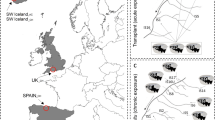
Abstract
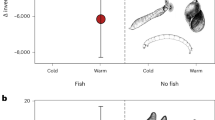
Metabolic rate determines the amount of energy an organism needs to survive, and it is typically predicted to increase with warming up to an optimum temperature for ectothermic organisms. Once their metabolic demands have been met, organisms can use the excess energy from feeding for enhanced growth and reproduction. Experimental evidence suggests that metabolic rate may increase more with warming than energy intake, which could lead to energetic inefficiency and population decline. Downregulating metabolic rates or enhancing feeding rates after chronic exposure to warmer environments could help overcome this problem, but populations and individuals may vary in their capacity for such change. Here, we experimentally measured the temperature-dependent metabolic and feeding rates of brown trout (Salmo trutta) originating from one cold and two warm streams in the same geothermally heated catchment, and examined their population genetic structure. We found a consistent increase in metabolic rate with temperature for all fish, but a stronger increase in feeding rate with temperature for those originating from warm streams. This resulted in the latter exhibiting a greater energetic efficiency with increasing temperature than the fish originating from the cold stream. We detected significant genetic differentiation at neutral markers between the cold and warm streams, implying limited gene flow across the thermal or geographic gradient, and thus scope for adaptive divergence. Collectively our results point towards important variation in eco-physiology within a single catchment that has implications for population persistence in the face of warming. These results highlight the importance of considering intraspecific variation in predictive models of biological responses to climate change. They moreover emphasise how energy intake versus expenditure can be differentially thermally sensitive even at fine spatial scales.
Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the University of Essex Research Data Repository105 at https://doi.org/10.5526/ERDR-00000243. Numerical source data underpinning the figures can be found in Supplementary Data. All other data are available from the corresponding authors on reasonable request.
Code availability
The code that supports the findings of this study is available from the University of Essex Research Data Repository105 at https://doi.org/10.5526/ERDR-00000243. All analyses were conducted in R v4.0.2.
References
He, F. et al. The global decline of freshwater megafauna. Glob. Chang. Biol. 25, 3883–3892 (2019).
Vörösmarty, C. J. et al. Global threats to human water security and river biodiversity. Nature 467, 555–561 (2010).
Dudgeon, D. et al. Freshwater biodiversity: importance, threats, status and conservation challenges. Biol. Rev. 81, 163–182 (2006).
Strayer, D. L. & Dudgeon, D. Freshwater biodiversity conservation: recent progress and future challenges. J. N. Am. Benthol. Soc. 29, 344–358 (2010).
Capon, S. J., Stewart-Koster, B. & Bunn, S. E. Future of freshwater ecosystems in a 1.5°C warmer world. Front. Environ. Sci. 9, 784642 (2021).
Woodward, G., Perkins, D. M. & Brown, L. E. Climate change and freshwater ecosystems: impacts across multiple levels of organization. Philos. Trans. R. Soc. B Biol. Sci. 365, 2093–2106 (2010).
IPCC. Climate Change 2022: Impacts, Adaptation and Vulnerability. Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change (Cambridge University Press, Cambridge, UK, 2022).
Abram, P. K., Boivin, G., Moiroux, J. & Brodeur, J. Behavioural effects of temperature on ectothermic animals: unifying thermal physiology and behavioural plasticity. Biol. Rev. 92, 1859–1876 (2017).
Willmer, P., Stone, G. & Johnston, I. Environmental Physiology of Animals (Blackwell Publishing, 2005).
Dell, A. I., Pawar, S. & Savage, V. M. Systematic variation in the temperature dependence of physiological and ecological traits. Proc. Natl. Acad. Sci. USA 108, 10591–10596 (2011).
Brown, J. H., Gillooly, J. F., Allen, A. P., Savage, V. M. & West, G. B. Toward a metabolic theory of ecology. Ecology 85, 1771–1789 (2004).
