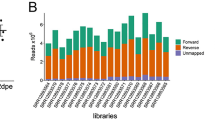
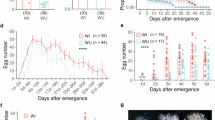
Abstract
Wolbachia is a widespread endosymbiont and a promising tool for pest control due to its ability to manipulate arthropod reproduction. However, how Wolbachia shapes host microRNA regulation remains poorly understood. Here, we profile the Wolbachia-driven host miRNAs using small RNA-seq in spider mite Tetranychus truncatus to uncover their roles in reproduction and symbiosis. Enrichment analyses of predicted miRNA targets suggest that Wolbachia-driven miRNAs may be involved in Wolbachia–host interactions. Functional assays show that several Wolbachia-driven miRNAs influence host fecundity, with some also affecting Wolbachia density. Inhibition of novel-33 or novel-40 reduces host fecundity and is accompanied by reduced Wolbachia density, whereas inhibition of novel-65 reduces fecundity without altering density. These findings indicate that specific miRNAs contribute to the regulation of reproduction and Wolbachia density in T. truncatus, highlighting host miRNAs regulated by Wolbachia as a mechanism of host manipulation and offering opportunities for Wolbachia-based pest management.

Similar content being viewed by others
Data availability
Raw sequencing data for the miRNA and mRNA libraries have been deposited in the NCBI Sequence Read Archive under BioProject IDs PRJNA1253370 and PRJNA1253798, respectively. Sample information is provided in Supplementary Data 3. Source data underlying the main figures are provided in Supplementary Data 4. All other data supporting the findings of this study are available within the paper and its Supplementary Information. Further information and requests for resources or materials should be directed to, and will be fulfilled by, the corresponding author.
References
Gotoh, T., Noda, H. & Hong, X. Y. Wolbachia distribution and cytoplasmic incompatibility based on a survey of 42 spider mite species (Acari: Tetranychidae) in Japan. Heredity 91, 208–216 (2003).
Porter, J. & Sullivan, W. The cellular lives of Wolbachia. Nat. Rev. Microbiol. 21, 750–766 (2023).
Hilgenboecker, K., Hammerstein, P., Schlattmann, P., Telschow, A. & Werren, J. H. How many species are infected with Wolbachia?-A statistical analysis of current data. FEMS Microbiol. Lett. 281, 215–220 (2008).
Ribeiro, P. et al. Pervasive horizontal transmission of Wolbachia in natural populations of closely related and widespread tropical skipper butterflies. BMC Microbiol. 25, 5 (2025).
Werren, J. H., Baldo, L. & Clark, M. E. Wolbachia: master manipulators of invertebrate biology. Nat. Rev. Microbiol. 6, 741–751 (2008).
Beckmann, J. F., Ronau, J. A. & Hochstrasser, M. A. deubiquitylating enzyme induces cytoplasmic incompatibility. Nat. Microbiol. 2, 17007 (2017).
Shropshire, J. D., Leigh, B. & Bordenstein, S. R. Symbiont-mediated cytoplasmic incompatibility: what have we learned in 50 years? eLife 9, e61989 (2020).
Lau, M. J., Ross, P. A. & Hoffmann, A. A. Infertility and fecundity loss of Wolbachia-infected Aedes aegypti hatched from quiescent eggs is expected to alter invasion dynamics. PLoS Negl. Trop. Dis. 15, e0009179 (2021).
Maciel-de-Freitas, R. et al. Wolbachia strains wMel and wAlbB differentially affect Aedes aegypti traits related to fecundity. Microbiol. Spectr. 12, e0012824 (2024).
Zug, R. & Hammerstein, P. Bad guys turned nice? A critical assessment of Wolbachia mutualisms in arthropod hosts. Biol. Rev. Camb. Philos. Soc. 90, 89–111 (2015).
Weeks, A. R., Turelli, M., Harcombe, W. R., Reynolds, K. T. & Hoffmann, A. A. From parasite to mutualist: rapid evolution of Wolbachia in natural populations of Drosophila. PLoS Biol. 5, e114 (2007).
Fry, A. J., Palmer, M. R. & Rand, D. M. Variable fitness effects of Wolbachia infection in Drosophila melanogaster. Heredity 93, 379–389 (2004).
Shropshire, J. D., Leigh, B. & Bordenstein, S. R. Male age and Wolbachia dynamics: investigating how fast and why bacterial densities and cytoplasmic incompatibility strengths vary. mBio 12, e02998-21 (2021).
