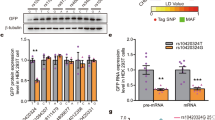
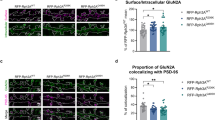
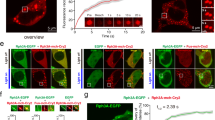
Abstract
The aberrant formation and function of neuronal synapses are recognized as major phenotypes in many cases of neurodevelopmental (NDDs) and -psychiatric disorders (NPDs). A growing body of research has identified an expanding number of susceptibility genes encoding proteins with synaptic function. Here, we present the first brain-focused characterization of a potential new susceptibility gene, ARHAGP8, which encodes a Rho GTPase activating protein (RhoGAP). Accumulating evidence suggests that ARHGAP8 plays a pivotal role in the pathogenesis of NPDs/NDDs. We provide the first evidence for ARHGAP8 as a novel player at excitatory synapses, with its synaptic localisation linked to the presence of the developmentally important NMDA receptor subunit GluN2B. By increasing ARHGAP8 levels in hippocampal neurons to mimic elevated levels found in subsets of patients, we observed reductions in dendritic complexity and spine volume, accompanied by a significant decrease in synaptic AMPA receptor-mediated transmission. These results suggest that ARHGAP8 plays a role in shaping the morphology and function of excitatory synapses, and prompt further investigation of ARHGAP8 as a candidate gene in NDDs/NPDs.
Similar content being viewed by others
Data availability
Numerical data set is made available on Zenodo92.
References
Caldeira, G. L., Peça, J. & Carvalho, A. L. New insights on synaptic dysfunction in neuropsychiatric disorders. Curr. Opin. Neurobiol. 57, 62–70 (2019).
Parenti, I., Rabaneda, L. G., Schoen, H. & Novarino, G. Neurodevelopmental disorders: from genetics to functional pathways. Trends Neurosci. 43, 608–621 (2020).
Chidambaram, S. B. et al. Dendritic spines: revisiting the physiological role. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 92, 161–193 (2019).
Huganir, R. L. & Nicoll, R. A. AMPARs and synaptic plasticity: the last 25 years. Neuron 80, 704–717 (2013).
Diering, G. H. & Huganir, R. L. The AMPA receptor code of synaptic plasticity. Neuron 100, 314–329 (2018).
Hansen, K. B. et al. Structure, function, and pharmacology of glutamate receptor ion channels. Pharm. Rev. 73, 1469–1658 (2021).
Matsuzaki, M., Honkura, N., Ellis-Davies, G. C. R. & Kasai, H. Structural basis of long-term potentiation in single dendritic spines. Nature 429, 761–766 (2004).
Oh, W. C., Hill, T. C. & Zito, K. Synapse-specific and size-dependent mechanisms of spine structural plasticity accompanying synaptic weakening. Proc. Natl. Acad. Sci. USA. 110, E305-12 (2013).
Phillips, M. & Pozzo-Miller, L. Dendritic spine dysgenesis in autism related disorders. Neurosci. Lett. 601, 30–40 (2015).
Forrest, M. P., Parnell, E. & Penzes, P. Dendritic structural plasticity and neuropsychiatric disease. Nat. Rev. Neurosci. 19, 215–234 (2018).
Kasri, N. N. & Van Aelst, L. Rho-linked genes and neurological disorders. Pflug. Arch. - Eur. J. Physiol. 455, 787–797 (2008).
Hall, A. & Lalli, G. Rho and Ras GTPases in axon growth, guidance, and branching. Cold Spring Harb. Perspect. Biol. 2, a001818–a001818 (2010).
Auer, M., Hausott, B. & Klimaschewski, L. Rho GTPases as regulators of morphological neuroplasticity. Ann. Anat. Anatomischer Anz. 193, 259–266 (2011).
Tolias, K. F., Duman, J. G. & Um, K. Control of synapse development and plasticity by Rho GTPase regulatory proteins. Prog. Neurobiol. 94, 133–148 (2011).
Ba, W., Van Der Raadt, J. & Nadif Kasri, N. Rho GTPase signaling at the synapse: Implications for intellectual disability. Exp. Cell Res. 319, 2368–2374 (2013).
