Abstract
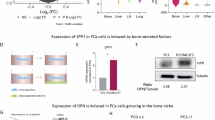
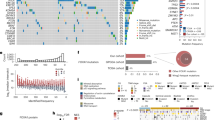
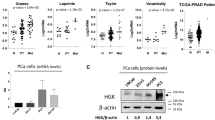
Bone metastases represent a critical phenotype of prostate cancer progression, driven by factors within the bone microenvironment. However, the molecular mechanisms underlying this progression remain poorly understood. In this study, we observed a significant accumulation of single-stranded DNA within the metastatic bone microenvironment of PCa patients. Through cell-SELEX methodology, we identified a PCa target-specific ssDNA, EHBP1. Specifically, EHBP1-ssDNA specifically captures PCa cells by binding to the transmembrane protein integrin α6, which subsequently activates the integrin α6-FAK signaling pathway. Functional studies revealed that knockdown of integrin-α6 expression effectively abrogated EHBP1-ssDNA mediated PCa bone metastatic capacity. Notably, these findings were recapitulated through pharmacological inhibition of FAK signaling using Defactinib, an FAK-specific inhibitor. Taken together, our findings reveal that bone-marrow ssDNA may represent a bone microenvironment factor that captures and promotes PCa homing to bone, further suggesting a potential therapeutic strategy for mitigating bone metastasis.

Similar content being viewed by others
Data availability
The data that support the findings of this study are available in Supplementary Data. The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE partner repository with the dataset identifier PXD074143 (Reviewer username: reviewer_pxd074143@ebi.ac.uk, password: 71smTS4HnxaP). Unedited blot/gel images can be seen in the Supplementary Fig. 1. The gating strategy of flow cytometry can be seen in Supplementary Fig. 2.
References
Siegel, R. L., Giaquinto, A. N. & Jemal, A. Cancer statistics, 2024. CA Cancer J. Clin. 74, 12–49 (2024).
Sung, H. et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71, 209–249 (2021).
Berish, R. B., Ali, A. N., Telmer, P. G., Ronald, J. A. & Leong, H. S. Translational models of prostate cancer bone metastasis. Nat. Rev. Urol. 15, 403–421 (2018).
Swami, U., McFarland, T. R., Nussenzveig, R. & Agarwal, N. Advanced prostate cancer: treatment advances and future directions. Trends Cancer 6, 702–715 (2020).
Ge, R. et al. Epigenetic modulations and lineage plasticity in advanced prostate cancer. Ann. Oncol. 31, 470–479 (2020).
Lang, C. et al. m(6) A modification of lncRNA PCAT6 promotes bone metastasis in prostate cancer through IGF2BP2-mediated IGF1R mRNA stabilization. Clin. Transl. Med. 11, e426 (2021).
Kang, J. et al. Tumor microenvironment mechanisms and bone metastatic disease progression of prostate cancer. Cancer Lett. 530, 156–169 (2022).
Fornetti, J., Welm, A. L. & Stewart, S. A. Understanding the bone in cancer metastasis. J. Bone Min. Res. 33, 2099–2113 (2018).
Kong, D. et al. Procoxacin bidirectionally inhibits osteoblastic and osteoclastic activity in bone and suppresses bone metastasis of prostate cancer. J. Exp. Clin. Cancer Res. 42, 45 (2023).
Yoneda, T. & Hiraga, T. Crosstalk between cancer cells and bone microenvironment in bone metastasis. Biochem. Biophys. Res. Commun. 328, 679–687 (2005).
Price, T. T. et al. Dormant breast cancer micrometastases reside in specific bone marrow niches that regulate their transit to and from bone. Sci. Transl. Med. 8, 340ra73 (2016).
Robinson, D. et al. Integrative clinical genomics of advanced prostate cancer. Cell 161, 1215–1228 (2015).
