Abstract
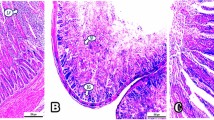
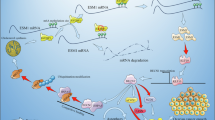
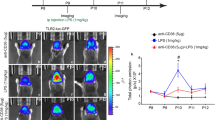
Exposure to zearalenone (ZEN), an estrogen-like mycotoxin produced by Fusarium fungi, impairs reproductive performance and farm-animal welfare; however, the underlying mechanism remains unclear. Using female pigs as the large animal model, we demonstrate that ZEN-induced reproductive lipotoxicity is associated with alterations in estrogen receptor-alpha (ERα) expression, diacylglycerol levels, and pro-inflammatory cytokine production. Mechanistically, upregulated ERα directly binds to the promoter regions of Cd36 and Tlr4 in the nucleus, leading to increased expression of the lipid transporter CD36 and the inflammatory receptor TLR4, as well as enhanced CD36-TLR4 interaction. This subsequently results in diacylglycerol accumulation and inflammation. Consistently, studies using Cd36-knockout mice and the TLR4-specific inhibitor TAK-242 reveal that ZEN-induced reproductive lipotoxicity is dependent on the ERα-CD36/TLR4 axis. Furthermore, dietary supplementation with the ZEN-degrading enzyme ZLHY-6 in female pigs also counteracts ZEN-induced reproductive lipotoxicity through the inhibition of the ERα-CD36/TLR4 signaling pathway. Together, these findings reveal a novel mechanism underlying ZEN-induced reproductive lipotoxicity, and highlight the ERα-CD36/TLR4 axis as a potential therapeutic target.

Similar content being viewed by others
Data availability
The RNA-seq dataset generated and analyzed in this study is publicly available for download in the National Center for Biotechnology Information Gene Expression Omnibus (BioProject ID: PRJNA1136154). The data that support the findings of this study are available from the corresponding author upon reasonable request. The internal standards for lipidomics are provided in Supplementary Data 1. The data source for all Graphs in this study is provided in Supplementary Data 2. Uncropped and unedited blot are provided in Supplementary Information as Supplementary Fig. 9.
References
Eskola, M. et al. Worldwide contamination of food-crops with mycotoxins: validity of the widely cited ‘FAO estimate’of 25%. Crit. Rev. Food Sci. Nutr. 60, 2773–2789 (2020).
Liu, J. & Applegate, T. Zearalenone (ZEN) in Livestock and Poultry: Dose, toxicokinetics, toxicity and estrogenicity. Toxins 12, 377 (2020).
Wang, J. et al. Mycotoxin contamination in organic and conventional cereal grain and products: a systematic literature review and meta-analysis. Compr. Rev. Food Sci. Food Saf. 23, e13363 (2024).
Awuchi, C. G. et al. Mycotoxins affecting animals, foods, humans, and plants: types, occurrence, toxicities, action mechanisms, prevention, and detoxification strategies—A revisit. Foods 10, 1279 (2021).
Kowalska, K., Habrowska-Górczyńska, D. E. & Piastowska-Ciesielska, A. W. Zearalenone as an endocrine disruptor in humans. Environ. Toxicol. Pharm. 48, 141–149 (2016).
Wang, S. et al. Zearalenone disturbs the reproductive-immune axis in pigs: the role of gut microbial metabolites. Microbiome 10, 1–24 (2022).
Binder, S. B. et al. Metabolism of zearalenone and its major modified forms in pigs. Toxins 9, 56 (2017).
Rai, A., Das, M. & Tripathi, A. Occurrence and toxicity of a Fusarium mycotoxin, zearalenone. Crit. Rev. Food Sci. Nutr. 60, 2710–2729 (2020).
Bai, J. et al. Roles of stress response-related signaling and its contribution to the toxicity of zearalenone in mammals. Compr. Rev. Food Sci. Food Saf. 21, 3326–3345 (2022).
Ropejko, K. & Twarużek, M. Zearalenone and its metabolites—general overview, occurrence, and toxicity. Toxins 13, 35 (2021).
Fu, W. et al. Enhanced glutathione production protects against zearalenone-induced oxidative stress and ferroptosis in female reproductive system. Food Chem. Toxicol. 185, 114462 (2024).
Gao, X. et al. Dietary silymarin supplementation alleviates zearalenone-induced hepatotoxicity and reproductive toxicity in rats. J. Nutr. 148, 1209–1216 (2018).
Drouault, M. et al. Deoxynivalenol enhances estrogen receptor alpha-induced signaling by ligand-independent transactivation. Food Chem. Toxicol. 165, 113127 (2022).
Björnström, L. & Sjöberg, M. Mechanisms of estrogen receptor signaling: convergence of genomic and nongenomic actions on target genes. Mol. Endocrinol. 19, 833–842 (2005).
Zhao, F. et al. Multigenerational exposure to dietary zearalenone (ZEA), an estrogenic mycotoxin, affects puberty and reproduction in female mice. Reprod. Toxicol. 47, 81–88 (2014).
Zatecka, E. et al. Effect of zearalenone on reproductive parameters and expression of selected testicular genes in mice. Reprod. Toxicol. 45, 20–30 (2014).
Zhou, J. et al. Zearalenone toxicosis on reproduction as estrogen receptor selective modulator and alleviation of zearalenone biodegradative agent in pregnant sows. J. Anim. Sci. Biotechnol. 13, 36 (2022).
González-Alvarez, M. E., McGuire, B. C. & Keating, A. F. Obesity alters the ovarian proteomic response to zearalenone exposure. Biol. Reprod. 105, 278–289 (2021).