Schmidt, A. & Hall, A. Guanine nucleotide exchange factors for Rho GTPases: turning on the switch. Genes Dev. 16, 1587–1609 (2002).
Tcherkezian, J. & Lamarche-Vane, N. Current knowledge of the large RhoGAP family of proteins. Biol. Cell 99, 67–86 (2007).
Duman, J. G. et al. Mechanisms for spatiotemporal regulation of Rho-GTPase signaling at synapses. Neurosci. Lett. 601, 4–10 (2015).
Kutsche, K. et al. Mutations in ARHGEF6, encoding a guanine nucleotide exchange factor for Rho GTPases, in patients with X-linked mental retardation. Nat. Genet 26, 247–250 (2000).
Govek, E.-E. et al. The X-linked mental retardation protein oligophrenin-1 is required for dendritic spine morphogenesis. Nat. Neurosci. 7, 364–372 (2004).
Kasri, N. N., Nakano-Kobayashi, A., Malinow, R., Li, B. & Van Aelst, L. The Rho-linked mental retardation protein oligophrenin-1 controls synapse maturation and plasticity by stabilizing AMPA receptors. Genes Dev. 23, 1289–1302 (2009).
Remmers, C., Sweet, R. A. & Penzes, P. Abnormal kalirin signaling in neuropsychiatric disorders. Brain Res. Bull. 103, 29–38 (2014).
Ba, W. & Nadif Kasri, N. RhoGTPases at the synapse: an embarrassment of choice. Small GTPases 8, 106–113 (2017).
Disciglio, V. et al. Interstitial 22q13 deletions not involving SHANK3 gene: A new contiguous gene syndrome. Am. J. Med Genet. Pt A 164, 1666–1676 (2014).
Donarum, E. A., Halperin, R. F., Stephan, D. A. & Narayanan, V. Cognitive dysfunction in NFI knock-out mice may result from altered vesicular trafficking of APP/DRD3 complex. BMC Neurosci. 7, 22 (2006).
Gong, X. et al. LncRNA THUMPD3-AS1 regulates behavioral and synaptic structural abnormalities in schizophrenia via miR-485-5p and ARHGAP8. Adv. Sci. 13, e08867 (2025).
Johnston, J. N. et al. Response of iPSC-derived neurons from individuals with treatment-resistant depression to (2 R,6 R)-hydroxynorketamine and reelin: an exploratory study. Transl. Psychiatry 15, 524 (2025).
McElroy, S. L. et al. Bipolar disorder with binge eating behavior: a genome-wide association study implicates PRR5-ARHGAP8. Transl. Psychiatry 8, 40 (2018).
Wong, M.-L. et al. The PHF21B gene is associated with major depression and modulates the stress response. Mol. Psychiatry 22, 1015–1025 (2017).
Hering, H. & Sheng, M. Activity-dependent redistribution and essential role of cortactin in dendritic spine morphogenesis. J. Neurosci. 23, 11759–11769 (2003).
Lua, B. L. & Low, B. C. BPGAP1 interacts with cortactin and facilitates its translocation to cell periphery for enhanced cell migration. MBoC 15, 2873–2883 (2004).
Lua, B. L. & Low, B. C. Activation of EGF receptor endocytosis and ERK1/2 signaling by BPGAP1 requires direct interaction with EEN/endophilin II and a functional RhoGAP domain. J. Cell Sci. 118, 2707–2721 (2005).
Chowdhury, S. et al. Arc/Arg3.1 interacts with the endocytic machinery to regulate AMPA receptor trafficking. Neuron 52, 445–459 (2006).
Catarino, T., Ribeiro, L., Santos, S. D. & Carvalho, A. L. Regulation of synapse composition by protein acetylation: the role of acetylated cortactin. J. Cell Sci. 126, 149–162 (2013).
Zhang, J. et al. Distinct functions of endophilin isoforms in synaptic vesicle endocytosis. Neural Plasticity 2015, 1–10 (2015).
Antonelli, R. et al. Pin1 modulates the synaptic content of NMDA receptors via prolyl-isomerization of PSD-95. J. Neurosci. 36, 5437–5447 (2016).