Zhou, Q. et al. Differentially expressed proteins identified by TMT proteomics analysis in bone marrow microenvironment of osteoporotic patients. Osteoporos. Int. 30, 1089–1098 (2019).
Feeley, B. T. et al. Influence of BMPs on the formation of osteoblastic lesions in metastatic prostate cancer. J. Bone Min. Res. 20, 2189–2199 (2005).
Gupta, N. et al. Clinical significance of circulatory microRNA-203 in serum as novel potential diagnostic marker for multiple myeloma. J. Cancer Res. Clin. Oncol. 145, 1601–1611 (2019).
Rostami, A. et al. Senescence, necrosis, and apoptosis govern circulating cell-free DNA release kinetics. Cell Rep. 31, 107830 (2020).
Lo, Y. M. D., Han, D. S. C., Jiang, P. & Chiu, R. W. K. Epigenetics, fragmentomics, and topology of cell-free DNA in liquid biopsies. Science 372, 6538 (2021).
Cheng, J. et al. Plasma contains ultrashort single-stranded DNA in addition to nucleosomal cell-free DNA. iScience 25, 104554 (2022).
Cheng, L. Y., Dai, P., Wu, L. R., Patel, A. A. & Zhang, D. Y. Direct capture and sequencing reveal ultra-short single-stranded DNA in biofluids. iScience 25, 105046 (2022).
Kordasht, H. K. & Hasanzadeh, M. Aptamer based recognition of cancer cells: Recent progress and challenges in bioanalysis. Talanta 220, 121436 (2020).
Schneider, A. et al. Bone turnover mediates preferential localization of prostate cancer in the skeleton. Endocrinology 146, 1727–1736 (2005).
Jiang, S. et al. Generic Diagramming Platform (GDP): a comprehensive database of high-quality biomedical graphics. Nucleic Acids Res. 53, D1670–d6 (2025).
Nakhjavani, M. et al. A flow cytometry-based cell surface protein binding assay for assessing selectivity and specificity of an anticancer aptamer. J. Vis. Exp. 187, e64304 (2022).
Kelly, L., Maier, K. E., Yan, A. & Levy, M. A comparative analysis of cell surface targeting aptamers. Nat. Commun. 12, 6275 (2021).
Li, Y. J. et al. YBX1 promotes type H vessel-dependent bone formation in an m5C-dependent manner. JCI Insight 9, 4 (2024).
Li, C. J. et al. Senescent immune cells release grancalcin to promote skeletal aging. Cell Metab. 33, 1957–1973.e6 (2021).
Xiao, Y. et al. Splicing factor YBX1 regulates bone marrow stromal cell fate during aging. EMBO J. 42, e111762 (2023).
Li, C. J. et al. Long noncoding RNA Bmncr regulates mesenchymal stem cell fate during skeletal aging. J. Clin. Investig. 128, 5251–5266 (2018).
Peng, H. et al. A mechanosensitive lipolytic factor in the bone marrow promotes osteogenesis and lymphopoiesis. Cell Metab. 34, 1168–1182.e6 (2022).
Feng, X. et al. Senescent immune cells accumulation promotes brown adipose tissue dysfunction during aging. Nat. Commun. 14, 3208 (2023).
van Bokhoven, A. et al. Molecular characterization of human prostate carcinoma cell lines. Prostate 57, 205–225 (2003).
Liu, C. W. et al. Resistin stimulates PC-3 prostate cancer cell growth through stimulation of SOCS3 and SOCS5 genes. Exp. Biol. Med. 248, 1695–1707 (2023).
Wengerter, B. C. et al. Aptamer-targeted antigen delivery. Mol. Ther. 22, 1375–1387 (2014).
Camorani, S. et al. Aptamer targeted therapy potentiates immune checkpoint blockade in triple-negative breast cancer. J. Exp. Clin. Cancer Res. 39, 180 (2020).
