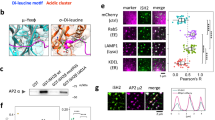
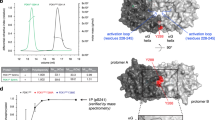
Abstract
Phosphoinositide-3 kinase (PI3K) is a central regulator of cellular metabolism and survival, and its dysregulation is implicated in major human diseases, particularly cancer. The p85 regulatory subunit of PI3K uses its C-terminal domains to stabilise the catalytic p110 subunit in an inhibited state. Certain Src homology 3 (SH3) domains activate p110 by binding to the proline-rich (PR) 1 motif at the N-terminus of p85. How this interaction leads to PI3K activation remains unclear. Moreover, the low specificity of SH3 domains raises the question about how they can selectively control PI3K activation. Combining structural, biophysical, and functional methods, we demonstrate that both questions are linked: PI3K-activating SH3 domains form additional ‘tertiary’ interactions with the C-terminal domains of p85, relieving p110 inhibition. SH3 domains lacking these tertiary contacts may bind p85 with similar affinity but fail to activate PI3K. Thus, p85 employs a selection mechanism that discriminates based on binding mode rather than binding strength, preventing nonspecific activation rather than nonspecific binding. This mechanism conveys a functional selectivity to SH3 domains that are otherwise considered promiscuous. These insights establish a mechanistic framework that will help to predict, modulate, and therapeutically target SH3-driven PI3K activation in disease.
Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article and its supplementary information files. The source data underlying graphs, plots and charts in the manuscript are presented in Supplementary Data 2. The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE partner repository with the dataset identifier PXD058464.
References
Zhang, J. Evolution by gene duplication: an update. Trends Ecol. Evol. 18, 292–298 (2003).
Ladbury, J. E. & Arold, S. Searching for specificity in SH domains. Chem. Biol. 7, R3–R8 (2000).
Ladbury, J. E. & Arold, S. T. Energetics of Src homology domain interactions in receptor tyrosine kinase-mediated signaling. in Methods in Enzymology vol. 488 (Elsevier, 2011).
Li, S. S. C. Specificity and versatility of SH3 and other proline-recognition domains: Structural basis and implications for cellular signal transduction. Biochem. J. 390, 641–653 (2005).
Ladbury, J. E. & Arold, S. T. Noise in cellular signaling pathways: causes and effects. Trends Biochem. Sci. 37, 173–178 (2012).
Milanesi, L. et al. Systematic analysis of human kinase genes: a large number of genes and alternative splicing events result in functional and structural diversity. BMC Bioinform. 6, S20 (2005).
Backer, J. M. The regulation of class IA PI 3-kinases by inter-subunit interactions. Curr. Top. Microbiol. Immunol. 346, 87–114 (2010).
Engelman, J. A., Luo, J. & Cantley, L. C. The evolution of phosphatidylinositol 3-kinases as regulators of growth and metabolism. Nat. Rev. Genet. 7, 606–619 (2006).
Rai, S. N. et al. The Role of PI3K/Akt and ERK in neurodegenerative disorders. Neurotox. Res. 35, 775–795 (2019).
Vanhaesebroeck, B., Guillermet-Guibert, J., Graupera, M. & Bilanges, B. The emerging mechanisms of isoform-specific PI3K signalling. Nat. Rev. Mol. Cell Biol. 11, 329–341 (2010).
Yu, J. et al. Regulation of the p85/p110 phosphatidylinositol 3′-Kinase: stabilization and inhibition of the p110α catalytic subunit by the p85 regulatory subunit. Mol. Cell. Biol. 18, 1379–1387 (1998).
Hale, B. G. et al. CDK/ERK-mediated phosphorylation of the human influenza A virus NS1 protein at threonine-215. Virology 383, 6–11 (2009).
Mandelker, D. et al. A frequent kinase domain mutation that changes the interaction between PI3Kα and the membrane. Proc. Natl. Acad. Sci. USA. 106, 16996–17001 (2009).
