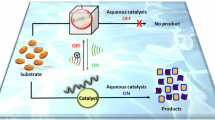
Strategies for controlling or increasing the yield of radical reactions generated by ultrasonic cavitation in aqueous media have been the object of research for many years. Past studies have focused on the role of organic solvents in increasing Reactive Oxygen Species (ROS) formation or have investigated the effect of ultrasound on accelerating the OH radicals generation from Fenton reactive. More recently, piezoelectric micro-nanoparticles have shown a synergistic effect in activating specific reactions and increasing radicals production from ultrasound. Here we report the generation of ROS together with H2 evolution or increase of oxidizing species during ultrasonic treatments of homogeneous concentrated aqueous solutions of simple salts as acidic phosphates, potassium sodium tartrate and alkaline nitrates. An increase in organic dye degradation efficiency, and the increase of reducing or oxidizing species compared with pure water has been found. The activation mechanism revealed a new, unexpected, approach to enhance the efficiency of sono-catalyzed reactions in aqueous media for environmental or energy applications.

Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author (a.troia@inrim.it) upon reasonable request.
References
Suslick, K. S. Sonochemistry. Science 247, 1439–1445 (1990).
Flint, E. B. & Suslick, K. S. The temperature of cavitation. Science 253, 13979 (1991).
Flannigan, D. J. & Suslick, K. S. Plasma formation and temperature measurement during single bubble cavitation. Nature 434, 52–55 (2005).
Taylor, K. J. & Jarman, P. D. The spectra of sonoluminescence. Aust. J. Phys. 23, 319 (1970).
Ndiaye, A. A. et al. Nonequilibrium vibrational excitation of oh radicals generated during multibubble cavitation in water *†. J. Phys. Chem. A. 116, 4860–4867 (2012).
Riesz, P. & Takashi, K. Free radical formation induced by ultrasound and its biological implications. Free Radic. Biol. Med. 13, 247–270 (1992).
Ohl, C.-D., Kurz, T., Geisler, R., Lindau, O. & Lauterborn, W. Bubble dynamics, shock waves and sonoluminescence. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 357, 269–294 (1999).
Suslick, K. S. & Price, G. J. Applications Of Ultrasound To Materials Chemistry. Annu. Rev. Mater. Res. 29, 295–326 (1999).
Mason, T. J. Sonochemistry and the environment – Providing a “green” link between chemistry, physics and engineering. Ultrason. Sonochem. 14, 476–483 (2007).
Wood, R. J., Lee, J. & Bussemaker, M. J. A parametric review of sonochemistry: Control and augmentation of sonochemical activity in aqueous solutions. Ultrason. Sonochem. 38, 351–370 (2017).
M. Singla, N. Sit energy aspects of acoustic cavitation and sonochemistry fundamentals and engineering2022, Pages 349-373 Chapter 21 - Raising challenges of ultrasound-assisted processes and sonochemistry in industrial applications based on energy efficiency, Book Chapter ISBN 978-032391937-1, 978-032398490-4 https://doi.org/10.1016/B978-0-323-91937-1.00017-7.
Asakura, Y. & Yasuda, K. Frequency and power dependence of the sonochemical reaction. Ultrason. Sonochem. 81, 105858 (2021).
Murali Krishna, C., Riesz,P. & Takashi, K. Free radical formation by ultrasound in aqueous solutions. Aspin trapping study. Free Radical Res. 10, 27–35 (1990).
Sharifishourabi, M., Dincer, I. & Mohany, A. Implementation of experimental techniques in ultrasound-driven hydrogen production: a comprehensive review. Int. J. Hydrog. Energy 62, 1183–1204 (2024).
Henglein, A. & Kormann, C. Scavenging of OH radicals produced in the sonolysis of water. Int. J. Radiat. Biol. Relat. Stud. Phys. Chem. Med. 48, 251–258 (1985).