Shang, X., Zhou, Y. T. & Low, B. C. Concerted regulation of cell dynamics by BNIP-2 and Cdc42GAP homology/Sec14p-like, Proline-rich, and GTPase-activating protein domains of a novel Rho GTPase-activating protein, BPGAP1. J. Biol. Chem. 278, 45903–45914 (2003).
Burnashev, N. & Szepetowski, P. NMDA receptor subunit mutations in neurodevelopmental disorders. Curr. Opin. Pharmacol. 20, 73–82 (2015).
Hu, C., Chen, W., Myers, S. J., Yuan, H. & Traynelis, S. F. Human GRIN2B variants in neurodevelopmental disorders. J. Pharmacol. Sci. 132, 115–121 (2016).
Duman, J. G. et al. Rac-maninoff and Rho-vel: the symphony of Rho-GTPase signaling at excitatory synapses. Small GTPases 13, 14–47 (2022).
Lohmann, C. & Kessels, H. W. The developmental stages of synaptic plasticity. J. Physiol. 592, 13–31 (2014).
Buttery, P. et al. The diacylglycerol-binding protein α1-chimaerin regulates dendritic morphology. Proc. Natl. Acad. Sci. USA 103, 1924–1929 (2006).
Ferreira, J. S. et al. GluN2B-containing NMDA receptors regulate AMPA receptor traffic through anchoring of the synaptic proteasome. J. Neurosci. 35, 8462–8479 (2015).
Ferreira, J. S., Kellermayer, B., Carvalho, A. L. & Groc, L. Interplay between NMDA receptor dynamics and the synaptic proteasome. Eur. J. Neurosci. 54, 6000–6011 (2021).
Mony, L. & Paoletti, P. Mechanisms of NMDA receptor regulation. Curr. Opin. Neurobiol. 83, 102815 (2023).
Dupuis, J. P., Nicole, O. & Groc, L. NMDA receptor functions in health and disease: Old actor, new dimensions. Neuron 111, 2312–2328 (2023).
Nakazawa, T. et al. p250GAP, a novel brain-enriched GTPase-activating Protein for Rho Family GTPases, is involved in the N -Methyl- d -aspartate receptor signaling. MBoC 14, 2921–2934 (2003).
Nakazawa, T. et al. Regulation of dendritic spine morphology by an NMDA receptor-associated Rho GTPase-activating protein, p250GAP. J. Neurochem. 105, 1384–1393 (2008).
Kiraly, D. D., Lemtiri-Chlieh, F., Levine, E. S., Mains, R. E. & Eipper, B. A. Kalirin binds the NR2B subunit of the NMDA receptor, altering its synaptic localization and function. J. Neurosci. 31, 12554–12565 (2011).
Lemtiri-Chlieh, F. et al. Kalirin-7 is necessary for normal NMDA receptor-dependent synaptic plasticity. BMC Neurosci. 12, 126 (2011).
Hodge, R. G. & Ridley, A. J. Regulating Rho GTPases and their regulators. Nat. Rev. Mol. Cell Biol. 17, 496–510 (2016).
Wong, D. C. P. et al. The scaffold RhoGAP protein ARHGAP8/BPGAP1 synchronizes Rac and Rho signaling to facilitate cell migration. MBoC 34, ar13 (2023).
Caetano-Anollés, K. et al. Cerebellum transcriptome of mice bred for high voluntary activity offers insights into locomotor control and reward-dependent behaviors. PLoS ONE 11, e0167095 (2016).
Firth, H. V. et al. DECIPHER: database of chromosomal imbalance and phenotype in humans using Ensembl resources. Am. J. Hum. Genet. 84, 524–533 (2009).
Matus, A. Actin-based plasticity in dendritic spines. Science 290, 754–758 (2000).
Okabe, S. Regulation of actin dynamics in dendritic spines: nanostructure, molecular mobility, and signaling mechanisms. Mol. Cell. Neurosci. 109, 103564 (2020).
Riedl, J. et al. Lifeact: a versatile marker to visualize F-actin. Nat. Methods 5, 605–607 (2008).