Zheng, X. et al. Incompatible and sterile insect techniques combined eliminate mosquitoes. Nature 572, 56–61 (2019).
Lim, J. T. et al. Efficacy of Wolbachia-mediated sterility to reduce the incidence of dengue: a synthetic control study in Singapore. Lancet Microbe 5, e422–e432 (2024).
Zabalou, S. et al. Wolbachia-induced cytoplasmic incompatibility as a means for insect pest population control. Proc. Natl. Acad. Sci. USA 101, 15042–15045 (2004).
Jin, P.-Y., Tian, L., Chen, L. & Hong, X.-Y. Spider mites of agricultural importance in China, with focus on species composition during the last decade (2008–2017). Syst. Appl. Acarol. 23, 2087 (2018).
Ullah, M. S., Gotoh, T. & Lim, U. T. Life history parameters of three phytophagous spider mites, Tetranychus piercei, T. truncatus and T. bambusae (Acari: Tetranychidae). J. Asia-Pac. Entomol. 17, 767–773 (2014).
Zhu, Y. X. et al. A change in the bacterial community of spider mites decreases fecundity on multiple host plants. MicrobiologyOpen 8, e00743 (2019).
Zhu, Y. X. et al. Incidence of facultative bacterial endosymbionts in spider mites associated with local environments and host plants. Appl. Environ. Microbiol. 84, e02546 (2018).
Zhang, Y. K., Chen, Y. T., Yang, K., Qiao, G. X. & Hong, X. Y. Screening of spider mites (Acari: Tetranychidae) for reproductive endosymbionts reveals links between co-infection and evolutionary history. Sci. Rep. 6, 27900 (2016).
Yang, K. et al. Wolbachia dominate Spiroplasma in the co-infected spider mite Tetranychus truncatus. Insect Mol. Biol. 29, 19–37 (2020).
Xia, X. et al. Wolbachia affects reproduction in the spider mite Tetranychus truncatus (Acari: Tetranychidae) by regulating chorion protein S38-like and Rop. Insect Mol. Biol. 30, 18–29 (2021).
Zhu, Y. X., Song, Z. R., Zhang, Y. Y., Hoffmann, A. A. & Hong, X. Y. Spider mites singly infected with either Wolbachia or Spiroplasma have reduced thermal tolerance. Front. Microbiol. 12, 706321 (2021).
Zhang, Q., Dou, W., Taning, C. N. T., Smagghe, G. & Wang, J. J. Regulatory roles of microRNAs in insect pests: prospective targets for insect pest control. Curr. Opin. Biotechnol. 70, 158–166 (2021).
Lucas, K. & Raikhel, A. S. Insect microRNAs: biogenesis, expression profiling and biological functions. Insect Biochem. Mol. Biol. 43, 24–38 (2013).
Asgari, S. Role of microRNAs in insect host-microorganism interactions. Front Physiol. 2, 48 (2011).
Lozano, J., Montanez, R. & Belles, X. MiR-2 family regulates insect metamorphosis by controlling the juvenile hormone signaling pathway. Proc. Natl. Acad. Sci. USA 112, 3740–3745 (2015).
Zhang, S. et al. Host miRNAs are involved in hormonal regulation of HaSNPV-triggered climbing behaviour in Helicoverpa armigera. Mol. Ecol. 27, 459–475 (2018).
Belles, X. MicroRNAs and the evolution of insect metamorphosis. Annu. Rev. Entomol. 62, 111–125 (2017).
Lu, M. Y. & Chtarbanova, S. The role of micro RNAs (miRNAs) in the regulation of Drosophila melanogaster’s innate immunity. Fly 16, 382–396 (2022).
Xie, J. et al. miR-275/305 cluster is essential for maintaining energy metabolic homeostasis by the insulin signaling pathway in Bactrocera dorsalis. PLoS Genet. 18, e1010418 (2022).
Zhang, Y. et al. Insect-specific microRNA Involved in the development of the silkworm Bombyx mori. PLoS ONE 4, e4677 (2009).
Roy, S., Saha, T. T., Zou, Z. & Raikhel, A. S. Regulatory pathways controlling female insect reproduction. Annu. Rev. Entomol. 63, 489–511 (2018).
Lucas, K. J., Zhao, B., Liu, S. & Raikhel, A. S. Regulation of physiological processes by microRNAs in insects. Curr. Opin. Insect Sci. 11, 1–7 (2015).