Xu, J., Ma, L., Wang, D. & Yang, J. Uncarboxylated osteocalcin promotes proliferation and metastasis of MDA-MB-231 cells through TGF-β/SMAD3 signaling pathway. BMC Mol. Cell Biol. 23, 18 (2022).
Panet, F. et al. PACE4 is an important driver of ZR-75-1 estrogen receptor-positive breast cancer proliferation and tumor progression. Eur. J. Cell Biol. 96, 469–475 (2017).
Liu, Z. J., Lee, W. J. & Zhu, B. T. Selective insensitivity of ZR-75-1 human breast cancer cells to 2-methoxyestradiol: evidence for type II 17beta-hydroxysteroid dehydrogenase as the underlying cause. Cancer Res. 65, 5802–5811 (2005).
de Pereda, J. M., Lillo, M. P. & Sonnenberg, A. Structural basis of the interaction between integrin alpha6beta4 and plectin at the hemidesmosomes. EMBO J. 28, 1180–1190 (2009).
Koivusalo, S., Schmidt, A., Manninen, A. & Wenta, T. Regulation of kinase signaling pathways by α6β4-integrins and plectin in prostate cancer. Cancers 15, 149 (2022).
Jiang, Q. et al. Lunasin suppresses the migration and invasion of breast cancer cells by inhibiting matrix metalloproteinase-2/-9 via the FAK/Akt/ERK and NF-κB signaling pathways. Oncol. Rep. 36, 253–262 (2016).
Deng, Z. et al. Disintegrin tablysin-15 suppresses cancer hallmarks in melanoma cells by blocking FAK/Akt/ERK and NF-κB signaling. Curr. Cancer Drug Targets 20, 306–315 (2020).
Calalb, M. B., Polte, T. R. & Hanks, S. K. Tyrosine phosphorylation of focal adhesion kinase at sites in the catalytic domain regulates kinase activity: a role for Src family kinases. Mol. Cell. Biol. 15, 954–963 (1995).
Hu, Z. et al. Histone deacetylase inhibitors promote breast cancer metastasis by elevating NEDD9 expression. Signal Transduct. Target. Ther. 8, 11 (2023).
Clark, P. E. & Torti, F. M. Prostate cancer and bone metastases: medical treatment. Clin. Orthop. Relat. Res. 415, S148–S157 (2003).
Clézardin, P. et al. Bone metastasis: mechanisms, therapies, and biomarkers. Physiol. Rev. 101, 797–855 (2021).
Sun, X. et al. Elucidation of CKAP4-remodeled cell mechanics in driving metastasis of bladder cancer through aptamer-based target discovery. Proc. Natl. Acad. Sci. USA 119, e2110500119 (2022).
Bates, P. J., Kahlon, J. B., Thomas, S. D., Trent, J. O. & Miller, D. M. Antiproliferative activity of G-rich oligonucleotides correlates with protein binding. J. Biol. Chem. 274, 26369–26377 (1999).
Bates, P. J., Laber, D. A., Miller, D. M., Thomas, S. D. & Trent, J. O. Discovery and development of the G-rich oligonucleotide AS1411 as a novel treatment for cancer. Exp. Mol. Pathol. 86, 151–164 (2009).
Dassie, J. P. et al. Targeted inhibition of prostate cancer metastases with an RNA aptamer to prostate-specific membrane antigen. Mol. Ther. 22, 1910–1922 (2014).
Yang, L. et al. DNA of neutrophil extracellular traps promotes cancer metastasis via CCDC25. Nature 583, 133–138 (2020).
Hawes, M. C., Wen, F. & Elquza, E. Extracellular DNA: a bridge to cancer. Cancer Res. 75, 4260–4264 (2015).
Lin, E. et al. Identification of somatic gene signatures in circulating cell-free DNA associated with disease progression in metastatic prostate cancer by a novel machine learning platform. Oncologist 26, 751–760 (2021).
Gourdin, T. & Sonpavde, G. Utility of cell-free nucleic acid and circulating tumor cell analyses in prostate cancer. Asian J. Androl. 20, 230–237 (2018).
