Abstract
Ecological adaptive radiation drives evolutionary innovation, yet intrinsic constraints and environmental stressors may dictate its limits. Heteroptera, repeatedly colonized aquatic realms, but their major terrestrial lineage Pentatomomorpha remained confined to land for least 230 million years. We shatter this paradigm with the first semi-aquatic Pentatomomorpha, Zekuformidae fam. nov., an Early Miocene semi-aquatic fossil from Tibetan Plateau. Convergent, it evolved trimerous antennae (extremely short) and subapically inserted pretarsal claws for water-land transitions, yet lacked key adaptations such as hydrodynamic body shapes, elongate legs for propulsion, and hydrophobic setae. Mid-Miocene aridification from Tibetan uplift extinguished its wetland habitat, dooming this “evolutionary dead-end.” This discovery reveals four independent aquatic origins in Heteroptera, exposing how selection and constraint govern ecological transitions.
Similar content being viewed by others
Introduction
The history of life is a story of dramatic ecological shifts. We see this in Devonian fish venturing onto land, and in insects later taking to the skies, each event a testament to evolutionary innovation1,2. But for every success, the fossil record holds evidence of failed experiments: Jurassic gliders that disappeared along with their membranous wings, or Triassic pseudosuchians outcompeted by their rivals3,4. These examples remind us that evolution is driven as much by boundless creativity as it is by relentless selection. Few groups showcase this dynamic better than insects, whose relentless adaptation has allowed them to permeate nearly every environment on Earth. Among them, the true bugs (Hemiptera: Heteroptera) stand out. With over 45,000 described species found from deserts to ocean spray zones, their evolutionary journey is marked by repeated habitat transitions5. While their ancestors were likely terrestrial, several lineages independently forged lives in or on water5. Against this backdrop of aquatic experimentation, the Pentatomomorpha, a massive group of over 16,000 species—have long stood as a bastion of terrestrial life6. Despite occupying diverse microhabitats including shrubs, arboreal strata, bark crevices, leaf litter, and even ant nests, ecological transitions into semi-aquatic or aquatic niches remain unverified in these lineages5,6. With very few exceptions (like Lipostemmata, which may live on floating ferns), their evolution has been confined to land6,7.
It is therefore surprising that we now report a fossil that breaks this long-standing pattern. From the Early Miocene of the Tibetan Plateau, we describe Zekuformidae fam. nov., a new family of true bugs that represents the first semi-aquatic lineage ever discovered within the superfamily Pentatomoidea. Its morphology tells a clear story: truncated antennae and claws positioned high on the tarsi—features that curiously mirror those of other insects that live at the water’s edge. This discovery does more than just add a new branch to the heteropteran family tree. It reveals a previously unknown ecological experiment, a foray into the semi-aquatic realm by a group thought to be strictly land-bound. Furthermore, the presence of this insect in Tibet paints a picture of a region that was warmer, wetter, and ecologically more complex before the major uplift of the plateau. The Zekuformidae ultimately went extinct, likely a casualty of the ensuing climatic upheaval.
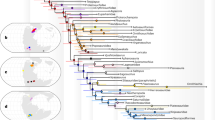
a–i Photographs of CNU-HET-QZ2015010 female. a In dorsal view. b In dorsal view with alcohol. c Hemelytron. d Head. e Antenna. f Reflective fluorescence imaging of the Antennomere II. g Tarsus. h Ovipositor. i Reflective fluorescence imaging of the scutellum. j Geometric morphometric analysis of tarsomere III and pretarsal claw in Heteroptera. k Reconstruction of macrohabitat associations across Heteroptera. (Modified from Weirauch et al.6). l Ancestral-state reconstruction of the number of antennomeres in Pentatomoidea. Scale bars = 5 mm for (a and b); 1 mm for (c, d, e), and i; 0.5 mm in (g and h).
Results
Family: Zekuformidae fam. nov. (Fig. 1, Supplementary Fig. S1)
ZooBank LSID: urn:lsid:zoobank.org:act:FABEC944-3E64-403B-892E-9E91AE8614F5.
Type genus: Zekuforma gen. nov
Diagnosis
Antenna trimerous (three antennomeres), shorter than pronotum (Fig. 1e); mesoscutellum narrow linguliform, posterior angle well-developed, extending posteriorly to cover clavus, exceeding half of abdominal length (Fig. 1i); tarsi trimerous, tarsomere III swollen (Fig. 1g); pretarsal claws inserted subapically on tarsomere III; tergite IX in female separated, equal to or longer than gonocoxite VIII (Fig. 1h); tergite IX in female visible dorsally, not concealed by subapically positioned tergite VIII (Fig. 1h).