Ravichandran, A. & Low, B. C. SmgGDS antagonizes BPGAP1-induced Ras/ERK activation and neuritogenesis in PC12 cell differentiation. MBoC 24, 145–156 (2013).
Tang, A.-H. et al. A trans-synaptic nanocolumn aligns neurotransmitter release to receptors. Nature 536, 210–214 (2016).
Martinez-Sanchez, A. et al. Trans-synaptic assemblies link synaptic vesicles and neuroreceptors. Sci. Adv. 7, eabe6204 (2021).
Adesnik, H., Li, G., During, M. J., Pleasure, S. J. & Nicoll, R. A. NMDA receptors inhibit synapse unsilencing during brain development. Proc. Natl. Acad. Sci. USA 105, 5597–5602 (2008).
Gray, J. A. et al. Distinct modes of AMPA receptor suppression at developing synapses by GluN2A and GluN2B: single-cell NMDA receptor subunit deletion in vivo. Neuron 71, 1085–1101 (2011).
Hall, B. J., Ripley, B. & Ghosh, A. NR2B signaling regulates the development of synaptic AMPA receptor current. J. Neurosci. 27, 13446–13456 (2007).
Hall, B. J. & Ghosh, A. Regulation of AMPA receptor recruitment at developing synapses. Trends Neurosci. 31, 82–89 (2008).
Ultanir, S. K. et al. Regulation of spine morphology and spine density by NMDA receptor signaling in vivo. Proc. Natl. Acad. Sci. USA 104, 19553–19558 (2007).
Wang, C.-C. et al. A critical role for GluN2B-containing NMDA receptors in cortical development and function. Neuron 72, 789–805 (2011).
Wang, S. & Yao, T. Multi-omics framework integrating genetics microbiome and immunity for understanding motor neuron degeneration pathogenesis. npj Biofilms Microbiomes 11, 236 (2025).
Racz, B. & Weinberg, R. J. The subcellular organization of cortactin in hippocampus. J. Neurosci. 24, 10310–10317 (2004).
Zhang, J. et al. Collapsin response mediator protein 2 and endophilin2 coordinate regulation of AMPA receptor GluA1 subunit recycling. Front. Mol. Neurosci. 13, 128 (2020).
Govek, E.-E., Newey, S. E. & Van Aelst, L. The role of the Rho GTPases in neuronal development. Genes Dev. 19, 1–49 (2005).
Guo, D., Yang, X. & Shi, L. Rho GTPase regulators and effectors in autism spectrum disorders: animal models and insights for therapeutics. Cells 9, 835 (2020).
Silva, M. M. et al. MicroRNA-186-5p controls GluA2 surface expression and synaptic scaling in hippocampal neurons. Proc. Natl. Acad. Sci. USA 116, 5727–5736 (2019).
Chen, H.-J., Rojas-Soto, M., Oguni, A. & Kennedy, M. B. A synaptic Ras-GTPase activating protein (p135 SynGAP) inhibited by CaM Kinase II. Neuron 20, 895–904 (1998).
Hamdan, F. F. et al. Mutations in SYNGAP1 in autosomal nonsyndromic mental retardation. N. Engl. J. Med. 360, 599–605 (2009).
Hamdan, F. F. et al. Excess of De novo deleterious mutations in genes associated with glutamatergic systems in nonsyndromic intellectual disability. Am. J. Hum. Genet. 88, 306–316 (2011).
Berryer, M. H. et al. Mutations in SYNGAP1 cause intellectual disability, autism, and a specific form of epilepsy by inducing haploinsufficiency. Hum. Mutat. 34, 385–394 (2013).
Yang, Y., Tao-Cheng, J.-H., Reese, T. S. & Dosemeci, A. SynGAP moves out of the core of the postsynaptic density upon depolarization. Neuroscience 192, 132–139 (2011).
Araki, Y., Zeng, M., Zhang, M. & Huganir, R. L. Rapid dispersion of SynGAP from synaptic spines triggers AMPA receptor insertion and spine enlargement during LTP. Neuron 85, 173–189 (2015).
Komiyama, N. H. et al. SynGAP regulates ERK/MAPK signaling, synaptic plasticity, and learning in the complex with postsynaptic density 95 and NMDA receptor. J. Neurosci. 22, 9721–9732 (2002).
