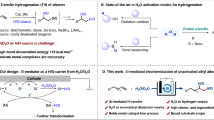
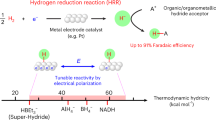
Abstract
Atomic partial charges are local, model-dependent descriptors that often fail to capture the global electrostatic environment governing chemical reactivity. This study demonstrates that the molecular electrostatic potential (ESP) at the Si–H hydrogen in trisubstituted silanes reliably predicts electrophilic versus nucleophilic behavior, whereas local charges alone can mislead. Electron-donating substituents generate hydridic hydrogens with negative ESP near hydrogen, favoring nucleophilicity. Electron-withdrawing substituents typically leave hydrogen with a negative local charge but generate a positive ESP region along the Si–H axis, promoting electrophilic character. Increasing solvent polarity amplifies these contrasts, driving minima more negative for electron-donating substituents and maxima more positive for electron-withdrawing substituents. The resulting positive region near hydrogen in electron-withdrawing silanes is directionally σ-hole-like, yet unlike classical σ-holes from lone-pair depletion, it reflects a collective molecular electrostatic effect. These findings highlight the importance of global electrostatics for understanding structure–reactivity relationships and predicting substituent-controlled behavior in silicon hydrides.

Similar content being viewed by others
Data availability
The data that support the findings of this study are available in the supporting information of this article. The publication data are available at ZENODO https://doi.org/10.5281/zenodo.18852906 under the same title.
References
Feynman, R. P. Forces in Molecules. Phys. Rev. 56, 340–343 (1939).
Suresh, C. H. & Anila, S. Molecular electrostatic potential topology analysis of noncovalent interactions. Acc. Chem. Res. 56, 1884–1895 (2023).
Cho, M. et al. The atomic partial charges arboretum: trying to see the forest for the trees. ChemPhysChem 21, 688–696 (2020).
Mahmoudi, S. et al. Experimental determination of partial charges with electron diffraction. Nature 645, 88–94 (2025).
Murray, J. S. & Politzer, P. The electrostatic potential: an overview. WIREs Comput. Mol. Sci. 1, 153–163 (2011).
Bulat, F. A., Toro-Labbé, A., Brinck, T., Murray, J. S. & Politzer, P. Quantitative analysis of molecular surfaces: areas, volumes, electrostatic potentials and average local ionization energies. J. Mol. Model 16, 1679–1691 (2010).
Politzer, P., Landry, S. J. & Waernheim, T. Proposed procedure for using electrostatic potentials to predict and interpret nucleophilic processes. J. Phys. Chem. 86, 4767–4771 (1982).
Politzer, P., Laurence, P. R. & Jayasuriya, K. Molecular electrostatic potentials: an effective tool for the elucidation of biochemical phenomena. Environ. Health Perspect. 61, 191–202 (1985).
Brinck, T., Murray, J. S. & Politzer, P. Surface electrostatic potentials of halogenated methanes as indicators of directional intermolecular interactions. Int. J. Quantum Chem. 44, 57–64 (1992).
Suresh, C. H., Remya, G. S. & Anjalikrishna, P. K. Molecular electrostatic potential analysis: a powerful tool to interpret and predict chemical reactivity. WIREs Comput. Mol. Sci. 12, e1601 (2022).
Haritha, M. & Suresh, C. H. The fundamental nature and importance of electrostatic potential in hydrogen bond formation: a case study of heterocycles frequently observed in drugs. N. J. Chem. 47, 17323–17332 (2023).
Shin, D. & Jung, Y. Molecular electrostatic potential as a general and versatile indicator for electronic substituent effects: statistical analysis and applications. Phys. Chem. Chem. Phys. 24, 25740–25752 (2022).
Mallada, B. et al. Real-space imaging of anisotropic charge of σ-hole by means of Kelvin probe force microscopy. Science 374, 863–867 (2021).