Genus: Zekuforma gen. nov
ZooBank LSID: urn:lsid:zoobank.org:act:7D8B42A4-C954-469A-B3D0-52271FFCA9C9
Type species: Zekuforma maculata sp. nov
Diagnosis
Body medium, approximately 20 mm; integument smooth with maculations (Supplementary Fig. S1b); head triangular, length subequal to width (Fig. 1d); clypeus prominent, surpassing anterior margins of mandibular plates; compound eyes small, diameter not exceeding 1/4 of head length (Fig.1d); pronotum collar less, calli absent, posterior angle rounded (Supplementary Fig. S1b); mesoscutellum reaching abdominal sternite V, posterior angles narrowing posteriorly; tarsomere III longest, tarsomere II shortest (Supplementary Fig. S1e).
Species included. Only the type species is known.
Zekuforma maculata sp. nov. (Fig. 1, Supplementary Fig. S1)
ZooBank LSID: urn:lsid:zoobank.org:act:5A0A5D6F-C3F3-431C-AFEE-48C507C19426.
Material
Holotype, female: CNU-HET-QZ2015010 (housed in the Key Lab of Insect Evolution and Environmental Changes, College of Life Sciences, Capital Normal University, Beijing, China).
Locality and age
Garang Formation; Early Miocene (~19.3 Ma); Caergen Village, Zeku County, Qinghai Province, China.
Diagnosis
Antennomere III longest, fusiformly swollen; labium reaching metacoxae, article I longest, article IV shortest (Supplementary Fig. S1a); pronotum transverse, width twice length, disc with four black circular maculae (Supplementary Fig. S1b); apices of all femora and tibiae darkened; proximal femoral regions near apices each bearing a black ring (Fig. 1a); apices of connexival tergites darkened; sternites each with a black circular macula surrounding spiracle (Supplementary Figs. S1g, h).
(More details see Supplementary Text: Results)
Discussion
Insect adaptive radiations often have tight links between antennal morphology and niche specialization, especially in heteropteran terrestrial-to-aquatic transitions. Fully aquatic Nepomorpha (e.g., Belostomatidae, Corixidae) exhibit extremely shortened antennae (≤3 antennomeres) concealed in cephalic grooves to reduce hydrodynamic drag, while semi-aquatic lineages (e.g., Gelastocoridae, Ochteridae) retain robust, laterally projecting antennae (3–4 antennomeres)5. Surface-dwelling Gerromorpha possess moderately elongated antennae (4 antennomeres), contrasting with terrestrial Pentatomomorpha, which rely on slender 4–5-articled antennae for phytochemical detection8. Within Pentatomoidea, most taxa possess slender, pentamerous antennae, with tetramerous antennae representing the ancestral character state (Fig. 1l). Critically, our phylogenetic analyses place Zekuforma maculata sp. nov. robustly within Pentatomoidea while revealing its exceptional divergence (Supplementary Fig. S2). This novel lineage is defined by the following synapomorphies warranting establishment of Zekuformidae fam. nov.: (1) Trimerous antennae (Character 12-2); (2) Subapically inserted pretarsal claws (58-1); (3) Dorsally exposed female abdominal tergite IX (43-0); (4) Membranous gonapophyses VIII with reduced first rami (45-1); (5) Completely fused gonocoxites IX forming a sclerotized plate (46-3); (6) Laterotergites IX equal to/exceeding gonocoxites VIII length (48-1). Notably, the trimerous antennae—markedly shortened and robust—represent a radical departure from the pentatomoid ground plan. Trimerous antennae occur only in Zekuformidae, Phloeidae, and the Australian pentatomid genus Omyta9. However, our phylogenetic analyses reveal distant relationships among these lineages, with ancestral-state reconstruction confirming independent derivations of the trimerous condition (Fig. 1l and Supplementary Fig. S2). Crucially, Zekuforma maculata exhibits extremely shortened, robust antennae not exceeding the head length, whereas both Omyta and Phloeidae possess elongate, slender antennae. In Omyta the antennae exceed the combined head-pronotum length, while Phloeidae’s slender antennae fold dorsoventrally beneath cephalic expansions for bark camouflage. Strikingly, Z. maculata—phylogenetically nested within terrestrial Pentatomoidea—evolved trimerous, robust antennae convergent with semi-aquatic groups. And, reflective fluorescence imaging shows sparse, stout sensilla in Z. maculata (Fig. 1f), analogous to aquatic mechanosensilla in Nepomorpha, contrasting with dense chemosensilla in terrestrial relatives, suggesting adaptation to humid substrates10,11. Zekuforma maculata further has unique pretarsal claws: subapical insertion on the third tarsomere with terminal expansion (Fig. 1g)—a configuration previously