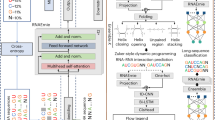
Abstract
The latent features of RNA sequences are crucial for our understanding of their functions. Thus, Transformer-based nucleotide language models have received widespread attention; however, the O(n²) complexity of Transformer limits their ability to process long sequences. In this work, we propose RNAret, an RNA language model based on Retention Network, which achieves training parallelism, low computational overhead, and long-sequence processing through a retention mechanism, with O(n) complexity. We pretrain RNAret using a self-supervised masked language modeling approach on 29.8 million RNA sequences. Experiments demonstrate the merit of RNAret as an RNA language model, achieving superior performance on a range of tasks, including RNA-RNA interaction prediction, RNA secondary structure prediction, and mRNA/lncRNA classification. RNAret shows strong potential for extracting latent features from RNA sequences and advancing our understanding of RNA biology.
Similar content being viewed by others
Data availability
All the datasets used for analyses in this work are publicly available online. The RNAcentral dataset is available at https://ftp.ebi.ac.uk/pub/databases/RNAcentral/releases/21.0/. Datasets for downstream fine-tuning tasks are available at https://bis.zju.edu.cn/rnaret/download/ and have been deposited in Zenodo (https://doi.org/10.5281/zenodo.18313475)54. Source data for figures are provided in Supplementary Data 1-3.
Code availability
The RNAret source code, including scripts for pretraining, training, and inference, is available on GitHub (https://github.com/DrBlackZJU/RNAret/) and archived on Zenodo (https://doi.org/10.5281/zenodo.18271233)55. The RNAret web server is accessible at https://bis.zju.edu.cn/rnaret/. Model weights are available at the project website (https://bis.zju.edu.cn/rnaret/download/) and have also been deposited on Zenodo (https://doi.org/10.5281/zenodo.18313475)54.
References
Caprara, M. G. & Nilsen, T. W. RNA: versatility in form and function. Nat. Struct. Biol. 7, 831–833 (2000).
Holbrook, S. R. RNA structure: the long and the short of it. Curr. Opin. Struct. Biol. 15, 302–308 (2005).
Chen, Z., Ain, N. U., Zhao, Q. & Zhang, X. From tradition to innovation: conventional and deep learning frameworks in genome annotation. Brief. Bioinform. 25, bbae138 (2024).
Vaswani, A. et al. Attention is all you need. In Adv. Neural Inf. Process. Syst. 30, 5998–6008 (NIPS, 2017).
Min, B. et al. Recent advances in natural language processing via large pre-trained language models: a survey. ACM Comput. Surv. 56, 30 (2023).
Chen, J. et al. Interpretable RNA foundation model from unannotated data for highly accurate RNA structure and function predictions. Preprint at https://arxiv.org/abs/2204.00300 (2022).
Zhang, Y. et al. Multiple sequence alignment-based RNA language model and its application to structural inference. Nucleic Acids Res 52, e3 (2024).
Wang, N. et al. Multi-purpose RNA language modelling with motif-aware pretraining and type-guided fine-tuning. Nat. Mach. Intell. 6, 548–557 (2024).
Shen, T. et al. Accurate RNA 3D structure prediction using a language model-based deep learning approach. Nat. Methods 21, 2287–2298 (2024).
Wang, X. et al. Uni-RNA: universal pre-trained models revolutionize RNA research. Preprint at https://www.biorxiv.org/content/10.1101/2023.07.11.548588v1 (2023).
Penić, R. J., Vlašić, T., Huber, R. G., Wan, Y. & Šikić, M. RiNALMo: general-purpose RNA language models can generalize well on structure prediction tasks. Nat. Commun. 16, 5671 (2025).
Dao, T., Fu, D., Ermon, S., Rudra, A. & Ré, C. FlashAttention: fast and memory-efficient exact attention with IO-Awareness. In Adv. Neural Inf. Process. Syst. 35, 16344–16359 (NIPS, 2022).
Dao, T. FlashAttention-2: faster attention with better parallelism and work partitioning. In International Conference on Learning Representations (ICLR, 2024).
Avsec, Ž et al. Effective gene expression prediction from sequence by integrating long-range interactions. Nat. Methods 18, 1196–1203 (2021).
