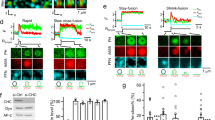
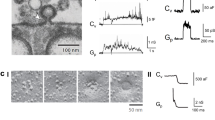
Abstract
For a long time, it remained an unresolved question how the fusion pore is regulated to close after fusion occurs during kiss-and-run. Our study focuses on the physiological function of NSF in modulating single vesicle fusion events in live cells, using whole-cell capacitance recording coupled with live-cell confocal imaging. Our results demonstrates that in both male Sprague-Dawley rat adrenal chromaffin endocrine cells and dorsal root ganglion neurons, inhibiting or diminishing the ATPase activity of NSF significantly impedes fusion pore closure, inhibits various forms of endocytosis, and disrupts the replenishment of readily releasable pool. Interestingly, NSF is exclusively required for calcium-dependent exo-endocytosis but not for the calcium-independent process. In response to calcium influx, NSF disassembles the trans-SNARE complex intermediate via its ATPase activity, which regulates fusion pore closure and subsequently mediates slow, fast, and overshoot endocytosis. Our findings revealed a key mechanism for fusion pore regulation in calcium-evoked exo-endocytosis coupling.
Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are included in the supplementary materials of this article. Original, uncropped, and unedited Western blot images are presented in Supplementary Figs. 7 and 8 within the Supplementary Information. Supplementary Data 1 provides a comprehensive list of nucleotide and peptide sequences, including all sgRNA, siRNAs, primers, and inhibitory peptide amino acid sequences. Supplementary Data 2 includes the numerical source data underlying the main and Supplementary Figs. Supplementary Data 3 contains the original Sanger sequencing trace files validating the genome editing results. Plasmids generated in this study have been deposited in Addgene with the following Addgene IDs: 252600, 252601, 252602, 252603, and 252604. Additional data supporting the findings of this study are available from the corresponding author upon reasonable request.
References
Rudolph, S., Tsai, M.-C., von Gersdorff, H. & Wadiche, J. I. The ubiquitous nature of multivesicular release. Trends Neurosci. 38, 428–438 (2015).
Wu, L.-G., Hamid, E., Shin, W. & Chiang, H.-C. Exocytosis and endocytosis: modes, functions, and coupling mechanisms. Annu. Rev. Physiol. 76, 301–331 (2014).
Saheki, Y. & De Camilli, P. Synaptic vesicle endocytosis. Cold Spring Harb. Perspect. Biol. 4, a005645 (2012).
Shin, W. et al. Preformed Ω-profile closure and kiss-and-run mediate endocytosis and diverse endocytic modes in neuroendocrine chromaffin cells. Neuron 109, 3119–3134 (2021).
Wu, L.-G. & Chan, C. Y. Membrane transformations of fusion and budding. Nat. Commun. 15, 21 (2024).
Shin, W. et al. Visualization of membrane pore in live cells reveals a dynamic-pore theory governing fusion and endocytosis. Cell 173, 934–945 (2018).
Chiang, H.-C. et al. Post-fusion structural changes and their roles in exocytosis and endocytosis of dense-core vesicles. Nat. Commun. 5, 3356 (2014).
Ge, L. et al. Sequential compound fusion and kiss-and-run mediate exo- and endocytosis in excitable cells. Sci. Adv. 8, eabm6049 (2022).
Fan, F. et al. Dynamin deficiency causes insulin secretion failure and hyperglycemia. Proc. Natl. Acad. Sci. USA 118, e2021764118 (2021).
Shin, W. et al. Molecular mechanics underlying flat-to-round membrane budding in live secretory cells. Nat. Commun. 13, 3697 (2022).
Zhao, M. & Brunger, A. T. Recent advances in deciphering the structure and molecular mechanism of the AAA+ ATPase N-ethylmaleimide-sensitive factor (NSF). J. Mol. Biol. 428, 1912–1926 (2016).
Zhao, C., Slevin, J. T. & Whiteheart, S. W. Cellular functions of NSF: not just SNAPs and SNAREs. FEBS Lett. 581, 2140–2149 (2007).
