Abstract
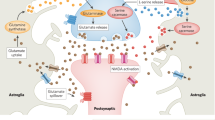
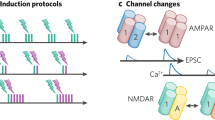
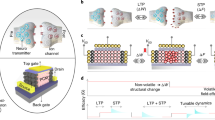
The dynamics of higher-order topological signals are increasingly recognized as a key aspect of the activity of complex systems. A paradigmatic example are synaptic dynamics: synaptic efficacy changes over time driven by different mechanisms. Beyond traditional node-driven short-term plasticity, the role of astrocyte modulation through higher-order interactions, in the tripartite synapse, is increasingly recognized. However, the competition and interplay between node-driven and higher-order mechanisms remain poorly understood. Here, we introduce a higher-order model of the tripartite synapse, accounting for astrocyte-synapse-neuron interactions in short-term plasticity, such that astrocyte gliotransmission and pre-synaptic facilitation jointly modulate neurotransmitter release, generalizing earlier short-term plasticity models. We study these mechanisms in a minimal recurrent motif—a directed ring of three excitatory neurons—where one neuron receives external stimulation. Due to strong recurrence, the circuit is prone to self-sustained activity, often ignoring external input. By introducing higher-order interactions via astrocyte modulation, we show this robustly stabilizes circuit dynamics and expands the parameter space supporting stimulus-driven activity. Our findings highlight how astrocytes reshape effective connectivity through higher-order interactions—even in simple recurrent circuits.
Similar content being viewed by others
Data availability
All data supporting the findings of this study were generated from numerical simulations of the model described in the “Methods” section. These data can be reproduced using the code provided and are therefore not separately archived. Example outputs are available from the corresponding author upon reasonable request.
Code availability
The code used to perform the simulations and generate the results is publicly available97.
References
Millán, A. P. et al. Topology shapes dynamics of higher-order networks. Nat. Phys. 21, 353–361 (2025).
Faskowitz, J., Betzel, R. F. & Sporns, O. Edges in brain networks: contributions to models of structure and function. Netw. Neurosci. 6, 1–28 (2022).
Herzog, R. et al. Genuine high-order interactions in brain networks and neurodegeneration. Neurobiol. Dis. 175, 105918 (2022).
Santoro, A., Battiston, F., Petri, G. & Amico, E. Higher-order organization of multivariate time series. Nat. Phys. 19, 221–229 (2023).
Santoro, A., Battiston, F., Lucas, M., Petri, G. & Amico, E. Higher-order connectomics of human brain function reveals local topological signatures of task decoding, individual identification, and behavior. Nat. Commun. 15, 10244 (2024).
Santos, F.A. et al. Emergence of high-order functional hubs in the human brain. bioRxiv, 2023–02. https://doi.org/10.1101/2023.02.10.528083 (2023).
Tewarie, P. K. et al. Non-reversibility outperforms functional connectivity in characterisation of brain states in meg data. NeuroImage 276, 120186 (2023).
Li, Q. et al. Higher-order triadic interactions: Insights into the multiscale network organization in schizophrenia. Hum. Brain Mapp. 46, https://doi.org/10.1002/hbm.70399 (2025).
Nartallo-Kaluarachchi, R. et al. Multilevel irreversibility reveals higher-order organization of nonequilibrium interactions in human brain dynamics. Proc. Natl. Acad. Sci. 122, https://doi.org/10.1073/pnas.2408791122 (2025).
Schaub, M. T., Benson, A. R., Horn, P., Lippner, G. & Jadbabaie, A. Random walks on simplicial complexes and the normalized Hodge 1-Laplacian. SIAM Rev. 62, 353–391 (2020).
Barbarossa, S. & Sardellitti, S. Topological signal processing over simplicial complexes. IEEE Trans. Signal Process. 68, 2992–3007 (2020).
Battiston, F. et al. Networks beyond pairwise interactions: Structure and dynamics. Phys. Rep. 874, 1–92 (2020).
Millán, A. P., Sun, H. & Torres, J. J. Spatio-temporal activity patterns induced by triadic interactions in an in silico neural medium. J. Phys.: Complex. 6, 015017 (2025).
Di Gaetano, L. et al. Neighbourhood topology unveils pathological hubs in the brain networks of epilepsy-surgery patients. Brain Commun. 7, fcaf431 (2025).
