Abstract
Retrospective cues in working memory (WM) prioritize relevant information and guide attention toward target items to optimize behavior. Although the frontoparietal attention network is known to support this top-down attention mechanism, the broader cortical network behind this process remains elusive. Here, by combining EEG source localization with Bayesian non-negative matrix factorization, we identify nine WM task-related subnetworks, including two newly identified networks: the left parahippocampal-insula network (lPIN) and the right parahippocampal-insula network (rPIN). Dynamic network similarity (DNS) analysis reveals that the brain rapidly transitions into a ‘silent’ state following memory encoding. During retention, both retro- and neutral-cues reactivate memory representation; however, retro-cues elicit stronger reactivation, primarily involving the lPIN and rPIN. Furthermore, this reactivation correlates positively with behavior in the alpha band and negatively with lateralized alpha power. Additionally, the global network configuration during encoding shows significant behavioral relevance. In contrast, during the retrospective phase, a sparser network pattern with lPIN and rPIN as hub nodes significantly influences both behavior and alpha lateralization. These findings highlight the parahippocampal-insula network’s crucial role in enhancing WM behavior through the flexible manipulation of target representations via retrospective attention, thereby supporting the attentional strengthening hypothesis of the retro-cue effect at the large-scale network level.
Similar content being viewed by others
Introduction
Working memory (WM) is a fundamental cognitive system responsible for the temporary storage, maintenance, and manipulation of external information to facilitate goal-directed behavior. Despite its crucial role, WM is constrained by a limited capacity for representation1. Behavioral studies have demonstrated that this limitation can be mitigated by the use of retro-cues, which manipulate memory representations, prioritize information, and direct internal attention toward target items to optimize behavioral outcomes2,3. However, the neural basis underlying the retro-cue effect remains largely unexplored. Therefore, this study aims to investigate the neural basis of the retro-cue effect to capture key brain activity patterns during flexible representation of WM that promotes behavioral gains.
Lateralized alpha power tracks covert spatial attention4 and changes when participants are retrospectively cued to an item previously presented in either the left or right visual hemifields5. These changes reflect the adjustment of attentional prioritization for the information representation prompted by retro-cues, and thereby serve as a gating mechanism for access to WM6,7. This selective attention relies on a top-down modulation mechanism of the frontoparietal network (FPN), through which internal attention selects which WM representation should currently be prioritized (i.e., activated in the sensory cortex) and thereby allowed to interact with sensory input, while other representations are deprioritized into an “activity-silent” state8. In regulating this attention mechanism, the FPN mobilizes various cortices, including the insula9, the premotor area10, and the medial temporal lobe (MTL)11, enabling selective integration of distributed information stored in the sensory cortex into a unified object representation that can be maintained for behavioral output12. Anatomical studies in primates indicate the existence of a parietal-middle temporal pathway originating from regions within the inferior posterior parietal cortex that project directly or indirectly to the hippocampal and parahippocampal cortex (PHC), via the posterior cingulate and retrosplenial cortices13,14. This neural circuitry is associated with spatial processing and memory15,16. Considering the crucial role of the parietal cortex in attention control, we hypothesize that this anatomical connection makes it possible for the PHC to also be involved in the internal attention process guided by retro-cues and collaborate with broader cortical networks to promote flexible selection and regulation of memory representations.
Studies have confirmed that spatially separated brain regions collaborate in a temporally dependent manner to form large-scale functional networks, such as the attention network, visual network, sensorimotor network, etc.17,18, which play a crucial role in executing various cognitive tasks19. It offers a more context-sensitive and comprehensive understanding of information processing at the higher level of neural networks during cognitive processes20. However, most existing studies on large-scale networks, including WM, primarily rely on functional magnetic resonance imaging (fMRI) due to its high spatial resolution. Although combining electroencephalogram (EEG) inverse solutions with spatially independent component analysis (ICA) can overcome the limitations of EEG’s low spatial resolution and fMRI’s low temporal resolution21, ICA poses challenges, such as identifying negatively activating subnetworks and assuming spatial independence among components22. These conflict with the non-negative nature of source power and the fact that one brain region may belong to multiple communities simultaneously23,24. To overcome these limitations, we previously proposed a framework that integrates EEG source localization with Bayesian nonnegative matrix factorization (Bayesian NMF)22. This approach successfully identified non-negative activating subnetworks with overlapping communities in decision-making and P300 tasks, including such important subnetworks as the central executive network, default mode network, etc. Additionally, although our previous work using scalp EEG elucidated the involvement of bilateral temporal lobes in WM information extraction and maintenance25, there is still a need to investigate these processes at higher spatial resolution. In this study, we extend this framework to WM tasks to identify best-fit subnetworks while exploring the potential role of the PHC in accessing internal representations during WM tasks.
The various subprocesses of WM are distinct in time course yet related in their cognitive functions, collectively contributing to the integrity of WM. Encoding involves the formation of meaningful representations through stimulus-induced perceptions, while maintenance entails sustained rehearsal, storage, and interference resistance26. Previous studies directly comparing the two subprocesses have demonstrated that activity patterns during maintenance partially resemble those observed during encoding, including both firing patterns of single neurons27,28 and functional coupling patterns between brain regions29. According to synaptic theory, WM can retain information through transient changes in synaptic weights without requiring persistent neuronal activity30. This notion aligns with the “activity-silent” theory of WM31,32, which posits that information may be stored in latent synaptic states and reactivated when necessary. Such a perspective raises a challenge in distinguishing between sustained encoding-evoked activity throughout maintenance and phasic encoding activity that is reconstituted during maintenance. Motivated by these studies, we constructed a dynamic large-scale network of WM across the entire temporal axis with high spatiotemporal resolution. Subsequently, we proposed a method called dynamic network similarity (DNS), which measures the correlation between the network at the encoding stage and the network at each subsequent moment point to dynamically explore how stimulus encoding- and maintenance-related (including retro-cue-guided information extraction) neural processes correspond to each other. This approach may provide a new framework for understanding how retro-cues direct attention as gating mechanisms for accessing mental representation.
In this study, we adopted a visual WM task with retro-cues and neutral-cues and utilized standardized low-resolution brain electromagnetic tomography (sLORETA)33 to reconstruct the cortical sources of event-related potentials from high temporal resolution EEG data. Subsequently, we employed a robust analytical approach—Bayesian NMF22—to identify functional subnetworks associated with the WM task and construct a time-varying large-scale cortical network. Based on this dynamic network, we further calculated DNS to investigate the dynamic activation mechanism of memory representation networks under retro-cue conditions and to explore how this cognitive process influences behavioral performance.
