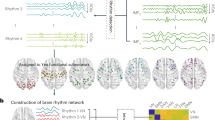
Abstract
Human social behaviors involve complex interactions between individuals, and understanding how interbrain neural activity reflects and predicts these interactions is critical for advancing social cognitive neuroscience. While electroencephalography (EEG) hyperscanning has been widely used to explore interpersonal neural dynamics, most studies focus on pairwise regional coupling, overlooking the brain’s intrinsic network-level organization. Here, we propose a spatiotemporal network analysis framework that combines Bayesian non-negative matrix factorization with EEG source imaging to identify interpretable subnetworks with spatiotemporal information. Applying this framework to dyadic EEG datasets from interactive decision-making tasks identifies eight task-relevant subnetworks, including the default mode network (DMN), somatosensory-motor network (SMN), and visual network (VN). Effective interpersonal coordination was associated with enhanced network-level time-domain interbrain synchrony and spatial-domain inter-subject similarity, and the fusion of these metrics reliably predicted interactive behaviors. Notably, synchrony and similarity involving DMN, VN, and SMN emerge as robust predictors of interactive behaviors, with spatiotemporal coupling most prominent within these subnetworks. These findings reveal spatiotemporal network signatures underlying interpersonal neural synchronization and demonstrate the importance of distributed subnetworks and their temporal and spatial alignment in achieving effective social interactions. This framework provides a useful computational tool for probing the neurobiological basis of social behaviors.
Similar content being viewed by others
Data availability
The source data used to generate all figures in this paper are provided in Supplementary Data. The raw data of this study are available from the corresponding author upon reasonable request.
Code availability
EEG data were pre-processed using EEGLAB_v2020.0 (https://sccn.ucsd.edu/eeglab/index.php) and REST_v1.2_20200818 (https://www.neuro.uestc.edu.cn/name/shopwap/do/index/content/96). Bayesian non-negative matrix factorization, Interbrain phase-locking value and network properties computation, and statistical procedures—including correlation analyses, one-way repeated-measures ANOVA, and t-tests—were conducted in MATLAB R2018b (https://jp.mathworks.com/products/matlab.html). Spatial distributions of subnetworks were rendered using LORETA v20170220 (https://www.uzh.ch/keyinst/loreta). Graphical representations of correlation results, interbrain synchrony connectivity, and inter-subject similarity were produced using MATLAB R2018b, Origin 2021 (https://www.originlab.com/), and GraphPad Prism 8.3 (https://www.graphpad.com/).
References
Boothby, E. J., Cooney, G. & Schweitzer, M. E. Embracing complexity: a review of negotiation research. Annu. Rev. Psychol. 74, 299–332 (2023).
Zhang, L. & Gläscher, J. A brain network supporting social influences in human decision-making. Sci. Adv. 6, eabb4159 (2020).
Yi, C. et al. Constructing large-scale cortical brain networks from scalp EEG with Bayesian nonnegative matrix factorization. Neural Netw. 125, 338–348 (2020).
Luft, C. D. B. et al. Social synchronization of brain activity increases during eye-contact. Commun. Biol. 5, 412 (2022).
Schippers, M. B., Roebroeck, A., Renken, R., Nanetti, L. & Keysers, C. Mapping the information flow from one brain to another during gestural communication. Proc. Natl. Acad. Sci. USA. 107, 9388–9393 (2010).
De Dreu, C. K. W., Gross, J., Fariña, A. & Ma, Y. Group cooperation, carrying-capacity stress, and intergroup conflict. Trends Cogn. Sci. 24, 760–776 (2020).
Czeszumski, A. et al. Hyperscanning: a valid method to study neural inter-brain underpinnings of social interaction. Front. Hum. Neurosci. 14, 39 (2020).
Schilbach, L. et al. Toward a second-person neuroscience. Behav. Brain Sci. 36, 393–414 (2013).
Valencia, A. L. & Froese, T. What binds us? Inter-brain neural synchronization and its implications for theories of human consciousness. Neurosci. Conscious. 2020, niaa010 (2020).
Kingsbury, L. et al. Correlated neural activity and encoding of behavior across brains of socially interacting animals. Cell 178, 429–446.e416 (2019).