Stolz, B., Emerson, T., Nahkuri, S., Porter, M. & Harrington, H. Topological data analysis of task-based fMRI data from experiments on schizophrenia. J. Phys.: Complex. 2, 035006 (2021).
Torres, L., Blevins, A. S., Bassett, D. & Eliassi-Rad, T. The why, how, and when of representations for complex systems. SIAM Rev. 63, 435–485 (2021).
Cencetti, G., Contreras, D. A., Mancastroppa, M. & Barrat, A. Distinguishing simple and complex contagion processes on networks. Phys. Rev. Lett. 130, 247401 (2023).
Zhou, D., Huang, J., Scholkopf, B. Learning with hypergraphs: clustering, classification, and embedding. In: NeurIPS https://doi.org/10.5555/2976456.2976657 (2006).
Sardellitti, S. & Barbarossa, S. Topological signal processing over generalized cell complexes. IEEE Transactions on Signal Processing 72,687–700 (2024).
Zhao, Y. et al. Dynamic hypergraph structure learning for traffic flow forecasting. In Proc. IEEE 39th International Conference on Data Engineering (ICDE) 2303–2316 (IEEE, 2023).
Ji, J., Ren, Y. & Lei, M. Fc–hat: hypergraph attention network for functional brain network classification. Inf. Sci. 608, 1301–1316 (2022).
Antelmi, A. et al. A survey on hypergraph representation learning. ACM Comput. Surv. 56, 1–38 (2023).
Abbott, L. F. & Regehr, W. G. Synaptic computation. Nature 431, 796–803 (2004).
Stevens, C. F. & Wang, Y. Facilitation and depression at single central synapses. Neuron 14, 795–802 (1995).
Markram, H. & Tsodyks, M. Redistribution of synaptic efficacy between neocortical pyramidal neurons. Nature 382, 807–810 (1996).
Lin, R. C. & Scheller, R. H. Mechanisms of synaptic vesicle exocytosis. Annu. Rev. Cell Dev. Biol. 16, 19–49 (2000).
Li, L. & Chin, L.-S. The molecular machinery of synaptic vesicle exocytosis. Cell. Mol. Life Sci. 60, 942–960 (2003).
Tsodyks, M., Pawelzik, K. & Markram, H. Neural networks with dynamic synapses. Neural Comput. 10, 821–835 (1998).
Torres, J.J., Kappen, H.J. Emerging phenomena in neural networks with dynamic synapses and their computational implications. Front. Comput. Neurosci. 7, https://doi.org/10.3389/fncom.2013.00030 (2013).
Torres, J. J. & Marro, J. Brain performance versus phase transitions. Sci. Rep. 5, 12216 (2015).
Pretel, J., Torres, J.J., Marro, J. Eegs disclose significant brain activity correlated with synaptic fickleness. Biology 10, https://doi.org/10.3390/biology10070647 (2021).
Menesse, G. & Torres, J. J. Information dynamics of in silico EEG brain waves: insights into oscillations and functions. PLoS Comput Biol. 20, 1–41 (2024).
Araque, A., Parpura, V., Sanzgiri, R. P. & Haydon, P. G. Tripartite synapses: glia, the unacknowledged partner. Trends Neurosci. 22, 208–215 (1999).
Pittà, M. D., Brunel, N. & Volterra, A. Astrocytes: orchestrating synaptic plasticity? Neuroscience 323, 43–61 (2016).
Bazargani, N. & Attwell, D. Astrocyte calcium signaling: the third wave. Nat. Neurosci. 19, 182–189 (2016).
DePittà, M., Berry, H. A Neuron–Glial Perspective for Computational Neuroscience. Springer, 2019). 3–35.
Oliveira, J. F. & Araque, A. Astrocyte regulation of neural circuit activity and network states. Glia 70, 1455–1466 (2022).
Lezmy, J. How astrocytic ATP shapes neuronal activity and brain circuits. Curr. Opin. Neurobiol. 79, 102685 (2023).
Dossi, E. et al. Astroglial gap junctions strengthen hippocampal network activity by sustaining afterhyperpolarization via KCNQ channels. Cell Rep. 43, 114158 (2024).
Millán, A. P., Torres, J. J. & Bianconi, G. Explosive higher-order Kuramoto dynamics on simplicial complexes. Phys. Rev. Lett. 124, 218301 (2020).