Results
Behavioral benefits from retro-cue
Sixty participants (32 males, aged 22 ± 2 years) performed the WM task under two cue conditions (retro-cues and neutral-cues) across multiple levels of task load (Fig. 1A; see “Methods” for detailed experimental procedures). Behavioral performance was measured by accuracy (% correct) and reaction time (RT). For accuracy (Fig. 1B), the ANOVA showed a significant main effect of cue type (F (1, 59) = 62.71, p < 0.001, ηp2 = 0.52), with higher accuracy observed in the retro-cue condition compared to the neutral-cue condition. The main effect of task load was also significant (F (2.55, 150.23) = 658.03, p < 0.001, ηp2 = 0.92), indicating that accuracy decreased as task difficulty increased. However, the interaction between cue type and task load was not statistically significant (F (2.44, 143.90) = 1.93, p > 0.05, ηp2 = 0.03). Regarding RT (Fig. 1C), a significant main effect of cue type was found (F (1, 59) = 176.27, p < 0.001, ηp2 = 0.749), with shorter RT in the retro-cue condition than in the neutral-cue condition. A significant main effect of task load was also observed (F (3, 177) = 107.29, p < 0.001, ηp2 = 0.645), showing that RT increased with greater task difficulty. Importantly, a significant interaction between cue type and task load emerged (F (3, 177) = 4.12, p < 0.01, ηp2 = 0.065), suggesting that the influence of task load on RT differed depending on cue type.
A Schematic illustration of the WM task with retro-cues and neutral-cues. B Accuracy and C RT comparisons between the retro-cue and neutral-cue conditions under different task loads. * indicates a statistically significant difference between conditions (p < 0.001, n = 60). Error bars represent mean ± SEM.
Task-related large-scale functional subnetwork
The EEG data analysis strategy and procedures adhered to the workflow outlined in Fig. 2. Preprocessed EEG signals were analyzed using source reconstruction technology, pattern splicing, and Bayesian NMF to identify the functional subnetworks associated with the WM task. These subnetworks were then used to construct dynamic large-scale networks and conduct DNS analysis.
A Source reconstruction was conducted on the preprocessed EEG data. B Pattern splicing was performed for each subject across different task loads and cue types along the time dimension, resulting in a single group-level 2D matrix. C Bayesian NMF was applied to identify functional subnetworks associated with the WM task. D Following the construction of the dynamic large-scale network, DNS was calculated. The correlation between DNS during the cue stage and behavioral performance was then analyzed, and the relationship between network patterns at different stages and behavioral performance was examined.
Nine NMF components were identified as the best-fit task-related subnetworks. These subnetworks include the central executive network (component 10, CEN), left parahippocampal-insula network (component 3, lPIN), somatosensory-motor network (component 13, SMN), primary visual network (component 42, PVN), visual network (component 40, VN), posterior default mode network (component 5, pDMN), self-referential network (component 1, SRN), right parahippocampal-insula network (component 9, rPIN), and anterior default mode network (component 26, aDMN). Fig. 3 illustrates the spatial distribution of these nine subnetworks, and further details, including activated regions, cluster voxel counts, peak activation values, and MNI coordinates, are provided in Table S1.
A Central executive network. B Left parahippocampal-insula network. C Somatosensory-motor network. D Primary visual network. E Visual network. F Posterior default mode network. G Self-referential network. H Right parahippocampal-insula network. I Anterior default mode network. The distinct colors indicate the activation in the related brain areas.
DNS and its behavioral relevance
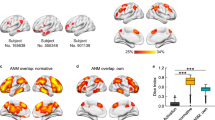
DNS analysis was performed across different frequency bands, including delta, theta, alpha, and beta. As shown in Fig. 4A, within the alpha band, DNS exhibited a sharp decline immediately after the offset of memory items, followed by a transient rebound upon the onset of both retro-cues and neutral-cues. Notably, the DNS during the retro-cue stage was significantly higher than that during the neutral-cue stage (paired t-tests, p < 0.05). However, this pattern was not observed in the other three frequency bands (i.e., delta, theta, and beta; Fig. S4). Further analyses of the alpha band revealed that, regardless of task load, DNS was consistently greater during the retro-cue stage compared to the neutral-cue stage (Fig. S3). Moreover, during the retro-cue stage rather than the neutral-cue stage, DNS rebound was primarily linked to functional connectivity (FC) involving the rPIN, lPIN, and pDMN (Pearson correlation, p < 0.05, FDR-corrected, Fig. 4B). Further investigation into the relationship between DNS during the cue stage and behavioral performance across frequency bands showed that only in the alpha band did the retro-cue DNS exhibit a significant positive correlation with behavioral outcomes (r = 0.399, p < 0.005, Fig. 4C). In contrast, no such correlation was found in the neutral-cue condition (r = 0.083, p > 0.05, Fig. 4D). Consequently, subsequent analyses focused exclusively on the alpha band.
A DNS across different cue types. The area within the dotted line represents the time window during which the retro-cue or neutral-cue was presented. The bar chart illustrates the differences in DNS between the retro-cue and neutral-cue conditions, with * denoting a statistically significant difference (p < 0.05, n = 60). Error bars represent mean ± SEM. B Large-scale network topology shows a significant positive correlation with DNS during the retro-cue stage. Different colors represent distinct subnetworks (n = 56). C Correlation between DNS during the retro-cue stage and behavioral accuracy. The distributions of behavioral accuracy and DNS are displayed on the Upper and Right margins of the plot frame (n = 56). D Correlation between DNS during the neutral-cue stage and behavioral accuracy (n = 56).
Relationship between DNS and lateralized alpha power
To further investigate the specificity of the alpha band, we analyzed the relationship between cortical DNS and scalp lateralized alpha power. Lateralized alpha power occurred approximately 400 ms after the onset of the retro-cue across different load conditions (Fig. 5A). Although lateralized alpha power tended to increase with task load, this trend did not reach statistical significance (paired t-test, p > 0.05). Importantly, lateralized alpha power showed significant negative correlations with both behavioral performance (r = −0.341, p < 0.01, Fig. 5B) and DNS during the retro-cue stage (r = −0.324, p < 0.05, Fig. 5C). Given that both lateralized alpha power and DNS were correlated with behavioral performance, a support vector regression (SVR) model incorporating these two variables effectively predicted individual performance (r = 0.376, p < 0.005, RMSE = 0.040, Fig. 5D).