Ling, L., Kokoza, V. A., Zhang, C., Aksoy, E. & Raikhel, A. S. MicroRNA-277 targets insulin-like peptides 7 and 8 to control lipid metabolism and reproduction in Aedes aegypti mosquitoes. Proc. Natl. Acad. Sci. USA 114, E8017–E8024 (2017).
Liu, L., Zhang, K. J., Rong, X., Li, Y. Y. & Liu, H. Identification of Wolbachia-responsive miRNAs in the small brown planthopper, Laodelphax striatellus. Front. Physiol. 10, 928 (2019).
Rong, X., Zhang, Y. K., Zhang, K. J. & Hong, X. Y. Identification of Wolbachia-responsive microRNAs in the two-spotted spider mite, Tetranychus urticae. BMC Genomics 15, 1122 (2014).
Bishop, C., Hussain, M., Hugo, L. E. & Asgari, S. Analysis of Aedes aegypti microRNAs in response to Wolbachia wAlbB infection and their potential role in mosquito longevity. Sci. Rep. 12, 15245 (2022).
Hussain, M., Frentiu, F. D., Moreira, L. A., O’Neill, S. L. & Asgari, S. Wolbachia uses host microRNAs to manipulate host gene expression and facilitate colonization of the dengue vector Aedes aegypti. Proc. Natl. Acad. Sci. USA 108, 9250–9255 (2011).
Osei-Amo, S., Hussain, M., O’Neill, S. L. & Asgari, S. Wolbachia-induced aae-miR-12 miRNA negatively regulates the expression of MCT1 and MCM6 genes in Wolbachia-infected mosquito cell line. PLoS ONE 7, e50049 (2012).
Wang, J. et al. XPO5 promotes primary miRNA processing independently of RanGTP. Nat. Commun. 11, 1845 (2020).
Fast, E. M. et al. Wolbachia enhance Drosophila stem cell proliferation and target the germline stem cell niche. Science 334, 990–992 (2011).
Iovino, N., Pane, A. & Gaul, U. miR-184 has multiple roles in Drosophila female germline development. Dev. Cell 17, 123–133 (2009).
Zhang, G., Hussain, M., O’Neill, S. L. & Asgari, S. Wolbachia uses a host microRNA to regulate transcripts of a methyltransferase, contributing to dengue virus inhibition in Aedes aegypti. Proc. Natl. Acad. Sci. USA 110, 10276–10281 (2013).
She, L. et al. Wolbachia mediates crosstalk between miRNA and Toll pathways to enhance resistance to dengue virus in Aedes aegypti. PLoS Pathog. 20, e1012296 (2024).
Xia, X., Peng, C.-W., Lu, Y.-J., Zheng, X.-Y. & Hong, X.-Y. Transfection and colonization of Tetranychus truncatus Wolbachia strain wTtru in cell lines of the mosquito Aedes albopictus. Syst. Appl. Acarol. 23, 2420–2431 (2018).
He, Z. et al. How do Wolbachia modify the Drosophila ovary? New evidences support the “titration-restitution” model for the mechanisms of Wolbachia-induced CI. BMC Genomics 20, 608 (2019).
He, K. et al. Multiple miRNAs jointly regulate the biosynthesis of ecdysteroid in the holometabolous insects, Chilo suppressalis. RNA 23, 1817–1833 (2017).
Sun, K., Jee, D., de Navas, L. F., Duan, H. & Lai, E. C. Multiple In vivo biological processes are mediated by functionally redundant activities of Drosophila mir-279 and mir-996. PLoS Genet. 11, e1005245 (2015).
Wang, Y. L. et al. The microRNA miR-184 regulates the CYP303A1 transcript level to control molting of Locusta migratoria. Insect Sci. 28, 941–951 (2021).
Feng, K. et al. Cuticle protein mediates the evolution of stress resistance by generating a decoy circular RNA in spider mite. Sci. Adv. 11, eads3361 (2025).
Baldo, L. et al. Multilocus sequence typing system for the endosymbiont Wolbachia pipientis. Appl. Environ. Microbiol. 72, 7098–7110 (2006).
Chen, L. et al. The genome sequence of a spider mite, Tetranychus truncatus, provides insights into interspecific host range variation and the genetic basis of adaptation to a low-quality host plant. Insect Sci. 30, 1208–1228 (2023).
Kim, D., Paggi, J. M., Park, C., Bennett, C. & Salzberg, S. L. Graph-based genome alignment and genotyping with HISAT2 and HISAT-genotype. Nat. Biotechnol. 37, 907–915 (2019).