exclusive to Gerromorpha (e.g., Veliidae, Gerridae) for surface propulsion12. In Gerromorpha, subapical claws (often with swimming fans) enhance hydroplaning, while apical claws correlate with damp-ground habitats12. Geometric morphometrics of tarsomere III and the claw confirms, Z. maculata clusters with semi-aquatic taxa (Fig. 1j, Supplementary Figs. S3, S4), supporting water-associated niche exploitation. Phylogenetically nested within basal Pentatomoidea, Z. maculata’s traits evolved independently from aquatic lineages, suggesting a bold ecological foray. However, unlike typical aquatic or semi-aquatic heteropterans inhabiting water surfaces or subaquatic environments—which often exhibit dense hydrophobic setae to prevent water infiltration—the specimen of Z. maculata lacks such cuticular specializations. Combined with its broad body and short, robust legs, we hypothesize that Z. maculata could not sustain prolonged hydroplaning on water surfaces (a trait enabled in most Veliidae and Gerridae by streamlined bodies and specialized elongate legs) nor survive fully submerged (as seen in lineages of Nepomorpha with natatorial legs and water-repellent pubescence). This “partial specialization” pattern closely mirrors transitional stages in ancestral gerromorphan evolution. Andersen (1982)8 proposed that early gerromorphan ancestors similarly inhabited semi-aquatic, humid habitats. Through progressive morphological innovations—such as gradual leg elongation, tarsal tip refinement, and subapical claw migration—they radiated into surface-dwelling niches, eventually evolving traits for sustained aquatic life (e.g., hydrophobic setae, streamlined bodies). Zekuformidae likely occupied analogous transitional habitats, bridging terrestrial and aquatic realms. However, the lifestyle of Zekuformidae likely differed from that of other groups of semi-aquatic phytophagous insects, such as the Donaciinae and certain Miridae species like Eccritotarsus catarinensis. Adults of species such as Donaciasta goeckei are associated with floating-leaved aquatic plants like Nymphaea13. Their tarsal tips bear setae and spurs for enhanced attachment to aquatic plants, and generally attach themselves to the surface of the plants and only descend below the water surface to lay eggs13. Similarly, E. catarinensis is a specialized phytophagous insect tightly associated with free-floating or emergent aquatic plants (e.g., Eichhornia crassipes), on which both adults and nymphs cling and feed gregariously14. In contrast, Zekuformidae do not have structures for attaching to plants, and the tips of their tarsomeres appear to be related to hydroplaning, like some gerromorphans. Shared environmental pressures at this interface drove convergent evolution in tarsal and pretarsal claw morphology between Zekuformidae and semi-aquatic Veliidae, despite their distant phylogenetic relationship. This “partial specialization” of morphological characteristics exemplifies the transitional nature of semi-aquatic adaptive experimentation.
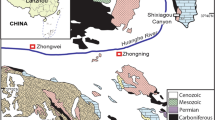
Zekuformidae were excavated from Early Miocene strata in the Zeku Basin, northeastern Tibetan Plateau (Fig. 2a–d). Today, this region exhibits complex geomorphology and ecology—dominated by vast Gobi deserts, barren plateaus, and extreme seasonality (arid climate, harsh winters)—driven by late Cenozoic tectonic uplift, resulting in limited extant biodiversity15. However, fossil evidence from coeval deposits (e.g., plant leaves, fruits, pollen, and diverse insects including Diptera, Mecoptera, Odonata, and Hemiptera) reveals a strikingly different past: a warm-temperate forest climate with lower paleoelevation (about 1200–1400 m), higher temperatures, and greater precipitation than today15,16,17,18. The associated fossil assemblage within the same stratum includes typical aquatic organisms such as odonate nymphs (Fig. 2f), corixid adults (Fig. 2g), and chironomid larvae (Fig. 2h) alongside previously documented plant fossils of pollen grains and other botanical remains18. These findings collectively confirm the existence of a stable freshwater wetland ecosystem in the Zeku Basin during the early Miocene. The temporal range of Zekuformidae aligns closely with the retreat of Paleo-Tethys remnant water systems (~25 Ma)19, suggesting their morphological innovations directly tracked wetland expansion driven by this hydrological shift. This humid phase imposed key selective pressures that catalyzed Pentatomomorpha’s first evolutionary foray beyond terrestrial niches.
