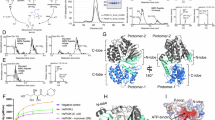
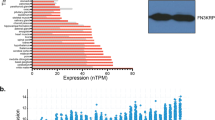
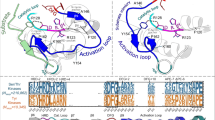
Abstract
Protein glycation is a detrimental byproduct of carbohydrate metabolism, and nearly all organisms encode kinases that facilitate the removal of early glycation products. In humans, these repair functions are performed by Fructosamine-3 and Ketosamine-3 kinases (FN3K, KT3K). Recent structural studies identified conserved residues essential for FN3K activity, but the molecular basis for substrate discrimination between FN3K and KT3K remains unknown. Here, we show that substrate-binding residues are highly conserved and do not confer specificity in the FN3K family. Using APR, we resurrected four ancestral fructosamine kinases that recapitulate the distinct substrate preferences of FN3K and KT3K. Through mutational studies and structural analysis, we identify an evolutionarily tuned allosteric network that modulates substrate selection through long-range intramolecular interactions. Our findings define the mechanism of substrate selectivity within the FN3K family and establish a framework for the development of selective FN3K inhibitors.
Similar content being viewed by others
Data availability
All data supporting the findings of this study are available within the paper and its Supplementary Information. Ancestral reconstruction and plasmid sequence data are available through Zenodo (https://doi.org/10.5281/zenodo.17965681). Uncropped blot images can be found in Supplementary Fig. 11. Experimental data generated in this study is available in Supplementary Data. Previously published structures used in this study can be accessed using PDB codes 8UE1 and 9CX8.
References
Ahmed, M. U., Thorpe, S. R. & Baynes, J. W. Identification of N epsilon-carboxymethyllysine as a degradation product of fructoselysine in glycated protein. J. Biol. Chem. 261, 4889–4894 (1986).
Teerlink, T., Barto, R., Ten Brink, H. J. & Schalkwijk, C. G. Measurement of Nε-(Carboxymethyl)lysine and Nε-(Carboxyethyl)lysine in human plasma protein by stable-isotope-dilution tandem mass spectrometry. Clin. Chem. 50, 1222–1228 (2004).
Krook, M., Ghosh, D., Strömberg, R., Carlquist, M. & Jörnvall, H. Carboxyethyllysine in a protein: native carbonyl reductase/NADP(+)-dependent prostaglandin dehydrogenase. Proc. Natl. Acad. Sci. USA 90, 502–506 (1993).
Sell, D. R., Lapolla, A., Odetti, P., Fogarty, J. & Monnier, V. M. Pentosidine formation in skin correlates with severity of complications in individuals with long-standing IDDM. Diabetes 41, 1286–1292 (1992).
Nagaraj, R. H., Shipanova, I. N. & Faust, F. M. Protein cross-linking by the Maillard reaction. Isolation, characterization, and in vivo detection of a lysine-lysine cross-link derived from methylglyoxal. J. Biol. Chem. 271, 19338–19345 (1996).
Martin, M. S., Jacob-Dolan, J. W., Pham, V. T. T., Sjoblom, N. M. & Scheck, R. A. The chemical language of protein glycation. Nat. Chem. Biol. https://doi.org/10.1038/S41589-024-01644-Y (2024).
Schmidt, A. M. et al. Rage: a novel cellular receptor for advanced glycation end products. Diabetes 45, S77–S80 (1996).
Laughlin, T. et al. Autophagy activators stimulate the removal of advanced glycation end products in human keratinocytes. J. Eur. Acad. Dermatol. Venereol. 34, 12–18 (2020).
Zawada, A. et al. Accumulation of advanced glycation end-products in the body and dietary habits. Nutrients 14, 3982 (2022).
Delpierre, G. et al. Identification, cloning, and heterologous expression of a mammalian fructosamine-3-kinase. Diabetes 49, 1627–1634 (2000).