Politzer, P., Murray, J. S. & Clark, T. Halogen bonding and other σ-hole interactions: a perspective. Phys. Chem. Chem. Phys. 15, 11178 (2013).
Clark, T., Hennemann, M., Murray, J. S. & Politzer, P. Halogen bonding: the σ-hole: Proceedings of “Modeling interactions in biomolecules II”, Prague, September 5th–9th, 2005. J. Mol. Model 13, 291–296 (2007).
Kolář, M. H. & Hobza, P. Computer modeling of Halogen Bonds and other σ-Hole interactions. Chem. Rev. 116, 5155–5187 (2016).
Manna, D. et al. Beyond deshielding: NMR evidence of shielding in hydridic and protonic hydrogen bonds. J. Chem. Theory Comput. 21, 7495–7502 (2025).
Civiš, S. et al. Hydrogen bonding with hydridic hydrogen–experimental low-temperature IR and computational study: is a revised definition of hydrogen bonding appropriate? J. Am. Chem. Soc. 145, 8550–8559 (2023).
Mirza, Y. K., Bera, P. S., Mohite, S. B., Pandey, A. K. & Bera, M. Silanes as a versatile hydride source for Ni–H catalysis: a promising tool for π-hydro functionalization. Org. Chem. Front. 11, 4290–4317 (2024).
Scheiner, S. Lower limits on hydrogen bond strength. Charge of bridging H atom. Phys. Chem. Chem. Phys. 27, 10283–10290 (2025).
Adamo, C. & Barone, V. Toward reliable density functional methods without adjustable parameters: the PBE0 model. J. Chem. Phys. 110, 6158–6170 (1999).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Weigend, F. & Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: design and assessment of accuracy. Phys. Chem. Chem. Phys. 7, 3297 (2005).
Mardirossian, N. & Head-Gordon, M. B97M-V: a combinatorially optimized, range-separated hybrid, density functional with VV10 nonlocal correlation. J. Chem. Phys. 144, 214110 (2016).
Zhao, Y. & Truhlar, D. G. The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition metals: stocktaking and recommendations for multivariate density functionals and tests of design-constrained functionals. Theor. Chem. Acc. 120, 215–241 (2008).
Riplinger, C., Pinski, P., Becker, U., Valeev, E. F. & Neese, F. Sparse maps—a systematic infrastructure for reduced-scaling electronic structure methods. II. Linear scaling domain based pair natural orbital coupled cluster theory. J. Chem. Phys. 144, 024109 (2016).
Saitow, M., Becker, U., Riplinger, C., Valeev, E. F. & Neese, F. A new near-linear scaling, efficient and accurate, open-shell domain-based local pair natural orbital coupled cluster singles and doubles theory. J. Chem. Phys. 146, 164105 (2017).
Guo, Y. et al. Communication: An improved linear scaling perturbative triples correction for the domain based local pair natural orbital based singles and doubles coupled cluster method [DLPNO-CCSD(T)]. J. Chem. Phys. 148, 011101 (2018).
Klamt, A. & Schüürmann, G. COSMO: a new approach to dielectric screening in solvents with explicit expressions for the screening energy and its gradient. J. Chem. Soc. Perkin Trans. 2, 799–805 (1993).
Qin, L. et al. The strongest dative bond in main-group compounds. Theoretical study of OAeF− (Ae = Be–Ba). Phys. Chem. Chem. Phys. 26, 24294–24313 (2024).
Benkeser, R. A., Foley, K. M., Grutzner, J. B. & Smith, W. E. Evidence for the existence of the trichlorosilyl anion. J. Am. Chem. Soc. 92, 3–7 (1970).
Lamanec, M., Špirko, V., Civiš, S. & Hobza, P. Blue-shifting hydridic hydrogen bonds in complexes of (Me3Si)3SiH. J. Phys. Chem. A 129, 11512–11522 (2025).
Wolinski, K., Hinton, J. F. & Pulay, P. Efficient implementation of the gauge-independent atomic orbital method for NMR chemical shift calculations. J. Am. Chem. Soc. 112, 8251–8260 (1990).