Cover, K. K. & Mathur, B. N. Axo-axonic synapses: Diversity in neural circuit function. J. Comp. Neurol. 529, 2391–2401 (2021).
Pan-Vazquez, A., Wefelmeyer, W., Sabater, V. G., Neves, G. & Burrone, J. Activity-dependent plasticity of axo-axonic synapses at the axon initial segment. Neuron 106, 265–276 (2020).
Sun, H., Radicchi, F., Kurths, J. & Bianconi, G. The dynamic nature of percolation on networks with triadic interactions. Nat. Commun. 14, 1308 (2023).
Ceglia, R. et al. Specialized astrocytes mediate glutamatergic gliotransmission in the CNS. Nature 622, 120–129 (2023).
Lenk, K. et al. Computational Models of Astrocyte Function at Glutamatergic Synapses. Springer, 2024). 229–263.
Malarkey, E. B. & Parpura, V. Mechanisms of glutamate release from astrocytes. Neurochem. Int. 52, 142–154 (2008).
Kovács, A. & Pál, B. Astrocyte-dependent slow inward currents (SICS) participate in neuromodulatory mechanisms in the pedunculopontine nucleus (ppn). Front. Cell. Neurosci. 11, https://doi.org/10.3389/fncel.2017.00016 (2017).
Csemer, A. et al. Astrocyte- and NMDA receptor-dependent slow inward currents differently contribute to synaptic plasticity in an age-dependent manner in mouse and human neocortex. Aging Cell 22, https://doi.org/10.1111/acel.13939 (2023).
Jo, S. et al. Gaba from reactive astrocytes impairs memory in mouse models of alzheimer’s disease. Nat. Med. 20, 886–896 (2014).
Pittà, M.D., Brunel, N. Multiple forms of working memory emerge from synapse-astrocyte interactions in a neuron-glia network model. Proc. Natl. Acad. Sci. 119, https://doi.org/10.1073/pnas.2207912119 (2022).
Durkee, C., Kofuji, P., Navarrete, M. & Araque, A. Astrocyte and neuron cooperation in long-term depression. Trends Neurosci. 44, 837–848 (2021).
Navarrete, M. & Araque, A. Endocannabinoids mediate neuron-astrocyte communication. Neuron 57, 883–893 (2008).
Pittà, M. D., Volman, V., Berry, H. & Ben-Jacob, E. A tale of two stories: astrocyte regulation of synaptic depression and facilitation. PLoS Comput Biol. 7, 1002293 (2011).
De Pitta’, M. et al. Computational quest for understanding the role of astrocyte signaling in synaptic transmission and plasticity. Front. Comput. Neurosci. 6, https://doi.org/10.3389/fncom.2012.00098 (2012).
Oschmann, F., Berry, H., Obermayer, K. & Lenk, K. From in silico astrocyte cell models to neuron-astrocyte network models: a review. Brain Res. Bull. 136, 76–84 (2018).
Manninen, T., Saudargiene, A., Linne, M.L. Astrocyte-mediated spike-timing-dependent long-term depression modulates synaptic properties in the developing cortex. PLoS Comput. Biol. 16, https://doi.org/10.1371/journal.pcbi.1008360 (2020).
Verisokin, A.Y., Verveyko, D.V., Postnov, D.E., Brazhe, A.R. Modeling of astrocyte networks: toward realistic topology and dynamics. Front. Cell. Neurosci. 15, https://doi.org/10.3389/fncel.2021.645068 (2021).
Lenk, K. et al. A computational model of interactions between neuronal and astrocytic networks: The role of astrocytes in the stability of the neuronal firing rate. Front. Comput. Neurosci. 13, https://doi.org/10.3389/fncom.2019.00092 (2020).
Tan, C. X. & Eroglu, C. Cell adhesion molecules regulating astrocyte-neuron interactions. Curr. Opin. Neurobiol. 69, 170–177 (2021).
Ventura, R. & Harris, K. M. Three-dimensional relationships between hippocampal synapses and astrocytes. J. Neurosci. 19, 6897–6906 (1999).
Duigou, C.L., Simonnet, J., Teleñczuk, M.T., Fricker, D., Miles, R. Recurrent synapses and circuits in the ca3 region of the hippocampus: an associative network. Front. Cell. Neurosci. 7, https://doi.org/10.3389/fncel.2013.00262 (2014).
Sammons, R.P. et al. Structure and function of the hippocampal CA3 module. Proc. Natl. Acad. Sci. 121, https://doi.org/10.1073/pnas.2312281120 (2024).