Di Santo, V. & Bennett, W. A. Effect of rapid temperature change on resting routine metabolic rates of two benthic elasmobranchs. Fish. Physiol. Biochem. 37, 929–934 (2011).
Gillooly, J. F., Brown, J. H., West, G. B., Savage, V. M. & Charnov, E. L. Effects of size and temperature on metabolic rate. Science 293, 2248–2251 (2001).
Kleiber, M. Body size and metabolism. Hilgardia 6, 315–353 (1932).
Savage, V. M. et al. The predominance of quarter-power scaling in biology. Funct. Ecol. 18, 257–282 (2004).
White, C. R., Cassey, P. & Blackburn, T. M. Allometric exponents do not support a universal metabolic allometry. Ecology 88, 315–323 (2007).
Bokma, F. Evidence against universal metabolic allometry. Funct. Ecol. 18, 184–187 (2004).
Lemoine, N. P. & Burkepile, D. E. Temperature-induced mismatches between consumption and metabolism reduce consumer fitness. Ecology 93, 2483–2489 (2012).
Rall, B.ÖC., Vucic-Pestic, O., Ehnes, R. B., EmmersoN, M. & Brose, U. Temperature, predator–prey interaction strength and population stability. Glob. Chang. Biol. 16, 2145–2157 (2010).
Vucic-Pestic, O., Ehnes, R. B., Rall, B. C. & Brose, U. Warming up the system: higher predator feeding rates but lower energetic efficiencies. Glob. Chang. Biol. 17, 1301–1310 (2011).
Huey, R. B. & Kingsolver, J. G. Climate warming, resource availability, and the metabolic meltdown of ectotherms. Am. Nat. 194, E140–E150 (2019).
Vasseur, D. A. & McCann, K. S. A mechanistic approach for modeling temperature-dependent consumer-resource dynamics. Am. Nat. 166, 184–198 (2005).
Arim, M., Bozinovic, F. & Marquet, P. A. On the relationship between trophic position, body mass and temperature: reformulating the energy limitation hypothesis. Oikos 116, 1524–1530 (2007).
Urban, M. C., Zarnetske, P. L. & Skelly, D. K. Searching for biotic multipliers of climate change. Integr. Comp. Biol. 57, 134–147 (2017).
Jordán, F. Keystone species and food webs. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 364, 1733–1741 (2009).
Allan, B. J. M., Domenici, P., Munday, P. L. & McCormick, M. I. Feeling the heat: the effect of acute temperature changes on predator-prey interactions in coral reef fish. Conserv. Physiol. 3, cov011 (2015).
O’Gorman, E. J. et al. Temperature effects on fish production across a natural thermal gradient. Glob. Chang. Biol. 22, 3206–3220 (2016).
Seebacher, F., White, C. R. & Franklin, C. E. Physiological plasticity increases resilience of ectothermic animals to climate change. Nat. Clim. Chang. 5, 61–66 (2015).
González-Ferreras, A. M. et al. Chronic exposure to environmental temperature attenuates the thermal sensitivity of salmonids. Nat. Commun. 14, 8309 (2023).
Pilakouta, N. et al. Multigenerational exposure to elevated temperatures leads to a reduction in standard metabolic rate in the wild. Funct. Ecol. 34, 1205–1214 (2020).
Sandblom, E. et al. Physiological constraints to climate warming in fish follow principles of plastic floors and concrete ceilings. Nat. Commun. 7, 1–8 (2016).
Sohlström, E. H. et al. Thermal acclimation increases the stability of a predator–prey interaction in warmer environments. Glob. Chang. Biol. 27, 3765–3778 (2021).
Román-Palacios, C. & Wiens, J. J. Recent responses to climate change reveal the drivers of species extinction and survival. Proc. Natl. Acad. Sci. USA 117, 4211–4217 (2020).