Yu, J., Wjasow, C. & Backer, J. M. Regulation of the p85/p110α phosphatidylinositol 3’-kinase: Distinct roles for the n-terminal and c-terminal sh2 domains. J. Biol. Chem. 273, 30199–30203 (1998).
Mutations, O. P. et al. The structure of a human p110a/p85a complex elucidates the effects of oncogenic PI3Ka mutations. Science 318, 1744–1748 (2007).
Zhang, X. et al. Structure of lipid kinase p110β/p85β elucidates an unusual SH2-domain-mediated inhibitory mechanism. Mol. Cell 41, 567–578 (2011).
Hart, J. R. et al. Nanobodies and chemical cross-links advance the structural and functional analysis of PI3Kα. Proc. Natl. Acad. Sci. USA. 119, e2210769119 (2022).
Hofmann, B. T. & Jücker, M. Activation of PI3K/Akt signaling by n-terminal SH2 domain mutants of the p85α regulatory subunit of PI3K is enhanced by deletion of its c-terminal SH2 domain. Cell. Signal. 24, 1950–1954 (2012).
Ito, Y., Vogt, P. K. & Hart, J. R. Domain analysis reveals striking functional differences between the regulatory subunits of phosphatidylinositol 3-kinase (PI3K), p85α and p85β. Oncotarget 8, 55863–55876 (2017).
Jücker, M. et al. Expression of a mutated form of the p85α regulatory subunit of phosphatidylinositol 3-kinase in a Hodgkin’s lymphoma-derived cell line (CO). Leukemia 16, 894–901 (2002).
Carpenter, C. L. et al. Phosphoinositide 3-kinase is activated by phosphopeptides that bind to the SH2 domains of the 85-kDa subunit. J. Biol. Chem. 268, 9478–9483 (1993).
Li, X. et al. Cancer-associated mutations in the p85α N-terminal SH2 domain activate a spectrum of receptor tyrosine kinases. Proc. Natl. Acad. Sci. USA. 118, e2101751118 (2021).
Axelsson, L. et al. Clustering of β2-integrins on human neutrophils activates dual signaling pathways to Ptdlns 3-kinase. Exp. Cell Res. 256, 257–263 (2000).
Kapeller, R. et al. Identification of two SH3-binding motifs in the regulatory subunit of phosphatidylinositol 3-kinase*. J. Biol. Chem. 269, 1927–1933 (1994).
Pleiman, C. M., Hertz, W. M. & Cambier, J. C. Activation of phosphatidylinositol-3′ kinase by Src-family kinase SH3 binding to the p85 subunit. Science 263, 1609–12 (1994).
Prasad, K. V. et al. Regulation of CD4-p56lck-associated phosphatidylinositol 3-kinase (PI 3-kinase) and phosphatidylinositol 4-kinase (PI 4-kinase). Philos. Trans. R. Soc. Lond. B. Biol. Sci. 342, 35–42 (1993).
Renzoni, D. A. et al. Structural and thermodynamic characterization of the interaction of the SH3 domain from Fyn with the proline-rich binding site on the p85 subunit of PI3-kinase. Biochemistry 35, 15646–15653 (1996).
Liu, X. et al. Cryo-EM structures of PI3Kα reveal conformational changes during inhibition and activation. Proc. Natl. Acad. Sci. USA. 118, e2109327118 (2021).
Arold, S. et al. The crystal structure of HIV-1 Nef protein bound to the Fyn kinase SH3 domain suggests a role for this complex in altered T cell receptor signaling. Structure 5, 1361–1372 (1997).
Collette, Y. et al. HIV-2 and SIV Nef proteins target different Src family SH3 domains than does HIV-1 Nef because of a triple amino acid substitution. J. Biol. Chem. 275, 4171–4176 (2000).
Horenkamp, F. A. et al. Conformation of the dileucine-based sorting motif in HIV-1 Nef revealed by intermolecular domain assembly. Traffic 12, 867–877 (2011).