a, b Environment of the Zeku Basin. c Corresponding author collecting fossils. d, e Geographical location, of the Zeku Basin in the Tibetan Plateau region, and the study site at Caergen. f Odonata nymphs. g Corixid adults. h Chironomid adults. Scale bars = 5 mm for (f); 2 mm for (g and h).
Recent studies demonstrate that the Zeku Basin experienced substantial uplift of 2200–2500 m since the Early Miocene, which, coupled with the Himalayan orogeny, triggered regional aridification characterized by reduced precipitation, temperature decline, and a transition to drier and colder local climatic conditions20. For Zekuformidae—dependent on nearshore aquatic vegetation—this climatic upheaval caused habitat fragmentation and ecological disruption, likely sealing their extinction. Or perhaps their broad body plan (lacking hydrophobic setae), shortened legs, and incomplete semi-aquatic adaptations created functional vulnerabilities. When compounded by mid-Miocene habitat loss, this dual pressure—morphological inadequacy + ecological collapse—likely funneled Zekuformidae into an evolutionary dead end. No extant relatives or ecological equivalents survive today, underscoring the precariousness of their transitional strategy. The semi-aquatic experiments represented by Zekuformidae indicate that while ecological opportunities can selectively preserve adaptive morphological innovations, their long-term adaptive success depends on the alignment between environmental stability and functional integration.
Heteropteran insects have adapted to nearly all known habitats, with recent studies suggesting their common ancestor was terrestrial6,20. Aquatic and semi-aquatic lineages independently colonized these habitats at least three times: transitions to aquatic (Nepomorpha), intertidal (Dipsocoridae within Dipsocoromorpha, Leptopodomorpha), and water surface-dwelling (Gerromorpha) environments20. Notably, some semi-aquatic groups like Ochteroidea, terrestrial genera within Mesoveliidae, Hydrometridae and Veliidae likely represent reversions from aquatic ancestors to terrestrial habitats, while Pentatomomorpha (stink bugs and allies) were traditionally considered strictly terrestrial (though some lineages occupy microhabitats like soil or beneath bark). Our findings challenge this paradigm. The discovery of Zekuformidae, a pentatomomorphan lineage potentially adapted to semi-aquatic environments, suggests that even groups historically regarded as terrestrial may have explored transitional habitats in their evolutionary past. This expands the known adaptive capacity of Heteroptera, implying at least four independent transitions from land to aquatic/semi-aquatic habitats (Fig. 1k). Such repeated ecological experiments highlight the group’s ability to test evolutionary boundaries, though success appears constrained by the depth of morpho-functional integration. For instance, selected groups of nepomorphans, the families Naucoridae, Potamocoridae, and Aphelocheiridae living in swiftly running waters evolved a plastron respiratory structure—a dense layer of water-repellent microsetae on their body surface that traps a permanent air layer. This adaptation allows direct oxygen uptake from water, bypassing the need for surfacing. Coupled with paddle-shaped appendages, this innovation established them as top aquatic predators. Gerromorpha achieved surface-dwelling through coordinated trait evolution: a streamlined body reduces drag, hydrophobic setae repel water to maintain buoyancy, elongate legs distribute weight across the water surface, and modified pretarsal claws enhance grip on unstable interfaces. Lastly, in Leptopodomorpha, certain groups like Saldidae (shore bugs) developed elevated compound eyes to expand horizontal visual range—critical for detecting predators and prey in intertidal zones—along with thickened setae on their appendages to increase adhesion on wet substrates, enabling survival in fluctuating tidal environments. By contrast, Zekuformidae exploited water-land interfaces via subapical claw positioning and shortened antennae but may failed to cross the ecological threshold due to isolated adaptations (e.g., the absence of critical features like hydrodynamic body shapes, elongate legs for propulsion, and hydrophobic setae. While we document the observable lack of specialized setae, we acknowledge that wax-producing glands, known in some recent representatives of Pentatomoidea (e.g., Tessaratoma javaniva)21, enabling hydrophobic cuticular secretions could have been present but remain undetectable in the fossil record).
