Abstract
Evolution has generated a sophisticated convergence of material components, ultrastructural designs, and fabrication processes in response to similar selective pressures across a diverse array of extinct and extant species. This review explores three key convergent design strategies: struts for lightweight structures with load-bearing efficiency, sutures for increased flexibility and stress management, and helicoids for impact resistance and fracture toughness. Through this examination, the review sheds light on how evolution can inspire innovative engineering approaches and technologies through the adoption of aspects of natural design. We foresee natural evolutive processes of construction as the informative harbingers of new, advanced, ecologically aware, and energy-efficient modes of human fabrication.
Similar content being viewed by others
Introduction
Organisms have developed a large variety of lightweight and robust mineralized structures, as a result of functional adaptation to withstand both biotic (e.g., body weight/gravitational loads and predatory attacks) and abiotic (e.g., fluid flow and pressure) mechanical challenges1,2,3,4. The processes by which organisms deposit minerals are referred to as “biomineralization”, which has emerged as a major adaptation in the history of life amongst animals, plants, and protists5,6. The earliest clear evidence of biomineralization has been accredited to the pre-Ediacaran/Ediacaran fossil records (717–812/550 Ma)7,8. Biomineralization became conspicuous during the Phanerozoic (i.e., after 539 Ma), likely due to escalated ecological pressures4,9. The construction of these structures, which often serve additional functions (e.g., locomotion, reproduction, reserve generation, buoyancy, and in the detection of gravity, magnetism, and light)5,6,9, is costly in terms of energy and time, and it is often limited by the selection and availability of raw materials. Hence, natural selection typically favors a compromise between minimizing material use and build time, while maximizing structural performance and multifunctionality.
Over the past few decades, considerable research has studied these structures in both extant and extinct organisms, seeking design inspiration for next-generation materials. Among the vast diversity of species, common strategies and designs have evolved iteratively over millions of years to create high-performance, multifunctional, and lightweight mineralized structures. This phenomenon of independent evolution of common traits in different phylogenetic lineages in response to similar environmental pressures is known as “convergent evolution” (or “adaptive convergence”)10. Examples are wings developed independently in various taxa (e.g., pterosaurs, bats, insects, and birds) as well as the streamlined body shapes that reduce fluid dynamic drag developed in Ichthyosaurus, sharks, penguins, and cetaceans11. In fact, evolution has generated a sophisticated convergence of material components, ultrastructural designs, and fabrication processes found in different taxa and arranged from the nano to the macroscale in response to similar selective pressures, leading to extremely effective load-bearing, impact, and crack-resistant structured materials12,13. The architecture of these biological components far exceeds current synthetically fabricated materials and thus provides motivation for the exploration of natural systems14. This approach that combines the understanding of natural solutions with the translation into technical applications is known as “biomimetics”15,16.
In this framework, convergent traits in extinct and extant organisms can offer valuable analogous stratagems and keys to problem-solving with respect to human applications because they suggest independently replicated solutions. Indeed, beyond the abiotic/biotic environment in which they live, the constitutive design of organisms is influenced by various factors, such as the constitutive mineral (e.g., calcium, salt, silica), the fabrication process (e.g., growth via accretion or molting), the phylogenetic history of the taxon (e.g., its fundamental bauplan), and their dimensional scale17,18. Convergent traits aid the process of biomimetic transfer by revealing, in-depth, the shared mechanical solutions and the underlying physical and chemical mechanisms that construct them during each independent episode of their evolution.
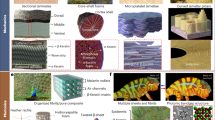
Following this approach, it is possible to recognize how certain structural challenges pervasive in evolution have been addressed evolutionarily by structural design solutions common to disparate groups of organisms. Naleway et al.19 provide examples of some common structural design solutions among extant organisms. This review aims to provide an evolutionary view of convergent mechanical strategies present in different biomineralized structures that are specifically associated with particular improvements in lightness and robustness in both extinct and extant species. Likely over 99% of species that have ever lived are extinct; therefore, the fossil record offers a huge expansion of the possible bioinspired-solution catalogues20,21. The inclusion of extinct organisms greatly increases the number of potential biological models and related strategies, leading to the emerging field of “paleomimetics”21. Because the field of structural convergence and related inspired materials is vast, this review focuses only on three convergent design strategies that are mechanically robust: struts, sutures, and helicoids, and their subsequent biomimetic translations (Fig. 1).
Brief introduction of the main convergent designs discussed in the review including representative existent and extinct examples: struts – echinoid, Pachycephalosaurus, and glyptodon; helicoids – mantis shrimp, Cuvierina and coelacanth; suture – beetle and ammonites.
Convergent design, mechanics, and biomimetics of struts
Convergent design
One of the most common strategies to decrease weight in biomineralized structures is to integrate voids/pores with complex networks of struts, the combination of which is deemed cellular solids. These structures are not only lightweight, but offer high specific stiffness/strength, high energy absorption, high permeability, good thermal insulation, and effective sound absorption (Fig. 2)22,23,24,25,26. Cellular solids can withstand large strains at nearly constant stress in compression, allowing them to absorb high impact forces. They are also capable of resisting buckling and bending, and/or increasing toughness19,22.
Main needs and sustained forces driving the evolution of the strut design strategy. Key extinct and extant examples of this convergent design: sea urchin skeletons feature a highly complex trabecular architecture, which offers a combination of lightness and mechanical robustness through variations in density, topology, and composition50; diatom thecae exhibit a unique hierarchical geometric porosity that provides multifunctional properties, such as lightness, mechanical strength, optical performance, and filtration capability139; Glyptodon body armor is composed of ossicles with a sandwich structure, which delivers impact absorption, space for vascular systems, and thermal insulation139.
A series of unique strategies have evolved over millions of years to optimize pore arrangement and the associated topology and geometry of these frameworks, increasing their mechanical performance. Cohen et al.8 described intricate scale-like microfossils with hexagonal networks of struts and oriented crystallites providing evidence for the earliest mineralized skeletons that emerged between 717–812 Ma, which perhaps was in response to protist predation27. Similar geometrical designs are also found in numerous living organisms at various length scales, some of which are known to optimize the spatial distribution of the biomineralized material (e.g., close-packed organization)28, thereby reducing mass and improving mechanical robustness, as found in diatom frustules, echinoid stereom microstructures, and sponge skeletons1,29,30.
Various cellular skeletal structures and hierarchical configurations can be found across different phyla, ranging from the skeletal lattices of sponges, the carbonate skeletons of corals and echinoderms to the complex trabecular structures of chordates24,31. The nature of these struts can vary from highly mineralized structures containing only a small amount of organic matter (i.e., less than 10% by volume) to less mineralized ones with larger amounts of organic matter, yielding structures like spicular skeletons1. Embedded spicules are widespread in nature and can be found not only in sponges, but also in coelenterates, echinoderms, mollusks, arthropods, and ascidians3. These spicular networks can consist of isolated, stiff, mineralized blocks embedded in a more compliant matrix. Interestingly, these lattice structures increasingly assume a structural tensegrity configuration, where short, isolated compression elements are immersed in multiple tension elements, which together can effectively accommodate broad-scale deformation (e.g., in the demospongiae species Tethya minuta and Spongilla lacustris)32,33. Even in highly mineralized structures, such as echinoids and trabecular bones, stiff compressive frameworks are combined with tensional elements that allow movement and deformation, including examples like collagen fibers, muscles, and tendons. The struts in nature are made of different types of basic minerals (such as calcite, silica and hydroxyapatite) and the organic matrix, besides being an important component of the struts and influencing their mechanical properties, also play a role in the nucleation of these minerals, their growth and final crystallographic configuration34. Indeed, mineral synthesis is often regulated by organic macromolecules, including mainly proteins and polysaccharides, which determine the shape of the crystallites and their spatial organization in well-defined microstructures35. Collagen and α-chitin are one of these major “framework” macromolecules, identified and extensively discussed in Chordata and in Mollusca and Arthropoda, respectively35.
Mechanics
Cellular structures are common in nature either as open cells, in which pores are interconnected, closed cells in which pores are isolated from each other, or as a porous central core architecture surrounded by dense walls, forming sandwich structures. The mechanical response of cellular solids in compression is characterized by three distinct regimes: (1) an initial linear elastic regime, related to cell edge bending (open-cell structures) or face stretching (closed-cell structures); (2) a constant stress plateau, corresponding to cell collapse by buckling, yielding, or fracture depending on the material; and (3) densification of the material, related to the collapse of the cells and the compression of cell edges and faces against each other, resulting in an increase in Young’s modulus23,24. The mechanical response of these structures during compression is sensitive to the geometry/topology, thickness, directionality, as well as nodal connectivity of the cellular structures36. Thus, these features are organized to effectively bear directionally applied loads. A well-known example is the trabecular system of vertebrate bones in which topology, thickness, and direction are aligned with the main stress trajectories, allowing the bone to resist both tensile and compressive forces37. Research by Julius Wolff in the 1870s investigated this relationship between the trabecular architecture of the proximal femur and the principal stress trajectories as determined by the “Graphical Statics” theory38. His studies demonstrated that this trabecular design is a mechanically optimized structure, achieving a balance of maximal strength with minimal weight.
