Abstract
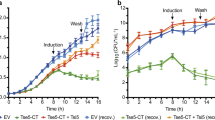
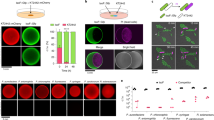
The soil bacterium Pseudomonas putida injects toxic proteins into neighbouring competitors, including resilient phytopathogens, using the Type VI secretion system (T6SS). The secretion of toxins endows P. putida with a significant fitness advantage, allowing this biocontrol agent to thrive in plant-related polymicrobial environments and prevent phytopathogen infections. Despite its agricultural significance, the toxin repertoire of P. putida, particularly those secreted via the K2- and K3-T6SSs, remains poorly understood. We present a comprehensive molecular study of Tke5, a potent toxin encoded within the K3-T6SS, which represents the initial biophysical and functional analysis of the BTH_I2691 family. Our data demonstrate that Tke5 is a pore-forming toxin that disrupts bacterial membranes through selective ion transport, inducing membrane depolarisation and cell death. Tke5 is neutralised by Tki5 in the inner membrane of Gram-negative bacteria. Unlike detergent-like pore-forming toxins, Tke5 preserves overall membrane integrity, avoiding large, non-specific disruptions. This mechanism offers a powerful approach to targeting resilient phytopathogens. This study reveals a previously undescribed mode of action within a widespread yet understudied toxin family. Our findings highlight the potential of P. putida as a biocontrol agent, offering alternatives to chemical pesticides by exploiting novel toxin mechanisms, crucial for developing effective strategies to combat plant pathogens.
Similar content being viewed by others
Data availability
The data are available within the manuscript. Supplementary Datasets 1–4 are available as additional supplementary files and contain: the list of proteins belonging to the BTH_I2691 family, the list of proteins identified by mass spectrometry from the Tke5 purified band, the numeric data for charts included within the article, and the flow cytometry raw data, respectively. Uncropped and unedited gel images are included in Supplementary Fig. 6. The gating strategy for all flow cytometry plots is included in Supplementary Fig. 7. The sequence of a newly generated vector available as a tool for the scientific community has been deposited into NCBI Genbank with accession number PQ628036 and into the SEVA repository https://seva-plasmids.com/.
References
Mondal, A.K. et al. Sequence diversity in the pore-forming motifs of the membrane-damaging protein toxins. J. Membr. Biology. 253, 469–478 (2020).
Carruthers, V. B. Apicomplexan pore-forming toxins. Annu. Rev. Microbiol. 78, 277–291 (2024).
Peraro, M. D., Van Der Goot, F. G. Pore-forming toxins: ancient, but never really out of fashion. Nat. Rev. Microbiol. 14, 77–92 (2016).
Allsopp, L. P. & Bernal, P. Killing in the name of: T6SS structure and effector diversity. Microbiology 169, 1367 (2023).
Rapisarda, C. et al. In situ and high-resolution cryo-EM structure of a bacterial type VI secretion system membrane complex. EMBO J. 38, e100886 (2019).
Cherrak, Y. et al. Biogenesis and structure of a type VI secretion baseplate. Nat. Microbiol. 3, 1404–1416 (2018).
Zoued, A. et al. Priming and polymerization of a bacterial contractile tail structure. Nature 531, 59–63 (2016).
Kudryashev, M. et al. Structure of the type VI secretion system contractile sheath. Cell 160, 952–962 (2015).
Basler, M., Pilhofer, M., Henderson, G. P., Jensen, G. J. & Mekalanos, J. J. Type VI secretion requires a dynamic contractile phage tail-like structure. Nature 483, 182–186 (2012).
Bernal, P. et al. A novel stabilization mechanism for the type VI secretion system sheath. Proc. Natl. Acad. Sci. USA 118, e2008500118 (2021).
Santin, Y. G. et al. In vivo TssA proximity labelling during type VI secretion biogenesis reveals TagA as a protein that stops and holds the sheath. Nat. Microbiol. 3, 1304–1313 (2018).
Salomon, D. et al. Marker for type VI secretion system effectors. Proc. Natl. Acad. Sci. USA 111, 9271–9276 (2014).
Bernal, P. T6SS-effector hunters uncover PIX: a novel delivery/marker domain. Trends Microbiol. 32, 617–619 (2024).
Kanarek, K., Fridman, C. M., Bosis, E. & Salomon, D. The RIX domain defines a class of polymorphic T6SS effectors and secreted adaptors. Nat. Commun. 14, 4983 (2023).
