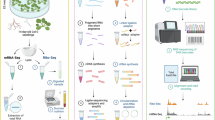
Abstract
Plants can adapt to environmental change through a variety of transcriptional and translational mechanisms. Although extensive knowledge exists about plant stress responses at the transcriptional level, understanding of these responses at the translational level is limited. This study aimed to evaluate the translational dynamics of plants in a simulated microgravity environment through the utilization of a ribosome profiling assay. A comparative analysis of ribosome profiling data and RNA-seq on a global scale demonstrated a modest correlation in the alteration of gene expression between transcriptional and translational levels. However, under one third of responsive genes exhibited concurrent transcriptional and translational activities. We found that simulated microgravity simulation elicited separate transcriptional and translational responses in plants. The findings indicated a significant alteration in the translational efficiency (TE) of 1081 genes under simulated microgravity conditions. Subsequent analysis identified specific sequence characteristics, including GC content and coding sequence length, as influential factors on gene TE. The abundance of upstream open reading frames (uORFs) within gene promoters indicates a possible influence on the translation efficiency of primary open reading frames located downstream. These results suggested that in response to simulated microgravity, plants had a responsive translational mechanism that interacted with transcription in a coordinated manner.
Similar content being viewed by others
Data availability
Source data for main Figs. 2a, b, 2d, 3a, 5a, 6a–c, and 7a–e, and Supplementary Fig. 5a, b and 6a–c, are available in Supplementary Data 4. The RNA-seq (GSA: CRA028515) and Ribo-seq (GSA: CRA028542) data reported in this paper have been deposited in the Genome Sequence Archive (GSA)65 in the National Genomics Data Center66, China National Center for Bioinformation/Beijing Institute of Genomics, Chinese Academy of Sciences, and are publicly accessible at https://ngdc.cncb.ac.cn/gsa.
References
Soga, K. Resistance of plants to gravitational force. J. Plant Res. 126, 589–596 (2013).
Tasaka, M., Kato, T. & Fukaki, H. Genetic regulation of gravitropism in higher plants. Int. Rev. Cytol. 206, 135–154 (2001).
Perbal, G. Plant development in space or in simulated microgravity. In Plant Biotechnology 2002 and Beyond, (ed. Vasil, I. k.) 351–357 (Springer Netherlands, 2003).
Kiss, J. Z., Wolverton, C., Wyatt, S. E., Hasenstein, K. H. & van Loon, J. J. W. A. Comparison of microgravity analogs to spaceflight in studies of plant growth and development. Front. Plant Sci. 10, 1577 (2019).
Silva, H. C. et al. Development of a system to submit sugarcane plants in real microgravity using the VSB30 sounding rocket. ESA Spec. Publ. 700, 185–188 (2011).
Farand, A. Legal environment for exploitation of the International Space Station (ISS). Space Stud. 4, 141–153 (2000).
Long, J. China’s space station project and international cooperation: potential models of jurisdiction and selected legal issues. Space Policy 36, 28–37 (2016).
Shimazu, T., Yuda, T., Miyamoto, K., Yamashita, M. & Ueda, J. Growth and development in higher-plants under simulated microgravity conditions on a 3-dimensional clinostat. Adv. Space Res. 27, 995–1000 (2001).
van Loon, J. J. W. A. Some history and use of the random positioning machine, RPM, in gravity related research. Adv. Space Res. 39, 1161–1165 (2007).
Xie, J. Y. & Zheng, H. Q. Arabidopsis flowering induced by photoperiod under 3-D clinostat rotational simulated microgravity. Acta Astronaut. 166, 567–572 (2020).
Ingolia, N. T., Brar, G. A., Rouskin, S., McGeachy, A. M. & Weissman, J. S. The ribosome profiling strategy for monitoring translation by deep sequencing of ribosome-protected mRNA fragments. Nat. Protoc. 7, 1534–1550 (2012).
Ingolia, N. T., Ghaemmaghami, S., Newman, J. R. S. & Weissman, J. S. Genome-wide analysis in vivo of translation with nucleotide resolution using ribosome profiling. Science 324, 218–223 (2009).
Chew, G. L. et al. Ribosome profiling reveals resemblance between long non-coding RNAs and 5′ leaders of coding RNAs. Development 140, 2828–2834 (2013).
Dunn, J. G., Foo, C. K., Belletier, N. G., Gavis, E. R. & Weissman, J. S. Ribosome profiling reveals pervasive and regulated stop codon readthrough in Drosophila melanogaster. eLife 2, e01179 (2013).
Michel, A. M. et al. Observation of dually decoded regions of the human genome using ribosome profiling data. Genome Res. 22, 2219–2229 (2012).
Liu, M. J. et al. Translational landscape of photomorphogenic Arabidopsis. Plant Cell 25, 3699–3710 (2013).
Hsu, P. Y. et al. Super-resolution ribosome profiling reveals unannotated translation events in Arabidopsis. Proc. Natl. Acad. Sci. USA 113, E7126–E7135 (2016).
