogenic avian influenza H5N1 by wild birds from Europe to North America in 2021. Sci. Rep. 12, 11729 (2022).
Alkie, T. N. et al. Recurring trans-atlantic incursion of clade 2.3.4.4b H5N1 viruses by long distance migratory birds from Northern Europe to Canada in 2022/2023. Viruses 15, 1836 (2023).
Gamarra-Toledo, V. et al. Avian flu threatens Neotropical birds. Science 379, 246–246 (2023).
Breed, A. et al. Southward expansion of high pathogenicity avian influenza H5 in wildlife in South America: estimated impact on wildlife populations, and risk of incursion into Antarctica. OFFLU Ad-Hoc Group on HPAI H5 in Wildlife of South America and Antarctica (2023).
Banyard, A. C. et al. Detection and spread of high pathogenicity avian influenza virus H5N1 in the Antarctic Region. Nat. Commun. 15, 7433 (2024).
Leguia, M. et al. Highly pathogenic avian influenza A (H5N1) in marine mammals and seabirds in Peru. Nat. Commun. 14, 5489 (2023).
Ariyama, N. et al. Highly Pathogenic Avian Influenza A(H5N1) Clade 2.3.4.4b Virus in Wild Birds, Chile. Emerg. Infect. Dis. 29, 1842–1845 (2023).
Adlhoch, C. et al. Avian influenza overview march–April 2023. Efsa J. 21, e08039 (2023).
Lisovski, S. et al. No evidence for highly pathogenic avian influenza virus H5N1 (clade 2.3.4.4b) in the Antarctic region during the austral summer 2022/23. BioRxiv https://doi.org/10.1101/2023.10.24.563692 (2023).
Bennison, A. et al. A case study of highly pathogenic avian influenza (HPAI) H5N1 at Bird Island, South Georgia: the first documented outbreak in the subantarctic region. Bird Study https://doi.org/10.1080/00063657.2024.2396563 (2024).
Clessin, A. et al. Mass mortality events in the sub-Antarctic Indian Ocean caused by long-distance circumpolar spread of highly pathogenic avian influenza H5N1 clade 2.3.4.4b. bioRxiv https://doi.org/10.1101/2025.02.25.640068 (2025).
SCAR. Sub-Antarctic and Antarctic Highly Pathogenic Avian Influenza H5N1 Monitoring Project https://scar.org/library-data/avian-flu#database (2025).
Campagna, C. et al. Catastrophic mortality of southern elephant seals caused by H5N1 avian influenza. Mar. Mamm. Sci. 40, 322–325 (2024).
Fedak, M. A., Arnbom, T. & Boyd, I. L. The relation between the size of Southern Elephant Seal Mothers, the Growth of Their Pups, and the Use of Maternal Energy, Fat, and Protein during Lactation. Physiol. Zool. 69, 887–911 (1996).
McConnell, B. J., Chambers, C. & Fedak, M. A. Foraging ecology of southern elephant seals in relation to the bathymetry and productivity of the Southern Ocean. Antarct. Sci. 4, 393–398 (1992).
Modig, A. O. Effects of body size and harem size on male reproductive behaviour in the southern elephant seal. Anim. Behav. 51, 1295–1306 (1996).
Arnbom, T., Fedak, M. & Boyd, I. L. Factors affecting maternal expenditure in southern elephant seals during lactation. Ecology 78, 471–483 (1997).
Lewis, M., Campagna, C., Quintana, F. & Falabella, V. Estado actual y distribución de la población del elefante marino del sur en la Península Valdés. Argent. Mastozool. Neotrop. 5, 29–40 (1998).
de Bruyn, P. J. N. et al. Sex at sea: alternative mating system in an extremely polygynous mammal. Anim. Behav. 82, 445–451 (2011).
Hindell, M. A. & Little, G. J. Longevity, fertility and philopatry of two female southern elephant seals (Mirounga leonina) at Macquarie Island. Mar. Mamm. Sci. 4, 168–171 (1988).
Bester, M. N. Movements of southern elephant seals and subantarctic fur seals in relation to Marion Island. Mar. Mamm. Sci. 5, 257–265 (1989).
McConnell, B. J., Chambers, C. & Fedak, M. A. Foraging ecology of southern elephant seals in relation to the bathymetry and productivity of the Southern Ocean. Antarct. Sci. 4, 393–398 (2004).
Labrousse, S. et al. Coastal polynyas: winter oases for subadult southern elephant seals in East Antarctica. Sci. Rep. 8, 3183 (2018).
De Bruyn, P. Life history studies of the southern elephant seal population at Marion Island. Doctor of Philosophy thesis, (University of Pretoria, 2009).
Pistorius, P. A. et al. Adult female survival, population trend, and the implications of early primiparity in a capital breeder, the southern elephant seal (Mirounga leonina). J. Zool. 263, 107–119 (2004).
Campagna, C. et al. Predicting population consequences of an epidemic of high pathogenicity Avian Influenza on Southern Elephant Seals. Mar. Mamm. Sci, https://doi.org/10.1111/mms.70009 (2025).
Pistorius, P. A., Taylor, F. E., Bester, M. N., Hofmeyr, G. J. G. & Kirkman, S. P. Evidence for density dependent population regulation in southern elephant seals in the southern Indian Ocean. Afr. Zool. 43, 75–80, 76 (2008).