Jana, B., Fridman, C. M., Bosis, E. & Salomon, D. A modular effector with a DNase domain and a marker for T6SS substrates. Nat. Commun. 10, 3595 (2019).
Carobbi, A. et al. PIX is an N-terminal delivery domain that defines a class of polymorphic T6SS effectors in Enterobacterales. Cell Rep. 43, 114015 (2024).
Fridman, C.M. et al. A new class of type VI secretion system effectors can carry two toxic domains and are recognized through the WHIX motif for export. PLoS Biol. 23, e3003053 (2025).
González-Magaña, A. et al. The P. aeruginosa effector Tse5 forms membrane pores disrupting the membrane potential of intoxicated bacteria. Commun. Biol. 5, 1189 (2022).
González-Magaña, A. et al. Structural and functional insights into the delivery of a bacterial Rhs pore-forming toxin to the membrane. Nat. Commun. 14, 7808 (2023).
Rojas-Palomino, J. et al. The Pseudomonas aeruginosa Tse4 toxin assembles ion-selective and voltage-sensitive ion channels to couple membrane depolarization with K+ efflux. PLoS Pathog. 21, e1012981 (2025).
Ruhe, Z. C., Low, D. A. & Hayes, C. S. Polymorphic toxins and their immunity proteins: diversity, evolution, and mechanisms of delivery. Annu. Rev. Microbiol. 74, 497–520 (2020).
Weller, D. M. Pseudomonas biocontrol agents of soilborne pathogens: looking back over 30 years. Phytopathology 97, 250–256 (2007).
Bernal, P., Allsopp, L. P., Filloux, A. & Llamas, M. A. The Pseudomonas putida T6SS is a plant warden against phytopathogens. ISME J. 11, 972–987 (2017).
Bernal, P. et al. Transcriptional organization and regulation of the Pseudomonas putida K1 type VI secretion system gene cluster. Microbiology 169, 001295 (2023).
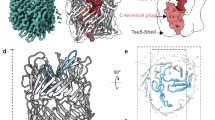
Velázquez, C. et al. Structural insights into the antibacterial function of the Pseudomonas putida effector Tke5. EMBO J. 45, 1229–1244 (2026).
Miyata, S. T., Kitaoka, M., Brooks, T. M., McAuley, S. B. & Pukatzki, S. Vibrio cholerae requires the type VI secretion system virulence factor VasX to kill Dictyostelium discoideum. Infect. Immun. 79, 2941–2949 (2011).
Dong, T. G., Ho, B. T., Yoder-Himes, D. R. & Mekalanos, J. J. Identification of T6SS-dependent effector and immunity proteins by Tn-seq in Vibrio cholerae. Proc. Natl. Acad. Sci. USA 110, 2623–2628 (2013).
Miyata, S. T., Unterweger, D., Rudko, S. P. & Pukatzki, S. Dual expression profile of type VI secretion system immunity genes protects pandemic Vibrio cholerae. PLoS Pathog. 9, e1003752 (2013).
van Kempen, M. et al. Fast and accurate protein structure search with Foldseek. Nat. Biotechnol. 42, 243–246 (2024).
Sockolosky, J. T. & Szoka, F. C. Periplasmic production via the pET expression system of soluble, bioactive human growth hormone. Protein Expr. Purif. 87, 129–135 (2013).
Calles, B., Goñi-Moreno, Á & Lorenzo, V. Digitalizing heterologous gene expression in Gram-negative bacteria with a portable ON/OFF module. Mol. Syst. Biol. 15, e8777 (2019).
Lomize, A. L., Todd, S. C. & Pogozheva, I. D. Spatial arrangement of proteins in planar and curved membranes by PPM 3.0. Protein Sci. 31, 209–220 (2022).
Buller, G. M., Bradford, J. A., Liu, J. & Godfrey, W. L. Novel reagents for the addition of viability measurements to immunostaining using flow cytometry. Blood 108, 3879–3879 (2006).
te Winkel, J. D., Gray, D. A., Seistrup, K. H., Hamoen, L. W. & Strahl, H. Analysis of antimicrobial-triggered membrane depolarization using voltage sensitive dyes. Front Cell Dev. Biol. 4, 29 (2016).
Montal, M. & Mueller, P. Formation of bimolecular membranes from lipid monolayers and a study of their electrical properties. Proc. Natl. Acad. Sci. USA 69, 3561–3566 (1972).