Since the type and magnitude of loads are different in terrestrial, flying, and aquatic animals, different trabecular designs can be identified, with clear convergence occurring among phylogenetically distant organisms adapted to similar environments. Flying animals experience more torsion, while terrestrial and aquatic animals are more subjected to compression39. Wings of flying vertebrates (e.g., birds and bats) tend to produce a predominance of thin, dense walls with a hollow or less compact interior40. This design allows for lightweight resistance to these torsional loadings39,41. Additional interesting arrangements of struts have been identified in extinct organisms such as pterosaurs, in which the microarchitecture of a well-preserved azhdarchid pterosaur vertebra forms a helix along the long axis of the bone42. This modification almost doubles the load capability of the vertebra, allowing pterosaurs to catch and lift their prey as well as withstand strong winds during flight or intra-/interspecific fights. In the osteoderms of Glyptotherium, an extinct group of large, heavily armored mammals, a sandwich configuration of dense compact layers, combined with a porous lattice core, increases the structural strength and prevents catastrophic failures43.
Another convergent strategy within strut-based architectures is the presence of “functional gradation”, which can be found in numerous mineralized structures and skeletons across phyla, especially in spines such as the Mg-calcite ones of sea urchins, mineralized collagen spines of lionfish and porcupine fish, swordfish sword’s bone, and keratinous spines of Echidna and hedgehogs44,45,46,47. Functionally graded materials are characterized by graded variation in composition and structure over their volume leading to localized changes in mechanical properties. These materials are generally engineered to have increased mechanical performance in tandem with a reduced secretion of biomineralized material, which has major applications in advanced engineered materials48,49. An outstanding example is the hierarchical, lightweight, and porous skeletal structure of sea urchins made of Mg-calcite. This is characterized by a complex lattice-like meshwork combining different anisotropic and isotropic microarchitectures creating a species-specific structural pattern (Fig. 3)50. In sea urchin spines, the microarchitectural design results in a cascading and graceful failure behavior under bulk compression, leading to an increased high-energy absorption capacity46,51,52. Hedgehog spines consist of graded fiber-reinforced structural composites with a sandwich structure revealing superior impact and buckling resistance mechanisms47. These spines allow these animals to withstand longitudinal and radial forces from 1 × 106 ∼ 3 × 106 times the gravity generated by their own weight and can thus effectively survive dropping on the ground at a speed of up to 15 m/s47. Koehl53 examined the role of size, shape, density, and orientation of more compliant spicules in cnidarians by subjecting them to stress-extension mechanical tests. Results suggested that small proportions of spiculae can dramatically increase stiffness, especially with higher densities or more elongated anisotropic structures. Another outstanding example is the bone design in the swordfish sword, which resists high bending forces and avoids catastrophic fractures. This functional strategy arises from a graded mineralization pattern along the length of the sword. Specifically, the degree of mineralization and particle size increase from the base to the tip, and fracture experiments reveal that crack-growth toughness significantly decreases toward the tip45.
a The Paracentrotus lividus skeleton and (b) the longitudinal section of the skeletal plate showing main hypothesized stresses (arrows) and different stereom types (i.e., microarchitectures) (circles), including tubercle galleried (gt), suture galleried (gs), labyrinthic (lab) and perforate (p). c Key characteristics of the different stereom types, including the topological characteristics of Node-Segment (N-S) configuration, thickness, mechanical response to normal stresses in vertical direction, and trabecular orientation50.
Biomimetics
Based on the benefits of porous, cellular structures, they are used in numerous applicative fields including product design, automotive, aerospace, civil engineering, and architecture (Fig. 4). To meet the requirements of the final application, design and digital fabrication methods have been developed to produce highly porous architectures inspired by organismal designs at different length scales54,55. Recent studies have highlighted 3D periodic or fractal sub-micrometer frame designs as a promising area of research due to their unique mechanical properties and multifunctional capabilities, finding major application for lightweight structures, thermal insulation, catalyst scaffolds, and electrodes55,56,57,58.
Gherkin in London and Aguahoja project by Neri Oxman and The Mediated Matter Group. a Euplectella glass sponge with an inset of the lattice structure. b Gherkin, bioinspired skyscraper in the City of London. Aguahoja project entails the creation of an environmentally adaptable and sustainable pavilion made of 3D lattice chitosan-based biopolymers. c Chitin and chitosan material extraction from shrimp shells. d Close-up image of Water-Based Digital Fabrication printing system. e Map of surface patterns with mechanical and optical properties. f Front view of the Aguahoja. Courtesy of the Mediated Matter Group63.
Inspired by the hierarchical, porous design of the echinoid spine, researchers have developed functionally graded ceramics and concrete with high-energy absorption properties46,51,52. As in the spines of the sea urchin Heterocentrotus mamillatus, brittle materials can be turned into a quasi-plastic composite by changing the microstructure leading to superior energy absorption capability.
At architectural scales, an interesting example of cellular structures with struts is provided by the Trabeculae Pavilion by ACTLAB, Politecnico di Milano59. The researchers used the hierarchical structure and the adaptive growth algorithm of bones as a model to design and develop a bioinspired multiscale shell pavilion. This pavilion, which was fabricated using additive manufacturing, consists of multiple cell structures with varied porosity and orientation, which have been aligned to the main stress trajectories. To achieve this, the authors designed a computational workflow using FEM and optimization algorithms on multiple, interdependent hierarchical levels for optimal material allocation in the shell structure.
A notable architectural design that reflects conceptual similarities to the biological principles of struts is the Gherkin skyscraper in London. Designed by Norman Foster and completed in 2003, the building features a diagonal grid structure akin to the lattice found in the silica-based skeleton of Euplectella. This arrangement of vertical, horizontal, and diagonal struts enhances mechanical resistance and stability60. The diagonal steel braces of the Gherkin not only create a visually striking pattern, but also improve the building’s stability and reduce the amount of steel required compared to traditional frameworks. Furthermore, research directly inspired by the lattice structures of Euplectella sponges includes a study by Fernandes et al.61, which demonstrated that diagonal reinforcement designs offer superior buckling resistance for a given amount of material and introduced an effective optimization algorithm for generating efficient sponge-inspired geometries.
Organismal structures entail more efficient architectures and adaptable material properties compared with synthetics, avoiding negative environmental impact. Using a limited palette of minerals, natural systems create a wide diversity of multifunctional structures that can thus provide key technical solutions to develop synthetic materials with both superior performance and sustainability. Montroni et al.62 developed chitin-based gel fibers obtained by crab shell dissolution, followed by direct ink write printing through controlled nozzle geometries, resulting in highly aligned, architectured, and biocompatible scaffolds. A fascinating example of sustainably inspired designs comes from the MIT Media Lab, which developed a water-based digital fabrication platform capable of converting cellulose, chitosan, pectin, and other biopolymers into high-performance sustainable hydrogels that can be printed into multiscale 3D objects, known as Aguahoja (Fig. 4)63. Specifically, the researchers created various bio-cement composites using natural polymers and, after analyzing their chemical, mechanical, and optical properties, utilized these results to guide the digital fabrication process and the development of a large-scale design. The material distribution and the related 3D lattice structure of the final object, together with its geometry and mechanical behavior, were computationally driven and additively manufactured by controlling physical properties (e.g., gravitational loadings) and environmental conditions (e.g., temperature, humidity, and light). The resulting products can not only adapt to the environment, but are also materially programmed to decay naturally, ultimately fueling new growth.
Convergent design, mechanics, and biomimetics of sutures
Convergent design
Across both invertebrates and vertebrates, it is possible to identify unique and complex morphological discontinuities between tissue structures, which are referred to as sutures or joints. The subdivision of structures such as mineralized shells or sclerotized exoskeletons into different segments/units using distinct morphologies and sutures can provide flexibility, dissipate energy, and permit relative motion3,64. Taking into consideration the wide variability of sutures found in organismal designs—from the exoskeletons of extinct trilobites and ammonoid shells to the complex cranial sutures of extinct and extant vertebrates – interesting similarities can be observed and investigated (Fig. 5). For example, suture morphologies, even non-homologous ones, evolved in response to specific biomechanical pressures from the external environment or due to the necessity for kinematic flexibility to accommodate locomotion, respiration, and growth3,64,65. Ammonoid shell suture morphology has been postulated to respond to ecological stresses such as hydrostatic pressure66, whereas vertebrate suture morphologies, like jaw movements, have been related to stresses imposed by behavior and ecology67,68. Internal and external forces can therefore alter the suture from a butt joint morphology to a complex interdigitated morphology across a variety of length scales (i.e., nano/micro/meso/macro). The biomineralization of these sutures spans various taxa and includes non-homologous growth processes and materials. In vertebrates such as fish and humans, cranial sutures, initially made of flexible fibrous joints, allow for the expansion of the skull and it becomes ossified over time. The sutures found in reptiles retain the flexibility in adulthood allowing for movements and impact resistance69. A completely different process takes place in arthropods, which involves the assembly of soft, flexible chitin that hardens over time via cross-linking process (i.e., sclerotization)70. In crustaceans specifically, mineral crystals, such as calcium carbonate, can be deposited among the chitin and protein molecules, increasing the hardness of the body segments and resistance to compression71. Due to their rigid exoskeletons, the development of key sutures (gap sutures) may represent functional gaps during molting processes allowing these animals to accommodate the shedding of the cuticle of the previous life stage. Hence, although the formation processes of the sutures are remarkably different, they similarly address and accommodate various stresses experienced during growth and formation.
Main needs and sustained forces driving the evolution of the suture design strategy. Key extinct and extant examples of this convergent design: diabolical ironclad beetles feature interdigitated sutures in their elytra, enhancing both impact resistance and flexibility78; sea urchins skeletons consist of multiple ossicles connected by protrusions and collagen fibers, which provide flexibility, energy absorption, and accommodate growth77; ammonites exhibit unique geometric sutural patterns that contribute to buoyancy as well as mechanical stability150.