Shalgi, R. et al. Widespread regulation of translation by elongation pausing in heat shock. Mol. Cell 49, 439–452 (2013).
Juntawong, P., Girke, T., Bazin, J. & Bailey-Serres, J. Translational dynamics revealed by genome-wide profiling of ribosome footprints in Arabidopsis. Proc. Natl. Acad. Sci. USA 111, E203–E212 (2014).
Tuller, T. et al. An evolutionarily conserved mechanism for controlling the efficiency of protein translation. Cell 141, 344–354 (2010).
Young, D. J., Guydosh, N. R., Zhang, F., Hinnebusch, A. G. & Green, R. Rli1/ABCE1 recycles terminating ribosomes and controls translation reinitiation in 3’UTRs in vivo. Cell 162, 872–884 (2015).
Barbosa, C., Peixeiro, I. & Romao, L. Gene expression regulation by upstream open reading frames and human disease. PLoS Genet. 9, e1003529 (2013).
Vélez-Bermúdez, I. C., Lin, W. D., Chou, S. J., Chen, A. P. & Schmidt, W. Transcriptome and translatome comparison of tissues from Arabidopsis thaliana. Sci. Data 12, 504 (2025).
Xu, P. et al. Pectin methylesterase gene AtPMEPCRA contributes to physiological adaptation to simulated and spaceflight microgravity in Arabidopsis. iScience 25, 104331 (2022).
Xu, P., Chen, H., Jin, J. & Cai, W. Single-base resolution methylome analysis shows epigenetic changes in Arabidopsis seedlings exposed to microgravity spaceflight conditions on board the SJ-10 recoverable satellite. NPJ Microgravity 4, 12 (2018).
Ogata, H. et al. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 27, 29–34 (1999).
Medina, F. J., Manzano, A., Villacampa, A., Ciska, M., & Herranz, R. Understanding reduced gravity effects on early plant development before attempting life-support farming in the Moon and Mars. Front. Astron. Space Sci. 8, 729154 (2021).
Okushima, Y. et al. Functional genomic analysis of the AUXIN RESPONSE FACTOR gene family members in Arabidopsis thaliana: unique and overlapping functions of ARF7 and ARF19. Plant Cell 17, 444–463 (2005).
Toyota, M., Furuichi, T., Tatsumi, H. & Sokabe, M. Cytoplasmic calcium increases in response to changes in the gravity vector in hypocotyls and petioles of Arabidopsis seedlings. Plant Physiol. 146, 505–514 (2008).
Albrecht, V. et al. The calcium sensor CBL1 integrates plant responses to abiotic stresses. Plant J.: Cell Mol. Biol. 36, 457–470 (2003).
Song, X. et al. CALMODULIN-LIKE-38 and PEP1 RECEPTOR 2 integrate nitrate and brassinosteroid signals to regulate root growth. Plant Physiol. 187, 1779–1794 (2021).
Barker, R. et al. Meta-analysis of the space flight and microgravity response of the Arabidopsis plant transcriptome. NPJ Microgravity 9, 21 (2023).
Lei, L. et al. Ribosome profiling reveals dynamic translational landscape in maize seedlings under drought stress. Plant J. 84, 1206–1218 (2015).
Lackner, D. H., Schmidt, M. W., Wu, S. D., Wolf, D. A. & Bähler, J. Regulation of transcriptome, translation, and proteome in response to environmental stress in fission yeast. Genome Biol. 13, R25 (2012).
Kozak, M. Point mutations define a sequence flanking the AUG initiator codon that modulates translation by eukaryotic ribosomes. Cell 44, 283–292 (1986).
Wiese, A., Elzinga, N., Wobbes, B. & Smeekens, S. A conserved upstream open reading frame mediates sucrose-induced repression of translation. Plant Cell 16, 1717–1729 (2004).
Sonenberg, N. & Hinnebusch, A. G. Regulation of translation initiation in eukaryotes: mechanisms and biological targets. Cell 136, 731–745 (2009).
Aravind, L. & Koonin, E. V. Eukaryote-specific domains in translation initiation factors: implications for translation regulation and evolution of the translation system. Genome Res. 10, 1172–1184 (2000).
Lee, M. V. et al. A dynamic model of proteome changes reveals new roles for transcript alteration in yeast. Mol. Syst. Biol. 7, 514 (2011).
Shenton, D. et al. Global translational responses to oxidative stress impact upon multiple levels of protein synthesis. J. Biol. Chem. 281, 29011–29021 (2006).
Bai, B. et al. Extensive translational regulation during seed germination revealed by polysomal profiling. N. Phytol. 214, 233–244 (2017).
Zhang, C. et al. Translational landscape and metabolic characteristics of the etiolated tea plant (Camellia sinensis). Sci. Hortic. 303, 111193 (2022).
Jian, H. et al. Dynamic translational landscape revealed by genome-wide ribosome profiling under drought and heat stress in potato. Plants 12, 2232 (2023).