LaCourse, K. D. et al. Conditional toxicity and synergy drive diversity among antibacterial effectors. Nat. Microbiol. 3, 440–446 (2018).
Aguilella, V. M., Queralt-Martín, M., Aguilella-Arzo, M. & Alcaraz, A. Insights on the permeability of wide protein channels: measurement and interpretation of ion selectivity. Integr. Biol. 3, 159–172 (2011).
Alcaraz, A. et al. Diffusion, exclusion, and specific binding in a large channel: a study of OmpF selectivity inversion. Biophys. J. 96, 56–66 (2009).
Hille, B. Ion Channels of Excitable Membranes, 3rd edn (Sinauer Associates, Inc, 2001).
Park, H. B., Kamcev, J., Robeson, L. M., Elimelech, M. & Freeman, B. D. Maximizing the right stuff: the trade-off between membrane permeability and selectivity. Science 356, 1138–1148 (2017).
Hodgkin, A. L. & Katz, B. The effect of sodium ions on the electrical activity of the giant axon of the squid. J. Physiol. 108, 37–77 (1949).
Mansfield, J. et al. Top 10 plant pathogenic bacteria in molecular plant pathology. Mol. Plant Pathol. 13, 614–629 (2012).
Vázquez-Arias, D. et al. The Pseudomonas putida type VI secretion systems shape the tomato rhizosphere microbiota. ISME Commun. 5, ycaf158 (2025).
Fridman, C. M., Keppel, K., Gerlic, M., Bosis, E. & Salomon, D. A comparative genomics methodology reveals a widespread family of membrane-disrupting T6SS effectors. Nat. Commun. 11, 1085 (2020).
Colautti, J. et al. Cryo-EM structure of a type VI secretion system-delivered membrane-depolarizing toxin involved in bacterial antagonism. Cell Rep. 44, 116263 (2025).
Rojas-Palomino, J. et al. Electrophysiological dissection of the ion channel activity of the Pseudomonas aeruginosa ionophore protein toxin Tse5. Chem. Phys. Lipids. 267, 105472 (2025).
Reglinski, M. et al. A widely-occurring family of pore-forming effectors broadens the impact of the Serratia type VI secretion system. EMBO J. 44, 6892–6918 (2025).
Mariano, G. et al. A family of type VI secretion system effector proteins that form ion-selective pores. Nat. Commun. 10, 1–15 (2019).
Halvorsen, T. M. et al. Contact-dependent growth inhibition (CDI) systems deploy a large family of polymorphic ionophoric toxins for inter-bacterial competition. PLoS Genet. 20, e1011494 (2024).
Ratner, H. K. et al. An electron transport complex required in the gut sensitizes Bacteroides to a pore-forming type VI secretion toxin. Proc. Natl. Acad. Sci. USA 122, e2523503122 (2025).
Bernal, P., Llamas, M. A. & Filloux, A. Type VI secretion systems in plant-associated bacteria. Environ. Microbiol. 20, 1–15 (2018).
Borrero-de-Acuña, J. M. & Bernal, P. Plant holobiont interactions mediated by the type VI secretion system and the membrane vesicles: promising tools for a greener agriculture. Environ. Microbiol. 23, 1830–1836 (2021).
Bourigault, Y. et al. Pseudomonas fluorescens MFE01 delivers a putative type VI secretion amidase that confers biocontrol against the soft-rot pathogen Pectobacterium atrosepticum. Environ. Microbiol. 25, 2564–2579 (2023).
Cassan, F. D. et al. The Azospirillum brasilense Type VI secretion system promotes cell aggregation, biocontrol protection against phytopathogens and attachment to the microalgae Chlorella sorokiniana. Environ. Microbiol. 23, 6257–6274 (2021).
Boak, E. N., Kirolos, S., Pan, H., Pierson, L. S. & Pierson, E. A. The type VI secretion systems in plant-beneficial bacteria modulate prokaryotic and eukaryotic interactions in the rhizosphere. Front. Microbiol. 13, 843092 (2022).
Durán, D. et al. Pseudomonas fluorescens F113 type VI secretion systems mediate bacterial killing and adaption to the rhizosphere microbiome. Sci. Rep. 11, 5772 (2021).
Marchi, M. et al. Genomic analysis of the biocontrol strain Pseudomonas fluorescens Pf29Arp with evidence of T3SS and T6SS gene expression on plant roots. Environ. Microbiol. Rep. 5, 393–403 (2013).