Pertea, M. et al. StringTie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat. Biotechnol. 33, 290–295 (2015).
Langmead, B., Trapnell, C., Pop, M. & Salzberg, S. L. Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol. 10, R25 (2009).
Friedlander, M. R., Mackowiak, S. D., Li, N., Chen, W. & Rajewsky, N. miRDeep2 accurately identifies known and hundreds of novel microRNA genes in seven animal clades. Nucleic Acids Res. 40, 37–52 (2012).
Wen, M., Shen, Y., Shi, S. & Tang, T. miREvo: an integrative microRNA evolutionary analysis platform for next-generation sequencing experiments. BMC Bioinforma. 13, 140 (2012).
Rehmsmeier, M., Steffen, P., Hochsmann, M. & Giegerich, R. Fast and effective prediction of microRNA/target duplexes. RNA 10, 1507–1517 (2004).
Enright, A. J. et al. MicroRNA targets in Drosophila. Genome Biol. 5, R1 (2003).
Mann, M., Wright, P. R. & Backofen, R. IntaRNA 2.0: enhanced and customizable prediction of RNA-RNA interactions. Nucleic Acids Res. 45, W435–W439 (2017).
Young, M. D., Wakefield, M. J., Smyth, G. K. & Oshlack, A. Gene ontology analysis for RNA-seq: accounting for selection bias. Genome Biol. 11, R14 (2010).
Mao, X., Cai, T., Olyarchuk, J. G. & Wei, L. Automated genome annotation and pathway identification using the KEGG Orthology (KO) as a controlled vocabulary. Bioinformatics 21, 3787–3793 (2005).
Sun, W., Jin, Y., He, L., Lu, W. C. & Li, M. Suitable reference gene selection for different strains and developmental stages of the carmine spider mite, Tetranychus cinnabarinus, using quantitative real-time PCR. J. Insect Sci. 10, 208 (2010).
Ghazy, N. A. et al. A leaf-mimicking method for oral delivery of bioactive substances into sucking arthropod herbivores. Front. Plant Sci. 11, 1218 (2020).
Phatak, P. & Donahue, J. M. Biotinylated micro-RNA pull down assay for identifying miRNA targets. Bio Protoc. 7, e2253 (2017).
Searle, S. R., Speed, F. M. & Milliken, G. A. Population marginal means in the linear model: an alternative to least squares means. Am. Stat. 34, 216–221 (2012).
Brooks, M. E. et al. glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. The R Journal 9, 378–400 (2017).
Lenth R. V. et al. emmeans: estimated marginal means, aka least-squares means. R package version 1.11.0. https://CRAN.R-project.org/package=emmeans. (2025).
Zhao, D. X., Zhang, X. F., Chen, D. S., Zhang, Y. K. & Hong, X. Y. Wolbachia-host interactions: host mating patterns affect Wolbachia density dynamics. PLoS ONE 8, e66373 (2013).
Acknowledgements
We thank Dr. Kaiyang Feng from the College of Plant Protection, Southwest University, for technical consultation and support with the RNA pull-down assay. We also appreciate the valuable suggestions and manuscript proofreading provided by Dr. Yu-Xi Zhu from Yangzhou University, Dr. Tong-Pu Li from Nanjing Forestry University, and Zhang-Rong Song from the University of Florida. This study was supported by the National Natural Science Foundation of China (Grants No. 32020103011, 32202290) and by the Postgraduate Research & Practice Innovation Program of Jiangsu Province, China (KYCX22_0760).
Author information
Authors and Affiliations
Contributions
Yi-Yin Zhang, Xin-Yu Shen, and Xiao-Yue Hong contributed to the conceptualization of the study. Yi-Yin Zhang, Xin-Yu Shen, and Xiao-Hui Xiong carried out the investigation. Methodology was developed by Yi-Yin Zhang, Xin-Yu Shen, Xiao-Hui Xiong, and Xiao-Yue Hong. Yi-Yin Zhang and Dian-Shu Zhao were responsible for formal analysis. The original draft was written by Yi-Yin Zhang, Xin-Yu Shen, and Dian-Shu Zhao. Xiao-Yue Hong supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Jan Hubert, Sassan Asgari and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editors: Hannes Schuler and Kaliya Georgieva. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, YY., Shen, XY., Xiong, XH. et al. Wolbachia-driven host miRNAs mediate arthropod reproduction in a Wolbachia density-dependent manner. Commun Biol (2026). https://doi.org/10.1038/s42003-026-10077-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-10077-3