One of the earliest examples of sutures can be found in the trilobite fossil record (~525–485 Ma) of the Cambrian period72, representing a useful adaptation to environmental and predatory stresses. Facial sutures and the cephalon structure allowed these animals to withstand greater stresses, enhancing their ability to withstand predatory pressures and contributing to the evolutionary success of this iconic group of arthropods1,73. In the large variety of suture types, an interesting example at the microscale can be seen in the actinopterygian boxfish (Lactoria cornuta). Among the various fish lineages, boxfishes stand out due to the disparity of their scales constituting a real armored carapace74. Thick and enlarged hexagonal plate-like scales, or scutes, are here strategically arranged to provide mechanical robustness75. Between these scutes, mineralized “teeth” with soft collagen fibers effectively dissipate energy under different loading modes (e.g., penetration, tension, and shearing)76. Interestingly, a similar design is also seen in the tasseled skeleton of echinoids (sea urchins), where a series of modules, called plates, are joined together by multiple skeletal protrusions and collagen fibers, preventing shear movements between individual plates and reducing bending moments77. Along the same tasseled design, complex 3D sutures can be found in the turtle shell in which the multi-bone arrangement and the interface morphologies amongst them allow these organisms to resist high-magnitude loadings (e.g., impacts and predatory attacks) while permitting low-rate deformations necessary during walking, breathing, swimming, and eating65.
A different design can be found in the terrestrial beetle, Phloeodes diabolicus, also known as the diabolical ironclad beetle, in which a jigsaw-like suture is present at the interface between the two halves of its permanently closed elytra, allowing them to withstand extreme flexural stresses (Fig. 6). While its predecessors may have deployed wings to fly and the elytra to protect them, the ironclad and other terrestrial beetle species adapted the microstructural design of the elytra to enhance crush resistance by dissipating stresses within their sutural interfaces, or blades78,79,80. This mechanical toughness has been described as a direct result of delamination of sub-microstructural features (lamellae) within these blades.
a Image of Diabolical Ironclad Beetle (DIB). Scale bar, 5 mm; b Optical micrograph of a transverse cross-section (green dashed arrow) of DIB indicating the location of the (c) medial suture; (d) False-colored Scanning Electron Microscopy (SEM) micrograph of uncompressed transverse cross-section from the elytra of P. diabolicus. Bl, blades; St, stops. Inset, entire cross-section of uncompressed elytra; e Laminated microstructure of blades before (top) and after (bottom) loading; f Charge contrast and false-colored SEM micrograph of compressed elytra, indicating crack initiation (green square) around blade regions depicted in d. Inset, entire cross-section of compressed elytra; g CT reconstruction of fractured suture, revealing multiple failure points (yellow arrows), including fiber bridging and delamination in the structure. Reproduced with permission from Nature, 201978.
While predatory factors may influence the development of external organismal skeletal structures, many internal sutures can also be found, such as within bone and tooth structures. The teeth of the Chiton articulatus and other marine mollusks have shown a nanostructural interface between iron oxide and calcium phosphate phases, enabling them to graze on hard surfaces and self-sharpen, without catastrophic failure81. This is similar to sutural interfaces in human teeth that join enamel with dentin82,83. Interestingly, other bone structures found in skulls and shells also demonstrate similar structures84,85,86.
Mechanics
The intricate geometric arrangement of sutures plays a key role in determining their mechanical behavior, influencing the overall stability, stiffness, and strength of these biological interfaces87,88. Sutures exhibit a diverse array of shapes with both regular and irregular behavior, contributing to their ability to withstand varying types and magnitudes of mechanical stress. Different structures have evolved as a result of this variance, such as simple sinusoid, zigzag, serrated, or complex interdigitated wavelength or fractal-like patterns, each designed to fulfill specific biomechanical functions3,88.
Failure in these sutures has been studied and modeled extensively. To gain a comprehensive understanding, various composite mechanical models for arbitrarily shaped, sinusoidal, and interdigitating suture interfaces have been considered1,3,86,87. This exploration aimed to assess the influence of wave-form geometry and the subsequent hierarchy of these arrangements on various aspects, including load transmission, deformation mechanisms, anisotropy, and the suture interface’s stiffness, strength, and toughness under both tensile and shear loading conditions78,87,88,89,90. The structural geometry of the suture waveform proved crucial in determining overall anisotropic mechanical properties. This design and degree of interdigitation allow for a tailored response across a range of orders of magnitude by strategically selecting geometric parameters and material composition for the suture (Fig. 7)89. Such adaptability enables sutures to fulfill their protective function effectively. Notably, the presence of sutures has been found to reduce the maximum strain energy density during high-strain events by more than an order of magnitude, significantly lowering the likelihood of material failure91,92. This is attributed to the increase in compliance with a minimum amount of displacement which allows these structures to remain rigid. In addition, interfacial debonding and plastic deformation of interfaces may also allow beneficial energy dissipation and fracture-resistant properties in some cases78,93,94.
a Micro computational tomography (micro-CT) images of the pelvic suture of a three-spined stickleback (red rectangle). b The schematics of an irregular suture model and the dimensions of a tooth unit. c Morphologies of irregular sutures and regular sutures (Rstdλ = 50%, RstdA = 20%, n = 30) with three different average tooth tip angles. d Theoretical prediction of the mechanical stress-strain curves under interface failure mode where the solid line represents an irregular suture and the dotted lines represent a regular suture; Error bars represent the influence of 100 morphological variations for the same level of irregularity. Reproduced with permission from Elsevier, 201789.
As previously reported, the mechanics of the sutures are associated not only with toughening mechanisms and energy absorption, but also with permitting deformations while resisting high loads, which optimize the functionality of some biomineralized structures. From this perspective, Krauss et al.65 explained how the complex shape of the sutures in the turtle shell allows for easy deformation under minor loads while avoiding large rotations due to the stiffening effect of the interlocking neighboring bone segments. A similar mechanism has been described for the echinoid interdigitated sutures in which the flexibility of the collagen fibers determines a reduction of the bending moments within the echinoid skeleton77. The sutures allow for a significant relative rotation between plates by transmitting small bending moments while avoiding large rotations. At the macroscale, this effect results in a significant reduction of bending moments compared to a monolithic structure, contributing to the equilibration of external loads mainly through membrane forces77.
Biomimetics
Biological sutures or suture-like interfaces have been widely studied and mimicked due to their utilization in nearly every engineering and construction-based field as joining dissimilar parts and materials continues to be a challenge even today, especially in additive manufacturing87,88,95. The high strength, flexibility, shock absorption, and resistance to delamination at these interfaces play a significant role in the fabrication of materials at every length scale96,97.
The tailoring of the mechanical behavior of an interlocking structure in dynamic loading scenarios is highly sought after in many modern engineering applications. Preliminary fabrication and testing of these interfaces at the macroscale have shown increased strength and toughness98. Synthetic analogs of the jigsaw-like interface of the diabolical ironclad beetle are also being developed to join dissimilar materials to replace traditional joints and fasteners in aerospace and automotive applications. Inspired by the unique design of the elytra in the diabolical ironclad beetle, Rivera et al.78 demonstrated the mechanical efficiency of bioinspired interdigitated sutures. They designed sutures with an ellipsoidal geometry and laminated microstructure constructed of continuous carbon fiber, which led to a similar strength, yet demonstrated more than a 100% increase in toughness compared to traditional engineering joints (i.e., titanium fasteners). The laminated microstructure exhibited a more gradual failure reducing stress concentration at the neck (Fig. 8)78.
a–d Images of the samples (left); DIC of strained samples (center); and DIC of fractured samples (right); e Ashby plot showing the strength and energy absorbance of composite-metal interfaces joined by laminated carbon fiber blades (‘Multilayered’), randomly oriented chopped carbon fiber blades (‘Random fiber’) and titanium-based engineering fasteners; f Suture-like designs used in turbine blades. Reproduced with permission from Nature, 201978.
Bioinspired suture designs can also be effectively utilized at larger scales, such as in building constructions and architecture. Numerical analysis showed that the reduction of bending moments determined by the sutures in the echinoid skeleton can also be transferred in both free-form and form-found shell structures improving their structural efficiency77. The 2015–2016 ICD/ITKE Research Pavilion shows an example of direct experimentation of biomimetic sutures in modern architecture (Fig. 9)99,100. Taking inspiration from the finger joints of regular echinoids (Cidaroidea) sutures as well as ones found in irregular echinoids (Clypeasteroida) interfaces, researchers were able to model and fabricate a modular timber pavilion that can undergo controlled deflection due to the morphology of the individual segments. The study of the procedural principles of growth allowed for an integrative design process. Scalable processing methods were used to fabricate and assemble the structure, highlighting the potential scalability of the process and implications for future development.
a The structural components inspired by various suture-like designs in natural systems. b A model of the localized maximum displacement based on the implementation of each component. c The finalized ICD/ITKE Research Pavilion. Courtesy of the Universität Stuttgart ITKE, 2016100.
Convergent design, mechanics, and biomimetics of helicoids
Convergent design
Impact resistance to predators requires a mechanically robust structure that can withstand high-rate loading via the ability to dissipate energy utilizing different toughening mechanisms. One of the most dominant hierarchically assembled architectures found in many biological materials is the “helicoid” or Bouligand structure (named after French scientist Yves Bouligand)101. This extremely robust structure is characterized by a periodic stacking of unidirectional fibrous layers that are stacked upon one another in a ‘helicoidal’ arrangement. A significant amount of research in the past several decades has focused on the identification of this impressive and mechanically robust structure in a broad diversity of organismal exoskeletons. These include mineralized structures in crustaceans (e.g., crabs, fish, and lobsters)102,103,104 to non-mineralized structures such as those found in insects (e.g., beetles) (Fig. 10)105,106,107,108.