O’Byrne, J. & Jerbi, K. How critical is brain criticality? Trends Neurosci. 45, 820–837 (2022).
Protachevicz, P. R., Batista, A. M., Caldas, I. L. & Baptista, M. S. Analytical solutions for the short-term plasticity. Chaos Solit. Fractals 181, 114678 (2024).
Ito, H.T., Schuman, E.M. Frequency-dependent signal transmission and modulation by neuromodulators. Front. Neurosci. 2, https://doi.org/10.3389/neuro.01.027.2008 (2008).
Hirase, H., Qian, L., Barthó, P. & Buzsáki, G. Calcium dynamics of cortical astrocytic networks in vivo. PLoS Biol. 2, 96 (2004).
Lallouette, J., Pittà, M.D. & Berry, H. In Astrocyte Networks and Intercellular Calcium Propagation (eds Pittà, M.D. & Berry, H.) 177–210. https://doi.org/10.1007/978-3-030-00817-8_7 (Springer, 2019).
Miguel-Quesada, C. et al. Astrocytes adjust the dynamic range of cortical network activity to control modality-specific sensory information processing. Cell Rep. 42, 112950 (2023).
Clarke, L. E. & Barres, B. A. Emerging roles of astrocytes in neural circuit development. Nat. Rev. Neurosci. 14, 311–321 (2013).
Ota, Y., Zanetti, A. T. & Hallock, R. M. The role of astrocytes in the regulation of synaptic plasticity and memory formation. Neural Plast. 2013, 1–11 (2013).
Squadrani, L. et al. Astrocytes enhance plasticity response during reversal learning. Commun. Biol. 7, 852 (2024).
Radulescu, A. R. et al. Estimating the glutamate transporter surface density in distinct sub-cellular compartments of mouse hippocampal astrocytes. PLoS Comput. Biol. 18, 1009845 (2022).
Lewerenz, J. & Maher, P. Chronic glutamate toxicity in neurodegenerative diseases-what is the evidence? Front. Neurosci. 9, 469 (2015).
Paniccia, J. E., Otis, J. M. & Scofield, M. D. Looking to the stars for answers: strategies for determining how astrocytes influence neuronal activity. Comput. Struct. Biotechnol. J. 20, 4146–4156 (2022).
Battiston, F. et al. The physics of higher-order interactions in complex systems. Nat. Phys. 17, 1093–1098 (2021).
Iacopini, I., Petri, G., Barrat, A. & Latora, V. Simplicial models of social contagion. Nat. Commun. 10, 2485 (2019).
Gatica, M. et al. High-order functional redundancy in ageing explained via alterations in the connectome in a whole-brain model. PLoS Comput. Biol. 18, 1010431 (2022).
Araque, A. et al. Gliotransmitters travel in time and space. Neuron 81, 728–739 (2014).
Hasselmo, M., Schnell, E. & Barkai, E. Dynamics of learning and recall at excitatory recurrent synapses and cholinergic modulation in rat hippocampal region CA3. J. Neurosci. 15, 5249–5262 (1995).
Sadeh, S. & Clopath, C. Inhibitory stabilization and cortical computation. Nat. Rev. Neurosci. 22, 21–37 (2021).
Millán, A. P., Sun, H., Torres, J. J. & Bianconi, G. Triadic percolation induces dynamical topological patterns in higher-order networks. PNAS Nexus 3, 270 (2024).
Kumar, R., Huang, Y.-T., Chen, C.-C., Tzeng, S.-F. & Chan, C.-K. Astrocytic regulation of synchronous bursting in cortical cultures: from local to global. Cereb. Cortex Commun. 1, https://doi.org/10.1093/texcom/tgaa053 (2020).
Flanagan, B., McDaid, L., Wade, J., Wong-Lin, K. & Harkin, J. A computational study of astrocytic glutamate influence on post-synaptic neuronal excitability. PLoS Comput. Biol. 14, 1006040 (2018).
Kwak, H. et al. Astrocytes control sensory acuity via tonic inhibition in the thalamus. Neuron 108, 691–70610 (2020).
Zonca, L. & Holcman, D. Modeling bursting in neuronal networks using facilitation-depression and afterhyperpolarization. Commun. Nonlinear Sci. Numer. Simul. 94, 105555 (2021).