A Lateralized alpha power (contralateral minus ipsilateral), averaged across electrode sites O1/2, PO7/8, PO3/4, P7/8, P5/6, P3/4, and P1/2, under different load conditions. The bar chart illustrates the average lateralized alpha power during the 400-1000 ms time window under different task loads (n = 60). Error bars represent mean ± SEM. B Correlation between lateralized alpha power and behavioral accuracy. The distributions of behavioral accuracy and lateralized alpha power are displayed on the Upper and Right margins of the plot frame (n = 56). C Correlation between lateralized alpha power and DNS during the retro-cue stage. The distributions of lateralized alpha power and DNS are shown on the Upper and Right margins of the plot frame (n = 56). D Correlation between predicted and actual behavioral accuracy. RMSE represents the root mean square error (n = 56).
Contribution of stage-specific network patterns to the relationship between brain and behavior
To further investigate how network patterns at different stages influence behavior, we employed partial least squares (PLS) regression to examine the relationships between stage-specific brain measures derived from interaction weights and behavioral performance. The first component (PLS-1) was defined as the spatial map that explained the greatest fraction of model weights derived from stage-specific functional connections (ppermutation < 0.001). Following bootstrapping, FDR corrections were used to adjust for multiple comparisons across functional connections. We found that during the encoding stage, nearly all network connections were significantly associated with individual behavioral accuracy (p < 0.05, FDR-corrected, Fig. 6A). In the retro-cue stage, connections primarily involving lPIN, rPIN, pDMN, and PVN showed the strongest contributions to behavior accuracy (p < 0.05, FDR-corrected, Fig. 6B). Additionally, we investigated whether the network pattern in the retro-cue stage was linked to lateralized alpha power. PLS analysis revealed significant correlations between lateralized alpha power and connections mainly associated with rPIN, lPIN, pDMN, and CEN (p < 0.05, FDR-corrected, Fig. 6C).
A Relationship between the network pattern during the encoding stage and behavioral accuracy (n = 60). B Relationship between the network pattern during the retro-cue stage and behavioral accuracy (n = 60). C Relationship between the network pattern during the retro-cue stage and lateralized alpha power (n = 60). Different colors represent distinct subnetworks.
Validation analyses
Our main findings were replicated using the validation datasets. First, we identified nine subnetworks—CEN, lPIN, SMN, PVN, VN, pDMN, SRN, rPIN, and aDMN—in the first dataset (Fig. S5 and Table S2), and eight subnetworks, excluding SRN, in the second dataset (Fig. S7 and Table S3). Second, DNS analysis across both datasets consistently revealed a rapid decline in network similarity following the offset of the memory items and a brief rebound after the onset of the retro-cue (Figs. S6 and S8). Moreover, DNS during the retro-cue stage was also specifically correlated with behavior in the alpha band. These replication results demonstrate the robustness of our findings.
Discussion
The attentional prioritization of the cued representation is a potential benefit of retro-cues on WM, and modulations in alpha activity over sensory cortices provide a plausible neural explanation for this hypothesis8. However, it is important to note that this local neural activity in response to specific events cannot be directly interpreted as an active WM representation. Further investigation into the neural mechanisms underlying the behavioral benefits of retrospective attention, particularly at the level of global information integration, is therefore warranted. In this study, we employed DNS analysis at the cortical large-scale network level to investigate whether and how retrospective attention enhances WM performance using a typical retro-cue WM task. Within this analytical framework, our observations were twofold: first, they aligned with the “activity-silent” theory of WM; second, they highlighted the critical role of the parahippocampal-insula network and its interactions with other cortical networks within the alpha band in facilitating the flexible prioritization of WM representations, thereby supporting the attentional strengthening hypothesis of the retro-cue effect.
The seven traditional subnetworks identified in this study, namely, CEN, SMN, PVN, VN, pDMN, SRN, and aDMN, are extensively involved in WM processes34,35,36,37,38. Importantly, we discovered a functional subnetwork comprising the PHC and insula. Previous studies using sLORETA have similarly highlighted the critical roles of these two regions in cognitive functions and diseases39,40,41. For example, Nguyen et al.42 demonstrated that the insula and PHC play essential roles in spatial navigation. While the PHC has traditionally been associated with episodic memory and visuospatial processing43,44, growing evidence suggests that it also contributes to various higher-order cognitive functions, including the active maintenance of information in WM45, via its functional interactions with brain regions implicated in executive control. Anatomically, the PHC is not only densely connected to MTL structures such as the entorhinal cortex and hippocampus but also maintains direct projection pathways to the lateral parietal cortex and dorsolateral prefrontal cortex46,47, which are key nodes of FPN. These structural connections provide a neuroanatomical basis for the PHC’s involvement in attentional regulation. Consistent with this view, recent findings also suggest an involvement of the hippocampus in attentional modulation, particularly during tasks requiring spatial relational processing and memory updating48,49. Collectively, these observations indicate that MTL may play a broader role in attention-demanding contexts beyond its conventional function in memory processing. Aminoff et al.50 proposed a “contextual association” framework, which posits that the core function of the PHC lies in processing associative links between stimuli and their spatial or contextual backgrounds. Given that retrospective attention (e.g., in retro-cue tasks) often relies on such contextual cues to reallocate attentional resources, the PHC may facilitate the redirection of attention toward task-relevant memory representations by linking current task demands with previously encoded contextual information. The insula, as a central hub of the salience network, plays a key role in allocating attentional resources and dynamically switching between internally and externally oriented attentional systems9. Importantly, the PHC and insula are anatomically connected through direct structural pathways51, providing a foundation for their functional interactions. Previous studies have shown that the connectivity between the PHC and insula is critically involved in cognitive flexibility and attentional control52,53. Therefore, we propose that the stable structural connectivity and functional coupling between the PHC and insula may serve as a crucial neural substrate supporting the regulation of retrospective attention. Through this interaction, the brain can flexibly adjust attentional prioritization toward goal-relevant memory content based on contextual information, thereby enhancing effective information maintenance and the execution of goal-directed behavior.
The rapid decline in DNS following the offset of the memory array indicates that the encoding of WM content is a highly dynamic and transient process. This decline is marked by reduced synchronous neuronal activity, leading the brain into a “silent” state. This observation aligns with recent theoretical models proposing that successful WM maintenance does not strictly rely on sustained neural activity during the delay period, but rather depends on transient changes in network configurations during encoding, which generate reactivatable memory traces54. In this study, we observed a significant DNS rebound under both retro- and neutral-cue conditions, further supporting the idea that external cues have a “ping” effect on the brain, reactivating functional networks in the “silent” state through external signals and thereby reawakening latent memory representations55. Notably, the DNS rebound induced by retro-cue exhibited greater amplitude and a later peak, potentially reflecting the critical role of retrospective attention in prioritizing the retrieval of target information. On one hand, retro-cue directs spatial attention to reduce memory load, facilitating more complete reactivation of target information from a latent state. On the other hand, the reallocation of attentional prioritization enhances the stability of target representation, thus promoting more efficient goal-directed behavior. Furthermore, we observed that DNS decreased as memory load increased, indicating that limitations in WM capacity constrain the amount of information that can be reactivated and used to guide behavior56. Importantly, further analysis revealed that the DNS rebound during the retrospective stage was significantly correlated with connectivity involving the rPIN, lPIN, and pDMN, indicating that the PIN regulates attentional prioritization through interactions with other cortical networks. The pDMN is regarded as playing an important role in context retrieval, implicit memory processing, and the handling of self-related information16. Therefore, the PIN may function as an integrative interface that combines context-driven memory signals (via PHC) with saliency detection (via insula), and collaborates with the pDMN to support the reactivation and maintenance of internal memory representations under conditions of limited cognitive resources.