Main needs and sustained forces driving the evolution of the helicoid design strategy. Key extinct and extant examples of this convergent design: Mantis shrimp (O. scyllarus) features a helicoidal arrangement of mineralized fibers in its dactyl clubs, which enhances toughness and improves impact-absorption during prey attacks114; Arapaima gigas is characterized by multiple scales with helicoidally arranged mineralized collagen fibrils, providing resistance to punctures (e.g., from piranha bites) and bending (e.g., from scale movements)102; Azhdarchid pterosaurs exhibited helically distributed trabeculae in their hyper-elongate cervical vertebrae, which offers lightness and increases buckling resistance by up to 90%, allowing them to capture heavy prey without risking cervical damage42.
The first instance of dermal armor in fish appeared quite early within the Paleozoic period (~470 Ma). Ancient fish scales prevent penetration from predators, but would have greatly hindered flexibility during movement109. One of the earliest examples of organismal propensity for optimizing structural designs in response to similar environmental pressures can be found in the scales of the coelacanth, a species of lobe-finned fish coined a ‘living fossil’, which has existed for 400 million years and dates back to the Devonian Period110. The gradual adaptation of biological materials to environmental conditions is shown in the evolution of fish scales, showcasing a classic case of how organisms evolve structural features for survival. Coelacanth scales have a rare double-twisted helicoidal structure arranged from a rotating orthogonal bilayer made of collagen. Another, more recent example of fish scales utilizing this strategy is found in Arapaima gigas102,111. Its scale is a biological composite that consists of a highly mineralized external layer and an inner foundation consisting of a protein fibrous helicoidal framework made of collagen, reinforced in a polysaccharide matrix. The added flexibility afforded by the architecture in its scales enables A. gigas to achieve a balance between protective armor against predators and the flexibility needed for swift, agile movement in freshwater environments. In arthropods, the exoskeleton similarly serves as protective armor against predators and environmental stresses112, but also functions as a weapon in many cases113. One outstanding example is found in Odontodactylus scyllarus, a highly aggressive marine crustacean that has developed a set of dactyl clubs used for imparting high-velocity impacts on heavily armored prey, exhibiting extraordinary damage mitigation and impact resistance13,113,114,115,116,117,118,119,120. The stomatopod’s thoracic appendage consists of a ceramic-polymer composite of calcium phosphate matrix surrounding helicoidally arranged organic fibers made of chitin. Interestingly, a similar structure is found in the tail segment (i.e., telson) of the mantis shrimp, which effectively acts as a shield during ritualized fighting117.
The composition and biomineralization processes of the helicoid in all these organisms are vastly different (e.g., formation and calcification of arthropod cuticles71 or the elasmoid fish scales protruding from the skin of A. gigas)111. Nonetheless, this architecture effectively confines damage to the local site of penetration. The presence of a collagenous system, in which proteinaceous collagen is embedded within a polysaccharide matrix in fish scales102, alongside the classic chitin-protein system observed in arthropod materials112—both exhibiting helicoidal architectures—highlights a noteworthy instance of convergent evolutionary adaptation. This juxtaposition reveals that different biological lineages can independently develop structurally and functionally similar material properties to meet comparable ecological challenges. The implication of these parallel developments is significant for understanding convergent evolution. Despite the distinct biochemical nature of the building blocks (i.e., collagen in fish and chitin in arthropods), the adoption of the helicoid architecture in both systems suggests a common strategy for enhancing material properties such as strength, toughness, and resistance to predation121. Moreover, recognizing these parallels in biological materials offers valuable insights into the principles of natural material design and evolution, informing biomimetic approaches in materials science and engineering.
Mechanics
Bouligand structures, characterized by their helicoidal stacking of fibrous materials, offer a unique combination of strength, toughness, and resilience due to the combination of material components and the unique directionality of crack propagation under high strain rate impacts afforded by its architecture13,19,114,115,116,117,118,119,120,122. This structure provides fracture toughening by guiding crack propagation in the biological composite, including crack deflection at interfaces between the fibers and matrix, which is dictated by the modulus mismatch between dissimilar materials31,123. Specifically, when a stiffness mismatch between two components (e.g., matrix and fiber) exceeds a factor of four, it is expected that crack deflection will occur at the interface, resulting in energy dissipation and thus, toughening of the structure. The propensity for a crack to either deflect or propagate through a layer of stiffer material depends on Dundurs’ parameter (α) as a function of modulus mismatch and the fracture toughness of the interface (GIF) and stiffer material (GC1). This is expressed as a critical ratio (approximately 1/4) where α = GIF/GC1, which indicates that cracks are deflected at the interface when GIF < αGC1 and propagate straight through the interface when the opposite condition is true124. A higher modulus mismatch raises α, resulting in a higher tendency for crack deflection, a characteristic mode of fracture toughening. Beyond this, the work has shown that additional toughening can occur via crack twisting as well as fiber bridging, which was observed in the mantis shrimp’s dactyl club after numerous impacts119,124. As a result of this pattern, these combined mechanisms considerably increase the surface area per unit of crack length along the primary direction of crack propagation and subsequently raise the total absorbed energy during impact. Moreover, the role of mineralization in Bouligand structures is critical as it provides a stiffer matrix enveloping the less stiff fibrous material. The helicoid design’s crack deflection Dundurs’ parameter, which facilitates the modulus mismatch, influences the energy of a deflected crack tip and is therefore modulated by the biomineralization process119,124.
The architecture’s capacity to introduce various fracture toughening mechanisms that enhance a material’s rigidity effectively prevents the rapid spread of cracks that could lead to catastrophic failure. An investigation of Odontodactylus scyllarus demonstrates the dactyl club’s ability to absorb and dissipate energy from high-velocity impacts with heavily armored prey attributed to the synergistic effect between the helicoidally arranged chitin fibers and the amorphous calcium phosphate matrix (Fig. 11)114. The fiber arrangement in its thoracic appendage amplifies the ability to distribute stresses and absorb impacts partially due to its anisotropic nature, allowing the organism to withstand forces from various directions13. This orientation may be tailored (in either natural or synthetic composites) to specific loading conditions, offering directional strength where needed while maintaining overall material flexibility. Moreover, the crack deflection mechanism inherent in the helicoidal structure improves the toughness of the exoskeleton12,13,119,125. When subjected to mechanical stress, cracks that form are guided along the spiraling layers of the helicoid, increasing their path length and the energy required for further propagation. This additional toughening mechanism, crack twisting, further inhibits crack propagation in composite materials. The effectiveness of crack twisting in toughening can be characterized by the pitch angle and interlayer spacing of fibers. The crack’s trajectory changes as it interacts with the fiber layers, where this path can be described mathematically as Y = -Z ∙ tan(dϕ/dX), where ϕ is the angle of twist and X is the crack’s position along the length of the material. Additionally, local stress intensity factors change at the crack front due to the twisting, impacting the local fracture mode and energy release rates. This inherent mechanism leads to increased toughness through complex crack paths, requiring greater energy for cracks to propagate, thus enhancing a material’s overall resistance to failure118. This feature is vital for maintaining the structural integrity of the composite. Lastly, it is important to note that in structures like those found in the fish scales of Arapaima gigas, the Bouligand architecture also allows for adaptive structural reorientation at smaller length scales, as lamellae of collagen fibrils reorient towards the tensile axis in a sliding mechanism102. In this regard, Zimmerman et al.102 were able to assess this mechanical deformation by using in situ synchrotron small-angle X-ray scattering during mechanical tensile tests. The adaptive behavior of the fibrils increasingly contributes to the bending resistance of the entire scale and thus its resistance to fracture.
(with periodicity: ~75 um). Comparisons between a generalized three-dimensional model of a helicoid (a) with an SEM fractograph (b) and (c) a polished surface from a transverse cross-section; d Visualization of the chitin fiber orientations from the x-ray scattering analysis of 92 separate diffractograms obtained through two superlayers; e Three representative χ plots of the α-chitin (110) reflection used to calculate fiber angles. The plots show changes in χ across the range of angles between each chitin fiber bundle and the x-ray beam. f Topological reference of the mantis club cross-section (longitudinal section, red square, and transversal section, blue arrow) followed by (g) charge contrast scanning electron micrograph with (h) false color and (i) a model of a helicoidal slice, which accurately reproduces the fracture patterns. Reproduced with permission from Science, 2012114.
Biomimetics
The study of helicoidal structures in natural systems offers valuable insights into designing advanced materials. By following these natural designs, researchers have developed a variety of novel composites that mimic the unique combination of impact resistance and toughness found in biological organisms at multiple length scales116,126,127,128,129,130,131,132. In fact, several patents were filed and granted133,134,135, which enabled translation to engineering applications. Such materials have direct market implications in designing composite parts for protective armor, aerospace, automotive, and even sporting industries (see Helicoid Industries)136.
The development of biomimetic helicoidal architectures has been explored in multiple composite materials shaped in laminated systems of fibers, which are primarily fabricated using traditional composite fabrication128, 3D printing, or even self-assembly processes13. For example, Rivera et al.129 demonstrated the role of helicoidal architecture with mismatched modulus in impact energy dissipation, using different fibers and matrix materials. Using glass or Kevlar fibers, along with epoxy or urethane as matrix materials, they carried out drop-weight impact testing on several samples. Results revealed that the helicoidal composite panels demonstrated significantly reduced dent depth and improved residual compressive strength compared with standard quasi-isotropic designs. Additionally, the epoxy-matrix composites showed the highest peak load on impact, but greater external damage, such as delamination and fiber breakage, compared to those with a urethane-based matrix. Other researchers are exploring the impact resistance of 3D-printed concrete materials–specimens manufactured with or without steel fibers, with helicoidal patterns varying in pitch angle. A study by Liu et al. shows that the change in pitch angles is negligible in the resulting performance of printed concrete specimens without steel fibers137. Conversely, the influence of pitch angle was much more significant in the specimens with steel fibers where helicoidal patterns outperformed a unidirectional design with respect to impact duration, peak impact force, and energy absorption. As in the dactyl club of Odontodactylus scyllarus, these printed concrete composites present complex crack propagation behaviors, including crack deflection, twisting, and crack bridging137. Notably, the bioinspired helicoid design can also be implemented with bio-based materials and natural fibers, as demonstrated by numerous design experiments in the Kisailus lab using flax (Fig. 12). This foreshadows the development of more sustainable products and significantly enhances the mechanical performance, durability, and thus usability of bio-based materials.
a Example schematics of (i) biaxial, (ii) quasi-isotropic, and (iii) helicoid architecture in a composite stacking sequence; b VARTM processing of a flax composite; c carbon fiber composite coupons for impact testing; d implementation of patented helicoid technology into CCM hockey sticks through Helicoid Industries, Inc151.