Chi-Hon, L., Saksela, K., Mirza, U. A., Chait, B. T. & Kuriyan, J. Crystal structure of the conserved core of HIV-1 Nef complexed with a Src family SH3 domain. Cell 85, 931–942 (1996).
Lee, C. H. et al. A single amino acid in the SH3 domain of Hck determines its high affinity and specificity in binding to HIV-1 Nef protein. EMBO J. 14, 5006–5015 (1995).
Aldehaiman, A. et al. Erratum: synergy and allostery in ligand binding by HIV-1 Nef. Biochem. J. 478, 1525–1545 (2021).
Arold, S. et al. RT loop flexibility enhances the specificity of Src family SH3 domains for HIV-1 Nef. Biochemistry 37, 14683–14691 (1998).
Klippel, A., Escobedo, J. A., Fantl, W. J. & Williams, L. T. The C-terminal SH2 domain of p85 accounts for the high affinity and specificity of the binding of phosphatidylinositol 3-kinase to phosphorylated platelet-derived growth factor beta receptor. Mol. Cell. Biol. 12, 1451–1459 (1992).
Malagrinò, F., Troilo, F., Bonetti, D., Toto, A. & Gianni, S. Mapping the allosteric network within a SH3 domain. Sci. Rep. 9, (2019).
Franke, T. F. et al. The protein kinase encoded by the Akt proto-oncogene is a target of the PDGF-activated phosphatidylinositol 3-kinase. Cell 81, 8279 (1995).
Guzmán-Vega, F. J. & ’Arold, S. T. Scaling up protein modeling with AlphaFold for SLURM-based HPC clusters. Preprint at https://github.com/strubelab/alphafold (2024).
Jumper, J. et al. Highly accurate protein structure prediction with AlphaFold. Nature 596, 583–589 (2021).
Penuel, E. & Martin, G. S. Transformation by v-Src: Ras-MAPK and PI3K-mTOR mediate parallel pathways. Mol. Biol. Cell 10, 1693–1703 (1999).
Sweeney, S. M. et al. AACR project genie: powering precision medicine through an international consortium. Cancer Discov 7, 818–831 (2017).
Wang, J., Auger, K. R., Jarvis, L., Shi, Y. & Roberts, T. M. Direct association of Grb2 with the p85 subunit of phosphatidylinositol 3-kinase. J. Biol. Chem. 270, 12774–80 (1995).
Haidar, M. et al. TGF-β2 induces Grb2 to recruit PI3-K to TGF-RII that activates JNK/AP-1-signaling and augments invasiveness of Theileria-transformed macrophages. Sci. Rep. 5, 15688 (2015).
Sastry, L., Cao, T. & King, C. R. Multiple Grb2-protein complexes in human cancer cells. Int. J. Cancer 70, 208–213 (1997).
Weinger, J. G. et al. In brain, Axl recruits Grb2 and the p85 regulatory subunit of PI3 kinase; in vitro mutagenesis defines the requisite binding sites for downstream Akt activation. J. Neurochem. 106, 134–146 (2008).
Cheung, L. W. T. et al. Regulation of the PI3K pathway through a p85α monomer–homodimer equilibrium. eLife 4, e06866 (2015).
Layton, M. J., Harpur, A. G., Panayotou, G., Bastiaens, P. I. H. & Waterfield, M. D. Binding of a diphosphotyrosine-containing peptide that mimics activated platelet-derived growth factor receptor β induces oligomerization of phosphatidylinositol 3-kinase. J. Biol. Chem. 273, 33379–33385 (1998).
Ge, Y., Paisie, T. K., Chen, S. & Concannon, P. UBASH3A regulates the synthesis and dynamics of TCR–CD3 complexes. J. Immunol. 203, 2827–2836 (2019).
Kowanetz, K. et al. Suppressors of T-cell receptor signaling Sts-1 and Sts-2 bind to Cbl and inhibit endocytosis of receptor tyrosine kinases. J. Biol. Chem. 279, 32786–32795 (2004).
Bertelsen, V., Breen, K., Sandvig, K., Stang, E. & Madshus, I. H. The Cbl-interacting protein TULA inhibits dynamin-dependent endocytosis. Exp. Cell Res. 313, 1696–1709 (2007).