The DNS in the retro-cue stage was significantly and positively correlated with behavior, specifically within the alpha band. This frequency-specific effect highlights the critical role of alpha oscillations in prioritizing and maintaining internal representations in WM57,58. To further investigate the functional relevance of alpha oscillations during WM maintenance, we analyzed their relationship with DNS and behavioral performance. Studies have shown that retro-cues facilitate the internal reallocation of attention toward task-relevant mnemonic representations, thereby focusing active WM retention on cued mental representations59,60. In this context, lateralized alpha power over posterior electrodes reflects the sustained prioritization of internal representations rather than the suppression of irrelevant information61,62. The observed negative correlations between lateralized alpha power and both behavioral performance and DNS suggest that alpha modulation not only signifies attentional redirection but more likely reflects the extent to which attentional resources are concentrated on target representations. In other words, stronger alpha lateralization indicates more effective selection of relevant WM content, thereby supporting improved memory-guided behavior. Interestingly, we observed that network reactivation, as indexed by DNS, preceded changes in lateralized alpha power. DNS reflects the processing of memory information at the level of the global brain network, whereas alpha lateralization represents activity in local brain regions, i.e., the posterior regions. This observation indicates that DNS captures neural responses related to retro-cues earlier at the network level before alpha lateralization occurs. Additionally, an SVR model incorporating both DNS and lateralized alpha power effectively predicted individual behavioral performance, underscoring the importance of large-scale network reactivation and attentional enhancement in improving memory-guided behavior.
The encoding stage of WM involves a series of cognitive processes, including the reception and integration of external visual information, the selection and planning of attentional resources and strategies, as well as the conversion and encoding of sensory inputs63. These processes necessitate a highly coordinated, cross-network integration mechanism to ensure efficient encoding and storage of information, thereby supporting goal-directed delay behavior19. For instance, VN and PVN jointly serve as the initial processing pathway for sensory input, facilitating the preliminary analysis of external information34,64. SMN may contribute to perception-action mapping related to task execution38. Meanwhile, higher-order control networks such as CEN and SRN are likely responsible for managing attentional resources, formulating encoding strategies, and extracting relevant target dimensions, thereby promoting the effective transformation and retention of information36,37. Notably, PIN also played a role during this stage, potentially supporting the transition from perceptual processing to memory representation through its interactions with both sensory and control networks. In contrast, during the retrospective stage, a sparser network structure was observed, with key nodes centered on lPIN, rPIN, pDMN, and PVN. The prominent role of PIN during this stage may reflect its critical involvement in the selective reactivation and integration of memory contents under retrospective attentional guidance. As an important hub for implicit memory and self-referential processing16, pDMN collaborated with PIN to promote implicit retrieval and recovery of the target representation. The sustained engagement of PVN suggests its role in the reprocessing of perceptual representations. Furthermore, we found that the network configuration involving rPIN, lPIN, pDMN, and CEN during the retrospective stage was significantly correlated with lateralized alpha power. This result indicates that the PIN also participates in the allocation of attentional prioritization through interaction with CEN. Specifically, the functional coupling between lPIN/rPIN and CEN may serve as an intermediary channel between intrinsic control systems and perceptual representations, aiding in the dynamic regulation of attentional resources across multiple memory items. Concurrently, the cooperation between pDMN and CEN reflects the integration of intrinsic and executive systems, supporting goal-directed prioritization of memory content. Notably, numerous previous studies have demonstrated that frontal and parietal regions play a critical role in modulating WM representations65,66,67,68. For example, recent transcranial magnetic stimulation (TMS) studies have provided causal evidence indicating that stimulation of the prefrontal cortex69 or the parietal cortex70 can modulate retro-cue benefits. Our results further extend these findings by identifying the PIN as a complementary hub that interacts with these control regions. By integrating perceptual input, mnemonic representations, and top-down control signals, the PIN mediates selective reactivation and prioritization at the network level, thereby expanding previous accounts of retro-cue mechanisms beyond the contributions of frontal and parietal regions. In other words, retro-cue benefits may arise from the interplay between causal control regions (as demonstrated by TMS) and integrative hubs such as the PIN, which coordinate network level reactivation to support goal-directed behavior.
Taken together, the target representation was enhanced by retro-cues during the visual WM process. This enhancement involved the parahippocampal-insula network and its interaction with other cortical networks in the alpha band. However, future research should focus on more in-depth analyses to further elucidate the hypotheses related to the retro-cue effect. Such analyses could include: applying the proposed analytical framework to additional WM tasks, such as verbal WM, to validate the generalizability of the current findings; refining source estimation techniques, for example, by developing novel deep learning-based source localization methods or assessing the reliability of localized cortical sources via multimodal data fusion (e.g., fMRI, MEG, or intracranial EEG); and employing advanced network construction approaches, such as dynamic causal network analysis, to investigate the temporal and directional dynamics of neural activity during retrospective attention enhancement.
Conclusion
In this study, we proposed a DNS analysis framework by combining EEG source reconstruction and Bayesian NMF to investigate the neural basis of the retro-cue effect. Our results support the “activity-silent” theory of WM, and further indicate that retrospective spatial attention enhances target-position binding through prioritization of target representation. Importantly, this process was closely associated with alpha oscillation-related activity in the parahippocampal-insula network. These findings provide compelling neurophysiological evidence at the level of cortical large-scale networks, reinforcing the attentional strengthening hypothesis of the retro-cue effect and highlighting the critical role of the parahippocampal-insula network in modulating WM information during retrospective attention.
Methods
Participants
The experimental protocol was approved by the Institution Research Ethics Board of the University of Electronic Science and Technology of China. Before the experiment, written informed consent was obtained from all participants after providing them with a comprehensive explanation of the study. A cohort of sixty healthy subjects (32 males, aged 22 ± 2 years) was recruited, and upon completion of the experiment, they received a reward of ¥ 200. All participants were right-handed individuals with normal or corrected-to-normal vision and no color-vision deficiencies.