Dai, C. et al. Increased NAD+ levels protect female mouse reproductive system against zearalenone-impaired glycolysis, lipid metabolism, antioxidant capacity and inflammation. Reprod. Toxicol. 124, 108530 (2024).
Balló, A. et al. Estrogenic and non-estrogenic disruptor effect of zearalenone on male reproduction: a review. Int. J. Mol. Sci. 24, 1578 (2023).
Lancaster, G. I. et al. Evidence that TLR4 is not a receptor for saturated fatty acids but mediates lipid-induced inflammation by reprogramming macrophage metabolism. Cell Metab. 27, 1096–1110 (2018).
Chang, X. et al. Zearalenone removal from corn oil by an enzymatic strategy. Toxins 12, 117 (2020).
Yang, C. et al. Betulinic acid alleviates zearalenone-induced uterine injury in mice. Environ. Pollut. 316, 120435 (2023).
Pavao, M. & Traish, A. M. Estrogen receptor antibodies: specificity and utility in detection, localization and analyses of estrogen receptor α and β. Steroids 66, 1–16 (2001).
Adibnia, E., Razi, M. & Malekinejad, H. Zearalenone and 17 β-estradiol induced damages in male rats reproduction potential; evidence for ERα and ERβ receptors expression and steroidogenesis. Toxicon 120, 133–146 (2016).
Metzler, M., Pfeiffer, E. & Hildebrand, A. Zearalenone and its metabolites as endocrine disrupting chemicals. World Mycotoxin J. 3, 385–401 (2010).
Wang, S. et al. Glycolysis-mediated activation of v-ATPase by nicotinamide mononucleotide ameliorates lipid-induced cardiomyopathy by repressing the CD36-TLR4 axis. Circ. Res. 134, 505–525 (2024).
Stewart, C. R. et al. CD36 ligands promote sterile inflammation through assembly of a Toll-like receptor 4 and 6 heterodimer. Nat. Immunol. 11, 155–161 (2010).
Lu, H. et al. Butyrate-producing Eubacterium rectale suppresses lymphomagenesis by alleviating the TNF-induced TLR4/MyD88/NF-κB axis. Cell Host Microbe 30, 1139–1150 (2022).
Sun, H., He, Z., Xiong, D. & Long, M. Mechanisms by which microbial enzymes degrade four mycotoxins and application in animal production: a review. Anim. Nutr. 15, 256–274 (2023).
Yang, W. C., Hsu, T. C., Cheng, K. C. & Liu, J. R. Expression of the clonostachys rosea lactonohydrolase gene by Lactobacillus reuteri to increase its zearalenone-removing ability. Environ. Pollut. 16, 1–11 (2017).
Takahashi-Ando, N., Kimura, M., Kakeya, H., Osada, H. & Yamaguchi, I. A novel lactonohydrolase responsible for the detoxification of zearalenone: enzyme purification and gene cloning. Biochem. J. 365, 1–6 (2002).
Bi, K., Zhang, W., Xiao, Z. & Zhang, D. Characterization, expression and application of a zearalenone degrading enzyme from Neurospora crassa. AMB Express 8, 1–10 (2018).
De Clercq K., Hennes A. & Vriens J. Isolation of mouse endometrial epithelial and stromal cells for in vitro decidualization. J. Vis. Exp. 121, 55168 (2017).
Wang, S., Wang, B., He, H., Sun, A. & Guo, C. A new set of reference housekeeping genes for the normalization RT-qPCR data from the intestine of piglets during weaning. PloS ONE 13, e0204583 (2018).
Zhong, X. et al. Microbial-driven butyrate regulates jejunal homeostasis in piglets during the weaning stage. Front. Microbiol. 9, 3335 (2019).
Acknowledgements
The work was supported by grants from the Strategic Priority Research Program of the Chinese Academy of Sciences (No. XD C0110304 to X.W), the National Key Research and Development Program of China (2023YFF1104600 to C.P.S), the National Natural Science Foundation of China (No. 32571297 to S.J.W and No. 82300970 to Y.L), the Sichuan Science and Technology Program (No. 2025ZNSFSC0170 to W.F).
Author information
Authors and Affiliations
Contributions
Chao Dai: Writing– original draft, Visualization, Validation, Methodology. Wei Fu: Writing– review & editing, Software, Methodology, Formal analysis, Data curation. YuanMeng Huang: Writing– review & editing, Formal analysis, Resources, Methodology. Tiantian Li: Formal analysis, Resources, Methodology. Zifeng Ma: Formal analysis, Methodology. Xiaojiao Chang: Data curation, Methodology. Changpo Sun: Resources, Project administration. Shujin Wang: Writing– original draft & review & editing, Resources, Funding acquisition, Software, Supervision, Conceptualization. Xin Wu: Writing– review & editing, Resources, Funding acquisition. Yang Li: Writing–review & editing, Resources, Project administration, Funding acquisition.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Compliance with Ethics Requirements
All Institutional and National Guidelines for the care and use of animals (fisheries) were followed. The animal experiments in this study received approval from the Animal Care and Use Committee at Chongqing Medical University (approval number: 2019201) and the Animal Welfare Committee of the Institute of Subtropical Agriculture, Chinese Academy of Sciences (approval number: 20160615-013).
Peer review
Peer review information
Communications Biology thanks the anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editors: Rupinder Kaur and Dario Ummarino. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Dai, C., Fu, W., Huang, Y. et al. Zearalenone causes female reproductive lipotoxicity through the ERα-CD36/TLR4 signaling pathway. Commun Biol (2026). https://doi.org/10.1038/s42003-026-09953-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-09953-9