Hansen, H. E., Seland, F., Sunde, S., Burheim, O. S. & Pollet, B. G. Optimum scavenger concentrations for sonochemical nanoparticle synthesis. Sci. Rep. 13, 6183 (2023).
Buettner, J., Gutierrez, M. & Henglein, A. Sonolysis of water-methanol mixtures. J. Phys. Chem. 95, 1528–1530 (1991).
Kohno, T., Mokudai, T. & Ozawa, Y. Niwano Free radical formation from sonolysis of water in the presence of different gases Masahiro 1,*. J. Clin. Biochem Nutr. 49, 96–101 (2011).
Gielen, B. et al. Influence of dissolved gases on sonochemistry and Sonoluminescence in a flow reactor. Ultrason. Sonochem. 31, 463–472 (2016).
Sheng Hong, K., Xu, H., Konishi, H. & Li, X. Direct Water Splitting Through Vibrating Piezoelectric Microfibers in Water. J. Phys. Chem. Lett. 1, 997–1002 (2010).
Sheng Hong, K., Xu, H., Konishi,†, H. & Li, X. Piezoelectrochemical effect: a new mechanism for azo dye decolorization in aqueous solution through vibrating piezoelectric microfibers. J. Phys. Chem. C. 116, 13045–13051 (2012).
Ahmad, M. et al. Photocatalytic, sonocatalytic and sonophotocatalytic degradation of Rhodamine B using ZnO/CNTs composites photocatalysts. Ultrason. Sonochem. 21, 761–773 (2014).
Morosini, V., Chave, T., Virot, M., Moisy, P. & Nikitenko, S. I. Sonochemical water splitting in the presence of powdered metal oxides. Ultrason. Sonochem. 29, 512–516 (2016).
Lianga, Z., Feng Yana, C., Rtimib, S. & Bandarac, J. Piezoelectric materials for catalytic/photocatalytic removal of pollutants: Recent advances and outlook. Appl. Catal. B Environ. 241, 256–269 (2019).
Bößl, F., Comyn, C. T. P., Cowin, P. I., García-García, F. R. & Tudela, I. Piezocatalytic degradation of pollutants in water: Importance of catalyst size, poling and excitation mode. Chem. Eng. J. Adv. 7, 100133 (2021).
You, H. et al. Harvesting the vibration energy of BiFeO3 nanosheets for hydrogen evolution. Angew. Chem. Int Ed. Engl. 58, 11779–11784 (2019).
Wang, Y. et al. Ultrasonic activation of inert poly (tetrafluoroethylene) enables piezocatalytic generation of reactive oxygen species. Nat. Commun. 12, 3508 (2021).
Thi, P. et al. Demonstration of enhanced piezo-catalysis for hydrogen generation and water treatment at the ferroelectric curie temperature. iScience 23, 101095 (2020).
Nie, G., Yao, Y., Duan, X., Xiao, L. & Wang, S. Advances of piezoelectric nanomaterials for applications in advanced oxidation technologies. Curr. Opin. Chem. Eng. 33, 100693 (2021).
Qian, W., Yang, W., Zhang, Y., Bowen, C. R. & Yang, Y. Piezoelectric materials for controlling electro-chemical processes. Nano-Micro Lett. 12, 149 (2020).
Li, S. et al. Mechanically induced highly efficient hydrogen evolution from water over piezoelectric snse nanosheets. Small 18, 2202507 (2022).
Wang, C. et al. Design strategies and effect comparisons toward efficient piezocatalytic system Author links open overlay panel. Nano Energy 107, 108093 (2023).
Wang, K. et al. The mechanism of piezocatalysis: energy band theory or screening charge effect? Angew. Chem. Int. Ed. 61, e202110429 (2022).
Zhang, Y. et al. High efficiency water splitting using ultrasound coupled to a BaTiO3 nanofluid. Adv. Sci. 9, 210524 (2022).
Bößl, F., Menzel, V. C., Chatzisymeon, E. I. & Tudela, I. Effect of frequency and power on the piezocatalytic and sonochemical degradation of dyes in water. Chem. Eng. J. Adv. 14, 100477 (2023).