Experimental procedures
Participants completed a change detection WM task with retro-cues and neutral-cues. Stimuli were presented using E-Prime 3.0 software on a 23-inch LCD monitor with a resolution of 1920 × 1080 pixels. A gray background ([128 128 128]) was maintained throughout the experiment. The memory array comprised 2 to 8 (load 2, 4, 6, and 8) colored squares (0.65° × 0.65°), symmetrically displayed within two 4° × 7.3° rectangular regions (containing 1 to 4 colored squares in each hemifield) centered 3° to the left and right of a central fixation cross. The display was viewed at a distance of 100 cm. The color of each square was randomly selected, without repetition, from a set of nine distinguishable colors (red, green, purple, blue, white, magenta, yellow, brown, and pink). Stimulus positions were randomized on each trial, with a minimum distance of 2° (center to center) between squares within each hemifield. In half of the trials, one square in the memory probe differed in color from its corresponding item in the memory array, while in the remaining trials, the colors in both arrays were identical.
Each trial began with a fixation cross presented at the screen center for 1000 ms, followed by the memory array, which was displayed for 500 ms. After a 1000 ms delay, an attentional cue appeared at the screen center for 250 ms. In half of the trials, a one-way arrow served as a retro-cue, indicating whether the left or right side of the previously shown items would be tested. The number of trials cued to each side was equal, with the order randomized for each participant. In the remaining trials, changing the one-way arrows to two-way arrows as neutral-cues, which provides no attentional benefits for a head start for retrieval. Trials were allocated to retro- and neutral-cue conditions randomly within each block. Following an additional 2250 ms delay, a probe array was presented on the side of the cued items (i.e., target) in the retro-cue trials, while a probe was randomly presented on either side of the memory array in the neutral-cue trials. Participants were instructed to press the “1” key as precisely as possible if the colors of the target matched between the memory array and the probe, or the “2” key if they did not. The subsequent trial began within a randomly determined interval ranging from 500 to 1000 ms. The experiment consisted of four blocks, with each comprising 120 trials. After each block, participants’ accuracy for that block was displayed on the screen, and they were allowed to take a rest. Before the experiment, participants completed a practice session of 30 trials to familiarize themselves with the task.
EEG acquisition
Participants were seated in a light-attenuated, electrically shielded room and instructed to remain relaxed while minimizing body movements throughout the data recording. EEG data were recorded using 64 Ag/AgCl electrodes (ANT Neuro, Berlin, Germany) positioned according to the international 10-10 system. The data were sampled at 1000 Hz with an online bandpass filter set between 0.1 and 100 Hz. The reference and ground electrodes were positioned at AFz and CPz, respectively, and electrode impedance was maintained below 5 kΩ throughout the recording.
EEG data preprocessing
Following conventional EEG data preprocessing procedures71, the preprocessing included 1-30 Hz bandpass filtering, ICA for removing eye movements, re-referencing by utilizing the Reference Electrode Standardization Technique (REST)72,73, [−200 ms, 4000 ms] (0 ms denotes the onset of the memory array) data segmenting, [−200 ms, 0 ms] baseline correction and bad trial removal with ±100 μV as the threshold. The preprocessed data were used for subsequent analyses.
EEG source reconstruction
The inherently distributed nature of WM emphasizes the rationality of using distributed source imaging techniques, such as sLORETA33 and minimum norm estimation (MNE)74. Among these methods, previous studies have consistently demonstrated that sLORETA effectively addresses the limitations of deep cortical source localization, achieving the lowest localization errors and minimal blurring33,75. Moreover, certain approaches like MNE present challenges in physiological situations requiring interactions between distributed sources76. These advantages position sLORETA as a robust tool for functional mapping, given its physiological consistency and accurate localization capabilities, which have significantly contributed to its widespread adoption in recent research77,78. Therefore, in our present study, we used sLORETA to capture the distributed cortical activities of WM. Additionally, to ensure the reliability of source localization, core parameters were adaptively calculated by sLORETA based on scalp EEG data, avoiding any arbitrary operations. Herein, for each participant, source activities corresponding to the scalp data (61 × 4200) were reconstructed using sLORETA, generating time courses (TCs) for each voxel (6239 × 4200) in the source space based on averaged EEG epochs across trials.
Bayesian NMF-based large-scale network construction
After reconstructing cortical activities for each subject, pattern splicing was performed along the temporal dimension to generate a single group-level 2D matrix. Before applying Bayesian NMF, cortical signals were down-sampled to 200 Hz (6239 × 840) to reduce computational demands. For each load condition, the TCs of cortical generators extracted from the EEG datasets were concatenated across subjects into a 2D matrix with dimensions of 50400 × 6239 (840 × 60 = 50400). The TCs from the four groups under both retro- and neutral-cue conditions were then combined into a new group-level matrix with a dimension of 403200 × 6239 (840 × 60 × 4 × 2 = 403200). This strategy enabled the integration of time-varying source patterns across all subjects and experimental conditions, thereby facilitating the detection of functional networks at the group-level. Following pattern splicing, temporal-spatial decomposition was carried out to identify subnetworks associated with the WM task. Given that EEG source activations are inherently nonnegative, it is more appropriate to extract positively activated subnetworks. Therefore, Bayesian NMF79 was employed in this study, decomposing signals into nonnegative components to construct large-scale cortical networks. Further methodological details regarding Bayesian NMF, including parameter settings, can be found in ref. 22.
DNS analysis
We sought to investigate the mechanism underlying the flexible manipulation of WM representation by examining dynamic changes in network resource allocation across distinct cognitive stages. To quantify these dynamics, we calculated the DNS. Specifically, based on the nine functional subnetworks identified in this study, we extracted TCs (9 × 840) for each of the four load conditions under both retro- and neutral-cue conditions per subject. We then constructed dynamic large-scale networks within different frequency bands (i.e., delta, theta, alpha, and beta) by utilizing wavelet coherence (WTC)80, generating adjacency matrices of dimension 9 × 9 × 840 per subject. WTC was chosen due to its high temporal and spectral resolution, as well as its robustness in capturing transient and frequency-specific functional couplings. It has been widely applied in signal analysis involving short-term, event-related, and non-stationary signals such as EEG81. Compared with conventional methods like sliding-window correlation or time-varying multivariate autoregressive models, WTC avoids the trade-off between temporal resolution and window size and does not require stationarity assumptions. Subsequently, for each load condition in both retro- and neutral-cue trials, we averaged the networks during the encoding stage ([1 ms, 500 ms]) within each frequency band to obtain baseline networks. Next, we computed the Spearman correlation coefficient between each subsequent time point ([501 ms, 4000 ms]) and the corresponding baseline network within each frequency band, separately for each load condition and cue type, thereby deriving the DNS for each participant. Then, we conducted further analysis on the DNS differences between cue types during the cue stage to elucidate how the brain dynamically regulates WM by reconfiguring network resources and how this process affects behavior.