Building on structural engineering design, modern architecture has also increasingly drawn inspiration from the helicoid architecture, characterized by its complex, spiral arrangements offering both aesthetically appealing and functional advantages. Among the interesting examples of implementing the helicoid in architectural design are the Turning Torso building (Sweden) and the Bramante Staircase in the Vatican City (Italy).
Discussion, limits, and challenges
Biomimetic technologies are continually developing, generating bio-inspired synthetic systems and processes that utilize the constructive laws of nature. From a biomimetic perspective, organisms are usually defined as functional models with efficient designs adapted to originate, live, and effectively perform in a specific environment. However, these designs are not a result of an intentional activity to realize optimized systems or the best absolute solution in terms of functional efficiency. Biological systems and processes are the consequence of the combined effects of chance and necessity, which are properties of evolution, and are constrained by numerous factors (e.g., phylogenetics, material composition and availability, fabrication processes, and environmental variables). The organismal design therefore emerges from a continuous adaptation with the available materials and processes, rather than an absolute invention or optimization138. Hence, it is possible to find traits that are imperfect, limited by other traits, or with no apparent functional role (e.g., trade-offs and vestigial traits)21 constituting traps or blind alleys for biomimetic transfers.
How, then, can we discern functional traits from incidental or sub-optimal ones for an effective biomimetic transfer? The key is convergence, i.e., shared derived traits that have evolved independently as solutions to similar structural problems. In this light, fossilized organisms together with extant ones can provide numerous examples of convergent evolution, offering a good understanding of the evolution of form and function, overcoming the species-specific morphological constraints. The inclusion of another dimension, time, allows for the identification of when, how, and why specific convergent traits evolved, including cases derived from taxa with fundamentally different original structures. This provides specific biomimetic solutions with minimal risk of distortion or misinterpretation of the structure-function relationship.
In this complex framework, the present review provides an overview of convergent organismal ultrastructural designs developed throughout time to create biomineralized (and non-mineralized) lightweight and robust structures, leading to unique bio-inspired applications. The overview entails convergent, mechanical, and biomimetic aspects of struts, sutures, and helicoids, including examples from extinct organisms, and an evolutionary perspective.
-
Struts and mineralized networks enable lightweight structures and generally include key strategies to ensure mechanical robustness, distributing and absorbing the applied stresses. This design emerged in different phyla ranging from the spicular skeleton of sponges to mineralized cellular solids present in vertebrate bones. The ratio of stiff to soft materials appears to be associated with the function: spicular skeletons with soft, tensegrity-like structures are found in sponges that need to contract, while skeletons in large animals must be sufficiently stiff to support gravitational loads and serve as levers and attachment sites for muscles to ensure body movement. While an increasing number of studies are emerging on the mechanics underlying biological struts1,4,11,13,19,22,23,24,30,31,32,33,36,37,38,39,40,41,42,43,44,45,46,47,48,49,51,52,53,54,55,56,57,58,59,60,61,62,63,139, there remains a limited understanding of their formation across different taxa, optimization strategies, and trade-offs, as well as scale effects. For instance, it is still unclear how the design and functionality of these struts change with scale and how these principles can be replicated and adapted in engineering materials and structures. Among the potential examples, can the geometric effectiveness of diatom porosity be translated to meso- or macroscales, or is its mechanical strategy limited to the microscale?
-
Sutural designs consist of interdigitating interfaces, which vary in geometry and related mechanics. Sutures provide more flexibility to otherwise stiff structures, allowing adaptability and movement. For example, bathymetrical compartments in ammonite shells provide flexibility for respiration, while similar structural adaptations enhance locomotion in turtle shells65. Although the biomimetic potential is promising, there are relatively few studies addressing the mechanics of joints, interface variability in nature, repair and regeneration mechanisms following damage, and interactions with organic and soft tissues. Notably, in some animals, such as echinoderms, the stiffness of connective tissues can change within a few seconds through nervous control, as exemplified by the “mutable collagenous tissue”140. This raises an intriguing question: Is this a convergent strategy observed in the joints of other organisms?
-
Helicoidal architectures consist of a spiraling arrangement of fibrous materials surrounded by a continuous matrix, providing structural strength, toughness, and resilience. This biological strategy can be found in a variety of protective mineralized and sclerotized structures that are subjected from quasi-static to high strain rate impacts, such as stomatopod dactyl clubs, which experience high-velocity impacts, or coelacanth and arapaima scales, which resist penetration102,103,104,111,114. Despite the functionalities of helicoid structures and the promising implications for biomimetic applications, several key gaps in knowledge persist. For instance, the effects of varying materials, pitch angles, thicknesses, gradients, and geometries on mechanical performance—observed across different species—and their relation to principal applied stresses, are not yet well understood. Furthermore, the performance of helicoid structures at different length scales, from microscopic to macroscopic, remains inadequately explored. Gaining a comprehensive understanding of this performance is crucial for advancing the scalability and biomimetic application of these designs.
Thus, despite the extensive existing literature, significant gaps in knowledge remain within each convergent design, representing opportunities for future research. More broadly, our understanding of how specific biomineralization processes, biomechanical principles, and evolutionary pathways have led to the diversity of struts, sutures, and helicoids across different organisms is still limited.
Although general processes of biomineralization are known5,141,142, more detailed studies on the specific proteins, organic matrices, and environmental conditions that control self-assembly and mineral deposition in organisms are not yet fully understood. The organic matrix is thought to play a central role in crystal growth, but the composition and mineral deposition can change across the different taxa, species, and even in different regions of the same organism (e.g., the different specialized macromolecules controlling the different mineral layers in the mollusk shell)143,144. Hence, key comparative studies are needed to understand how different taxa and organisms can achieve similar structural functions through varying biomineralization strategies. More biomechanical research is certainly needed to understand how convergent designs in different organismal bauplan respond to various mechanical stresses, including dynamic and cyclic loading. This includes investigating how these architectures adapt to different geometric parameters and scales (such as strut structure and dimensions; sutural shapes and multiplicity; Bouligand layers and pitch angles) and their influence on mechanical performance. Finally, more comprehensive phylogenetic studies are necessary to identify how these designs have evolved in response to different environmental pressures, functional requirements, and possible trade-offs. This includes understanding the role of genetic and environmental factors in shaping these structures, in which the study of convergent designs can effectively support the identification of related evolutive constraints.
Finally, it is interesting to notice how each convergent design can absolve multiple functionalities in the organism besides the specific mechanical purpose. For instance, the unique hierarchical geometric porosity in diatom thecae provides lightness and mechanical strength, as well as key optical performance and filtration capability139,145. The sandwich structure in the Glyptodon ossicles provides impact absorption, as well as space for vascular systems and thermal insulation (Fig. 2)43. Moreover, different convergent designs are often integrated within the same organism to address multiple functional challenges simultaneously. In the echinoid skeleton, the lightweight trabecular structure is combined with the presence of multiple sutures that serve mechanical purposes by reducing bending moments and optimizing stress distribution77. Additionally, these sutures facilitate organismal growth by providing space between plate margins for the addition and accretion of new plates. Another example is the microarchitecture of the Azhdarchid pterosaur vertebra, which forms a helix along the long axis of the bone, providing lightness useful for flight while also improving structural strength and toughness (see section “Mechanics”)42.
Outlook
The evolving structural patterns in nature offer valuable insights that drive technological progress and new research pathways. Human-made products, created solely for human needs, often harm ecosystems, lack sustainability, and fuel environmental crises. There is an urgent need to develop real-world solutions with deep respect for nature. By understanding why certain multiscale architectures were derived as well as how they are constructed, we can revolutionize engineering, moving beyond simple part assembly to integrated, ecologically aware growth146.
As we consider the future of structural design, it is important to explore how new models can predict architectures that evolve under constraints vastly different from those found in nature. Ecological pressures have long driven the selection of the material and structural features of organisms. For example, competition for food guided the evolution of divergent feeding appendages in stomatopod crustaceans: elongated spear-like appendages to ambush soft-bodied prey (spearers) or hammer-like appendages (smashers) to break hard-bodied prey147,148. However, when we replace these natural constraints with modern challenges, such as hypersonic vehicles or satellites, entirely new design paradigms must be considered. Figure 13 illustrates how ancestral spearing stomatopods evolved a subspecies of smashers with helicoidally reinforced dactyl clubs capable of hunting hard-bodied prey, demonstrating nature’s ability to adapt structurally over time149. Extrapolating this concept to modern engineering challenges, we can potentially utilize artificial intelligence and machine learning, combined with additive manufacturing, to explore new design spaces or “morphospaces”. This approach may lead to the development of entirely new, non-native structures that are optimized for the unique challenges of our era, surpassing the limitations of biological evolution (e.g., trade-offs or native constraints).