Osaka, N. et al. How two brains make one synchronized mind in the inferior frontal cortex: fNIRS-based hyperscanning during cooperative singing. Front. Psychol. 6, 1811 (2015).
Goldstein, P., Weissman-Fogel, I., Dumas, G. & Shamay-Tsoory, S. G. Brain-to-brain coupling during handholding is associated with pain reduction. Proc. Natl. Acad. Sci. USA. 115, E2528–e2537 (2018).
Leong, V. et al. Speaker gaze increases information coupling between infant and adult brains. Proc. Natl. Acad. Sci. USA. 114, 13290–13295 (2017).
Gvirts, H. Z. & Perlmutter, R. What guides us to neurally and behaviorally align with anyone specific? A neurobiological model based on fNIRS hyperscanning studies. Neuroscientist 26, 108–116 (2020).
Wass, S. V., Whitehorn, M., Marriott Haresign, I., Phillips, E. & Leong, V. Interpersonal neural entrainment during early social interaction. Trends Cogn. Sci. 24, 329–342 (2020).
Dikker, S. et al. Brain-to-brain synchrony tracks real-world dynamic group interactions in the classroom. Curr. Biol. 27, 1375–1380 (2017).
Liu, H., Zhao, C., Wang, F. & Zhang, D. Inter-brain amplitude correlation differentiates cooperation from competition in a motion-sensing sports game. Soc. Cogn. Affect. Neurosci. 16, 552–564 (2021).
Sporns, O., Chialvo, D. R., Kaiser, M. & Hilgetag, C. C. Organization, development and function of complex brain networks. Trends Cogn. Sci. 8, 418–425 (2004).
Wang, M. Y. et al. Concurrent mapping of brain activation from multiple subjects during social interaction by hyperscanning: a mini-review. Quant. Imaging Med. Surg. 8, 819–837 (2018).
Martin-Raugh, M. P. et al. Negotiation as an interpersonal skill: generalizability of negotiation outcomes and tactics across contexts at the individual and collective levels. Comput. Hum. Behav. 104, 105966 (2020).
Bressler, S. L. & Menon, V. Large-scale brain networks in cognition: emerging methods and principles. Trends Cogn. Sci. 14, 277–290 (2010).
Feng, C. et al. Common brain networks underlying human social interactions: evidence from large-scale neuroimaging meta-analysis. Neurosci. Biobehav. Rev. 126, 289–303 (2021).
Cole, M. W. et al. Multi-task connectivity reveals flexible hubs for adaptive task control. Nat. Neurosci. 16, 1348–1355 (2013).
Menon, V. Large-scale brain networks and psychopathology: a unifying triple network model. Trends Cogn. Sci. 15, 483–506 (2011).
Sockeel, S., Schwartz, D., Pélégrini-Issac, M. & Benali, H. Large-scale functional networks identified from resting-state EEG using spatial ICA. PloS one 11, e0146845 (2016).
Li, F. et al. The construction of large-scale cortical networks for P300 from scalp EEG. IEEE Access 6, 68498–68506 (2018).
Engel, A. K., Gerloff, C., Hilgetag, C. C. & Nolte, G. Intrinsic coupling modes: multiscale interactions in ongoing brain activity. Neuron 80, 867–886 (2013).
Townsend, R. G. & Gong, P. Detection and analysis of spatiotemporal patterns in brain activity. PLoS Comput. Biol. 14, e1006643 (2018).
Forbes, C. E. On the neural networks of self and other bias and their role in emergent social interactions. Cortex 177, 113–129 (2024).
Ronde, M., van der Zee, E. A. & Kas, M. J. H. Default mode network dynamics: an integrated neurocircuitry perspective on social dysfunction in human brain disorders. Neurosci. Biobehav. Rev. 164, 105839 (2024).
Power, J. D. et al. Functional network organization of the human brain. Neuron 72, 665–678 (2011).
Yan, X. et al. Homotopic local-global parcellation of the human cerebral cortex from resting-state functional connectivity. NeuroImage 273, 120010 (2023).
Yeo, B. T. et al. The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J. Neurophysiol. 106, 1125–1165 (2011).
Ni, J., Yang, J. & Ma, Y. Social bonding in groups of humans selectively increases inter-status information exchange and prefrontal neural synchronization. PLoS Biol. 22, e3002545 (2024).