Collard, F., Delpierre, G., Stroobant, V., Matthijs, G. & Van Schaftingen, E. A mammalian protein homologous to fructosamine-3-kinase is a ketosamine-3-kinase acting on psicosamines and ribulosamines but not on fructosamines. Diabetes 52, 2888–2895 (2003).
Fortpied, J., Gemayel, R., Stroobant, V. & Van Schaftingen, E. Plant ribulosamine/erythrulosamine 3-kinase, a putative protein-repair enzyme. Biochem. J. 388, 795–802 (2005).
Gemayel, R. et al. Many fructosamine 3-kinase homologues in bacteria are ribulosamine/erythrulosamine 3-kinases potentially involved in protein deglycation. FEBS J. 274, 4360–4374 (2007).
Wu, S., Lu, H. & Bai, Y. Nrf2 in cancers: a double-edged sword. Cancer Med. 8, 2252 (2019).
Huang, Y., Li, W., Su, Z. Y. & Kong, A. N. The complexity of the Nrf2 pathway: beyond the antioxidant response. J. Nutr. Biochem. 26, 1401–1413 (2015).
Sanghvi, V. R. et al. The oncogenic action of NRF2 depends on de-glycation by fructosamine-3-kinase. Cell 178, 807–819.e21 (2019).
Garg, A. et al. The molecular basis of Human FN3K mediated phosphorylation of glycated substrates. Nat. Commun. 16, 1–14 (2025).
Lokhandwala, J. et al. Structural basis for FN3K-mediated protein deglycation. Structure 32, 1711–1724.e5 (2024).
Harms, M. J. & Thornton, J. W. Analyzing protein structure and function using ancestral gene reconstruction. Curr. Opin. Struct. Biol. 20, 360–366 (2010).
Thornton, J. W. Resurrecting ancient genes: experimental analysis of extinct molecules. Nat. Rev. Genet 5, 366–375 (2004).
Trudeau, D. L., Kaltenbach, M. & Tawfik, D. S. On the potential origins of the high stability of reconstructed ancestral proteins. Mol. Biol. Evol. 33, 2633–2641 (2016).
Taylor, S. S. & Kornev, A. P. Protein kinases: evolution of dynamic regulatory proteins. Trends Biochem. Sci. 36, 65–77 (2011).
Kornev, A. P. & Taylor, S. S. Dynamics driven allostery in protein kinases. Trends Biochem. Sci. 40, 628 (2015).
Shim, G. seong, Manandhar, S., Shin, D. ha, Kim, T. H. & Kwak, M. K. Acquisition of doxorubicin resistance in ovarian carcinoma cells accompanies activation of the NRF2 pathway. Free Radic. Biol. Med. 47, 1619–1631 (2009).
Hayden, A. et al. The Nrf2 transcription factor contributes to resistance to cisplatin in bladder cancer. Urol. Oncol. 32, 806–814 (2014).
Niture, S. K. & Jaiswal, A. K. Nrf2-induced antiapoptotic Bcl-xL protein enhances cell survival and drug resistance. Free Radic. Biol. Med. 57, 119–131 (2013).
Wu, J., Zhang, L., Li, H., Wu, S. & Liu, Z. Nrf2 induced cisplatin resistance in ovarian cancer by promoting CD99 expression. Biochem. Biophys. Res. Commun. 518, 698–705 (2019).
Dinkova-Kostova, A. T. & Copple, I. M. Advances and challenges in therapeutic targeting of NRF2. Trends Pharmacol. Sci. 44, 137–149 (2023).
Buda, K., Miton, C. M., Fan, X. C. & Tokuriki, N. Molecular determinants of protein evolvability. Trends Biochem. Sci. 48, 751–760 (2023).
Bloom, J. D., Labthavikul, S. T., Otey, C. R. & Arnold, F. H. Protein stability promotes evolvability. Proc. Natl. Acad. Sci. USA 103, 5869–5874 (2006).
Tokuriki, N., Stricher, F., Schymkowitz, J., Serrano, L. & Tawfik, D. S. The stability effects of protein mutations appear to be universally distributed. J. Mol. Biol. 369, 1318–1332 (2007).
