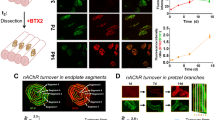
Abstract
The neuromuscular junction (NMJ) is the peripheral synapse controlling muscle contraction. Although aging and neurodegeneration result in NMJ denervation and synaptic dismantling, early indicators of this process remain elusive. Here, we analyzed the organization and dynamics of postsynaptic nicotinic acetylcholine receptors (nAChR) following muscle denervation. Using fluorescent conjugates of α-bungarotoxin (BTX), we found that loss of nAChR stability preceded morphological disintegration. Early after denervation, the combined use of receptor labeling and lectin staining revealed a rearrangement of long-lasting or newly inserted receptors that resulted in a novel compartmentalized postsynaptic pattern in which stable, pre-existing nAChRs concentrated centrally, while newly inserted, dynamic receptors localized peripherally. Small ectopic, highly dynamic nAChR clusters emerged since early denervation. Additionally, intracellular ring-like nAChR aggregates emerged since early denervation stages and were distributed in perinuclear regions, co-localizing with the lysosomal marker LAMP1, consistent with a degradative fate. Altogether, specific combinations of nAChR dynamics and morphologies serve as early markers of NMJ dismantling. These novel criteria to assess NMJ integrity may help define therapeutic windows to promote reinnervation in degenerative neuromuscular conditions.
Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files (Supplementary Data 1).
Code availability
No custom code was used in this study. Analyses were performed using standard software as described in the “Methods” section.
References
Sanes, J. R. & Lichtman, J. W. Induction, assembly, maturation and maintenance of a postsynaptic apparatus. Nat. Rev. Neurosci. 2, 791–805 (2001).
Zelada, D., Bermedo-Garcia, F., Collao, N. & Henriquez, J. P. Motor function recovery: deciphering a regenerative niche at the neuromuscular synapse. Biol. Rev. Camb. Philos. Soc. 96, 752–766 (2021).
Fatt, P. & Katz, B. The electric activity of the motor end-plate. Proc. R. Soc. Lond. B Biol. Sci. 140, 183–186 (1952).
Katz, B. Microphysiology of the neuromuscular junction; the chemo-receptor function of the motor end-plate. Bull. Johns. Hopkins Hosp. 102, 296–312 (1958).
Birks, R., Huxley, H. E. & Katz, B. The fine structure of the neuromuscular junction of the frog. J. Physiol. 150, 134–144 (1960).
Katz, B. Quantal mechanism of neural transmitter release. Science 173, 123–126 (1971).
Marques, M. J., Conchello, J. A. & Lichtman, J. W. From plaque to pretzel: fold formation and acetylcholine receptor loss at the developing neuromuscular junction. J. Neurosci. Off. J. Soc. Neurosci. 20, 3663–3675 (2000).
Balice-Gordon, R. J. & Lichtman, J. W. In vivo visualization of the growth of pre- and postsynaptic elements of neuromuscular junctions in the mouse. J. Neurosci. Off. J. Soc. Neurosci. 10, 894–908 (1990).
Kummer, T. T., Misgeld, T., Lichtman, J. W. & Sanes, J. R. Nerve-independent formation of a topologically complex postsynaptic apparatus. J. Cell Biol. 164, 1077–1087 (2004).
Tapia, J. C. et al. Pervasive synaptic branch removal in the mammalian neuromuscular system at birth. Neuron 74, 816–829 (2012).
Favero, M., Busetto, G. & Cangiano, A. Spike timing plays a key role in synapse elimination at the neuromuscular junction. Proc. Natl. Acad. Sci. USA 109, E1667–E1675 (2012).
Turney, S. G. & Lichtman, J. W. Reversing the outcome of synapse elimination at developing neuromuscular junctions in vivo: evidence for synaptic competition and its mechanism. PLoS Biol. 10, e1001352 (2012).
Valdez, G. et al. Attenuation of age-related changes in mouse neuromuscular synapses by caloric restriction and exercise. Proc. Natl. Acad. Sci. USA 107, 14863–14868 (2010).
Robbins, N. Compensatory plasticity of aging at the neuromuscular junction. Exp. Gerontol. 27, 75–81 (1992).
Ko, C. P. & Robitaille, R. Perisynaptic schwann cells at the neuromuscular synapse: adaptable, multitasking glial cells. Cold Spring Harb. Perspect. Biol. 7, a020503 (2015).
Sakuma, M. et al. Lack of motor recovery after prolonged denervation of the neuromuscular junction is not due to regenerative failure. Eur. J. Neurosci. 43, 451–462 (2016).