Horowitz, M. Heat acclimation: phenotypic plasticity and cues to the underlying molecular mechanisms. J. Therm. Biol. 26, 357–363 (2001).
Bautista, N. M. & Crespel, A. Within- and trans-generational environmental adaptation to climate change: perspectives and new challenges. Front. Mar. Sci. 8, 729194 (2021).
Rabaiotti, D. & Woodroffe, R. Coping with climate change: limited behavioral responses to hot weather in a tropical carnivore. Oecologia 189, 587–599 (2019).
Ficke, A. D., Myrick, C. A. & Hansen, L. J. Potential impacts of global climate change on freshwater fisheries. Rev. Fish. Biol. Fish. 17, 581–613 (2007).
Pörtner, H. O., Schulte, P. M., Wood, C. M. & Schiemer, F. Niche dimensions in fishes: an integrative view. Physiol. Biochem. Zool. 83, 808–826 (2010).
Hendry, A. P. & Gonzalez, A. Whither adaptation? Biol. Philos. 23, 673–699 (2008).
Kavanagh, K. D., Haugen, T. O., Gregersen, F., Jernvall, J. & Vøllestad, L. A. Contemporary temperature-driven divergence in a Nordic freshwater fish under conditions commonly thought to hinder adaptation. BMC Evol. Biol. 10, 350 (2010).
Nati, J. J. H. et al. Intraspecific variation in thermal tolerance differs between tropical and temperate fishes. Sci. Rep. 11, 21272 (2021).
McKenzie, D. J. et al. Intraspecific variation in tolerance of warming in fishes. J. Fish. Biol. 98, 1536–1555 (2021).
Munday, P. L. Transgenerational acclimation of fishes to climate change and ocean acidification. F1000Prime Rep. 6, 99 (2014).
Perrier, C. & Charmantier, A. On the importance of time scales when studying adaptive evolution. Evol. Lett. 3, 240–247 (2019).
Dunne, J. A., Saleska, S. R., Fischer, M. L. & Harte, J. Integrating experimental and gradient methods in ecological climate change research. Ecology 85, 904–916 (2004).
O’Gorman, E. J. et al. Climate change and geothermal ecosystems: natural laboratories, sentinel systems, and future refugia. Glob. Chang. Biol. 20, 3291–3299 (2014).
Moffett, E. R., Fryxell, D. C. & Simon, K. S. Multigenerational exposure to increased temperature reduces metabolic rate but increases boldness in Gambusia affinis. Ecol. Evol. 12, e8853 (2022).
Johansson, M. P., Quintela, M. & Laurila, A. Genetic divergence and isolation by thermal environment in geothermal populations of an aquatic invertebrate. J. Evol. Biol. 29, 1701–1712 (2016).
Orsini, L., Vanoverbeke, J., Swillen, I., Mergeay, J. & De Meester, L. Drivers of population genetic differentiation in the wild: Isolation by dispersal limitation, isolation by adaptation and isolation by colonization. Mol. Ecol. 22, 5983–5999 (2013).
Sinclair, B. J. et al. Can we predict ectotherm responses to climate change using thermal performance curves and body temperatures? Ecol. Lett. 19, 1372–1385 (2016).
Réveillon, T., Rota, T., Chauvet, É., Lecerf, A. & Sentis, A. Energetic mismatch induced by warming decreases leaf litter decomposition by aquatic detritivores. J. Anim. Ecol. 91, 1975–1987 (2022).
Pawar, S., Dell, A. I., Savage, V. M. & Knies, J. L. Real versus artificial variation in the thermal sensitivity of biological traits. Am. Nat. 187, E41–E52 (2016).
Fugère, V., Mehner, T. & Chapman, L. J. Impacts of deforestation-induced warming on the metabolism, growth and trophic interactions of an afrotropical stream fish. Funct. Ecol. 32, 1343–1357 (2018).