Validation analyses
To evaluate the robustness of our findings, we conducted validation analyses on two different datasets. The first validation dataset was derived from another WM task we designed, which did not include neutral-cues, while the second dataset was obtained from the study by Wolfe et al.55. The differences in memory content attributes between Wolfe et al.’s task and ours (i.e., orientation and color) allowed us to assess the generalizability of our conclusions. For further details on the validation datasets, please refer to the Supplementary Materials.
Statistical analyses
The behavioral performance was analyzed using a 2 (cue type: retrospective and neutral) × 4 (task load: load 2, load 4, load 6, and load 8) repeated-measures ANOVA in IBM SPSS Statistics 26. Paired t-tests were conducted to compare DNS values between the two cue conditions as well as lateralized alpha power across different task loads. Pearson correlation analyses were performed to explore the relationships between DNS and network pattern, DNS and behavior, lateralized alpha power and behavior, DNS and lateralized alpha power, as well as predicted and actual behavioral accuracy. To enhance the robustness of the potential relationships, the Mahalanobis distance82 method was applied to identify and exclude four participants, following established procedures in previous studies83,84.
PLS regression was used to determine the relationship between network patterns at different stages and individual behavior85. FC between all pairs of subnetworks was used as predictor variables for behavioral accuracy in the PLS regression. The first component of the PLS (PLS-1) represented the linear combination of FC across subnetworks that was most strongly correlated with behavior. A permutation test (n = 1000) was conducted to test the null hypothesis that PLS-1 did not explain more covariance between stage-specific network patterns and behavior than that expected by chance86. Bootstrapping (bootstrap samples = 500) was used to estimate the variability of each FC weight in PLS-1. The ratio of FC to its bootstrap SE was used to calculate z values. Functional connections that reliably contributed to PLS-1 were identified after FDR correction (p < 0.05) for multiple comparisons.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The source data used to generate all figures in this paper are provided in Supplementary Data. The raw data of this study are available from the corresponding author upon reasonable request.
Code availability
The EEG pre-processing toolboxes are freely available (EEGLAB v2020.0, https://sccn.ucsd.edu/eeglab/index.php; REST_v1.2_20200818, https://www.neuro.uestc.edu.cn/name/shopwap/do/index/content/96). Bayesian NMF, WTC, and statistical analyses—including correlation analyses and t-tests—were performed using MATLAB R2021a (https://jp.mathworks.com/products/matlab.html). Subnetwork distribution images were visualized using LORETA v20170220 (https://www.uzh.ch/keyinst/loreta). Statistical visualizations for correlation analyses and network connectivity were generated using Origin2024 (https://www.originlab.com/), while those of DNS and lateralized alpha power were conducted using GraphPad Prism 8.3 (https://www.graphpad.com/).
References
Ma, W. J., Husain, M. & Bays, P. M. Changing concepts of working memory. Nat. Neurosci. 17, 347–356 (2014).
Myers, N. E., Stokes, M. G. & Nobre, A. C. Prioritizing information during working memory: beyond sustained internal attention. Trends Cogn. Sci. 21, 449–461 (2017).
Ester, E. F., Nouri, A. & Rodriguez, L. Retrospective cues mitigate information loss in human cortex during working memory storage. J. Neurosci. 38, 8538–8548 (2018).
Bacigalupo, F. & Luck, S. J. Lateralized suppression of alpha-band EEG activity as a mechanism of target processing. J. Neurosci. 39, 900–917 (2019).
Schneider, D., Göddertz, A., Haase, H., Hickey, C. & Wascher, E. Hemispheric asymmetries in EEG alpha oscillations indicate active inhibition during attentional orienting within working memory. Behav. Brain Res. 359, 38–46 (2019).
van Moorselaar, D. et al. Spatially selective alpha oscillations reveal moment-by-moment trade-offs between working memory and attention. J. Cogn. Neurosci. 30, 256–266 (2018).
Myers, N. E., Walther, L., Wallis, G., Stokes, M. G. & Nobre, A. C. Temporal dynamics of attention during encoding versus maintenance of working memory: complementary views from event-related potentials and alpha-band oscillations. J. Cogn. Neurosci. 27, 492–508 (2015).
de Vries, I. E., Slagter, H. A. & Olivers, C. N. Oscillatory control over representational states in working memory. Trends Cogn. Sci. 24, 150–162 (2020).
Menon, V. & Uddin, L. Q. Saliency, switching, attention and control: a network model of insula function. Brain Struct. Funct. 214, 655–667 (2010).
Mayer, J. S. et al. Common neural substrates for visual working memory and attention. Neuroimage 36, 441–453 (2007).
Soto, D., Greene, C. M., Kiyonaga, A., Rosenthal, C. R. & Egner, T. A parieto-medial temporal pathway for the strategic control over working memory biases in human visual attention. J. Neurosci. 32, 17563–17571 (2012).
Wheeler, M. E. & Treisman, A. M. Binding in short-term visual memory. J. Exp. Psychol.: Gen. 131, 48 (2002).
Vann, S. D., Aggleton, J. P. & Maguire, E. A. What does the retrosplenial cortex do?. Nat. Rev. Neurosci. 10, 792–802 (2009).
Kravitz, D. J., Saleem, K. S., Baker, C. I. & Mishkin, M. A new neural framework for visuospatial processing. Nat. Rev. Neurosci. 12, 217–230 (2011).
Grön, G., Wunderlich, A. P., Spitzer, M., Tomczak, R. & Riepe, M. W. Brain activation during human navigation: gender-different neural networks as substrate of performance. Nat. Neurosci. 3, 404–408 (2000).
Buckner, R. L., Andrews-Hanna, J. R. & Schacter, D. L. The brain’s default network: anatomy, function, and relevance to disease. Ann. N. Y. Acad. Sci. 1124, 1–38 (2008).
Cai, W., Ryali, S., Pasumarthy, R., Talasila, V. & Menon, V. Dynamic causal brain circuits during working memory and their functional controllability. Nat. Commun. 12, 3314 (2021).
Soreq, E., Leech, R. & Hampshire, A. Dynamic network coding of working-memory domains and working-memory processes. Nat. Commun. 10, 936 (2019).
Bressler, S. L. & Menon, V. Large-scale brain networks in cognition: emerging methods and principles. Trends Cogn. Sci. 14, 277–290 (2010).