Bößl, F. & Tudela, I. Piezocatalysis: can catalysts really dance? Curr. Opin. Green. Sustain. Chem. 32, 100537 (2021).
Jiang Y. et al. Unveiling mechanically driven catalytic processes: beyond piezocatalysis to synergetic effects https://doi.org/10.1021/acsnano.5c02660.
Mohanty, R., Mansingh, S. & Parida, K. Boosting sluggish photocatalytic hydrogen evolution through piezo-stimulated polarization: a critical review. Mater. Horiz. 9, 1332–1355 (2022).
Jing, L. et al. Piezo-photocatalysts in the field of energy and environment: designs, applications, and prospects. Nano Energy 112, 108508 (2023).
Dong, H., Zhou, Y., Wang, L., Chen, L. & Zhu, M. Oxygen vacancies in piezocatalysis: a critical review. Chem. Eng. J. 487, 150480 (2024).
Domingo, N. Understanding piezocatalysis, pyrocatalysis and ferrocatalysis. Front. Nanotechnol. 6, 1320503 (2024).
Hossain, I. & Blanchard, G. J. Ionic liquids exhibit the piezoelectric effect Md. J. Phys. Chem. Lett. 14, 2731–2735 (2023).
Mason, W. P. Piezoelectricity, its history and applications. J. Acoust. Soc. Am. 70, 1561–1566 (1981).
Nagamiya, T. On the theory of the dielectric, piezoelectric, and elastic properties of NH4H2PO4. Prog. Theor. Phys. 7, 275–284 (1952).
Scott, J. F., Duiker, H. M., Beale, P. D., Butler, D. & Eaton, S. Properties of ceramic KNO3 thin-film memories. Phys. B+C. 150, 160–167 (1988).
Samara, G. A.;The effects of deuteration on the static ferroelectric properties of kh2p04 (KDP). Ferroelectrics 5, 25–37 (1973).
Koda, S., Kimura, T., Kondo, T. & Mitome, H. A standard method to calibrate sonochemical efficiency of an individual reaction system. Ultrason. Sonochem. 10, 149–156 (2003).
Bhai Rajamma, D. et al. Sonochemical dosimetry: a comparative study of Weissler, Fricke and terephthalic acid methods. Ultrason. Sonochem. 72, 105413 (2021).
Nan Liu, Y. et al. Hydrogenation/oxidation induced efficient reversible color switching between methylene blue and leuco-methylene blue. RSC Adv. 7, 30080 (2017).
Zalata, A., Hafez, T., Mahmoud, A. & Comhaire, F. Andrology: relationship between resazurin reduction test, reactive oxygen species generation, and γ-glutamyltransferase. Hum. Reprod. 10, 1136–1140 (1995).
Merabet, N., Kerboua, K. Sonolytic and ultrasound-assisted techniques for hydrogen production: a review based on the role of ultrasound. Int. J. Hydrogen Energy 47, 17879e17893 (2022).
Ma, R. et al. Thermally-enhanced sono-photo-catalysis by defect and facet modulation of Pt-TiO2 catalyst for high-efficient hydrogen evolution. Ultrason. Sonochem. 90, 106222 (2022).
Ruszczycky, M. W. & Anderson, V. E. Tartrate dehydrogenase reductive decarboxylation: stereochemical generation of diastereotopically deuterated hydroxymethylenes. Bioorg. Chem. 32, 51–61 (2004).
Mathivanana, V., Harisb, M. & Chandrasekaran, J. Thermal, magnetic, dielectric and anti microbial properties of solution-grown pure and doped sodium potassium tartrate crystals crystals. Optik 127, 1804–1808 (2016).
Freeman, E. liS. The Kinetics of the Thermal Decomposition of Potassium Nitrate and of the Reaction between Potassium Nitrite and Oxygen113. J. Am. Chem. Soc. 79, 838–842 (1957).