Fulmer, G. R. et al. NMR chemical shifts of trace impurities: common laboratory solvents, organics, and gases in deuterated solvents relevant to the organometallic chemist. Organometallics 29, 2176–2179 (2010).
Goyary, S., Sarmah, M. J., Goswami, H. P. & Nath, N. Solvent-induced 1H NMR chemical shifts of annulenes. Comput. Theor. Chem. 1236, 114601 (2024).
Harris, R. K. et al. Further conventions for NMR shielding and chemical shifts (IUPAC Recommendations 2008). Magn. Reson. Chem. 46, 596–622 (2008).
Ramsey, N. F. Magnetic shielding of nuclei in molecules. Physica 17, 303–307 (1951).
McGarvey, B. R. Temperature dependence of the pseudocontact shift in lanthanide shift reagents. J. Magn. Reson. 33, 445–455 (1979).
Kurland, R. J. & McGarvey, B. R. Isotropic NMR shifts in transition metal complexes: The calculation of Fermi contact and pseudocontact terms. J. Magn. Reson. 2, 286–301 (1970).
M. J. Frisch. et al. Gaussian 16, Revision C.01; Gaussian, Inc.: Wallingford, CT, 2016.
Reed, A. E., Curtiss, L. A. & Weinhold, F. Intermolecular interactions from a natural bond orbital, donor-acceptor viewpoint. Chem. Rev. 88, 899–926 (1988).
Hirshfeld, F. L. Bonded-atom fragments for describing molecular charge densities. Theor. Chim. Acta 44, 129–138 (1977).
Marenich, A. V., Jerome, S. V., Cramer, C. J. & Truhlar, D. G. Charge model 5: an extension of Hirshfeld population analysis for the accurate description of molecular interactions in gaseous and condensed phases. J. Chem. Theory Comput. 8, 527–541 (2012).
Lu, T. & Chen, F. Atomic dipole moment corrected Hirshfeld population method. J. Theor. Comput. Chem. 11, 163–183 (2012).
Bader, R. F. W. Atoms in Molecules: A Quantum Theory(Clarendon Press, 1990).
Lu, T. & Chen, F. Multiwfn: a multifunctional wavefunction analyzer. J. Comput. Chem. 33, 580–592 (2012).
Breneman, C. M. & Wiberg, K. B. Determining atom-centered monopoles from molecular electrostatic potentials. The need for high sampling density in formamide conformational analysis. J. Comput. Chem. 11, 361–373 (1990).
Neese, F. Software update: the ORCA program system—Version 5.0. WIREs Comput. Mol. Sci. 12, e1606 (2022).
Martyna, G. J., Klein, M. L. & Tuckerman, M. Nosé–Hoover chains: the canonical ensemble via continuous dynamics. J. Chem. Phys. 97, 2635–2643 (1992).
Martyna, G. J., Tuckerman, M. E., Tobias, D. J. & Klein, M. L. Explicit reversible integrators for extended systems dynamics. Mol. Phys. 87, 1117–1157 (1996).
Acknowledgements
This article has been produced with the financial support of the European Union under the REFRESH – Research Excellence for Region Sustainability and High-tech Industries project number CZ.10.03.01/00/22_003/0000048 via the Operational Programme Just Transition (P.H.). V.H. acknowledges the support from ERDF/ESF Project TECHSCALE (Grant CZ.02.01.01/00/22_008/0004587).
Author information
Authors and Affiliations
Contributions
P.H. supervised the project. R.L. and D.M. carried out the quantum chemical calculations. V.H. performed the experiments. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Chemistry thanks Rodrigo Cormanich and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hrubý, V., Manna, D., Lo, R. et al. Ambiphilic behavior of hydrogen in trisubstituted silanes induced by substituent controlled polarity inversion. Commun Chem (2026). https://doi.org/10.1038/s42004-026-01980-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42004-026-01980-1