Kan, Y., Zhang, Y., Lin, W. & Dong, T. Differential plant cell responses to Acidovorax citrulli T3SS and T6SS reveal an effective strategy for controlling plant-associated pathogens. mBio 14, e0045923 (2023).
Reyes-Pérez, P. J. et al. The type VI secretion system of Sinorhizobium fredii USDA257 is required for successful nodulation with Glycine max cv pekin. Micro. Biotechnol. 18, e70112 (2025).
Sambrook, J., Fritsch, E.F. & Maniatis, T. Molecular Cloning: A Laboratory Manual, 2nd edn (Cold Spring Harbor Laboratory Press, 1989).
Choi, K.-H., Kumar, A. & Schweizer, H. P. A 10-min method for preparation of highly electrocompetent Pseudomonas aeruginosa cells: application for DNA fragment transfer between chromosomes and plasmid transformation. J. Microbiol. Methods 64, 391–397 (2006).
Ramos-Gonzalez, M. I., Duque, E. & Ramos, J. L. Conjugational transfer of recombinant DNA in cultures and in soils: host range of Pseudomonas putida TOL plasmids. Appl. Environ. Microbiol. 57, 3020–3027 (1991).
Lei, S.-P., Lin, H.-C., Wang, S.-S., Callaway, J. & Wilcox, G. Characterization of the Erwinia carotovora pelB gene and its product pectate lyase. J. Bacteriol. 169, 4379–4383 (1987).
Tsirigotaki, A., De Geyter, J., Šoštarić, Economou, N. & Karamanou, A. S. Protein export through the bacterial Sec pathway. Nat. Rev. Microbiol. 15, 21–36 (2017).
Silva-Rocha et al. The Standard European Vector Architecture (SEVA): a coherent platform for the analysis and deployment of complex prokaryotic phenotypes. Nucleic Acids Res. 41, D666–D675 (2013).
Martínez-García, E. et al. SEVA 4.0: an update of the Standard European Vector Architecture database for advanced analysis and programming of bacterial phenotypes. Nucleic Acids Res. 51, D1558–D1567 (2023).
Nikel, P. I., Benedetti, I., Wirth, N. T., de Lorenzo, V. & Calles, B. Standardization of regulatory nodes for engineering heterologous gene expression: a feasibility study. Micro. Biotechnol. 15, 2250–2265 (2022).
Winsor, G. L. et al. Enhanced annotations and features for comparing thousands of Pseudomonas genomes in the Pseudomonas genome database. Nucleic Acids Res. 44, D646–D653 (2016).
Boratyn, G. M. et al. BLAST: a more efficient report with usability improvements. Nucleic Acids Res. 41, W29–W33 (2013).
Letunic, I., Doerks, T. & Bork, P. SMART: recent updates, new developments and status in 2015. Nucleic Acids Res. 43, D257–D260 (2015).
Finn, R. D. et al. The Pfam protein families database: towards a more sustainable future. Nucleic Acids Res. 44, D279–D285 (2016).
Wang, J. et al. The conserved domain database in 2023. Nucleic Acids Res. 51, D384–D388 (2023).
Zimmermann, L. et al. A completely reimplemented MPI bioinformatics toolkit with a new HHpred server at its core. J. Mol. Biol. 430, 2237–2243 (2018).
Krogh, A., Larsson, B., Von Heijne, G. & Sonnhammer, E. L. L. Predicting transmembrane protein topology with a hidden Markov model: application to complete genomes. J. Mol. Biol. 305, 567–580 (2001).
Tamura, K., Stecher, G. & Kumar, S. MEGA11: molecular evolutionary genetics analysis version 11. Mol. Biol. Evol. 38, 3022–3027 (2021).
Jones, D. T., Taylor, W. R. & Thornton, J. M. The rapid generation of mutation data matrices from protein sequences. Comput. Appl. Biosci. 8, 275–282 (1992).
Letunic, I. & Bork, P. Interactive tree of life (iTOL) v4: recent updates and new developments. Nucleic Acids Res. 47, W256–W259 (2019).
Madeira, F. et al. The EMBL-EBI job dispatcher sequence analysis tools framework in 2024. Nucleic Acids Res. 52, W521–W525 (2024).
Meng, E. C. et al. UCSF ChimeraX: tools for structure building and analysis. Protein Sci. 32, e4792 (2023).