Nguyen, E. et al. Sequence modeling and design from molecular to genome scale with Evo. Science 386, eado9336 (2024).
Sun, Y. et al. Retentive Network: a successor to Transformer for large language models. Preprint at https://arxiv.org/abs/2307.08621 (2023).
Sweeney, B. et al. RNAcentral 2021: secondary structure integration, improved sequence search and new member databases. Nucleic Acids Res. 49, D212–D220 (2021).
Kirk, J. M. et al. Functional classification of long non-coding RNAs by k-mer content. Nat. Genet. 50, 1474–1482 (2018).
van der Maaten, L. & Hinton, G. Visualizing Data using t-SNE. J. Mach. Learn. Res. 9, 2579–2605 (2008).
Wen, M., Cong, P., Zhang, Z., Lu, H. & Li, T. DeepMirTar: a deep-learning approach for predicting human miRNA targets. Bioinformatics 34, 3781–3787 (2018).
Pla, A., Zhong, X. & Rayner, S. miRAW: A deep learning-based approach to predict microRNA targets by analyzing whole microRNA transcripts. PLoS Comput. Biol. 14, e1006185 (2018).
Gu, T., Zhao, X., Barbazuk, W. B. & Lee, J. miTAR: a hybrid deep learning-based approach for predicting miRNA targets. BMC Bioinform 22, 96 (2021).
Akiyama, M. & Sakakibara, Y. Informative RNA base embedding for RNA structural alignment and clustering by deep representation learning. NAR Genom. Bioinform. 4, lqac12 (2022).
Bartel, D. P. MicroRNAs: target recognition and regulatory functions. Cell 136, 215–233 (2009).
Li, J., Liu, S., Zhou, H., Qu, L. & Yang, J. starBase v2.0: decoding miRNA-ceRNA, miRNA-ncRNA and protein-RNA interaction networks from large-scale CLIP-Seq data. Nucleic Acids Res, 42, D92–D97 (2014).
Tan, Z., Fu, Y., Sharma, G. & Mathews, D. H. TurboFold II: RNA structural alignment and secondary structure prediction informed by multiple homologs. Nucleic Acids Res. 45, 11570–11581 (2017).
Sloma, M. F. & Mathews, D. H. Exact calculation of loop formation probability identifies folding motifs in RNA secondary structures. RNA 22, 1808–1818 (2016).
Danaee, P. et al. bpRNA: large-scale automated annotation and analysis of RNA secondary structure. Nucleic Acids Res. 46, 5381–5394 (2018).
Szikszai, M., Wise, M., Datta, A., Ward, M. & Mathews, D. H. Deep learning models for RNA secondary structure prediction (probably) do not generalize across families. Bioinformatics 38, 3892–3899 (2022).
Fu, L. et al. UFold: fast and accurate RNA secondary structure prediction with deep learning. Nucleic Acids Res. 50, e14 (2022).
Gruber, A. R., Lorenz, R., Bernhart, S. H., Neuböck, R. & Hofacker, I. L. The Vienna RNA websuite. Nucleic Acids Res. 36, W70–W74 (2008).
Kerpedjiev, P., Hammer, S. & Hofacker, I. L. Forna (force-directed RNA): Simple and effective online RNA secondary structure diagrams. Bioinformatics 31, 3377–3379 (2015).
Zuker, M. & Stiegler, P. Optimal computer folding of large RNA sequences using thermodynamics and auxiliary information. Nucleic Acids Res 9, 133–148 (1981).
Frankish, A. et al. GENCODE 2021. Nucleic Acids Res. 49, D916–D923 (2021).
Kang, Y. et al. CPC2: a fast and accurate coding potential calculator based on sequence intrinsic features. Nucleic Acids Res. 45, W12–W16 (2017).
Wang, L. et al. CPAT: Coding-Potential Assessment Tool using an alignment-free logistic regression model. Nucleic Acids Res. 41, e74 (2013).
Subramanian, K., Payne, B., Feyertag, F. & Alvarez-Ponce, D. The codon statistics database: a database of codon usage bias. Mol. Biol. Evol. 39, msac157 (2022).