Alberto-Payet, F., Lassus, R., Isla, A., Daufresne, M. & Sentis, A. Nine years of experimental warming did not influence the thermal sensitivity of metabolic rate in the medaka fish Oryzias latipes. Freshw. Biol. 67, 577–585 (2022).
Elliott, J. M. & Elliott, J. A. Temperature requirements of Atlantic salmon Salmo salar, brown trout Salmo trutta and Arctic charr Salvelinus alpinus: predicting the effects of climate change. J. Fish. Biol. 77, 1793–1817 (2010).
Mayer, N. B., Hinch, S. G. & Eliason, E. J. Thermal tolerance in Pacific salmon: a systematic review of species, populations, life stages and methodologies. Fish Fish. 25, 283–302 (2024).
Gillooly, J. F. et al. Response to Clarke and Fraser: effects of temperature on metabolic rate. Funct. Ecol. 20, 400–404 (2006).
Cloyed, C. S., Dell, A. I., Hayes, T., Kordas, R. L. & O’Gorman, E. J. Long-term exposure to higher temperature increases the thermal sensitivity of grazer metabolism and movement. J. Anim. Ecol. 88, 833–844 (2019).
Kontopoulos, D. G., Smith, T. P., Barraclough, T. G. & Pawar, S. Adaptive evolution shapes the present-day distribution of the thermal sensitivity of population growth rate. PLoS Biol. 18, e3000894 (2020).
Moffett, E. R., Fryxell, D. C., Palkovacs, E. P., Kinnison, M. T. & Simon, K. S. Local adaptation reduces the metabolic cost of environmental warming. Ecology 99, 2318–2326 (2018).
King, N. G. et al. Evidence for different thermal ecotypes in range centre and trailing edge kelp populations. J. Exp. Mar. Biol. Ecol. 514–515, 10–17 (2019).
Kenkel, C. D. et al. Evidence for a host role in thermotolerance divergence between populations of the mustard hill coral (Porites astreoides) from different reef environments. Mol. Ecol. 22, 4335–4348 (2013).
Barret, L., Miron, G., Ouellet, P. & Tremblay, R. Settlement behavior of American lobster (Homarus americanus): effect of female origin and developmental temperature. Fish. Oceanogr. 26, 69–82 (2017).
Austin, C. J. & Moehring, A. J. Local thermal adaptation detected during multiple life stages across populations of Drosophila melanogaster. J. Evol. Biol. 32, 1342–1351 (2019).
Jonsson, B. & Jonsson, N. Early environment influences later performance in fishes. J. Fish. Biol. 85, 151–188 (2014).
Donelson, J. M., Munday, P. L., McCormick, M. I. & Pitcher, C. R. Rapid transgenerational acclimation of a tropical reef fish to climate change. Nat. Clim. Chang. 2, 20–32 (2012).
Liu, S., Jonsson, B., Greenberg, L. & Hansen, M. M. Limited persistence of temperature-induced methylation compared to significant parental effects in juvenile brown trout (Salmo trutta). Authorea https://doi.org/10.22541/au.174177206.67961270/v1 (2025).
Sánchez-Hernández, J. & Cobo, F. Adaptive flexibility in the feeding behaviour of brown trout: optimal prey size. Zool. Stud. 54, 26 (2015).
Keeley, E. R. & Grant, J. W. A. Prey size of salmonid fishes in streams, lakes, and oceans. Can. J. Fish. Aquat. Sci. 58, 1122–1132 (2001).
Cummins, K. W. & Wuycheck, J. C. Caloric equivalents for investigations in ecological energetics. Int. Ver. Theor. Angew. Limnol. Mitt. 18, 1–158 (1971).
Wotton, R. S. Feeding by blackfly larvae (Diptera: Simuliidae) forming dense aggregations at lake outlets. Freshw. Biol. 27, 139–149 (1992).
Schmitz, O. Predator and prey functional traits: understanding the adaptive machinery driving predator–prey interactions. F1000Res 6, 1767 (2017).