Vidal, J., Godbillon, G. & Gadal, P. Recovery of active, highly purified phosphoenolpyruvate carboxylase from specific immunoadsorbent column. FEBS Lett. 118, 31–34 (1980).
Morein, S., Andersson, A.-S., Rilfors, L. & Ran Lindblom, G. Wild-type Escherichia coli cells regulate the membrane lipid composition in a “Window” between gel and non-lamellar structures*. J. Biol. Chem. 271, 6801–6809 (1996).
Rojas-Palomino, J. et al. The Pseudomonas aeruginosa Tse4 toxin assembles ion-selective and voltage-sensitive ion channels to couple membrane depolarisation with K + efflux. PLoS Pathog. 21, e1012981 (2025).
Reglinski, M. et al. A widely-occurring family of pore-forming effectors broadens the impact of the Serratia Type VI secretion system. EMBO J. 44, 6892–6918 (2024).
Buist, G., Steen, A., Kok, J. & Kuipers, O. P. LysM, a widely distributed protein motif for binding to (peptido)glycans. Mol. Microbiol. 68, 838–847 (2008).
Shneider, M. M. et al. PAAR-repeat proteins sharpen and diversify the type VI secretion system spike. Nature 500, 350–353 (2013).
Pukatzki, S. et al. Identification of a conserved bacterial protein secretion system in Vibrio cholerae using the Dictyostelium host model system. Proc. Natl. Acad. Sci. USA 103, 1528–1533 (2006).
Unterweger, D. et al. Chimeric adaptor proteins translocate diverse type VI secretion system effectors in Vibrio cholerae. EMBO J. 34, 2198–2210 (2015).
Storey, D. et al. Klebsiella pneumoniae type VI secretion system-mediated microbial competition is PhoPQ controlled and reactive oxygen species dependent. PLoS Pathog. 16, e1007969 (2020).
Acknowledgements
This publication is part of the project PID2021-123000OB-100, funded by MICIU/AEI/10.13039/501100011033 and by ERDF/EU. P.B. acknowledges the financial support received from the Spanish Minister of Science, Innovation and Universities (MICIU/AEI/10.13039/501100011033) through the Ramón y Cajal Program (RYC2019-026551-I, ESF Investing in your future), the research grant from the State Subprogram for Knowledge Generation PID2021-123000OB-I00 and PID2024-159235OB-I00 (ERDF/EU), and the research grant from the State Subprogram for Promotion of Research Consolidation CNS2022-135585 (European Union NextGenerationEU/PRTR). D.A.-J. acknowledges support by the MICIU Contract PID2021-127816NB-I00, PID2024-155225NB-I00, and the Basque Government’s Department of Education IT1745-22. A.A. and M.Q.-M. acknowledge financial support by the Spanish Government MICIU/AEI/10.13039/501100011033/FEDER, UE (Project 2019-108434GB-I00 and Project PID2022-142795 NB-I00), Generalitat Valenciana (project CIGRIS/2021/021), and Universitat Jaume I (Project UJI-B2022-42). M.Q.-M acknowledges support from the Spanish Ministry of Science and Innovation (Project IJC2018-035283-I funded by MICIU/AEI/10.13039/501100011033) and Universitat Jaume I (project UJI-A2020-21). We thank Cayo Ramos, Emilia López Solanilla, Francisco M. Cazorla, Ehr Min Lai, María Milagros López, and Inmaculada Sampedro for their kind gifts of phytopathogenic strains.
Author information
Authors and Affiliations
Contributions
Conceptualisation: A.A., D.A.-J., P.B. Methodology: C.V., A.A.-R., M.Q.-M., J.A.-A., J.R.-P., A.F.-C., C.C.-N., A.R., J.P.-N., and C.C. Investigation: A.A., D.A.-J., and P.B. Visualisation: C.V., A.A.-R., M.Q.-M., and P.B. Supervision: M.Q.-M., A.G.-M., A.A.-R., A.A., D.A.-J., and P.B. Writing—original draft: A.A., D.A.-J., and P.B. Writing—review and editing: C.V., A.A.-R., and M.Q.-M.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Alain Filloux, Long Lin, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editors: Heejin Yoo and David Favero. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Velázquez, C., Arce-Rodríguez, A., Altuna-Alvarez, J. et al. Tke5 is a Pseudomonas putida toxin that kills plant pathogens by depolarising membranes. Commun Biol (2026). https://doi.org/10.1038/s42003-026-09863-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-09863-w