Spearing mantis shrimp evolves specialized smashing club-like appendages in response to the emergence of hard-bodied prey. The concept is extrapolated to modern challenges (e.g., extreme armored vehicles), proposing AI and machine learning algorithms coupled with additive manufacturing for future structural design innovations.
Accordingly, by viewing nature as both a measure and mentor, we can learn how to code advanced design sustainably while addressing specific and/or extreme technical needs (e.g., resistance to extreme temperatures, radiation, and other conditions in space) that define the new technical morphospace. This morphospace comprises: (i) coding, the information (“genetics”) guiding design conception and generation; (ii) functionality or multifunctionality, including specific purposes; (iii) fabrication, material choice, and processes; and (iv) environmental awareness and integration. In this framework, nature can inform human technology with new functional designs (e.g., bioinspiration and bio/paleo-mimetics) and with intrinsically sensitive, active, and programmable materiality (e.g., bio-based and living engineered materials), all of which are ecologically integrated. In this regard, evolutionary convergence can effectively indicate what is functionally significant in nature and how designs were adapted to specific environments. The inclusion of extinct organisms can drastically increase the number of natural models and solutions for bioinspired innovation. Certain extinct organisms lived in contexts unrepresented today, including extreme climates, expanding the repertoire of convergent adaptive solutions and potentially revealing solutions relevant to the climatic changes we ourselves have initiated. Nonetheless, while the use of living organisms is becoming a common practice in biomimetics, the identification of solutions independently derived within multiple organismal lineages, living and extinct, presents challenges as well as opportunities for bioinspired research driven by the historical record of evolution and the strategies adopted by extinct organisms. In this context, this review represents an initial attempt to anchor the biomimetic approach to iteratively evolved, natural, convergent design solutions and to promote a new hybrid approach that marries convergent evolution with biomimetic design. This approach aims to lead to sustainable and programmable technologies that themselves evolve.
References
Wainwright, S. A. Mechanical design in organisms (Princeton University Press, 1982).
Wegst, U. G. & Ashby, M. F. The mechanical efficiency of natural materials. Philos. Mag. 84, 2167–2186 (2004).
Vogel, S. Comparative biomechanics: life’s physical world (Princeton University Press, 2013).
Hamm, C. Evolution of Lightweight Structures: Analyses and Technical Applications. (Springer, Berlin/Heidelberg, Germany, 2015).
Lowenstam, H. A. Minerals formed by organisms. Science 211, 1126–1131 (1981).
Addadi, L. & Weiner, S. Biomineralization: mineral formation by organisms. Phys. Scr. 89, 098003 (2014).
Clites, E. C., Droser, M. L. & Gehling, J. G. The advent of hard-part structural support among the ediacara biota: Ediacaran harbinger of a Cambrian mode of body construction. Geology 40, 307–310 (2012).
Cohen, P. A., Schopf, J. W., Butterfield, N. J., Kudryavtsev, A. B. & Macdonald, F. A. Phosphate Biomineralization in Mid-Neoproterozoic Protists. Geology 39, 539–542 (2011).
Gilbert, P. U. P. A. et al. Biomineralization: integrating mechanism and evolutionary history. Sci. Adv. 8, 9653 (2022).
Bels, V. L., Russell, A. P. Convergent Evolution (Springer, 2023).
Hildebrand, M., Goslow, G. E. Analysis of vertebrate structure. In Analysis of vertebrate structure pp 125–155 (Wiley, 2001).
Huang, W. et al. Multiscale toughening mechanisms in biological materials and bioinspired designs. Adv. Mater. 31, 1901561 (2019).
Yaraghi, N. A. & Kisailus, D. Biomimetic structural materials: inspiration from design and assembly. Annu. Rev. Phys. Chem. 69, 23–57 (2018).
Barthelat, F. Architectured materials in engineering and biology: fabrication, structure, mechanics and performance. Int. Mater. Rev. 60, 413–430 (2015).
Vincent, J. F., Bogatyreva, O. A., Bogatyrev, N. R., Bowyer, A. & Pahl, A. K. Biomimetics: its practice and theory. J. R. Soc. Interface 3, 471–482 (2006).
Bhushan, B. Biomimetics: lessons from nature–an overview. Philos. Trans. R. Soc. Math. Phys. Eng. Sci. 367, 1445–1486 (2009).
Seilacher, A. Gishlick A. D. Morphodynamics (CRC Press, 2014).
Perricone, V., Santulli, C., Rendina, F. & Langella, C. Organismal design and biomimetics: a problem of scale. Biomimetics 6, 56 (2021).
Naleway, S. E., Porter, M. M., McKittrick, J. & Meyers, M. A. Structural design elements in biological materials: application to bioinspiration. Adv. Mater. 27, 5455–5476 (2015).
Jablonski, D. Extinction: past and present. Nature 427, 589 (2004).
Perricone, V., Grun, T., Raia, P. & Langella, C. Paleomimetics: a conceptual framework for a biomimetic design inspired by fossils and evolutionary processes. Biomimetics 7, 89 (2022).
Ashby, M. F., Medalist, R. M. The mechanical properties of cellular solids. https://doi.org/10.1007/BF02645546 (1983).
Gibson, L. J. Cellular solids. MRS Bull. 28, 270–274 (2003).
Gibson, L. J. Biomechanics of cellular solids. J. Biomech. 38, 377–399 (2005).
Meyers, M. A., Chen, P. Y., Lin, A. Y. M. & Seki, Y. Biological materials: structure and mechanical properties. Prog. Mater. Sci. 53, 1–206 (2008).
Meyers, M. A., McKittrick, J. & Chen, P.-Y. Structural biological materials: critical mechanics-materials connections. Science 339, 773–779 (2013).
Porter, S. The rise of predators. Geology 39, 607–608 (2011).
Pearce, P. Structure in nature is a strategy for design (MIT Press, 1990).
Perricone, V. et al. Hexagonal Voronoi pattern detected in the microstructural design of the echinoid skeleton. J. R. Soc. Interface 19, 20220226 (2022).
Weaver, J. C. et al. Hierarchical assembly of the siliceous skeletal lattice of the hexactinellid sponge. Euplectella Aspergillum. J. Struct. Biol. 158, 93–106 (2007).
Fratzl, P. & Weinkamer, R. Nature’s hierarchical materials. Prog. Mater. Sci. 52, 1263–1334 (2007).
Nickel, M., Bullinger, E. & Beckmann, F. Functional morphology of Tethya species (Porifera): 2. three-dimensional morphometrics on spicules and skeleton superstructures of T. Minuta. Zoomorphology 125, 225–239 (2006).
Woźnica, A., Karczewski, J., Bernaś, T., Świątek, P., Drab, M., Surma, R., Babczyńska, A. The spatial structure (3D) and mechanical properties of the sponge Spongilla Lacustris L. (Porifera: Spongillida) skeleton as a potential tensegral architecture. Eur. Zool. J. 89, 1002–1017 (2022).
Marin, F., Bundeleva, I., Takeuchi, T., Immel, F. & Medakovic, D. Organic matrices in metazoan calcium carbonate skeletons: composition, functions, evolution. J. Struct. Biol. 196, 98–106 (2016).
Lowenstam, H. A. & Weiner, S. On biomineralization (Oxford Univ. Press, 1989).
Fleck, N. A., Deshpande, V. S. & Ashby, M. F. Micro-architectured materials: past, present and future. Proc. R. Soc. Math. Phys. Eng. Sci. 466, 2495–2516 (2010).
Von Meyer, G. Die Architektur. der Spongiosa. Archs Anat. Physiol. Wiss. Med 34, 615–628 (1867).
Wolff, J. Ueber die innere Architectur der Knochen und ihre Bedeutung für die Frage vom Knochenwachsthum. Clin. Orthop. Relat. Res. 468, 1056–1065 (2010).
Padian, K., Lamm, E. T. Why study the bone microstructure of fossil tetrapods. bone histology of fossil tetrapods: advancing methods, analysis, and interpretation (University of California Press, 2013).
Swartz, S. M., Bennett, M. B., Carrier, D. R. Wing bone stresses in free flying bats and the evolution of skeletal design for flight. Nature https://doi.org/10.1038/359726a0 (1992).
de Margerie, E. Laminar bone as an adaptation to torsional loads in flapping flight. Anat. Rec. 201, 521–526 (2002).
Williams, C. J. et al. Helically arranged cross struts in azhdarchid pterosaur cervical vertebrae and their biomechanical implications. Science 24. https://doi.org/10.1016/j.isci.2021.102338 (2021).
Du Plessis, A., Broeckhoven, C., Yadroitsev, I., Yadroitsava, I. & Roux, S. G. Analyzing nature’s protective design: the glyptodont body armor. J. Mech. Behav. Biomed. Mater. 82, 218–223 (2018).
Su, F. Y. et al. Spines of the porcupine fish: structure, composition, and mechanical properties. J. Mech. Behav. Biomed. Mater. 73, 38–49 (2017).
Schmidt, F. N. et al. On the origins of fracture toughness in advanced teleosts: how the swordfish sword’s bone structure and composition allow for slashing under water to kill or stun prey. Adv. Sci. 6, 1900287 (2019).
Toader, N., Sobek, W. & Nickel, K. G. Energy absorption in functionally graded concrete bioinspired by sea urchin spines. J. Bionic Eng. 14, 369–378 (2017).
Li, Y. et al. Porous morphology and graded materials endow hedgehog spines with impact resistance and structural stability. Acta Biomater. 147, 91–101 (2022).
Torelli, G., Fernández, M. G. & Lees, J. M. Functionally graded concrete: design objectives, production techniques, and analysis methods for layered and continuously graded elements. Constr. Build. Mater. 242, 118040 (2020).