Zhao, H., Zhang, C., Tao, R., Duan, H. & Xu, S. Distinct inter-brain synchronization patterns underlying group decision-making under uncertainty with partners in different interpersonal relationships. NeuroImage 272, 120043 (2023).
Endevelt-Shapira, Y., Djalovski, A., Dumas, G. & Feldman, R. Maternal chemosignals enhance infant-adult brain-to-brain synchrony. Sci. Adv. 7, eabg6867 (2021).
Balters, S., Heaton, S. & Reiss, A. L. in Design Thinking Research: Innovation–Insight–Then and Now (eds Meinel, C. & Leifer, L.) 213–225 (Springer Nature Switzerland, 2023).
Guo, X. et al. Increased neural responses to unfairness in a loss context. Neuroimage 77, 246–253 (2013).
Güroğlu, B., van den Bos, W., van Dijk, E., Rombouts, S. A. & Crone, E. A. Dissociable brain networks involved in development of fairness considerations: understanding intentionality behind unfairness. Neuroimage 57, 634–641 (2011).
Khani, A. Partially dissociable roles of OFC and ACC in stimulus-guided and action-guided decision making. J. Neurophysiol. 111, 1717–1720 (2014).
Plassmann, H., O’Doherty, J. P. & Rangel, A. Appetitive and aversive goal values are encoded in the medial orbitofrontal cortex at the time of decision making. J. Neurosci. 30, 10799–10808 (2010).
Wei, C. et al. Social support modulates neural responses to unfairness in the ultimatum game. Front. Psychol. 9, 182 (2018).
Minati, L., Grisoli, M., Seth, A. K. & Critchley, H. D. Decision-making under risk: a graph-based network analysis using functional MRI. Neuroimage 60, 2191–2205 (2012).
Blakemore, S. J. The social brain in adolescence. Nat. Rev. Neurosci. 9, 267–277 (2008).
Vaziri-Pashkam, M. & Xu, Y. Goal-directed visual processing differentially impacts human ventral and dorsal visual representations. J. Neurosci. 37, 8767–8782 (2017).
Xu, Y. A tale of two visual systems: invariant and adaptive visual information representations in the primate brain. Annu. Rev. Vis. Sci. 4, 311–336 (2018).
Qin, P. & Northoff, G. How is our self related to midline regions and the default-mode network? NeuroImage 57, 1221–1233 (2011).
Qin, P. et al. Dissociation between anterior and posterior cortical regions during self-specificity and familiarity: a combined fMRI–meta-analytic study. Hum. Brain Mapp. 33, 154–164 (2012).
Harvey, P.-O., Lee, J., Horan, W. P., Ochsner, K. & Green, M. F. Do patients with schizophrenia benefit from a self-referential memory bias? Schizophrenia Res. 127, 171–177 (2011).
Fuentes-Claramonte, P. et al. Shared and differential default-mode related patterns of activity in an autobiographical, a self-referential and an attentional task. PLoS ONE 14, e0209376 (2019).
Coutinho, J. F. et al. Default mode network dissociation in depressive and anxiety states. Brain Imaging Behav. 10, 147–157 (2016).
D’argembeau, A. et al. Self-referential reflective activity and its relationship with rest: a PET study. Neuroimage 25, 616–624 (2005).
Reniers, R. L. et al. Moral decision-making, ToM, empathy and the default mode network. Biol. Psychol. 90, 202–210 (2012).
Boncompagni, I. & Casagrande, M. Executive control of emotional conflict. Front. Psychol. 10, 359 (2019).
Lima, C. F., Krishnan, S. & Scott, S. K. Roles of supplementary motor areas in auditory processing and auditory imagery. Trends Neurosci. 39, 527–542 (2016).
Gabay, A. S., Radua, J., Kempton, M. J. & Mehta, M. A. The Ultimatum Game and the brain: a meta-analysis of neuroimaging studies. Neurosci. Biobehav. Rev. 47, 549–558 (2014).
Alcalá-López, D. et al. Computing the social brain connectome across systems and states. Cereb. Cortex 28, 2207–2232 (2018).
Wang, Y. & Olson, I. R. The original social network: white matter and social cognition. Trends Cogn. Sci. 22, 504–516 (2018).