Araki, Y. et al. SynGAP regulates synaptic plasticity and cognition independently of its catalytic activity. Science 383, eadk1291 (2024).
Kaech, S. & Banker, G. Culturing hippocampal neurons. Nat. Protoc. 1, 2406–2415 (2006).
Kutsuwada, T. et al. Impairment of suckling response, trigeminal neuronal pattern formation, and hippocampal LTD in NMDA receptor epsilon 2 subunit mutant mice. Neuron 16, 333–344 (1996).
Tovar, K. R., Sprouffske, K. & Westbrook, G. L. F. astN. M. D. A. Receptor–mediated synaptic currents in neurons from mice lacking the ε2 (NR2B) subunit. J. Neurophysiol. 83, 616–620 (2000).
Truett, G. E. et al. Preparation of PCR-quality mouse genomic DNA with hot sodium hydroxide and tris (HotSHOT). BioTechniques 29, 52–54 (2000).
Stoppini, L., Buchs, P.-A. & Muller, D. A simple method for organotypic cultures of nervous tissue. J. Neurosci. Methods 37, 173–182 (1991).
Jiang, M., Deng, L. & Chen, G. High Ca2+-phosphate transfection efficiency enables single neuron gene analysis. Gene Ther. 11, 1303–1311 (2004).
Jiang, M. & Chen, G. High Ca2+-phosphate transfection efficiency in low-density neuronal cultures. Nat. Protoc. 1, 695–700 (2006).
Woods, G. & Zito, K. Preparation of Gene Gun Bullets and Biolistic Transfection of Neurons in Slice Culture. JoVE 675, https://doi.org/10.3791/675 (2008).
Longair, M. H., Baker, D. A. & Armstrong, J. D. Simple neurite tracer: open source software for reconstruction, visualization and analysis of neuronal processes. Bioinformatics 27, 2453–2454 (2011).
Sholl, D. A. Dendritic organization in the neurons of the visual and motor cortices of the cat. J. Anat. 87, 387–406 (1953).
Peça, J. et al. Shank3 mutant mice display autistic-like behaviours and striatal dysfunction. Nature 472, 437–442 (2011).
Schmidt, J. & Carvalho, A. L. Numerical source data for ‘Neuronal ARHGAP8 controls synapse structure and AMPA receptor-mediated synaptic transmission’. Zenodo https://doi.org/10.5281/zenodo.18776119 (2026).
Acknowledgements
We would like to acknowledge Professor Boon Chuan Low of the Cell Signalling and Developmental Biology Group, Mechanobiology Institute of National University of Singapore, for the kind gift of the GFP-BPGAP1 and HA-BPGAP1 plasmids (including the mutants). The project leading to these results has received funding from “la Caixa” Foundation (ID 100010434), and FCT, I.P under the project code LCF/PR/HP20/52300003. The work was also funded by the European Regional Development Fund (ERDF), through the COMPETE 2020 — Operational Programme for Competitiveness and Internationalisation and Portuguese national funds via FCT, under projects UIDB/04539/2020, UIDP/04539/2020, LA/P/0058/2020, SFRH/BD/51960/2012, PTDC/BIA-CEL/2286/2020, Marie Skłodowska-Curie Action Individual Fellowship 101031398, IBRO Return Home Fellowship 2021, 2020.01761. CEECIND/CP1609/CT0003, 2022.05386.PTDC and European Union Horizon 2020 Research and Innovation programme under grant agreement 857524.
Author information
Authors and Affiliations
Contributions
J.S., Â.S.I., J.F., D.S., R.S., N.B., L.F.R., P.P., J.B.R., A.L.C. – research design; J.S., Â.S.I., J.F., D.S., R.S., N.B. performance of experiments and analysis; Manuscript written by J.S., J.F. and A.L.C. with contributions made by all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Cláudia Almeida and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Benjamin Bessieres. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Schmidt, J., Inácio, Â.S., Ferreira, J. et al. Neuronal ARHGAP8 controls synapse structure and AMPA receptor-mediated synaptic transmission. Commun Biol (2026). https://doi.org/10.1038/s42003-026-09884-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-09884-5