Ribeiro, J., Abby, E., Livera, G. & Martini, E. RPA homologs and ssDNA processing during meiotic recombination. Chromosoma 125, 265–276 (2016).
Lam, F. C., Kong, Y. W. & Yaffe, M. B. Inducing DNA damage through R-loops to kill cancer cells. Mol. Cell. Oncol. 8, 1848233 (2020).
Schneider, J. G., Amend, S. R. & Weilbaecher, K. N. Integrins and bone metastasis: integrating tumor cell and stromal cell interactions. Bone 48, 54–65 (2011).
Soung, Y. H., Gil, H. J., Clifford, J. L. & Chung, J. Role of α6β4 integrin in cell motility, invasion and metastasis of mammary tumors. Curr. Protein Pept. Sci. 12, 23–29 (2011).
Tagliabue, E. et al. Prognostic value of alpha 6 beta 4 integrin expression in breast carcinomas is affected by laminin production from tumor cells. Clin. Cancer Res. 4, 407–410 (1998).
Tennenbaum, T. et al. The suprabasal expression of alpha 6 beta 4 integrin is associated with a high risk for malignant progression in mouse skin carcinogenesis. Cancer Res. 53, 4803–4810 (1993).
Russell, A. J. et al. Alpha 6 beta 4 integrin regulates keratinocyte chemotaxis through differential GTPase activation and antagonism of alpha 3 beta 1 integrin. J. Cell Sci. 17, 3543–3556 (2003). 116.
Shaw, L. M., Rabinovitz, I., Wang, H. H., Toker, A. & Mercurio, A. M. Activation of phosphoinositide 3-OH kinase by the alpha6beta4 integrin promotes carcinoma invasion. Cell 91, 949–960 (1997).
Song, X. et al. Focal adhesion kinase (FAK) promotes cholangiocarcinoma development and progression via YAP activation. J. Hepatol. 75, 888–899 (2021).
Jeong, K., Murphy, J. M., Ahn, E. E. & Lim, S. S. FAK in the nucleus prevents VSMC proliferation by promoting p27 and p21 expression via Skp2 degradation. Cardiovasc. Res. 118, 1150–1163 (2022).
Alanko, J. & Ivaska, J. Endosomes: emerging platforms for integrin-mediated FAK signalling. Trends Cell Biol. 26, 391–398 (2016).
Alanko, J. et al. Integrin endosomal signalling suppresses anoikis. Nat. Cell Biol. 17, 1412–1421 (2015).
Gao, S. et al. Role of overexpression of MACC1 and/or FAK in predicting prognosis of hepatocellular carcinoma after liver transplantation. Int. J. Med. Sci. 11, 268–275 (2014).
Wang-Gillam, A. et al. Defactinib, pembrolizumab, and gemcitabine in patients with advanced treatment refractory pancreatic cancer: a Phase I dose escalation and expansion study. Clin. Cancer Res. 28, 5254–5262 (2022).
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (Grant No.82471619) and the Natural Science Foundation of Hunan Province (Grant No.2024JJ5459).
Author information
Authors and Affiliations
Contributions
X.C. and Y.J.-L. performed the major experiments and analyzed overall experimental data. Y.J.-L. and M.S.-Y. revised the manuscript. M.S.-Y. and Z.L.-P. assisted with the overall experiments and interpreted data. F.Y. guided the animal experiment of left ventricular injection in mice. Y.X. and Y.X. designed the experiments and revised the manuscript. Y.X. designed the experiments, curated data, and acquired funding.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Brian Thomas, Raffaella Gallo, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Johannes Stortz. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, X., Ye, Ms., Peng, ZL. et al. Single-stranded DNA in the bone microenvironment promotes prostate cancer bone metastasis via the ITGA6-FAK pathway. Commun Biol (2026). https://doi.org/10.1038/s42003-026-09929-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-09929-9