Tokuriki, N., Stricher, F., Serrano, L. & Tawfik, D. S. How protein stability and new functions trade off. PLoS Comput Biol. 4, e1000002 (2008).
Mashiba, S., Uchida, K., Okuda, S. & Tomita, S. Measurement of glycated albumin by the nitroblue tetrazolium colorimetric method. Clin. Chim. Acta 212, 3–15 (1992).
Walker, S. W., Howie, A. F. & Smith, A. F. The measurement of glycosylated albumin by reduction of alkaline nitro-blue tetrazolium. Clin. Chim. Acta 156, 197–206 (1986).
Consortium, T. U. et al. UniProt: the Universal Protein Knowledgebase in 2025. Nucleic Acids Res. 53, D609–D617 (2025).
Potter, S. C. et al. HMMER web server: 2018 update. Nucleic Acids Res. 46, W200–W204 (2018).
Li, W. et al. The EMBL-EBI bioinformatics web and programmatic tools framework. Nucleic Acids Res. 43, W580–W584 (2015).
Madeira, F. et al. The EMBL-EBI Job Dispatcher sequence analysis tools framework in 2024. Nucleic Acids Res. 52, W521–W525 (2024).
Guindon, S. et al. New algorithms and methods to estimate maximum-likelihood phylogenies: assessing the performance of PhyML 3.0. Syst. Biol. 59, 307–321 (2010).
Lefort, V., Longueville, J. E. & Gascuel, O. SMS: smart model selection in PhyML. Mol. Biol. Evol. 34, 2422–2424 (2017).
Yang, Z. PAML 4: phylogenetic analysis by maximum likelihood. Mol. Biol. Evol. 24, 1586–1591 (2007).
Sievers, F. et al. Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal Omega. Mol. Syst. Biol. 7, 539 (2011).
Crooks, G. E., Hon, G., Chandonia, J. M. & Brenner, S. E. WebLogo: a sequence logo generator. Genome Res. 14, 1188–1190 (2004).
Abramson, J. et al. Accurate structure prediction of biomolecular interactions with AlphaFold 3. Nature 630, 493–500 (2024).
Felline, A., Seeber, M. & Fanelli, F. webPSN v2.0: a webserver to infer fingerprints of structural communication in biomacromolecules. Nucleic Acids Res. 48, W94–W103 (2020).
Ferruz, N., Schmidt, S. & Höcker, B. ProteinTools: a toolkit to analyze protein structures. Nucleic Acids Res. 49, W559–W566 (2021).
Acknowledgements
We thank members of the Binning and Luca labs for their suggestions and technical assistance. The authors also acknowledge the Chemical Biology Core at the H. Lee Moffitt Cancer Center & Research Institute, which is supported in part by a National Cancer Institute (NCI) support grant (P30-CA076292). The protein structure network was adapted using Biorender.com. This study was supported by the U.S. National Institutes of Health grant R35 GM143004 to J.M.B. Figure 6B was created in BioRender. https://BioRender.com/0xc7owf.
Author information
Authors and Affiliations
Contributions
J.K.M. and J.M.B. designed the research and wrote the manuscript. J.K.M. performed all biochemical studies of FN3Ks and variants, ancestral protein reconstruction, structural analysis, and protein structure network analysis. R.E.M. performed the colorimetric assay on glycated substrates and cloned HsFN3K variants. J.L. performed conservation analysis of HsFN3K, cloned HsFN3K variants, and aided in the design of the research. J.M.B. conceptualized, supervised, provided resources, reviewed, and edited the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Gareth Surman and the other anonymous reviewer(s) for their contribution to the peer review of this work. Primary Handling Editors: Dr. Nilanjan Banerjee and Dr. Dr Michele Repetto.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Matlack, J.K., Miner, R.E., Lokhandwala, J. et al. Ancestral protein reconstruction reveals the mechanism of substrate specificity in FN3K-mediated deglycation. Commun Biol (2026). https://doi.org/10.1038/s42003-026-09967-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-09967-3