Buckner, R. L. & DiNicola, L. M. The brain’s default network: updated anatomy, physiology and evolving insights. Nat. Rev. Neurosci. 20, 593–608 (2019).
Corradi-Dell’Acqua, C., Civai, C., Rumiati, R. I. & Fink, G. R. Disentangling self-and fairness-related neural mechanisms involved in the ultimatum game: an fMRI study. Soc. Cogn. Affect. Neurosci. 8, 424–431 (2012).
Yamagishi, T. et al. Rejection of unfair offers in the ultimatum game is no evidence of strong reciprocity. Proc. Natl. Acad. Sci. USA. 109, 20364–20368 (2012).
van Schie, C. C., Chiu, C.-D., Rombouts, S. A., Heiser, W. J. & Elzinga, B. M. When compliments do not hit but critiques do: an fMRI study into self-esteem and self-knowledge in processing social feedback. Soc. Cogn. Affect. Neurosci. 13, 404–417 (2018).
Kong, X. et al. Sensory-motor cortices shape functional connectivity dynamics in the human brain. Nat. Commun. 12, 6373 (2021).
Bhatt, M. A., Lohrenz, T., Camerer, C. F. & Montague, P. R. Neural signatures of strategic types in a two-person bargaining game. Proc. Natl. Acad. Sci. USA. 107, 19720–19725 (2010).
Samson, D., Apperly, I. A., Kathirgamanathan, U. & Humphreys, G. W. Seeing it my way: a case of a selective deficit in inhibiting self-perspective. Brain 128, 1102–1111 (2005).
Li, R., Mayseless, N., Balters, S. & Reiss, A. L. Dynamic inter-brain synchrony in real-life inter-personal cooperation: a functional near-infrared spectroscopy hyperscanning study. NeuroImage 238, 118263 (2021).
Toppi, J. et al. In 2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC) 2211–2214 (2015).
Gao, J. et al. Interbrain synchronization in classroom during high-entropy music listening and meditation: a hyperscanning EEG study. Front. Neurosci. 19, 1557904 (2025).
Arnal, L. H. & Giraud, A.-L. Cortical oscillations and sensory predictions. Trends Cogn. Sci. 16, 390–398 (2012).
Müller, R. A. & Fishman, I. Brain connectivity and neuroimaging of social networks in autism. Trends Cogn. Sci. 22, 1103–1116 (2018).
Dikker, S. et al. Crowdsourcing neuroscience: inter-brain coupling during face-to-face interactions outside the laboratory. NeuroImage 227, 117436 (2021).
Krueger, F., Barbey, A. K. & Grafman, J. The medial prefrontal cortex mediates social event knowledge. Trends Cogn. Sci. 13, 103–109 (2009).
Oberwelland, E. et al. Look into my eyes: investigating joint attention using interactive eye-tracking and fMRI in a developmental sample. NeuroImage 130, 248–260 (2016).
Fareri, D. S., Niznikiewicz, M. A., Lee, V. K. & Delgado, M. R. Social network modulation of reward-related signals. J. Neurosci. 32, 9045–9052 (2012).
Lu, K., Teng, J. & Hao, N. Gender of partner affects the interaction pattern during group creative idea generation. Exp. Brain Res. 238, 1157–1168 (2020).
Chen, M. et al. Neural alignment during face-to-face spontaneous deception: does gender make a difference? Hum. Brain Mapp. 41, 4964–4981 (2020).
Rotter, J. B. A new scale for the measurement of interpersonal trust. J Pers. 35, 651–665 (1967).
Gouveia, V. V., Athayde, R., Gouveia, R., Gomes, A. I. A. S. D. B. & Souza, R. Self-report Altruism Scale: evidences of construct validity. Aletheia, 30–44 (2010).
Yao, D. A method to standardize a reference of scalp EEG recordings to a point at infinity. Physiol. Meas. 22, 693–711 (2001).
He, T., Clifford, G. & Tarassenko, L. Application of independent component analysis in removing artefacts from the electrocardiogram. Neural Comput. Appl. 15, 105–116 (2006).
Winkler, I., Haufe, S. & Tangermann, M. Automatic classification of artifactual ICA-components for artifact removal in EEG signals. Behav. Brain Funct. 7, 30 (2011).