Such “partial adaptations” leading to evolutionary dead-ends are widespread: Early actinopterygian fish evolved limb-like fins but failed to transition terrestrially due to incomplete skeletal-respiratory integration22. Similarly, gliding frogs (Rhacophorus spp.) possess webbed feet yet lack aerodynamic control, restricting them to arboreal niches, while early land plants like Cooksonia succeeded via synergistic cuticle-stomata systems, whereas lineages with isolated adaptations perished23,24. These cases, alongside Zekuformidae’s extinction, demonstrate that transcending habitat boundaries requires not isolated trait innovations but deep developmental-functional integration of multiple adaptations. Repeated adaptive experiments in Heteroptera and other lineages suggest evolutionary plasticity is bounded by morpho-functional integration depth and eco-developmental constraints. When innovations remain isolated within functional networks, or environmental stressors (e.g., habitat instability, niche preemption) exceed adaptive capacity, such experiments culminate in “evolutionary dead-ends.” This principle extends beyond insects, permeating all branches of the Tree of Life. The discovery of Zekuformidae reshapes the ecological paradigm of Heteroptera: traditional views confined Pentatomoidea to terrestrial niches, yet Zekuformidae reveals a bold semi-aquatic adaptive experiment. Despite its ultimate failure—attributed to shifting habitats or morphogenetic thresholds—this “failed adaptation” provides critical empirical evidence for understanding limits to cross-habitat evolution.
Methods
Localities and repositories
The compression fossils were collected from the Garang Formation (<16-19 Ma) at Caergen Village, Duohemao Town, Zeku County, eastern Qinghai Province, Northwest China (34°56’N, 101°48’E, 3700 m a.s.l.). The type specimen CNU-HET-QZ 2015010, and other specimens (Odonata nymphs: CNU-ODO-QZ 2015016, Corixid adults: CNU-HET-QZ 2015089, Chironomid adults: CNU-DIP-QZ 2015131) housed in the Key Lab of Insect Evolution and Environmental Changes, College of Life Sciences, Capital Normal University, Beijing, China (CNUB; Yunzhi Yao, Curator).
Nomenclatural acts
This published work and the nomenclatural acts it contains have been registered in ZooBank, the proposed online registration system for the International Code of Zoological Nomenclature (ICZN). The ZooBank LSIDs (Life Science Identifiers) can be resolved and the associated information viewed through any standard web browser by appending the LSID to the prefix http://zoobank.org/. The LSID for this publication are:urn:lsid:zoobank.org:pub:00210184-1C81-41A5-A98C-945AAB6D9FE7
Specimen imaging
To clearly observe the specimen impressions, we employed three imaging techniques:
-
1.
Light Microscopy: The specimens were examined and photographed using a Nikon SMZ25 dissecting microscope and a Nikon ECLIPSE Ni microscope, both equipped with a Nikon DS-Ri 2 digital camera system (Nikon, Japan).
-
2.
Extended Depth-of-Field Imaging: The GW-11A high-depth micro-specimen digital imaging system was used to capture wing venation details through multi-angle illumination.
-
3.
Confocal Laser Microscopy: For the first time, a LEICA SP8 confocal laser microscope was employed to image the fossil impressions. Ultrahigh-resolution reflected fluorescence revealed the boundaries of the scutellum and densely packed sensory receptors on the antennae.
Statistics and reproducibility
Measurements and terminology
Morphological terminology mainly follows Grazia et al.25. Body length was measured along the midline from the apex of the head to the apex of the abdomen. Body width was measured at the maximum width of the body. The length of the fore wing was measured from its base to its apex. All measurements are given in millimeters (mm).
Phylogenetic analyses
In addition to our fossil, Zekuforma maculata, the characters and taxa used are those of Grazia et al.25, which is the reliable phylogenetic analysis of the Pentatomoidea (see Supplementary Appendix 1). Cladistic analysis was performed using TNT 1.6 with running the script guoyi.run v. 0.6 provided by Zhang (2023)26,27,28. First, Maximum-parsimony analysis was performed with implied weighting with the weighting set as 12 according to recommendations and tree searching with tree bisection reconnection (TBR)27. Bootstrap analysis was undertaken for MP with 1000 replicates, and the consistency index (CI) and retention index (RI) were calculated using the script guoyi.run v. 0.6 by TNT. The character mapping for the phylogenetic tree was performed by WinClada 2.0. CI = 0.497;RI = 0.749;Tree Length (TL) is 169 (see Supplementary Appendix 2, Supplementary Data 1).