Gilbert, B. et al. A bioenergetic framework for the temperature dependence of trophic interactions. Ecol. Lett. 17, 902–914 (2014).
Dell, A. I., Pawar, S. & Savage, V. M. Temperature dependence of trophic interactions are driven by asymmetry of species responses and foraging strategy. J. Anim. Ecol. 83, 70–84 (2014).
Meehan, M. L. & Lindo, Z. Mismatches in thermal performance between ectothermic predators and prey alter interaction strength and top-down control. Oecologia 201, 1005–1015 (2023).
Álvarez-Codesal, S. et al. Thermal mismatches explain consumer–resource dynamics in response to environmental warming. Ecol. Evol. 13, e10179 (2023).
Holling, C. S. Some characteristics of simple types of predation and parasitism. Can. Entomol. 91, 385–398 (1959).
Archer, L. C. et al. Consistent temperature dependence of functional response parameters and their use in predicting population abundance. J. Anim. Ecol. 88, 1670–1683 (2019).
O’Gorman, E. J. et al. Unexpected changes in community size structure in a natural warming experiment. Nat. Clim. Chang. 7, 659–663 (2017).
Barneche, D. R. et al. Warming impairs trophic transfer efficiency in a long-term field experiment. Nature 592, 76–79 (2021).
Sokolova, I. Bioenergetics in environmental adaptation and stress tolerance of aquatic ectotherms: linking physiology and ecology in a multi-stressor landscape. J. Exp. Biol. 224, jeb236802 (2021).
Melián, C. J. et al. Eco-evolutionary Dynamics of Individual-Based Food Webs. Adv. Ecol. Res. 45, 225–268 (2011).
Bozinovic, F. & Naya, D. E. Linking physiology, climate, and species distributional ranges. In Integrative Organismal Biology (eds. Martin, L. B. et al.) 277–290 (John Wiley & Sons, Ltd, 2015).
Buckley, L. B. & Kingsolver, J. G. Evolution of thermal sensitivity in changing and variable climates. Annu. Rev. Ecol. Evol. Syst. 52, 563–586 (2021).
Harding, L., Jackson, A. L. & Payne, N. Energetic costs increase with faster heating in an aquatic ectotherm. Conserv. Physiol. 11, coad042 (2023).
Terblanche, J. S., Nyamukondiwa, C. & Kleynhans, E. Thermal variability alters climatic stress resistance and plastic responses in a globally invasive pest, the Mediterranean fruit fly (Ceratitis capitata). Entomol. Exp. Appl. 137, 304–315 (2010).
Friberg, N. et al. Relationships between structure and function in streams contrasting in temperature. Freshw. Biol. 54, 2051–2068 (2009).
Woodward, G. et al. Sentinel systems on the razor’s edge: effects of warming on Arctic geothermal stream ecosystems. Glob. Change Biol. 16, 1979–1991 (2010).
Kordas, R. L., Pawar, S., Kontopoulos, D. G., Woodward, G. & O’Gorman, E. J. Metabolic plasticity can amplify ecosystem responses to global warming. Nat. Commun. 13, 1–8 (2022).
Arnason, B., Theodorsson, P., Björnsson, S. & Saemundsson, K. Hengill, a high temperature thermal area in Iceland. Bull. Volcanol. 33, 245–259 (1969).
O’Gorman, E. J. et al. Impacts of warming on the structure and function of aquatic communities: individual- to ecosystem-level responses. Adv. Ecol. Res. 47, 81–176 (2012).
Archer, L. C. et al. Metabolic traits in brown trout (Salmo trutta) vary in response to food restriction and intrinsic factors. Conserv. Physiol. 8, coaa096 (2020).
Ikeda, T. Routine metabolic rates of pelagic marine fishes and cephalopods as a function of body mass, habitat temperature and habitat depth. J. Exp. Mar. Biol. Ecol. 480, 74–86 (2016).