Pistorius, P. A., de Bruyn, P. J. N. & Bester, M. N. Population dynamics of southern elephant seals: a synthesis of three decades of demographic research at Marion Island. Afr. J. Mar. Sci. 33, 523–534 (2011).
Dewar, M. et al. The risk of highly pathogenic avian influenza in the Southern Ocean: a practical guide for operators and scientists interacting with wildlife. Antar. Sci. 35, 407–414 (2023).
Coleman, J. et al. A comparison of established and digital surface model (DSM)-based methods to determine population estimates and densities for king penguin colonies, using fixed-wing drone and satellite imagery. Remote Sens. Ecol. Conserv. https://doi.org/10.1002/rse2.424 (2024).
McCann, T. S. & Rothery, P. Population size and status of the southern elephant seal (Mirounga leonina) at South Georgia, 1951–1985. Polar Biol. 8, 305–309 (1988).
Fenney, N. et al. Aerial survey of the St Andrews Bay, South Georgia, elephant seal and king penguin colonies during the 2022/23 season, flight number DPLUS_109_37. NERC EDS UK Polar Data Centre. https://doi.org/10.5285/f79b6577-d4cd-4eb2-9fec-b71e4e1d2389 (2024).
Fenney, N. et al. Aerial survey of the St Andrews Bay, South Georgia, elephant seal and king penguin colonies during the 2022/23 season, flight number DPLUS_109_36. NERC EDS UK Polar Data Centre. https://doi.org/10.5285/8189ed89-f36d-43c5-ae33-7e1c9ba0564d (2024).
Fenney, N. et al. Aerial survey of the Hound Bay, South Georgia, elephant seal colony during the 2022/23 season, flight number DPLUS_109_35. NERC EDS UK Polar Data Centre. https://doi.org/10.5285/eb210711-85e1-4671-97a8-8c2791979a19 (2024).
Fenney, N. et al. Aerial survey of the Gold Harbour, South Georgia, elephant seal colony during the 2022/23 season, flight number DPLUS_109_43. NERC EDS UK Polar Data Centre. https://doi.org/10.5285/00cf7916-5c3f-42a2-8b98-38fa99b5aeb8 (2024).
Fenney, N., Coleman, J. & Bamford, C. C. G. Aerial survey of St Andrews Bay, South Georgia, elephant seal colony during the 2023/24 season, flight DLPUS_214_22. NERC EDS UK Polar Data Centre. https://doi.org/10.5285/e238f84e-63b2-4019-8308-7398a8ea204f (2025).
Fenney, N., Coleman, J. & Bamford, C. C. G. Aerial survey of Hound Bay, South Georgia, elephant seal colony during the 2023/24 season, flight DLPUS_214_23. NERC EDS UK Polar Data Centre. https://doi.org/10.5285/85de16ff-a3b2-42b4-a898-aea94ee47b83 (2025).
Fenney, N., Coleman, J. & Bamford, C. C. G. Aerial survey of Gold Harbour, South Georgia, elephant seal colony during the 2023/24 season, flight DLPUS_214_23. NERC EDS UK Polar Data Centre. https://doi.org/10.5285/47d0718d-7146-44d3-965c-60e62a48b8cc (2025).
Acknowledgements
This work was funded by the Biodiversity Challenge Fund Darwin Plus Main stage grants DPLUS109 and DPLUS214. Fieldwork was conducted under Government of South Georgia and the South Sandwich Islands Regulated Activity Permit Numbers 2022/021 and 2024/028 and ASSI permits P/314 and P/444 & 445 for the 2022 and 2024 seasons, respectively. These permits permitted BVLOS (Beyond Visual Line of Sight) UAV flights. Thanks also go to colleagues at BAS and partners at GSGSSI for their comments on this manuscript. We would also like to thank GSGSSI and the crew of MV Pharos SG for their logistical support during both field seasons. Thanks also go to Lindblad National Geographic Expeditions and the crew of the NG Explorer, who kindly supported the field team in 2022 and 2024. Both fieldwork seasons underwent review by the animal ethical approvals board at the British Antarctic Survey, with work approved and permitted under AWREB:1071 & 1109 in 2022 and 2024, respectively.
Author information
Authors and Affiliations
Contributions
This work was co-led by C.B., N.F. and J.C. Conceptualisation: C.B., N.F., J.C., L.H., M.F. and P.H. Methodology: C.B., N.F., J.C., P.F., M.F. and L.H. Data collection: 2024: C.B., N.F., J.C., C.F.C. and J.D. 2022: N.F. and J.C. Analysis: J.C. and N.F. in 2022, J.C., N.F. and C.B. in 2024. Drafting: C.B., with all authors contributing to the final review of this paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks John van Den Hoff, Christophe Guinet and Marthán N. Bester for their contribution to the peer review of this work. Primary Handling Editor: Michele Repetto. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bamford, C.C.G., Fenney, N., Coleman, J. et al. Highly Pathogenic Avian Influenza Viruses (HPAIV) Associated with Major Southern Elephant Seal Decline at South Georgia. Commun Biol 8, 1493 (2025). https://doi.org/10.1038/s42003-025-09014-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s42003-025-09014-7