Bermedo-Garcia, F., Zelada, D., Martinez, E., Tabares, L. & Henriquez, J. P. Functional regeneration of the murine neuromuscular synapse relies on long-lasting morphological adaptations. BMC Biol. 20, 158 (2022).
Tremblay, E., Martineau, E. & Robitaille, R. Opposite synaptic alterations at the neuromuscular junction in an ALS mouse model: when motor units matter. J. Neurosci. Off. J. Soc. Neurosci. 37, 8901–8918 (2017).
Clark, J. A., Southam, K. A., Blizzard, C. A., King, A. E. & Dickson, T. C. Axonal degeneration, distal collateral branching and neuromuscular junction architecture alterations occur prior to symptom onset in the SOD1(G93A) mouse model of amyotrophic lateral sclerosis. J. Chem. Neuroanat. 76, 35–47 (2016).
Pratt, S. J. P., Valencia, A. P., Le, G. K., Shah, S. B. & Lovering, R. M. Pre- and postsynaptic changes in the neuromuscular junction in dystrophic mice. Front. Physiol. 6, 252 (2015).
van der Pijl, E. M. et al. Characterization of neuromuscular synapse function abnormalities in multiple Duchenne muscular dystrophy mouse models. Eur. J. Neurosci. 43, 1623–1635 (2016).
Murray, L. M. et al. Selective vulnerability of motor neurons and dissociation of pre- and post-synaptic pathology at the neuromuscular junction in mouse models of spinal muscular atrophy. Hum. Mol. Genet. 17, 949–962 (2008).
Ling, K. K., Gibbs, R. M., Feng, Z. & Ko, C. P. Severe neuromuscular denervation of clinically relevant muscles in a mouse model of spinal muscular atrophy. Hum. Mol. Genet. 21, 185–195 (2012).
Slater, C. R. ‘Fragmentation’ of NMJs: a sign of degeneration or regeneration? A long journey with many junctions. Neuroscience. https://doi.org/10.1016/j.neuroscience.2019.05.017 (2019).
Akaaboune, M., Culican, S. M., Turney, S. G. & Lichtman, J. W. Rapid and reversible effects of activity on acetylcholine receptor density at the neuromuscular junction in vivo. Science 286, 503–507 (1999).
Bruneau, E., Sutter, D., Hume, R. I. & Akaaboune, M. Identification of nicotinic acetylcholine receptor recycling and its role in maintaining receptor density at the neuromuscular junction in vivo. J. Neurosci. Off. J. Soc. Neurosci. 25, 9949–9959 (2005).
Khan, M. M. et al. Role of autophagy, SQSTM1, SH3GLB1, and TRIM63 in the turnover of nicotinic acetylcholine receptors. Autophagy 10, 123–136 (2014).
Wild, F., Khan, M. M., Straka, T. & Rudolf, R. Progress of endocytic CHRN to autophagic degradation is regulated by RAB5-GTPase and T145 phosphorylation of SH3GLB1 at mouse neuromuscular junctions in vivo. Autophagy 12, 2300–2310 (2016).
Andreose, J. S., Xu, R., Lomo, T., Salpeter, M. M. & Fumagalli, G. Degradation of two AChR populations at rat neuromuscular junctions: regulation in vivo by electrical stimulation. J. Neurosci. Off. J. Soc. Neurosci. 13, 3433–3438 (1993).
Ojeda, J. et al. The mouse levator auris longus muscle: an amenable model system to study the role of postsynaptic proteins to the maintenance and regeneration of the neuromuscular synapse. Front. Cell. Neurosci. 14, 225 (2020).
Zelada, D., Barrantes, F. J. & Henriquez, J. P. Lithium causes differential effects on postsynaptic stability in normal and denervated neuromuscular synapses. Sci. Rep. 11, 17285 (2021).
Roder, I. V. et al. Myosin Va cooperates with PKA RIalpha to mediate maintenance of the endplate in vivo. Proc. Natl. Acad. Sci. USA 107, 2031–2036 (2010).
Haddix, S. G., Lee, Y. I., Kornegay, J. N. & Thompson, W. J. Cycles of myofiber degeneration and regeneration lead to remodeling of the neuromuscular junction in two mammalian models of Duchenne muscular dystrophy. PloS One 13, e0205926 (2018).
Strack, S. et al. A novel labeling approach identifies three stability levels of acetylcholine receptors in the mouse neuromuscular junction in vivo. PloS One 6, e20524 (2011).
Jones, R. A. et al. NMJ-morph reveals principal components of synaptic morphology influencing structure-function relationships at the neuromuscular junction. Open Biol. https://doi.org/10.1098/rsob.160240 (2016).