Blaize, G. et al. CD5 signalosome coordinates antagonist TCR signals to control the generation of Treg cells induced by foreign antigens. Proc. Natl. Acad. Sci. USA. 117, 12969–12979 (2020).
Xu, W., Harrison, S. C. & Eck, M. J. Three-dimensional structure of the tyrosine kinase c-Src. Nature 385, 595–602 (1997).
Sicheri, F., Moarefi, I. & Kuriyan, J. Crystal structure of the Src family tyrosine kinase Hck. Nature 385, 602–609 (1997).
Mitra, S. K. & Schlaepfer, D. D. Integrin-regulated FAK–Src signaling in normal and cancer cells. Curr. Opin. Cell Biol. 18, 516–523 (2006).
Boggon, T. J. & Eck, M. J. Structure and regulation of Src family kinases. Oncogene 23, 7918–7927 (2004).
Seet, B. T. et al. Efficient T-cell receptor signaling requires a high-affinity interaction between the Gads C-SH3 domain and the SLP-76 RxxK motif. EMBO J. 26, 678–689 (2007).
Kärkkäinen, S. et al. Identification of preferred protein interactions by phage-display of the human Src homology-3 proteome. EMBO Rep. 7, 186–191 (2006).
Smith, J. S., Lefkowitz, R. J. & Rajagopal, S. Biased signalling: from simple switches to allosteric microprocessors. Nat. Rev. Drug Discov. 17, 243–260 (2018).
Bodenhausen, G. & Ruben, D. J. Natural abundance nitrogen-15 NMR by enhanced heteronuclear spectroscopy. Chem. Phys. Lett. 69, 185–189 (1980).
Muhandiram, D. R. & Kay, L. E. Gradient-enhanced triple-resonance three-dimensional NMR experiments with improved sensitivity. J. Magn. Reson. B 103, 203–216 (1994).
Ikura, M., Kay, L. E. & Bax, A. A novel approach for sequential assignment of 1H, 13C, and 15N spectra of larger proteins: heteronuclear triple-resonance three-dimensional NMR spectroscopy. Application to calmodulin. Biochemistry 29, 4659–4667 (1990).
Wittekind, M. & Mueller, L. HNCACB, a high-sensitivity 3D NMR experiment to correlate amide-proton and nitrogen resonances with the alpha- and beta-carbon resonances in proteins. J. Magn. Reson. B 101, 201–205 (1993).
Grzesiekt, S. & Bax, A. Correlating backbone amide and side chain resonances in larger proteins by multiple relayed triple resonance NMR. J. Am. Chem. Soc. 114, 6291–6293 (1992).
Clubb, R. T., Thanabal, V. & Wagner, G. A constant-time three-dimensional triple-resonance pulse scheme to correlate intraresidue 1HN, 15N, and 13C′ chemical shifts in 15N13C-labelled proteins. J. Magn. Reson. 97, 213–217 (1992).
Williamson, M. P. Using chemical shift perturbation to characterise ligand binding. Prog. Nucl. Magn. Reson. Spectrosc. 73, 1–16 (2013).
Delaglio, F. et al. NMRPipe: a multidimensional spectral processing system based on UNIX pipes. J. Biomol. NMR 6, 277–93 (1995).
Kazimierczuk, K. & Orekhov, V. Y. Accelerated NMR spectroscopy by using compressed sensing. Angew. Chem. Int. Ed. 50, 5556–5559 (2011).
Orekhov, V. Y. & Jaravine, V. A. Analysis of non-uniformly sampled spectra with multi-dimensional decomposition. Prog. Nucl. Magn. Reson. Spectrosc. 59, 271–292 (2011).
Momin, A. A. et al. PYK2 senses calcium through a disordered dimerization and calmodulin-binding element. Commun. Biol. 5, 800 (2022).