Ancestral-state reconstruction
We reconstructed the ancestral states of antennal segment number across Pentatomoidea lineages using equally weighted parsimony methods in Mesquite (Version 3.70). The coding protocol recognized three characteristic states: (0) four-segmented antennae, (1) five-segmented antennae, and (2) three-segmented antennae, following the taxonomic framework for hemipteran antennal segmentation. State transition patterns were resolved through iterative optimization algorithms, with minimum step lengths calculated for each node.
Geometric morphometric analyses
To quantify morphological differences in tarsal claw attachment positions, we selected the third tarsomere and a single claw (to avoid interference from multiple claw morphologies) from 34 species for landmark-based analysis (see Supplementary Appendix 3, Supplementary Data 2). The landmark configuration consisted of 30 semilandmarks along the tarsal edge and 12 semilandmarks covering the claw base to tip. Specimens were categorized into three functional groups: (1) subapical claw attachment (adapted for surface-dwelling and newly described species, n = 12), (2) apical claw attachment (terrestrial and semi-aquatic groups, n = 19), and (3) elongated claws and tarsomeres (specialized aquatic taxa, n = 3). Dorsal view images were digitized using tps-DIG 2.31, with shape configurations standardized through Procrustes superimposition to remove non-shape variations. Principal component analysis (PCA) and canonical variate analysis (CVA) implemented in MorphoJ 1.07 were employed to evaluate geometric shape divergence among the three groups.
Data availability
The datasets generated during and/or analysed during the current study are available in the Supplementary information
References
Shubin, N. H., Daeschler, E. B. & Jenkins, J.r F. A. The pectoral fin of Tiktaalik roseae and the origin of the tetrapod limb. Nature 440, 764–771 (2006).
Engel, M. S. & Grimaldi, D. A. New light shed on the oldest insect. Nature 427, 627–630 (2004).
Meng, L. et al. A Mesozoic gliding mammal from northeastern China. Nature 444, 889–893 (2006).
Nesbitt, S. J. The early evolution of archosaurs: Relationships and the origin of major clades. Bull. Am. Mus. Nat. Hist. 352, 1–292 (2011).
Schuh, R. T. & Weirauch, C. True Bugs of the World (Hemiptera: Heteroptera). Classification and Natural History. In Monogr. Ser., second ed., vol. 8. (Siri Scientific Press 2020).
Weirauch, C. et al. Revisiting habitat and lifestyle transitions in Heteroptera (Insecta: Hemiptera): insights from a combined morphological and molecular phylogeny. Cladistics 35, 67–105 (2019).
Baranowski, R. M. & Bennett, F. D. Biology, host specificity, and descriptions of the immatrure stages of Lipostemmata major Ashlock and L. humeralis Berg (Hemiptera: Lygaeidae). Fla. Entomol. 62, 383–389 (1979).
Andersen, N. M. The Semiaquatic Bugs (Hemiptera, Gerromorpha). Phylogeny Adaptations, Biogeography, and Classification. Entomonograph vol. 3. (Klampenborg, Denmark: Scandinavian Science Press 1982).
Gross, G. F. Plant-feeding and other bugs (Hemiptera) of South Australia. Heteroptera-Part II. (Handbook of the Flora and Fauna of South Australia 1976).
Nowińska, A. & Brożek, J. Insect evolution toward aquatic habitats; reassessment of antennal sensilla in the water bug families Chteridae, Gelastocoridae and Aphelocheiridae (Hemiptera: Heteroptera: Nepomorpha). Contrib. Zool. 89, 412–433 (2020).
Nowińska, A. Report on the types and distribution of antennal sensilla in Lygaeidae (Heteroptera: Lygaeoidea) and their putative functions. Insects 16, 44 (2025).
Chen, P. P., Nieser, N. & Zettel, H. The aquatic and semi-aquatic bugs (Heteroptera: Nepomorpha & Gerromorpha) of Malesia (Vol. 5). (Brill 2021).
Grobbelaar, E. Research on Chrysomelidae (Vol. 2). (Brill 2009).
Henry, T. J. Description of a Cryptic New Species of the Plant Bug Genus Eccritotarsus (Heteroptera: Miridae: Bryocorinae) from Peru, a New Biocontrol Agent of Water Hyacinth, Eichhornia crassipes (Pontederiaceae). Proc. Entomol. Soc. Wash. 119, 398–407 (2017).