Scott, L. J., Bacou, F. & Sanes, J. R. A synapse-specific carbohydrate at the neuromuscular junction: association with both acetylcholinesterase and a glycolipid. J. Neurosci. Off. J. Soc. Neurosci. 8, 932–944 (1988).
Martinez-Pena, Y. V. I., Aittaleb, M., Chen, P. J. & Akaaboune, M. The knockdown of alphakap alters the postsynaptic apparatus of neuromuscular junctions in living mice. J. Neurosci. Off. J. Soc. Neurosci. 35, 5118–5127 (2015).
Bruneau, E. G. & Akaaboune, M. The dynamics of recycled acetylcholine receptors at the neuromuscular junction in vivo. Development 133, 4485–4493 (2006).
Medina-Moreno, A. & Henriquez, J. P. Maturation of a postsynaptic domain: Role of small Rho GTPases in organising nicotinic acetylcholine receptor aggregates at the vertebrate neuromuscular junction. J. Anat. 241, 1148–1156 (2022).
Apel, E. D., Lewis, R. M., Grady, R. M. & Sanes, J. R. Syne-1, a dystrophin- and Klarsicht-related protein associated with synaptic nuclei at the neuromuscular junction. J. Biol. Chem. 275, 31986–31995 (2000).
Grady, R. M., Starr, D. A., Ackerman, G. L., Sanes, J. R. & Han, M. Syne proteins anchor muscle nuclei at the neuromuscular junction. Proc. Natl. Acad. Sci. USA 102, 4359–4364 (2005).
Liu, W., Wei-LaPierre, L., Klose, A., Dirksen, R. T. & Chakkalakal, J. V. Inducible depletion of adult skeletal muscle stem cells impairs the regeneration of neuromuscular junctions. eLife. https://doi.org/10.7554/eLife.09221 (2015).
Murphy, M. M., Lawson, J. A., Mathew, S. J., Hutcheson, D. A. & Kardon, G. Satellite cells, connective tissue fibroblasts and their interactions are crucial for muscle regeneration. Development 138, 3625–3637 (2011).
Muzumdar, M. D., Tasic, B., Miyamichi, K., Li, L. & Luo, L. A global double-fluorescent Cre reporter mouse. Genesis 45, 593–605 (2007).
Sanes, J. R. & Cheney, J. M. Lectin binding reveals a synapse-specific carbohydrate in skeletal muscle. Nature 300, 646–647 (1982).
Martinez-Pena, Y. V. I. & Akaaboune, M. The metabolic stability of the nicotinic acetylcholine receptor at the neuromuscular junction. Cells. https://doi.org/10.3390/cells10020358 (2021).
Magill, C. K. et al. Reinnervation of the tibialis anterior following sciatic nerve crush injury: a confocal microscopic study in transgenic mice. Exp. Neurol. 207, 64–74 (2007).
Levitt, T. A. & Salpeter, M. M. Denervated endplates have a dual population of junctional acetylcholine receptors. Nature 291, 239–241 (1981).
Salpeter, M. M., Cooper, D. L. & Levitt-Gilmour, T. Degradation rates of acetylcholine receptors can be modified in the postjunctional plasma membrane of the vertebrate neuromuscular junction. J. Cell Biol. 103, 1399–1403 (1986).
Strack, S., Khan, M. M., Wild, F., Rall, A. & Rudolf, R. Turnover of acetylcholine receptors at the endplate revisited: novel insights into nerve-dependent behavior. J. Muscle Res. Cell Motil. 36, 517–524 (2015).
Kumari, S. et al. Nicotinic acetylcholine receptor is internalized via a Rac-dependent, dynamin-independent endocytic pathway. J. Cell Biol. 181, 1179–1193 (2008).
Green, W. N. & Claudio, T. Acetylcholine receptor assembly: subunit folding and oligomerization occur sequentially. Cell 74, 57–69 (1993).
Green, W. N. & Wanamaker, C. P. Formation of the nicotinic acetylcholine receptor binding sites. J. Neurosci. Off. J. Soc. Neurosci. 18, 5555–5564 (1998).
Mitra, M., Wanamaker, C. P. & Green, W. N. Rearrangement of nicotinic receptor alpha subunits during formation of the ligand binding sites. J. Neurosci. Off. J. Soc. Neurosci. 21, 3000–3008 (2001).
Smith, M. M., Lindstrom, J. & Merlie, J. P. Formation of the alpha-bungarotoxin binding site and assembly of the nicotinic acetylcholine receptor subunits occur in the endoplasmic reticulum. J. Biol. Chem. 262, 4367–4376 (1987).
Wild, F., Khan, M. M. & Rudolf, R. Evidence for the subsynaptic zone as a preferential site for CHRN recycling at neuromuscular junctions. Small GTPases 10, 395–402 (2019).