Rodgers, G. G., Tenzing, P. & Clark, T. D. Experimental methods in aquatic respirometry: the importance of mixing devices and accounting for background respiration. J. Fish. Biol. 88, 65–80 (2016).
Peters, R. H. The Ecological Implications of Body Size (Cambridge University Press, 1983).
Lang, B., Ehnes, R. B., Brose, U. & Rall, B. C. Temperature and consumer type dependencies of energy flows in natural communities. Oikos 126, 1717–1725 (2017).
Weir, B. S. & Cockerham, C. C. Estimating F-Statistics for the analysis of population structure. Evolution 38, 1358–1370 (1984).
Keenan, K., Mcginnity, P., Cross, T. F., Crozier, W. W. & Prodöhl, P. A. diveRsity: an R package for the estimation and exploration of population genetics parameters and their associated errors. Methods Ecol. Evol. 4, 782–788 (2013).
Jost, L. Partitioning diversity into independent alpha and beta components. Ecology 88, 2427–2439 (2007).
Wang, J. On the measurements of genetic differentiation among populations. Genet. Res. 94, 275–289 (2012).
Rousset, F. GENEPOP’007: a complete re-implementation of the GENEPOP software for Windows and Linux. Mol. Ecol. Resour. 8, 103–106 (2008).
Rousset, F., Lopez, J. & Belkhir, K. Package ‘genepop’. R package version, v 1.2.14 (2020).
Waples, R. S. & Gaggiotti, O. What is a population? An empirical evaluation of some genetic methods for identifying the number of gene pools and their degree of connectivity. Mol. Ecol. 15, 1419–1439 (2006).
Jombart, T. adegenet: a R package for the multivariate analysis of genetic markers. Bioinformatics 24, 1403–1405 (2008).
O’Gorman, E. J. et al. Warming effects on trout energetic efficiency. [Data Collection]. Colchester, University of Essex. https://doi.org/10.5526/ERDR-00000243 (2026).
Robinson, S. I., McLaughlin, ÓB., Marteinsdóttir, B. & O’Gorman, E. J. Soil temperature effects on the structure and diversity of plant and invertebrate communities in a natural warming experiment. J. Anim. Ecol. 87, 634–646 (2018).
Robinson, S. I., Mikola, J., Ovaskainen, O. & O’Gorman, E. J. Temperature effects on the temporal dynamics of a subarctic invertebrate community. J. Anim. Ecol. 90, 1217–1227 (2021).
Acknowledgements
We thank Gísli Már Gíslason, Jón S. Ólafsson, Guðni Guðbergsson and Sigriður Ásgeirsdóttir for providing research support and facilities. We acknowledge the funding support of NERC (NE/L011840/1, NE/M020843/1), the Royal Society (RG140601), Imperial College London, the Government of Cantabria through the Fénix Programme, grant RYC2023-045780-I funded by MICIU/AEI/10.13039/501100011033 and ESF +, the European Research Council (ERC Starting Grant 639192), and Science Foundation Ireland (SFI/15/IA/3028).
Author information
Authors and Affiliations
Contributions
E.J.O. secured funding and designed the study. E.J.O., P.S.A.B., and J.H. conducted the fieldwork. E.J.O. and A.M.G.F. analysed the data. J.C., K.P.P., P.M., and T.E.R. conducted the genetics analyses. E.J.O. and A.M.G.F. wrote the first draft of the paper. All authors edited the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests. E.J.O. is an Editorial Board Member for Communications Biology, but was not involved in the editorial review of, nor the decision to publish this article.
Peer review
Peer review information
Communications Biology thanks the anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editors: Johannes Stortz. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
O’Gorman, E.J., González-Ferreras, A.M., Blyth, P.S.A. et al. Brown trout (Salmo trutta) originating from warmer streams in Iceland exhibit increased energetic efficiency. Commun Biol (2026). https://doi.org/10.1038/s42003-026-09911-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-09911-5