Boulton, S., Akimoto, M., Selvaratnam, R., Bashiri, A. & Melacini, G. A tool set to map allosteric networks through the NMR chemical shift covariance analysis. Sci. Rep. 4, 7306 (2014).
Thureau, A., Roblin, P. & Pérez, J. BioSAXS on the SWING beamline at synchrotron SOLEIL. J. Appl. Crystallogr. 54, 1698–1710 (2021).
Manalastas-Cantos, K. et al. ATSAS 3.0: expanded functionality and new tools for small-angle scattering data analysis. J. Appl. Crystallogr. 54, 343–355 (2021).
Trnka, M. J., Baker, P. R., Robinson, P. J. J., Burlingame, A. L. & Chalkley, R. J. Matching cross-linked peptide spectra: only as good as the worse identification. Mol. Cell. Proteom. 13, (2014).
Evans, R. et al. Protein complex prediction with AlphaFold-multimer. Preprint at bioRxiv https://doi.org/10.1101/2021.10.04.463034 (2022).
Abramson, J. et al. Accurate structure prediction of biomolecular interactions with AlphaFold 3. Nature 630, 493–500 (2024).
Feng, S., Chen, J. K., Yu, H., Simon, J. A. & Schreiber, S. L. Two binding orientations for peptides to the Src SH3 domain: development of a general model for SH3-ligand interactions. Science 266, 1241–1247 (1994).
Sievers, F. & Higgins, D. G. Clustal Omega. Curr. Protoc. Bioinform. 2014, 3.13.1–3.13.16 (2014).
Robert, X. & Gouet, P. Deciphering key features in protein structures with the new ENDscript server. Nucleic Acids Res. 42, W320–W324 (2014).
Acknowledgements
The research reported in this publication was supported by King Abdullah University of Science and Technology (KAUST) through the baseline fund to S.T.A. and M.J. J.E.L. acknowledges support from the Cancer Research UK Grant C57233/A22356. L.W.C. was supported by the Hong Kong Research Grants Council (17122021, 17104022), and J.W.B. was supported by NCI 1P01CA257885. We acknowledge SOLEIL for provision of synchrotron radiation facilities and would like to thank P. Legrand, S. Sirigu, M. Savko and B. Shepard for assistance in using the beamlines PROXIMA 1 and PROXIMA 2 A (on crystalline material from p85 fragments), and Aurelien Thureau and Javier Perez for assistance using the beamline SWING. For computer time, this research used the resources of the KAUST Supercomputing Laboratory, and experimental research was supported by the Bioscience Core Lab, ACL Proteomics Lab and the Imaging and Characterisation Core Lab at King Abdullah University of Science & Technology (KAUST) in Thuwal, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
Conception of the project: S.T.A. Recombinant protein production: S.S.A., A.A., A.S., S.A., A.A.M., A.L., S.T.A.; Experimental data acquisition and analysis for ITC: S.S.A., A.A., A.S., A.A.M., S.A., S.T.A., J.E.L.; Experimental data acquisition and analysis for NMR: S.S.A., A.L., A.S., S.A., U.S., A.A.M., X.M., Ł.J., M.J., M.N.; XLMS: A.S., A.A.M.; DSF: A.S., A.A.; SAXS: A.S.; Activity assays: H.W., J.B., P.W., V.C.Y.M., L.W.T.C. AlphaFold modelling and analysis: S.T.A.; Contributed reagents, instrument time and lab facilities: J.B., Ł.J., M.J., S.T.A., J.B., X.M., L.W.T.C. Supervision: S.T.A., J.B., L.W.T.C., J.E.L., Ł.J., M.J., M.N., A.A.M., A.S., X.M. Writing of the initial manuscript: S.T.A. All authors read and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Tomonori Kaneko and the other anonymous reviewer(s) for their contribution to the peer review of this work. Primary handling editors: Laura Rodriguez Pérez. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Aljedani, S.S., Sandholu, A., Aldehaiman, A. et al. SH3 domains selectively activate the PI3 kinase through non-conventional tertiary contacts. Commun Biol (2026). https://doi.org/10.1038/s42003-026-09540-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-09540-y