Graham, T. R. & Emr, S. D. Compartmental organization of golgi-specific protein modification and vacuolar protein sorting events defined in a yeast sec18 (NSF) mutant. J. Cell Biol. 114, 207–218 (1991).
Littleton, J. T. et al. Temperature-sensitive paralytic mutations demonstrate that synaptic exocytosis requires SNARE complex assembly and disassembly. Neuron 21, 401–413 (1998).
Matsushita, K. et al. Nitric oxide regulates exocytosis by S-nitrosylation of N-ethylmaleimide-sensitive factor. Cell 115, 139–150 (2003).
Matsushita, K. et al. Hydrogen peroxide regulation of endothelial exocytosis by inhibition of N-ethylmaleimide sensitive factor. J. Cell Biol. 170, 73–79 (2005).
Huynh, H. et al. Control of vesicle fusion by a tyrosine phosphatase. Nat. Cell Biol. 6, 831–839 (2004).
Morrell, C. N. et al. Regulation of platelet granule exocytosis by S-nitrosylation. Proc. Natl. Acad. Sci. USA 102, 3782–3787 (2005).
Michaut, M., Tomes, C. N., De Blas, G., Yunes, R. & Mayorga, L. S. Calcium-triggered acrosomal exocytosis in human spermatozoa requires the coordinated activation of Rab3A and N-ethylmaleimide-sensitive factor. Proc. Natl. Acad. Sci. USA 97, 9996–10001 (2000).
Kawasaki, F., Mattiuz, A. M. & Ordway, R. W. Synaptic physiology and ultrastructure in comatose mutants define an in vivo role for NSF in neurotransmitter release. J. Neurosci. 18, 10241–10249 (1998).
Littleton, J. T. et al. SNARE-complex disassembly by NSF follows synaptic-vesicle fusion. Proc. Natl. Acad. Sci. USA 98, 12233–12238 (2001).
Xu, Y.-F. et al. PTP-MEG2 regulates quantal size and fusion pore opening through two distinct structural bases and substrates. EMBO Rep. 22, e52141 (2021).
Das, D., Bao, H., Courtney, K. C., Wu, L. & Chapman, E. R. Resolving kinetic intermediates during the regulated assembly and disassembly of fusion pores. Nat. Commun. 11, 231 (2020).
Bao, H. et al. Dynamics and number of trans-SNARE complexes determine nascent fusion pore properties. Nature 554, 260–263 (2018).
Zhang, Q. et al. Differential co-release of two neurotransmitters from a vesicle fusion pore in mammalian adrenal chromaffin cells. Neuron 102, 173–183 (2019).
Glick, B. S. & Rothman, J. E. Possible role for fatty acyl-coenzyme A in intracellular protein transport. Nature 326, 309–312 (1987).
Jiang, Y. H. et al. Phenylarsine oxide can induce the arsenite-resistance mutant PML protein solubility changes. Int. J. Mol. Sci. 18, 247 (2017).
Haucke, V., Neher, E. & Sigrist, S. J. Protein scaffolds in the coupling of synaptic exocytosis and endocytosis. Nat. Rev. Neurosci. 12, 127–138 (2011).
Smith, C. & Neher, E. Multiple forms of endocytosis in bovine adrenal chromaffin cells. J. Cell Biol. 139, 885–894 (1997).
Wu, X.-S. et al. Actin is crucial for all kinetically distinguishable forms of endocytosis at synapses. Neuron 92, 1020–1035 (2016).
Zhang, C. & Zhou, Z. Ca2+-independent but voltage-dependent secretion in mammalian dorsal root ganglion neurons. Nat. Neurosci. 5, 425–430 (2002).
Zhang, C. et al. Calcium- and dynamin-independent endocytosis in dorsal root ganglion neurons. Neuron 42, 225–236 (2004).
Liu, T. et al. Two distinct vesicle pools for depolarization-induced exocytosis in somata of dorsal root ganglion neurons. J. Physiol. 589, 3507–3515 (2011).
Zheng, H. et al. Action potential modulates Ca2+-dependent and Ca2+-independent secretion in a sensory neuron. Biophys. J. 96, 2449–2456 (2009).
Lam, A. J. et al. Improving FRET dynamic range with bright green and red fluorescent proteins. Nat. Methods 9, 1005–1012 (2012).