Yi, C. et al. A novel method for constructing EEG large-scale cortical dynamical functional network connectivity (dFNC): WTCS. IEEE Trans. Cybern. 52, 12869–12881 (2021).
Liu, Q., Farahibozorg, S., Porcaro, C., Wenderoth, N. & Mantini, D. Detecting large-scale networks in the human brain using high-density electroencephalography. Hum. Brain Mapp. 38, 4631–4643 (2017).
Yi, C. et al. Constructing large-scale cortical brain networks from scalp EEG with Bayesian nonnegative matrix factorization. Neural Netw. 125, 338–348 (2020).
Cole, M. W. et al. Multi-task connectivity reveals flexible hubs for adaptive task control. Nat. Neurosci. 16, 1348–1355 (2013).
Pessoa, L. Understanding brain networks and brain organization. Phys. Life Rev. 11, 400–435 (2014).
Chen, C. et al. The dynamic causality brain network reflects whether the working memory is solidified. Cereb. Cortex 34, bhad467 (2024).
Eriksson, J., Vogel, E. K., Lansner, A., Bergström, F. & Nyberg, L. Neurocognitive architecture of working memory. Neuron. 88, 33–46 (2015).
Lewis-Peacock, J. A., Drysdale, A. T., Oberauer, K. & Postle, B. R. Neural evidence for a distinction between short-term memory and the focus of attention. J. Cogn. Neurosci. 24, 61–79 (2012).
Harrison, S. A. & Tong, F. Decoding reveals the contents of visual working memory in early visual areas. Nature 458, 632–635 (2009).
Cohen, J. R., Sreenivasan, K. K. & D’Esposito, M. Correspondence between stimulus encoding-and maintenance-related neural processes underlies successful working memory. Cereb. Cortex. 24, 593–599 (2014).
Mongillo, G., Barak, O. & Tsodyks, M. Synaptic theory of working memory. Science 319, 1543–1546 (2008).
Stokes, M. G. Activity-silent’working memory in prefrontal cortex: a dynamic coding framework. Trends Cogn. Sci. 19, 394–405 (2015).
Rose, N. S. et al. Reactivation of latent working memories with transcranial magnetic stimulation. Science 354, 1136–1139 (2016).
Pascual-Marqui, R. D. Standardized low-resolution brain electromagnetic tomography (sLORETA): technical details. Methods Find. Exp. Clin. Pharm. 24, 5–12 (2002).
Xu, Y. A tale of two visual systems: invariant and adaptive visual information representations in the primate brain. Annu. Rev. Vis. Sci. 4, 311–336 (2018).
Fuentes-Claramonte, P. et al. Shared and differential default-mode related patterns of activity in an autobiographical, a self-referential and an attentional task. Plos one 14, e0209376 (2019).
Yin, S., Sui, J., Chiu, Y.-C., Chen, A. & Egner, T. Automatic prioritization of self-referential stimuli in working memory. Psychol. Sci. 30, 415–423 (2019).
Sridharan, D., Levitin, D. J. & Menon, V. A critical role for the right fronto-insular cortex in switching between central-executive and default-mode networks. Proc. Natl. Acad. Sci. USA 105, 12569–12574 (2008).
Marvel, C. L., Morgan, O. P. & Kronemer, S. I. How the motor system integrates with working memory. Neurosci. Biobehav. Rev. 102, 184–194 (2019).
Jiang, L. et al. Information transmission velocity-based dynamic hierarchical brain networks. NeuroImage 270, 119997 (2023).
Justen, C., Herbert, C., Werner, K. & Raab, M. Self vs. other: neural correlates underlying agent identification based on unimodal auditory information as revealed by electrotomography (sLORETA). Neuroscience 259, 25–34 (2014).
De Ridder, D. & Vanneste, S. Targeting the parahippocampal area by auditory cortex stimulation in tinnitus. Brain Stimul. 7, 709–717 (2014).
Nguyen, H. M., Matsumoto, J., Tran, A. H., Ono, T. & Nishijo, H. sLORETA current source density analysis of evoked potentials for spatial updating in a virtual navigation task. Front. Behav. Neurosci. 8, 66 (2014).
Aminoff, E., Gronau, N. & Bar, M. The parahippocampal cortex mediates spatial and nonspatial associations. Cereb. Cortex 17, 1493–1503 (2007).
Epstein, R. A. Parahippocampal and retrosplenial contributions to human spatial navigation. Trends Cogn. Sci. 12, 388–396 (2008).
Schon, K., Newmark, R. E., Ross, R. S. & Stern, C. E. A working memory buffer in parahippocampal regions: evidence from a load effect during the delay period. Cereb. Cortex 26, 1965–1974 (2016).
Blatt, G. J., Pandya, D. N. & Rosene, D. L. Parcellation of cortical afferents to three distinct sectors in the parahippocampal gyrus of the rhesus monkey: an anatomical and neurophysiological study. J. Comp. Neurol. 466, 161–179 (2003).
Lavenex, P., Suzuki, W. A. & Amaral, D. G. Perirhinal and parahippocampal cortices of the macaque monkey: projections to the neocortex. J. Comp. Neurol. 447, 394–420 (2002).
Ruiz, N. A., Meager, M. R., Agarwal, S. & Aly, M. The medial temporal lobe is critical for spatial relational perception. J. Cogn. Neurosci. 32, 1780–1795 (2020).
Aly, M. & Turk-Browne, N. B. Attention promotes episodic encoding by stabilizing hippocampal representations. Proc. Natl. Acad. Sci. USA 113, E420–E429 (2016).
Aminoff, E. M., Kveraga, K. & Bar, M. The role of the parahippocampal cortex in cognition. Trends Cogn. Sci. 17, 379–390 (2013).
Ghaziri, J. et al. The corticocortical structural connectivity of the human insula. Cereb. Cortex 27, 1216–1228 (2017).
Chen, B. et al. Structural and functional abnormalities of olfactory-related regions in subjective cognitive decline, mild cognitive impairment, and Alzheimer’s disease. Int. J. Neuropsychopharmacol. 25, 361–374 (2022).
Farb, N. A., Segal, Z. V. & Anderson, A. K. Attentional modulation of primary interoceptive and exteroceptive cortices. Cereb. Cortex 23, 114–126 (2013).
Barbosa, J. et al. Interplay between persistent activity and activity-silent dynamics in the prefrontal cortex underlies serial biases in working memory. Nat. Neurosci. 23, 1016–1024 (2020).
Wolff, M. J., Jochim, J., Akyürek, E. G. & Stokes, M. G. Dynamic hidden states underlying working-memory-guided behavior. Nat. Neurosci. 20, 864–871 (2017).