Ahamed, M. K., Wang, H. & Hazell, P. J. From biology to biomimicry: using nature to build better structures–a review. Constr. Build. Mater. 320, 126195 (2022).
Perricone, V. et al. Echinoid skeleton: an insight on the species-specific pattern of the Paracentrotus lividus plate and its microstructural variability. J. R. Soc. Interface 20, 20220673 (2023).
Presser, V., Schultheiß, S., Berthold, C. & Nickel, K. G. Sea urchin spines as a model-system for permeable, light-weight ceramics with graceful failure behaviour. Part I. Mechanical behaviour of sea urchin spines under compression. J. Bionic Eng. 6, 203–213 (2009).
Grossmann, J. N. & Nebelsick, J. H. Comparative morphological and structural analysis of selected cidaroid and camarodont sea urchin spines. Zoomorph 132, 301–315 (2013).
Koehl, M. A. R. Mechanical design of spicule-reinforced connective tissue: stiffness. J. Exp. Biol. 98, 239–267 (1982).
Hu, Z. et al. Design of ultra-lightweight and high-strength cellular structural composites inspired by biomimetics. Compos. B Eng. 121, 108–121 (2017).
Ramakrishna, D. & Bala Murali, G. Bio-inspired 3d-printed lattice structures for energy absorption applications: a review. Proc. Inst. Mech. Eng. J Mater. Des. Appl. 237, 503–542 (2023).
Jang, J. H. et al. 3D polymer microframes that exploit length‐scale‐dependent mechanical behavior. Adv. Mater. 18, 2123–2127 (2006).
Zhao, N. et al. Bioinspired materials: from low to high dimensional structure. Adv. Mater. 26, 6994–7017 (2014).
Zhang, H. K., Wu, W. J., Kang, Z. & Feng, X. Q. Topology optimization method for the design of bioinspired self-similar hierarchical microstructures. Comput. Methods Appl. Mech. Eng. 372, 113399 (2020).
Naboni, R. & Kunic, A. Bone-Inspired 3D printed structures for construction applications. Gest. Tecnol. Proj. 14, 111–124 (2019).
Weaver, J. C. et al. Hierarchical assembly of the siliceous skeletal lattice of the hexactinellid sponge Euplectella aspergillum. J. Struct. Biol. 158, 93–106 (2007).
Fernandes, M. C., Aizenberg, J., Weaver, J. C. & Bertoldi, K. Mechanically robust lattices inspired by deep-sea glass sponges. Nat. Mater. 20, 237–241 (2021).
Montroni, D. et al. Direct ink write printing of chitin-based gel fibers with customizable fibril alignment, porosity, and mechanical properties for biomedical applications. J. Funct. Biomater. 13, 83 (2022).
Duro-Royo, J., Zak, J., Tai, Y. J., Ling, A. S., Oxman, N. Parametric chemistry reverse engineering biomaterial composites for additive manufacturing of bio-cement structures across scales. In Challenges for Technology Innovation: An Agenda for the Future 217–223 (CRC Press, 2017).
Li, Y., Ortiz, C. & Boyce, M. C. Bioinspired, mechanical, deterministic fractal model for hierarchical suture joints. Phys. Rev. E 85, 031901 (2012).
Krauss, S., Monsonego‐Ornan, E., Zelzer, E., Fratzl, P. & Shahar, R. Mechanical function of a complex three‐dimensional suture joining the bony elements in the shell of the red‐eared slider turtle. Adv. Mater. 21, 407–412 (2009).
Hewitt, R. A., Westermann & Gerd, E. G. Mechanical significance of ammonoid septa with complex sutures. Lethaia 30, 205–212 (1997).
Jaslow, C. R. & Biewener, A. A. Strain patterns in the horncores, cranial bones and sutures of goats (Capra Hircus) during impact loading. J. Zool. 235, 193–210 (1995).
Buezas, G., Becerra, F. & Vassallo, A. Cranial suture complexity in caviomorph rodents (Rodentia; Ctenohystrica). J. Morphol. 278, 1125–1136 (2017).
Shahar, R., Kraus, S., Monsonego-Ornan, E. & Fratzl, P. Mechanical function of a complex three-dimensional suture joining the bony elements in the shell of the red-eared slider turtle. MRS Online Proc. Libr. 1187, 1187–KK01 (2009).
Andersen, S. O. Insect cuticular sclerotization: a review. Insect Biochem. Mol. Biol. 40, 166–178 (2010).
Luquet, G. Biomineralizations: insights and prospects from crustaceans. ZooKeys 176, 103 (2012).
Paterson, J. R. & Edgecombe, G. D. The early Cambrian trilobite family emuellidae Pocock, 1970: systematic position and revision of Australian species. J. Paleontol. 80, 496–513 (2006).
Esteve, J., Marcé‐Nogué, J., Pérez‐Peris, F. & Rayfield, E. Cephalic biomechanics underpins the evolutionary success of trilobites. Palaeontology 64, 519–530 (2021).
Santini, F., Sorenson, L., Marcroft, T., Dornburg, A. & Alfaro, M. E. A multilocus molecular phylogeny of boxfishes (Aracanidae, Ostraciidae; Tetraodontiformes). Mol. Phylogenet. Evol. 66, 153–160 (2013).
Yang, W., Naleway, S. E., Porter, M. M., Meyers, M. A. & McKittrick, J. The armored carapace of the Boxfish. Acta Biomater. 23, 1–10 (2015).
Garner, S. N. et al. The role of collagen in the dermal armor of the boxfish. J. Mater. Res. Technol. 9, 13825–13841 (2020).
Marmo, F., Perricone, V., Cutolo, A., Daniela Candia Carnevali, M., Langella, C., Rosati, L. Flexible sutures reduce bending moments in shells: from the echinoid test to tessellated shell structures. R. Soc. Open Sci. 9, 211972 (2022).
Rivera, J. et al. Toughening mechanisms of the elytra of the diabolical ironclad beetle. Nature 586, 543–548 (2020).
Rivera, J. et al. Structural design variations in beetle elytra. Adv. Funct. Mater. 31, 2106468 (2021).
Rivera, J., Yaraghi, N., Arango, D., Zavattieri, P., Kisailus, D. Compression resistant designs from the exoskeleton of a tough beetle. Proceedings of the 21st International Conference on Composite Materials, August 20–25 https://api.semanticscholar.org/CorpusID:211199922 (2017).
Montroni, D., et al. The Multiphasic Teeth of Chiton Articulatus, an Abrasion‐resistant and Self‐sharpening Tool for Hard Algae Collection. Adv. Funct. Mater. https://doi.org/10.1002/adfm.202401658 (2024).
Beniash, E., et al. The hidden structure of human enamel. Nat. Commun. 10 https://doi.org/10.1038/s41467-019-12185-7 (2019).
Al-Mosawi, M., et al. Crystallographic texture and mineral concentration quantification of developing and mature human incisal enamel. Sci. Rep. 8. https://doi.org/10.1038/s41598-018-32425-y (2018).
Mao, J. J. Mechanobiology of craniofacial sutures. J. Dent. Res. 81, 810–816 (2002).
Miura, T. et al. Mechanism of skull suture maintenance and interdigitation. J. Anat. 215, 642–655 (2009).
Alheit, B., Bargmann, S. & Reddy, B. D. Computationally modelling the mechanical behaviour of turtle shell sutures—a natural interlocking structure. J. Mech. Behav. Biomed. Mater. 110, 103973 (2020).
Li, Y., Ortiz, C. & Boyce, M. C. A generalized mechanical model for suture interfaces of arbitrary geometry. J. Mech. Phys. Solids 61, 1144–1167 (2013).
Li, Y., Ortiz, C. & Boyce, M. C. Stiffness and strength of suture joints in nature. Phys. Rev. E 84, 062904 (2011).
Liu, L. et al. The effects of morphological irregularity on the mechanical behavior of interdigitated biological sutures under tension. J. Biomech. 58, 71–78 (2017).
Wu, D. et al. Stiffness and toughness of soft/stiff suture joints in biological composites. Appl. Math. Mech. 43, 1469–1484 (2022).
Alheit, B., Bargmann, S. & Reddy, B. How suture networks improve the protective function of natural structures: a multiscale investigation. Acta Biomater. 145, 283–296 (2022).
Lee, N. et al. Stress wave mitigation at suture interfaces. Biomed. Phys. Amp Eng. Express 3, 035025 (2017).
Zhang, J. & An, B. Contribution of energy dissipation to dynamic fracture resistance of the turtle carapace. Eng. Fract. Mech. 290, 109505 (2023).
Zavattieri, P. D., Hector, L. G. & Bower, A. F. Cohesive zone simulations of crack growth along a rough interface between two elastic-plastic solids. Eng. Fract. Mech. 75, 4309–4332 (2008).
Gibbons, M. M. & Chen, D. A. Bio-inspired sutures: using finite element analysis to parameterize the mechanical response of dovetail sutures in simulated bending of a curved structure. Biomimetics 7, 82 (2022).
Lazarus, B. S., Velasco-Hogan, A., Gómez-del Río, T., Meyers, M. A. & Jasiuk, I. A review of impact resistant biological and bioinspired materials and structures. J. Mater. Res. Technol. 9, 15705–15738 (2020).
Huang, W. et al. Nanoarchitected tough biological composites from assembled chitinous scaffolds. Acc. Chem. Res. 55, 1360–1371 (2022).
Malik, I. A., Mirkhalaf, M. & Barthelat, F. Bio-inspired “Jigsaw”-like interlocking sutures: modeling, optimization, 3D printing and testing. J. Mech. Phys. Solids 102, 224–238 (2017).
Nikookar, N. Design through digital making: a human-system collaboration framework. Master’s Thesis, Carnegie Mellon University, Pittsburgh, PA. https://doi.org/10.13140/RG.2.2.25739.92964 (2020).