Dominguez, D. et al. Sequence, structure, and context preferences of human RNA binding proteins. Mol. Cell. 70, 854–867 (2018).
Ji, Y., Zhou, Z., Liu, H. & Davuluri, R. V. DNABERT: pre-trained Bidirectional Encoder Representations from Transformers model for DNA-language in genome. Bioinformatics 37, 2112–2120 (2021).
Ba, J. L., Kiros, J. R. & Hinton, G. E. Layer normalization. Preprint at https://arxiv.org/abs/1607.06450 (2016).
He, K., Zhang, X., Ren, S. & Sun, J. Deep residual learning for image recognition. In Proc. IEEE Conference on Computer Vision and Pattern Recognition 770–778 (IEEE, 2016).
Sun, Y. et al. A length-extrapolatable Transformer. In 61st Annual Meeting of the Association-for-Computational-Linguistics 14590-14604 (ACL, 2023)
Fan, Q., Huang, H., Chen, M., Liu, H. & He, R. RMT: Retentive Networks meet Vision Transformers. In IEEE Conference on Computer Vision and Pattern Recognition (CVPR, 2024).
Hendrycks, D. & Gimpel, K. Gaussian error linear units (GELUs). Preprint at https://arxiv.org/abs/1606.08415 (2016).
Kenton, J. & Toutanova, L. BERT: pre-training of deep bidirectional transformers for language understanding. In Proc. NAACL-HLT 2019 Vol. 1 (eds Burstein, J. et al.) 4171–4186 (Association for Computational Linguistics, 2019).
Loshchilov, I. & Hutter, F. Decoupled weight decay regularization. In International Conference on Learning Representations (ICLR, 2019).
Ning, W. CatIIIIIIII/RNAErnie: v.1.0. Zenodo https://doi.org/10.5281/zenodo.10847621 (2024).
Nowakowski, J. & Tinoco, I. RNA structure and stability. Semin. Virol. 8, 153–165 (1997).
Chen, X., Li, Y., Umarov, R., Gao, X. & Song, L. RNA secondary structure prediction by learning unrolled algorithms. In International Conference on Learning Representations (ICLR, 2020).
Kuhn, H. The Hungarian Method for the assignment problem. Nav. Res. Logist. 52, 7–21 (2005).
Ventola, G. M. M. et al. Identification of long non-coding transcripts with feature selection: a comparative study. BMC Bioinform. 18, 187 (2017).
Cock, P. J. A. et al. Biopython: freely available Python tools for computational molecular biology and bioinformatics. Bioinformatics 25, 1422–1423 (2009).
Pedregosa, F. et al. Scikit-learn: Machine Learning in Python. J. Mach. Learn. Res. 12, 2825–2830 (2011).
Shen, Y. RNAret - Datasets and Model Weights [Data set]. Zenodo https://doi.org/10.5281/zenodo.18313475 (2026).
Shen, Y. DrBlackZJU/RNAret: Retentive Network promotes efficient RNA language modeling of long sequences (v1.0). Zenodo https://doi.org/10.5281/zenodo.18271233 (2026).
Acknowledgements
This work was partially supported by the National Key Research and Development Program of China [2023YFE0112300]; National Science Foundation of China [32261133526; 32570787]; the Science and Technology Innovation Leading Scientist [2022R52035], the 151 talent project of Zhejiang Province (first level); and Collaborative Innovation Center for Modern Crop Production co-sponsored by the province and the ministry. The authors are grateful to the members of Ming Chen’s laboratory for helpful discussions and valuable comments, and to Jianghong Wu for assistance with computational resources.
Author information
Authors and Affiliations
Contributions
M.C. and D.C. supervised and designed the study. Y.S. designed the study, implemented the model, and performed data analysis with support from J.W. and Y.H. Y.S. wrote the manuscript with input from G.C. and Y.H. S.Z. helped to build up the web server. All authors reviewed and approved the submitted manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks the anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editors: Professor Maria Anisimova and Dr. Nilanjan Banerjee, Dr. Aylin Bircan, Dr. Kaliya Georgieva. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Shen, Y., Cao, G., Hu, Y. et al. Retentive Network promotes efficient RNA language modeling of long sequences. Commun Biol (2026). https://doi.org/10.1038/s42003-026-09757-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-09757-x