Kimura T. et al. Standardization of ultrasonic power for sonochemical reaction. Ultrasonics Sonochem. 3, S157–S161 1996
Fraím, E., Serna-Galvis, A. & Torres-Palma,R. A. An experimental class to illustrate the physical and chemical effects of ultrasound as an introduction to practical advanced oxidation processes. Ultrasonics Sonochem. 112, 107196 2025
Bannov, S. I. & Nevostruev, V. A. Formation and properties of NO32−, NO3 and ONOO radicals in nitrate-containing matrices. Radiat. Phys. Chem. 68, 917–924 (2003).
Bhai Rajamma, D., Anandan, S., Saadah, N., Yusof, M. & Pollet, B. G. Muthupandian Ashokkumar; Sonochemical dosimetry: a comparative study of Weissler, Fricke and terephthalic acid methods. Ultrason. Sonochem. 72, 105413 (2021).
Wang, Y. et al. Inhibition effect of KHCO3 and KH2PO4 on ethylene explosion. ACS Omega 8, 7566–7574 (2023).
Jiang, H., Bi, M., Li, B., Ma, D. & Gao, W. Flame inhibition of aluminum dust explosion by NaHCO3 and NH4H2PO4. Combust. Flame 200, 97–114 (2019).
Wang, Q. et al. Analysis of the effectiveness of Mg(OH)2/ NH4H2PO4 composite dry powder in suppressing methane explosion. Powder Technol. 417, 118255 (2023).
Yu, M. et al. Macroscopic behaviors and chemical reaction mechanism for the inhibition of NH4H2PO4 powder in hybrid methane/coal dust deflagrations. Fuel 356, 129558 (2024).
Fijołek, L. & Nawrocki, J. Phosphate helps to recover from scavenging effect of chloride in selfenhanced ozonation. Chemosphere 212, 802–810 (2018).
Rosso, J. A., Rodriguez Nieto, F. J. & Gonzalez, M. C. Reactions of phosphate radicals with substituted benzenes. J. Photochem. Photobiol. A Chem. 116, 21–25 (1998).
Vlase, T., Vlase, G. & Doca, N. Kinetics of thermal decomposition of alkaline phosphates. J. Therm. Anal. Calorim. 80, 207–210 (2005).
Wang, Q. et al. Inhibiting effects of gas– particle mixtures containing CO2, Mg(OH)2 particles, and NH4H2PO4 particles on methane explosion in a 20-L closed vessel. J. Loss Prev. Process Indus. 64, 104082 (2020).
Korobeinichev, O. P., Shvartsberg, V. M., Shmakov, A. G. & Knyazkov, D. A. Rybitskaya Inhibition of atmospheric lean and rich CH4/O2/Ar flames by phosphorus-containing compound. Proc. Combust. Inst. 31, 2741–2748 (2007).
Li, Z. et al. The characteristics and mechanism of core-shell structure fly ash/KH2PO4 composite inhibitor in suppressing coal dust explosions J. Loss Prevention Process Indus. 94, 105533 (2024).
Yu, C. F. et al. Ren Inhibiting effect investigation of ammonium dihydrogen phosphate on oxidative pyrolysis characteristics of bituminous coal. Fuel 333, 126352 (2023).
Jayaweera, T. M. et al. Flame inhibition by phosphorus- containing compounds over a range of equivalence ratios. Combust. Flame 140, 103–115 (2005).
Xu, H., Glumac, N. G. & Suslick, K. S. Temperature inhomogeneity during multibubble sonoluminescence**. Angew. Chem. Int. 49, 1079–1082 (2010).
Wang, D., Bao, Q. W. & Han, H. Dancing bubble sonoluminescence in phosphoric acid solution. Chin. Phys. B 33, 117803 (2024).
Aghelmaleki, A., Afarideh, H., Cairós, C., Pflieger, R. & Mettin, R. Effect of mechanical stirring on sonoluminescence and sonochemiluminescence. Ultrason. Sonochem. 111, 107145 (2024).