De Pittà, M. In Neuron-Glial Interactions (eds Jaeger, D., Jung, R.) 1–30. https://doi.org/10.1007/978-1-4614-7320-6_100691-1 (Springer, 2020).
Allen, N. J. & Eroglu, C. Cell biology of astrocyte-synapse interactions. Neuron 96, 697–708 (2017).
Semyanov, A. & Verkhratsky, A. Astrocytic processes: from tripartite synapses to the active milieu. Trends Neurosci. 44, 781–792 (2021).
Pasti, L., Pozzan, T. & Carmignoto, G. Long-lasting changes of calcium oscillations in astrocytes. J. Biol. Chem. 270, 15203–15210 (1995).
Verkhratsky, A., Rodríguez, J. J. & Parpura, V. Calcium signalling in astroglia. Mol. Cell. Endocrinol. 353, 45–56 (2012).
Sasaki, T. et al. Astrocyte calcium signalling orchestrates neuronal synchronization in organotypic hippocampal slices. J. Physiol. 592, 2771–2783 (2014).
Meriney, S.D., Fanselow, E.E. Metabotropic G-protein-coupled receptors and their cytoplasmic signaling pathways. In Synaptic Transmission (eds Meriney, S.D., Fanselow, E.E.) Ch. 12, 245–273. https://doi.org/10.1016/B978-0-12-815320-8.00012-0 (Academic Press, 2019).
Pittà, M.D. et al. Coexistence of amplitude and frequency modulations in intracellular calcium dynamics. Phys. Rev. E Stat. Nonlinear Soft Matter Phys. 77, https://doi.org/10.1103/PhysRevE.77.030903 (2008).
Graupner, M. & Brunel, N. Calcium-based plasticity model explains sensitivity of synaptic changes to spike pattern, rate, and dendritic location. Proc. Natl. Acad. Sci. 109, 3991–3996 (2012).
Chindemi, G. et al. A calcium-based plasticity model for predicting long-term potentiation and depression in the neocortex. Nat. Commun. 13, https://doi.org/10.1038/s41467-022-30214-w (2022).
Halassa, M. M. & Haydon, P. G. Integrated brain circuits: Astrocytic networks modulate neuronal activity and behavior. Annu. Rev. Physiol. 72, 335–355 (2010).
Menesse, G., Millan, A.P., Torres, J.J. Astrocyte-Mediated Higher-Order Synaptic Plasticity. Zenodo https://doi.org/10.5281/zenodo.19260114 (2026).
Gerstner, W., Kistler, W. M., Naud, R., Paninski, L. Neuronal Dynamics: From Single Neurons to Networks and Models of Cognition. Cambridge University Press, 2014.
Acknowledgements
This work has been supported by Grant No. PID2023-149174NB-I00 financed by the Spanish Ministry and Agencia Estatal de Investigación MICIU/AEI/10.13039/501100011033 and ERDF funds (European Union) (to A.P.M. and J.J.T.). A.P.M. also acknowledges financial support by the ‘Ramón y Cajal’ program of the Spanish Ministry of Science and Innovation (Grant RYC2021-031241-I). G.M. would like to thank the “Programa Nacional de Becas de Postgrados en el Exterior “Don Carlos Antonio López”—BECAL” of the Ministry of Economy and Finance of Paraguay for the financial sponsorship to pursue his doctoral studies in the Physics and Mathematics Program of the University of Granada. We thank Maurizio De Pittà for his valuable feedback on this manuscript, which helped enhance both its clarity and depth through his thoughtful comments and suggestions.
Author information
Authors and Affiliations
Contributions
A.P.M., G.M. and J.J.T. conceived the study. G.M. and J.J.T. developed the theoretical framework and computational methodology, and designed the numerical experiments. G.M. implemented the model, performed the simulations, conducted the data analysis, visualization and interpretation of the results. G.M., A.P.M. and J.J.T. wrote the original draft of the manuscript. G.M. and J.J.T. contributed to manuscript revision and provided critical feedback. J.J.T. supervised the project, and acquired funding.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Lou Zonca and Tim LaRock for their contribution to the peer review of this work. Primary Handling Editors: Jonathan Touboul and Jasmine Pan.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Menesse, G., Millán, A.P. & Torres, J.J. Astrocyte-mediated higher-order control of synaptic plasticity. Commun Biol (2026). https://doi.org/10.1038/s42003-026-10044-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-10044-y