Xiong, Q. et al. Ribosome profiling reveals the effects of nitrogen application translational regulation of yield recovery after abrupt drought-flood alternation in rice. Plant Physiol. Biochem. 155, 42–58 (2020).
Zhao, G. et al. Ribosome profiling and RNA sequencing reveal translation and transcription regulation under acute heat stress in rainbow trout (Oncorhynchus mykiss, Walbaum, 1792) liver. Int. J. Mol. Sci. 25, 8848 (2024).
Cao, M. et al. Comparative analysis of testicular transcriptional and translational landscapes in yak and cattle-yak: implications for hybrid male sterility. Biomolecules 15, 1080 (2025).
Spriggs, K. A., Bushell, M. & Willis, A. E. Translational regulation of gene expression during conditions of cell stress. Mol. Cell 40, 228–237 (2010).
Calvo, S. E., Pagliarini, D. J. & Mootha, V. K. Upstream open reading frames cause widespread reduction of protein expression and are polymorphic among humans. Proc. Natl. Acad. Sci. USA 106, 7507–7512 (2009).
Chang, K. S., Lee, S. H., Hwang, S. B. & Park, K. Y. Characterization and translational regulation of the arginine decarboxylase gene in carnation (Dianthus caryophyllus L.). Plant J. 24, 45–56 (2000).
Ebina, I. et al. Identification of novel upstream open reading frames that control expression of the main coding sequences in a peptide sequence-dependent manner. Nucleic Acids Res. 43, 1562–1576 (2015).
Hu, J. et al. A protocol for measuring the response of Arabidopsis roots to gravity and treatment for simulated microgravity. STAR Protoc. 4, 102099 (2023).
Morlan, J. D., Qu, K. B. & Sinicropi, D. V. Selective depletion of rRNA enables whole transcriptome profiling of archival fixed tissue. PLoS ONE 7, e42882 (2012).
Lauria, F. et al. riboWaltz: optimization of ribosome P-site positioning in ribosome profiling data. PLoS Comput. Biol. 14, e1006169 (2018).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Kanehisa, M. & Goto, S. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 28, 27–30 (2000).
Bazzini, A. A. et al. Identification of small ORFs in vertebrates using ribosome footprinting and evolutionary conservation. EMBO J. 33, 981–993 (2014).
Guttman, M., Russell, P., Ingolia, N. T., Weissman, J. S. & Lander, E. S. Ribosome profiling provides evidence that large noncoding RNAs do not encode proteins. Cell 154, 240–251 (2013).
Wang, L. et al. CPAT: coding-potential assessment tool using an alignment-free logistic regression model. Nucleic Acids Res. 41, e74 (2013).
Lorenz, R. et al. ViennaRNA package 2.0. Algorithm Mol. Biol. 6, 26 (2011).
Kozak, M. Pushing the limits of the scanning mechanism for initiation of translation. Gene 299, 1–34 (2002).
Chen, Y. J. et al. PsORF: a database of small ORFs in plants. Plant Biotechnol. J. 18, 2158–2160 (2020).
Chen, S. F., Zhou, Y. Q., Chen, Y. R. & Gu, J. fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 34, 884–890 (2018).
Olexiouk, V., Van Criekinge, W. & Menschaert, G. An update on sORFs.org: a repository of small ORFs identified by ribosome profiling. Nucleic Acids Res. 46, D497–D502 (2018).
Olexiouk, V. et al. sORFs.org: a repository of small ORFs identified by ribosome profiling. Nucleic Acids Res. 44, D324–D329 (2016).
Chen, T. et al. The genome sequence archive family: toward explosive data growth and diverse data types. Dev. Reprod. Biol. 19, 578–583 (2021).
CNCB-NGDC Members and Partners Database resources of the national genomics data center, China National Center for Bioinformation in 2022. Nucleic Acids Res. 50, D27–D38 (2022).
Acknowledgements
We thank Professor WR Hu for organizing the Strategic Priority Research Program of CAS, CSU of CAS for organizing the China’s Manned Space Program. This work was supported by the National Natural Science Foundation of China (Grant Nos. 32171232, 31570859, 31500236, 31070237, 90917009, and 31971172), Natural Science Foundation of Shanghai (Grant No. 22ZR1469500), CAS (No. XDA04020202-15), and China’s Manned Space Program.
Author information
Authors and Affiliations
Contributions
P.X., H.Z., and W.C. conceived and designed the research project. T.Z., L.Z., and Y.Y. performed the experiments, and J.H. provided technical recommendations. T.Z. and P.X. analyzed the data. T.Z. and P.X. wrote and revised the manuscript. All authors have read and approved the final version of the manuscript for submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Isabel Cristina Vélez-Bermúdez, Imara Perera and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: David Favero.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, T., Zhang, L., Yang, Y. et al. Genome-wide ribosome profiling reveals a dynamic translational landscape in Arabidopsis seedling roots under simulated microgravity. Commun Biol (2026). https://doi.org/10.1038/s42003-026-09991-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-09991-3