Cowan, N. Working Memory Capacity (Psychology Press, 2012).
Li, D. et al. Prioritizing flexible working memory representations through retrospective attentional strengthening. NeuroImage 269, 119902 (2023).
Foster, J. J. & Awh, E. The role of alpha oscillations in spatial attention: limited evidence for a suppression account. Curr. Opin. Psychol. 29, 34–40 (2019).
Griffin, I. C. & Nobre, A. C. Orienting attention to locations in internal representations. J. Cogn. Neurosci. 15, 1176–1194 (2003).
Lepsien, J. & Nobre, A. C. Attentional modulation of object representations in working memory. Cereb. Cortex 17, 2072–2083 (2007).
Rihs, T. A., Michel, C. M. & Thut, G. A bias for posterior α-band power suppression versus enhancement during shifting versus maintenance of spatial attention. NeuroImage 44, 190–199 (2009).
Sauseng, P. et al. A shift of visual spatial attention is selectively associated with human EEG alpha activity. Eur. J. Neurosci. 22, 2917–2926 (2005).
Baddeley, A. Working memory: theories, models, and controversies. Annu. Rev. Psychol. 63, 1–29 (2012).
Vaziri-Pashkam, M. & Xu, Y. Goal-directed visual processing differentially impacts human ventral and dorsal visual representations. J. Neurosci. 37, 8767–8782 (2017).
Wallis, G., Stokes, M., Cousijn, H., Woolrich, M. & Nobre, A. C. Frontoparietal and cingulo-opercular networks play dissociable roles in control of working memory. J. Cogn. Neurosci. 27, 2019–2034 (2015).
Kuo, B.-C., Stokes, M. G., Murray, A. M. & Nobre, A. C. Attention biases visual activity in visual short-term memory. J. Cogn. Neurosci. 26, 1377–1389 (2014).
Pessoa, L., Gutierrez, E., Bandettini, P. A. & Ungerleider, L. G. Neural correlates of visual working memory: fMRI amplitude predicts task performance. Neuron 35, 975–987 (2002).
Santangelo, V., Di Francesco, S. A., Mastroberardino, S. & Macaluso, E. Parietal cortex integrates contextual and saliency signals during the encoding of natural scenes in working memory. Hum. Brain Mapp. 36, 5003–5017 (2015).
Han, S., Zhou, H., Tian, Y. & Ku, Y. Early top-down control of internal selection induced by retrospective cues in visual working memory: advantage of peripheral over central cues. Prog. Neurobiol. 230, 102521 (2023).
Botta, F., Lupianez, J., Santangelo, V. & Martin-Arevalo, E. Transcranial magnetic stimulation of the right superior parietal lobule modulates the retro-cue benefit in visual short-term memory. Brain Sci. 11, 252 (2021).
Jiang, L. et al. Spatial–rhythmic network as a biomarker of familial risk for psychotic bipolar disorder. Nat. Ment. Health 1, 887–899 (2023).
Yao, D. A method to standardize a reference of scalp EEG recordings to a point at infinity. Physiol. Meas. 22, 693 (2001).
Yao, D. Is the surface potential integral of a dipole in a volume conductor always zero? A cloud over the average reference of EEG and ERP. Brain Topogr. 30, 161–171 (2017).
He, B. et al. eConnectome: a MATLAB toolbox for mapping and imaging of brain functional connectivity. J. Neurosci. Methods 195, 261–269 (2011).
Grech, R. et al. Review on solving the inverse problem in EEG source analysis. J. Neuroeng. Rehabil. 5, 25 (2008).
Michel, C. M. & He, B. EEG source localization. Handb. Clin. Neurol. 160, 85–101 (2019).
Imperatori, C. et al. Aberrant EEG functional connectivity and EEG power spectra in resting state post-traumatic stress disorder: a sLORETA study. Biol. Psychol. 102, 10–17 (2014).
Keeser, D. et al. Prefrontal direct current stimulation modulates resting EEG and event-related potentials in healthy subjects: a standardized low resolution tomography (sLORETA) study. NeuroImage 55, 644–657 (2011).
Schmidt, M. N., Winther, O. & Hansen, L. K. Bayesian non-negative matrix factorization. International Conference on Independent Component Analysis and Signal Separation. Berlin, Heidelberg: Springer Berlin Heidelberg, 540–547 (2009).
Grinsted, A., Moore, J. C. & Jevrejeva, S. Application of the cross wavelet transform and wavelet coherence to geophysical time series. Nonlinear Process. Geophys. 11, 561–566 (2004).
Lachaux, J.-P. et al. Estimating the time-course of coherence between single-trial brain signals: an introduction to wavelet coherence. Neurophysiol. Clin. 32, 157–174 (2002).
Leys, C., Klein, O., Dominicy, Y. & Ley, C. Detecting multivariate outliers: use a robust variant of the Mahalanobis distance. J. Exp. Soc. Psychol. 74, 150–156 (2018).
Dickhaus, T. Neurophysiological predictor of SMR-based BCI performance. NeuroImage 51, 1303–1309 (2010).
Jiang, L. et al. Altered temporal variability in brain functional connectivity identified by fuzzy entropy underlines schizophrenia deficits. J. Psychiatr. Res. 148, 315–324 (2022).
Abdi, H. Partial least squares regression and projection on latent structure regression (PLS Regression). Wiley Interdiscip. Rev.: Comput. Stat. 2, 97–106 (2010).
Váša, F. et al. Adolescent tuning of association cortex in human structural brain networks. Cereb. Cortex 28, 281–294 (2018).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (#W2411084, #82372084), the STI 2030-Major Projects (#2022ZD0208500), Sichuan Science and Technology Program (#2025YFHZ0261), the Key R&D projects of the Science & Technology Department of Chengdu (#2024-YF08-00072-GX), China Postdoctoral Science Foundation (#2024M760359) and the Postdoctoral Fellowship Program of CPSF (#GZC20240211).
Author information
Authors and Affiliations
Contributions
Conceptualization: C.C. and P.X.; methodology: C.C., C.Y.; investigation: C.C., Y.X., J.Z., S.X. and C.L.; writing: C.C.; review and editing: C.C., X.Z., F.L. and P.X.; funding acquisition: D.Y. and P.X.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Swati Agrawal and Valerio Santangelo for their contribution to the peer review of this work. Primary Handling Editors: Christian Beste and Benjamin Bessieres.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, C., Xie, Y., Zhao, J. et al. Prioritizing flexible working memory representations relies on the involvement of the parahippocampal-insula network during memory reactivation. Commun Biol 8, 1648 (2025). https://doi.org/10.1038/s42003-025-09029-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s42003-025-09029-0