Whiteheart, S. W. & Matveeva, E. A. Multiple binding proteins suggest diverse functions for the N-ethylmaleimide sensitive factor. J. Struct. Biol. 146, 32–43 (2004).
Henley, J. M. Proteins interactions implicated in AMPA receptor trafficking: a clear destination and an improving route map. Neurosci. Res. 45, 243–254 (2003).
Zhang, Q., Li, Y. & Tsien, R. W. The dynamic control of kiss-and-run and vesicular reuse probed with single nanoparticles. Science 323, 1448–1453 (2009).
Xu, T., Ashery, U., Burgoyne, R. D. & Neher, E. Early requirement for α-SNAP and NSF in the secretory cascade in chromaffin cells. EMBO J. 18, 3293–3304 (1999).
Barszczewski, M. et al. A novel site of action for α-SNAP in the SNARE conformational cycle controlling membrane fusion. Mol. Biol. Cell 19, 776–784 (2008).
Kasai, H., Takahashi, N. & Tokumaru, H. Distinct initial SNARE configurations underlying the diversity of exocytosis. Physiol. Rev. 92, 1915–1964 (2012).
Alabi, A. A. & Tsien, R. W. Perspectives on kiss-and-run: role in exocytosis, endocytosis, and neurotransmission. Annu. Rev. Physiol. 75, 393–422 (2013).
Chai, Z. et al. CaV2.2 gates calcium-independent but voltage-dependent secretion in mammalian sensory neurons. Neuron 96, 1317–1326 (2017).
Cong, M. et al. Binding of the beta2 adrenergic receptor to N-ethylmaleimide-sensitive factor regulates receptor recycling. J. Biol. Chem. 276, 45145–45152 (2001).
Wang, C. et al. Synaptotagmin-11 is a critical mediator of parkin-linked neurotoxicity and Parkinson’s disease-like pathology. Nat. Commun. 9, 81 (2018).
Huang, L.-Y. M. & Neher, E. Ca2+-dependent exocytosis in the somata of dorsal root ganglion neurons. Neuron 17, 135–145 (1996).
Lindau, M. & Neher, E. Patch-clamp techniques for time-resolved capacitance measurements in single cells. Pflug. Arch. 411, 137–146 (1988).
Guo, X. et al. Real-time visualization of exo- and endocytosis membrane dynamics with confocal and super-resolution microscopy. STAR Protoc. 3, 101404 (2022).
Verboogen, D. R. J., González Mancha, N., ter Beest, M. & van den Bogaart, G. Fluorescence lifetime imaging microscopy reveals rerouting of SNARE trafficking driving dendritic cell activation. eLife 6, e23525 (2017).
Wen, P. J. et al. Actin dynamics provides membrane tension to merge fusing vesicles into the plasma membrane. Nat. Commun. 7, 12604 (2016).
Acknowledgements
This work was supported by the Key Project of Gansu Province Science and Technology, China [Grant No. 22ZD6FA053]; National Natural Science Foundation of China [Grant No. 82073825]; Natural Science Foundation of Gansu Province, China [20JR10RA619] and Medical Innovation and Development Project of Lanzhou University [Grant No. lzuyxcx-2022-197].
Author information
Authors and Affiliations
Contributions
Y.W.: Writing - original draft, Formal analysis, Methodology. S.M.: Formal analysis, Methodology, Validation. S.S.: Writing - original draft, Formal analysis, Visualization. C.Z.: Investigation. Yu Wang: Methodology, Validation. B.L.: Resources. W.H.: Validation. H.L.: Writing - review & editing, Supervision. X.W.: Writing - review & editing, Methodology, Supervision. D.Z.: Writing - original draft, Writing - review & editing, Supervision, Project administration. D.W.: Writing - review & editing, Project administration, Funding acquisition, Conceptualization.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Neha Upmanyu, David Perrais and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Ophelia Bu. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, Y., Ma, S., Song, S. et al. ATPase N-ethylmaleimide-sensitive factor mediated calcium dependent fusion pore closure and endocytosis. Commun Biol (2026). https://doi.org/10.1038/s42003-026-09743-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-09743-3