Bagri, K. M. et al. Lysosomes accumulate at the perinuclear region of muscle cells during chick myogenesis. Cell Biol. Int. 48, 1625–1636 (2024).
Vainshtein, A., Desjardins, E. M., Armani, A., Sandri, M. & Hood, D. A. PGC-1alpha modulates denervation-induced mitophagy in skeletal muscle. Skelet. Muscle 5, 9 (2015).
Libelius, R., Lundquist, I., Tagerud, S. & Thesleff, S. Endocytosis and lysosomal enzyme activities in dystrophic muscle: the effect of denervation. Acta Physiol. Scand. 113, 259–261 (1981).
Triolo, M., Bhattacharya, D. & Hood, D. A. Denervation induces mitochondrial decline and exacerbates lysosome dysfunction in middle-aged mice. Aging 14, 8900–8913 (2022).
Tagerud, S. & Libelius, R. Lysosomes in skeletal muscle following denervation. Time course of horseradish peroxidase uptake and increase of lysosomal enzymes. Cell Tissue Res. 236, 73–79 (1984).
Triolo, M., Oliveira, A. N., Kumari, R. & Hood, D. A. The influence of age, sex, and exercise on autophagy, mitophagy, and lysosome biogenesis in skeletal muscle. Skelet. Muscle 12, 13 (2022).
Cantor, S. et al. Preserving neuromuscular synapses in ALS by stimulating MuSK with a therapeutic agonist antibody. eLife. https://doi.org/10.7554/eLife.34375 (2018).
Xu, R. & Salpeter, M. M. Acetylcholine receptors in innervated muscles of dystrophic mdx mice degrade as after denervation. J. Neurosci. Off. J. Soc. Neurosci. 17, 8194–8200 (1997).
Willadt, S., Nash, M. & Slater, C. R. Age-related fragmentation of the motor endplate is not associated with impaired neuromuscular transmission in the mouse diaphragm. Sci. Rep. 6, 24849 (2016).
Jones, R. A. et al. Cellular and molecular anatomy of the human neuromuscular junction. Cell Rep. 21, 2348–2356 (2017).
Boehm, I. et al. Comparative anatomy of the mammalian neuromuscular junction. J. Anat. 237, 827–836 (2020).
Martinez-Pena y Valenzuela, I., Pires-Oliveira, M. & Akaaboune, M. PKC and PKA regulate AChR dynamics at the neuromuscular junction of living mice. PloS One 8, e81311 (2013).
Roder, I. V. et al. Participation of myosin Va and Pka type I in the regeneration of neuromuscular junctions. PloS One 7, e40860 (2012).
Kang, H., Tian, L. & Thompson, W. J. Schwann cell guidance of nerve growth between synaptic sites explains changes in the pattern of muscle innervation and remodeling of synaptic sites following peripheral nerve injuries. J. Comp. Neurol. 527, 1388–1400 (2019).
Liu, W. et al. Loss of adult skeletal muscle stem cells drives age-related neuromuscular junction degeneration. eLife 6, e26464 (2017).
Love, F. M. & Thompson, W. J. Glial cells promote muscle reinnervation by responding to activity-dependent postsynaptic signals. J. Neurosci. Off. J. Soc. Neurosci. 19, 10390–10396 (1999).
Ham, A. S. et al. Single-nuclei sequencing of skeletal muscle reveals subsynaptic-specific transcripts involved in neuromuscular junction maintenance. Nat. Commun. 16, 2220 (2025).
Acknowledgements
We thank the highly collaborative and stimulating environment of the NeSt Lab members for their useful discussion and comments on this work. Our research has been supported by research grants FONDECYT 1170614, 1221213 to JPH and 3240731 to JM, AFM-Telethon 24878, and FONDECYT 1241240 to HO. DZ, FB-G, and JM have been ANID fellows.
Author information
Authors and Affiliations
Contributions
D.Z. designed the work, performed experiments and quantifications, analyzed, and interpreted data, and was a major contributor in writing the manuscript. F.B.-G. performed experiments, quantifications, and contributed to writing the manuscript. J.M. performed quantifications. H.O. assisted with experiments and contributed to writing the manuscript. J.P.H. designed the work, analyzed, and interpreted data, and was a major contributor in writing the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Mohammed Akaaboune and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editors: Ivo Lieberam and Benjamin Bessieres. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zelada, D., Bermedo-García, F., Mella, J. et al. Early postsynaptic instability and acetylcholine receptor compartmentalization precede neuromuscular synapse dismantling. Commun Biol (2026). https://doi.org/10.1038/s42003-026-09816-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-09816-3