Pion-Tonachini, L., Kreutz-Delgado, K. & Makeig, S. ICLabel: an automated electroencephalographic independent component classifier, dataset, and website. NeuroImage 198, 181–197 (2019).
Kim, H., Kim, S., Jun, S. C. & Nam, C. S. Is what I think what you think? Multilayer network-based inter-brain synchrony approach. Soc. Cogn. Affect. Neurosci. https://doi.org/10.1093/scan/nsaf028 (2025).
Schwartz, L. et al. Technologically-assisted communication attenuates inter-brain synchrony. NeuroImage 264, 119677 (2022).
Koul, A., Ahmar, D., Iannetti, G. D. & Novembre, G. Spontaneous dyadic behavior predicts the emergence of interpersonal neural synchrony. NeuroImage 277, 120233 (2023).
Pascual-Marqui, R. D. Standardized low-resolution brain electromagnetic tomography (sLORETA): technical details. Methods Find. Exp. Clin. Pharm. 24, 5–12 (2002).
Schmidt, M. N., Winther, O. & Hansen, L. K. In International Conference on Independent Component Analysis and Signal Separation. 540–547 (Springer, 2009).
Calhoun, V. D., Adali, T., Pearlson, G. D. & Pekar, J. J. Spatial and temporal independent component analysis of functional MRI data containing a pair of task-related waveforms. Hum. Brain Mapp. 13, 43–53 (2001).
Burgess, A. P. On the interpretation of synchronization in EEG hyperscanning studies: a cautionary note. Front. Hum. Neurosci. 7, 881 (2013).
Deng, X., Lin, M., Zhang, L., Li, X. & Gao, Q. Relations between family cohesion and adolescent-parent’s neural synchrony in response to emotional stimulations. Behav. Brain Funct. 18, 11 (2022).
Dumas, G., Nadel, J., Soussignan, R., Martinerie, J. & Garnero, L. Inter-brain synchronization during social interaction. PloS one 5, e12166 (2010).
Gugnowska, K. et al. Endogenous sources of interbrain synchrony in duetting pianists. Cereb. Cortex 32, 4110–4127 (2022).
Jahng, J., Kralik, J. D., Hwang, D. U. & Jeong, J. Neural dynamics of two players when using nonverbal cues to gauge intentions to cooperate during the Prisoner’s Dilemma Game. NeuroImage 157, 263–274 (2017).
Rubinov, M. & Sporns, O. Complex network measures of brain connectivity: uses and interpretations. NeuroImage 52, 1059–1069 (2010).
Xiang-Wei, H., Ming-An, S. & Horton, R. Estimating van Genuchten model parameters of undisturbed soils using an integral method. Pedosphere 20, 55–62 (2010).
Leys, C., Klein, O., Dominicy, Y. & Ley, C. Detecting multivariate outliers: use a robust variant of the Mahalanobis distance. J. Exp. Soc. Psychol. 74, 150–156 (2018).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (#W2411084, #82372084), the Lingang Laboratory (#LG-TKN-202205-01), the Key R&D projects of the Science & Technology Department of Chengdu (#2024-YF08-00072-GX), the STI 2030-Major Projects (#2022ZD0208500), the AI Program of Shanghai Municipal Education Commission (#JWAIZD-4), and the Postdoctoral Fellowship Program of CPSF (#GZC20240211).
Author information
Authors and Affiliations
Contributions
Yuqin Li: conceptualization, data curation, methodology, software, formal analysis, investigation, validation, visualization, writing—original draft, writing—review and editing; Senqi Li: data curation, methodology; Yanggong Li: data curation; Xiaoya Pang: investigation; Chanlin Yi: methodology; Lin Jiang: software; Dezhong Yao: supervision; Wei Wu: writing—review and editing; Fali Li: conceptualization, investigation, writing—review and editing; Peng Xu: methodology, funding acquisition, project administration, resources, supervision, writing—review and editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Takahiko Koike and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Jasmine Pan.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, Y., Li, S., Li, Y. et al. Temporal synchrony and spatial similarity of interbrain subnetworks predict dyadic social interaction. Commun Biol (2026). https://doi.org/10.1038/s42003-026-09854-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-09854-x