Menges, A. et al. ICD/ITKE Research Pavillion 2015-16 Project Report. (Presskit: Stuttgart, Germany, 2016). https://www.itke.uni-stuttgart.de/research/icd-itke-research-pavilions/icd-itke-research-pavilion-2015-16/.
Bouligand, Y. On a twisted fibrillar arrangement common to several biological structures. C R Hebd. Séances Acad. Sci. Sci. Nat. 261, 4864 (1965).
Zimmermann, E. A. et al. Mechanical adaptability of the bouligand-type structure in natural dermal armour. Nat. Commun. 4, 1–7 (2013).
Cheng, C., Shao, Z. & Vollrath, F. Silk fibroin-regulated crystallization of calcium carbonate. Adv. Funct. Mater. 18, 2172–2179 (2008).
Sachs, C., Fabritius, H. & Raabe, D. Influence of microstructure on deformation anisotropy of mineralized cuticle from the lobster Homarus Americanus. J. Struct. Biol. 161, 120–132 (2008).
Cheng, L., Wang, L. & Karlsson, A. M. Mechanics-based analysis of selected features of the exoskeletal microstructure of Popillia japonica. J. Mater. Res. 24, 3253–3267 (2009).
Libby, E. et al. Light reflection by the cuticle of C. Aurigans scarabs: a biological broadband reflector of left handed circularly polarized light. J. Opt. 16, 082001 (2014).
Campos-Fernández, C., Azofeifa, D. E., Hernández-Jiménez, M., Ruiz-Ruiz, A. & Vargas, W. E. Visible light reflection spectra from cuticle layered materials. Opt. Mater. Express 1, 85–100 (2011).
Jewell, S. A., Vukusic, P. & Roberts, N. W. Circularly polarized colour reflection from helicoidal structures in the beetle plusiotis boucardi. N. J. Phys. 9, 99 (2007).
Bruet, B. J. F., Song, J., Boyce, M. C. & Ortiz, C. Materials design principles of ancient fish armour. Nat. Mater. 7, 748–756 (2008).
Yin, S. et al. Toughening mechanism of coelacanth-fish-inspired double-helicoidal composites. Compos. Sci. Technol. 205, 108650 (2021).
Yang, W. et al. Protective role of Arapaima Gigas fish scales: structure and mechanical behavior. Acta Biomater. 10, 3599–3614 (2014).
Sun, J. & Bhushan, B. Structure and mechanical properties of beetle wings: a review. RSC Adv. 2, 12606 (2012).
Taylor, J. R. A. & Patek, S. N. Ritualized fighting and biological armor: the impact mechanics of the Mantis Shrimp’s Telson. J. Exp. Biol. 213, 3496–3504 (2010).
Weaver, J. C. et al. The stomatopod dactyl club: a formidable damage-tolerant biological hammer. Science 336, 1275–1280 (2012).
Guarín-Zapata, N., Gomez, J., Yaraghi, N., Kisailus, D. & Zavattieri, P. D. Shear wave filtering in naturally-occurring bouligand structures. Acta Biomater. 23, 11–20 (2015).
Yaraghi, N. A. et al. A sinusoidally architected helicoidal biocomposite. Adv. Mater. 28, 6835–6844 (2016).
Yaraghi, N. A. et al. The stomatopod telson: convergent evolution in the development of a biological shield. Adv. Funct. Mater. 29, 1902238 (2019).
Suksangpanya, N., Yaraghi, N. A., Kisailus, D. & Zavattieri, P. Twisting cracks in bouligand structures. J. Mech. Behav. Biomed. Mater. 76, 38–57 (2017).
Suksangpanya, N., Yaraghi, N. A., Pipes, R. B., Kisailus, D. & Zavattieri, P. Crack twisting and toughening strategies in bouligand architectures. Int. J. Solids Struct. 150, 83–106 (2018).
Grunenfelder, L. K. et al. Ecologically driven ultrastructural and hydrodynamic designs in stomatopod cuticles. Adv. Mater. 30, 1705295 (2018).
Zaheri, A. Convergent evolution in biological lamellar systems: multiscale structures and mechanics. Ph.D. Diss. (Northwestern University, 2019).
Launey, M. E., Buehler, M. J. & Ritchie, R. O. On the mechanistic origins of toughness in bone. Annu. Rev. Mater. Res. 40, 25–53 (2010).
Kolednik, O., Predan, J., Fischer, F. D. & Fratzl, P. Bioinspired design criteria for damage-resistant materials with periodically varying microstructure. Adv. Funct. Mater. 21, 3634–3641 (2011).
He, M. & Hutchinson, J. W. Crack deflection at an interface between dissimilar elastic materials. Int. J. Solids Struct. 25, 1053–1067 (1989).
Greenfeld, I., Kellersztein, I. & Wagner, H. D. Nested helicoids in biological microstructures. Nat. Commun. 11, 224 (2020).
Cheng, L., Thomas, A., Glancey, J. L. & Karlsson, A. M. Mechanical behavior of bio-inspired laminated composites. Compos. A 42, 211–220 (2011).
Grunenfelder, L. K. et al. Bio-inspired impact-resistant composites. Acta Biomater. 10, 3997–4008 (2014).
Grunenfelder, L. K., Herrera, S. & Kisailus, D. Crustacean-derived biomimetic components and nanostructured composites. Small 10, 3207–3232 (2014).
Rivera, J., Yaraghi, N. A., Huang, W., Gray, D. & Kisailus, D. Modulation of impact energy dissipation in biomimetic helicoidal composites. J. Mater. Res. Technol. 9, 14619–14629 (2020).
Koizumi, H. et al. Surface roughness and gloss of current CAD/CAM resin composites before and after toothbrush abrasion. Dent. Mater. J. 34, 881–887 (2015).
Nan, N., DeVallance, D. B., Xie, X. & Wang, J. The effect of bio-carbon addition on the electrical, mechanical, and thermal properties of polyvinyl alcohol/biochar composites. J. Compos. Mater. 50, 1161–1168 (2016).
Nguyen, L. H. et al. A methodology for hydrocode analysis of ultra-high molecular weight polyethylene composite under ballistic impact. Compos. Part Appl. Sci. Manuf. 84, 224–235 (2016).
Kisailus, D., Milliron, G. Shock and impact resistant materials. US Pat. No. 9,452,587 September 27, (2016).
Bert, A., Mencattelli, L., Kisailus, D., Wasilenkoff, C. High impact-resistant, reinforced fiber for leading edge protection of aerodynamic structures. US Pat. No. 11,952,103, April 9 (2024).
McCarville, D., Wasilenkoff, C., Des Ouches, P. J., Kisailus, D. Shock and impact resistant structures. US Pat. No. 11,376,812, July 5, (2022).
Helicoid Industries. https://www.helicoidind.com
Liu, J., Li, S., Fox, K. & Tran, P. T. 3D concrete printing of bioinspired bouligand structure: a study on impact resistance. Addit. Manuf. 50, 102544 (2022).
Pievani, T. The Natural History of Imperfection (Raffaello Cortina Editore, 2021).
Hamm, C. E. et al. Architecture and material properties of diatom shells provide effective mechanical protection. Nature 421, 841–843 (2003).
Wilkie, I. C. Mutable collagenous tissue: overview and biotechnological perspective. Echinodermata 39, 221–250 (2005).
Weiner, S. Biomineralization: a structural perspective. J. Struct. Biol. 163, 229–234 (2008).
Weiner, S. & Addadi, L. Crystallization pathways in biomineralization. Annu. Rev. Mater. Res. 41, 21–40 (2011).
Weiner, S. Organization of organic matrix components in mineralized tissues. Am. Zool. 24, 945–951 (1984).
Suzuki, M. & Nagasawa, H. Mollusk shell structures and their formation mechanism. Can. J. Zool. 91, 349–366 (2013).
Ferrara, M. A. et al. Optical properties of diatom nanostructured biosilica in Arachnoidiscus sp: micro-optics from mother nature. PLoS One 9, e103750 (2014).
Oxman, N. Material-based design computation. Ph.D. Diss., Massachusetts Institute of Technology, Boston, MA (2010).
deVries, M. S. The role of feeding morphology and competition in governing the diet breadth of sympatric stomatopod crustaceans. Biol. Lett. 13, 20170055 (2017).
Ahyong, S. T. & Harling, C. The phylogeny of the stomatopod Crustacea. Aust. J. Zool. 48, 607–642 (2000).
Koga, C. & Rouse, G. W. Mitogenomics and the phylogeny of mantis shrimps (Crustacea: Stomatopoda). Diversity 13, 647 (2021).
Peterman, D. J. et al. Buoyancy control in ammonoid cephalopods refined by complex internal shell architecture. Sci. Rep. 11, 8055 (2021).
CCM Hochey Industry. https://us.ccmhockey.com/homepage.html
Acknowledgements
The authors would like to acknowledge funding from the Air Force Office of Scientific Research (FA9550-15-1-0009) and (FA9550-23-1-0292), as well as the National Science Foundation (NSF EAR-1849963).
Author information
Authors and Affiliations
Contributions
D.K. and V.P. conceptualized the review; V.P., E.S., and A.N. prepared the initial and revised manuscript; D.K. and N.H. revised the manuscript; D.K. acquired funding for this work, and supervised V.P., E.S., and A.N.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications materials thanks the anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editor: John Plummer.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Perricone, V., Sarmiento, E., Nguyen, A. et al. The convergent design evolution of multiscale biomineralized structures in extinct and extant organisms. Commun Mater 5, 227 (2024). https://doi.org/10.1038/s43246-024-00669-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43246-024-00669-z