Li, X. C. et al. Fossil fruits of Koelreuteria (Sapindaceae) from the Miocene of northeastern Tibetan Plateau and their palaeoenvironmental, phytogeographic and phylogenetic implications. Rev. Palaeobot. Palynol. 234, 125–135 (2016).
Wu, S. et al. New fossil representative of the genus Helius (Diptera, Limoniidae) from the little known and newly discovered locality Caergen Village of northeastern Tibetan Plateau (China). Palaeontol. Electron. 22, 1–8 (2019).
Lai, Y. J. et al. Integrating fossil and extant plant communities to calibrate paleoelevation of the Qinghai-Tibet Plateau. J. Syst. Evol. 63, 25–38 (2025).
Hui, Z. C. et al. Miocene pollen assemblages from the Zeku Basin, northeastern Tibetan Plateau, and their palaeoecological and palaeoaltimetric implications. Palaeogeogr. Palaeoclimatol. Palaeoecol. 511, 419–432 (2018).
Rögl, Mediterranean, F. & Paratethys. Facts and hypotheses of an Oligocene to Miocene paleogeography. Geologica. Carpath. 50, 339–349 (1999).
Wang, Y. H. et al. Phylogenetic divergences of the true bugs (Insecta: Hemiptera: Heteroptera), with emphasis on the aquatic lineages: the last piece of the aquatic insect jigsaw originated in the Late Permian/Early Triassic. Cladistics 32, 390–405 (2016).
Kumari, A. et al. A Journey into Entomolgy (Vol. 1). (Lekh Publications 2023)
Coates, M. I. & Clack, J. A. Romer’s gap: tetrapod origins and terrestriality. Bull. Mus. Nat. Hist. Nat. Par. 17, 373–388 (1995).
Harvey, M. B., Pemberton, A. J. & Smith, E. N. New and poorly known parachuting frogs (Rhacophoridae: Rhacophorus) from Sumatra and Java. Herpetol. Monogr. 16, 46–92 (2002).
Bateman, R. M. et al. Early evolution of land plants: phylogeny, physiology, and ecology of the primary terrestrial radiation. Annu. Rev. Ecol. Syst. 29, 263–292 (1998).
Grazia, J., Schuh, R. T. & Wheeler, W. C. Phylogenetic relationships of family groups in Pentatomoidea based on morphology and DNA sequences (Insecta: Heteroptera). Cladistics 24, 932–976 (2008).
Zhang, G. Y. et al. Systematics and biogeography of the genera Pseudiberus Ancey, 1887 and Platypetasus Pilsbry, 1895 (Stylommatophora: Camaenidae), with the description of Pseudiberus shanheicus n. sp. J. Conchol. 45, 77–90 (2024).
Goloboff, P. A., Torres, A. & Arias, J. S. Weighted parsimony outperforms other methods of phylogenetic inference under models appropriate for morphology. Cladistics 34, 407–437 (2018).
Goloboff, P. A. & Morales, M. E. TNT version 16 with a granbicalinterface for MacOS and Linux, including new routines in parallel. Cladistics 39, 144–153 (2023).
Acknowledgements
We are deeply grateful to the anonymous reviewers and editors for their invaluable comments and suggestions, which have significantly improved this manuscript. We also thank to Drs. Feng Qi, Jiawei Chao, Xiaoyue Yang, and Zhaoqi Leng for their technical support. We are also grateful Yue Wang (Institute of Zoology, Chinese Academy of Sciences) for the laser scanning and analysis of specimens. This project has been supported by the National Natural Science Foundation of China (42288201; 42472001; 32200366).
Author information
Authors and Affiliations
Contributions
Y.Y. and R.D. conceived and designed the study; R.D. and Y.Y. performed the analyses and wrote the manuscript; R.D. and S.D. prepared the figures; M.S.E., D.R., and S.L. revised the manuscript. All authors read and approve the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Petr Kment and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editors: Katie Davis and Johannes Stortz.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Dai, R., Engel, M.S., Du, S. et al. Rare fossil insect (Zekuforma maculata sp. nov) from the Tibetan plateau unveils adaptive innovations and extinction mechanisms. Commun Biol 8, 1754 (2025). https://doi.org/10.1038/s42003-025-09134-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s42003-025-09134-0