Peng,K., Tian, S., Zhang, Y., He, Q. & Wang, Q. Penetration of hydroxyl radicals in the aqueous phase surrounding a cavitation bubble. Ultrasonics Sonochem. 91, 106235 (2022).
Cairós, C., Schneider, J., Pflieger, R. & Mettin, R. Effects of argon sparging rate, ultrasonic power, and frequency on multibubble sonoluminescence spectra and bubble dynamics in NaCl aqueous solutions. Ultrason. Sonochem. 21, 2044–2051 (2014).
Nikitenko, S. I. & Venault, L. Moisy Scavenging of OH radicals produced from H2O sonolysis with nitrate ions. Ultrason. Sonochem. 11, 139–142 (2004).
Dalodière, E., Virot, M., Moisy, P. & Nikitenko, S. I. Effect of ultrasonic frequency on H2O2 sonochemical formation rate in aqueous nitric acid solutions in the presence of oxygen. Ultrason. Sonochem. 29, 198–204 (2016).
Pardo, A., Romero, J. & Ortiz, E. High-temperature behaviour of ammonium dihydrogen phosphate IOP Conf. Ser.: J. Phys.: Conf. Ser. 935, 012050 (2017).
Margulis, M. A. & Margulis, I. M. Theory of local electrification of cavitation bubbles: new approaches. Ultrason. Sonochem. 6, 15–20 (1999).
Margulis, M. A. The kinetic equations of the electron-diffusion model of the space–time distribution of radicals in a cavitation field. Russian J. Phys. Chem. A 82, 1407–1411 (2008).
Margulis, M. A. & Margulis, I. M. Current state of the theory of local electrification of cavitation bubbles. Russian J. Phys. Chem. A 81, 129–138 (2007).
Pilgunov, V. & Efremova, K. Electro-hydromechanical processes in mineral oil flow IOP. Conf. Ser.: Mater. Sci. Eng. 779, 012032 (2020).
Cerreta, M. K. & Berglund, K. A. The structure of aqueous solutions of somedihydrogen orthophosphates by laser Raman spectroscopy. J. Cryst. Growth 84, 577–588 (1987).
Sun, C. & Xue, D. Hydrogen bonding nature during ADP crystallization. J. Mol. Struct. 1059, 338–342 (2014).
Alexandru, H. V. Second-order transition in Rochelle-salt saturated solutions. Phys. Rev. A 45, 15 (1992).
Troia a, A., Galati a, S., Vighetto b, V. & Cauda, V. Piezo/sono-catalytic activity of ZnO micro/nanoparticles for ROS generation as function of ultrasound frequencies and dissolved gases. Ultrason. Sonochem. 97, 106470 (2023).
Troia, A., Olivetti, E. S., Martino, L. & Basso, V. Sonochemical hydrogenation of metallic microparticles. Ultrason. Sonochem. 55, 1–7 (2019).
Troia, A. & Madonna Ripa, D. The role of vapour pressure in multibubble sonoluminescence from organic solvents. Ultrason. Sonochem. 18, 1180–1184 (2011).
Acknowledgements
The authors would like to thank Prof. R. Spagnolo for financial support of these experimental investigations and Dott. M. Pelassa for his scientific and technical contribution.
Author information
Authors and Affiliations
Contributions
A.T. conceived the study, performed the experiments and formulated hypothesis and conclusion of the work. V.C. and V.V. supported the experiments (EPR) and theoretical evaluation. S.H. and M.G support the experiments (GC) and statistical analysis V.M. and F.P. contributed to theoretical model of radical reactions and for the analysis of the measurements. All authors discussed the results and contributed to the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Chemistry thanks Timothy Mason and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Troia, A., Gallone, M., Vighetto, V. et al. Modulation of sonochemical reactions by cavitation driven thermal degradation of aqueous salts solutions. Commun Chem (2026). https://doi.org/10.1038/s42004-026-01961-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42004-026-01961-4