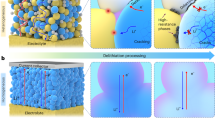
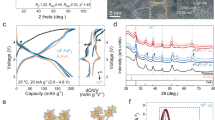
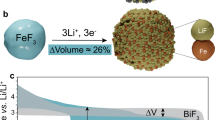
Abstract
Transition-metal fluorides (TMFs) are attracting attention as alternative lithium-ion battery cathodes, primarily focusing on Fe-based systems. Here, we report chromium as a previously unexplored transition metal (TM) for TMF cathodes in rechargeable lithium batteries. Utilizing a thin-film solid-state platform, we mitigate the common shortcomings of TMF cathodes, such as sluggish kinetics and electrolyte incompatibility. Coevaporation of Cr and LiF produces a heterogeneous thin film of Cr-LiF with a 1.1:2 stoichiometric ratio, delivering an initial capacity of 435 mAh/g and an energy density of 0.71 Wh/g at a C/10 cycling rate. Experimental measurements and first-principles calculations identify CrF2 as the dominant delithiated phase. The cathode maintains a capacity of 208 mAh/g at both 1C and 5C discharge rates after 1500 cycles. Compared to Fe-LiF (FeF2) analogs, Cr-LiF demonstrates a higher rate capability with 0.255 Wh/g at 3.80 W/g. This work introduces chromium fluorides as a new high-energy conversion cathode, expanding the options of viable positive electrode materials for next-generation batteries.
Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author, Yaroslav E. Romanyuk (yaroslav.romanyuk@empa.ch), upon reasonable request.
References
Janek, J. & Zeier, W. G. A solid future for battery development. Nat. Energy 1, 16141 (2016).
Wu, R. et al. Transition metal fluorides as advanced cathodes for lithium/sodium-ion batteries: rational enhancement strategies and underlying electrochemical mechanisms. Adv. Funct. Mater. 35, 2424603 (2025).
Dayah, M. Periodic Table—Ptable. (Michael Dayah, 1997).
Wolfram Research. Elementdata. (Wolfram Research, 2014).
Dong, K., Dai, W., Allison, J., Viola, J. & Leah, Z. Metal Prices. Shanghai Metal Market (SMM) (2025).
Wu, F. & Yushin, G. Conversion cathodes for rechargeable lithium and lithium-ion batteries. Energy Environ. Sci. 10, 435–459 (2017).
Sun, L., Li, Y. & Feng, W. Metal fluoride cathode materials for lithium rechargeable batteries: Focus on iron fluorides. Small Methods 7, 2201152 (2023).
Yan, D., Yang, H. Y. & Bai, Y. Tactics to optimize conversion-type metal fluoride/sulfide/oxide cathodes toward advanced lithium metal batteries. Nano Res. 16, 8173–8190 (2023).
Wang, X., Wang, Z., Chen, L., Li, H. & Wu, F. High-capacity sulfide all-solid-state lithium battery with a conversion-type iron fluoride cathode. J. Mater. Chem. A 11, 4142–4154 (2023).
Lai, C. et al. Highly reversible iron fluoride conversion cathodes enabled by deep-eutectic solvent method and heterostructure design. Adv. Funct. Mater. 34, 2312415 (2024).
Zhang, Y. et al. Enabling metal fluorides cathodes at elevated temperatures using a molten salt electrolyte. Adv. Energy Mater. 15, 2500293 (2025).
Xiao, A. W. et al. Understanding the conversion mechanism and performance of monodisperse FeF2 nanocrystal cathodes. Nat. Mater. 19, 644–654 (2020).
Omenya, F. et al. Intrinsic challenges to the electrochemical reversibility of the high energy density copper(ii) fluoride cathode material. ACS Appl. Energy Mater. 2, 5243–5253 (2019).
Zhao, Y. et al. Lif splitting catalyzed by dual metal nanodomains for an efficient fluoride conversion cathode. ACS Nano 13, 2490–2500 (2019).
Wang, F. et al. Ternary metal fluorides as high-energy cathodes with low cycling hysteresis. Nat. Commun. 6, 6668 (2015).
Fan, X. et al. In situ lithiated FeF3/C nanocomposite as high energy conversion-reaction cathode for lithium-ion batteries. J. Power Sources 307, 435–442 (2016).
Prakash, R. et al. A ferrocene-based carbon-iron lithium fluoride nanocomposite as a stable electrode material in lithium batteries. J. Mater. Chem. 20, 1871 (2010).
Kim, S. W. et al. Energy storage in composites of a redox couple host and a lithium ion host. Nano Today 7, 168–173 (2012).
Wu, T. et al. Catalysis of nickel nanodomains on Li-F dissociation for high-capacity fluoride cathodes with prior delithiation ability. Nano Energy 103, 107843 (2022).
Wei, K. et al. Low-overpotential lif splitting in lithiated fluoride conversion cathode catalyzed by spinel oxide. Adv. Funct. Mater. 31, 2009133 (2021).
Casella, J. et al. Electrochemical activation of Fe-LiF conversion cathodes in thin-film solid-state batteries. ACS Nano 18, 4352–4359 (2024).
Mishra, S. & Bharagava, R. N. Toxic and genotoxic effects of hexavalent chromium in environment and its bioremediation strategies. J. Environ. Sci. Health Part C. 34, 1–32 (2016).
Hope, E. G., Levason, W. & Ogden, J. S. Is chromium hexafluoride octahedral? Experiment still suggests “yes!”. Inorg. Chem. 30, 4873–4874 (1991).
Riedel, S. & Kaupp, M. The highest oxidation states of the transition metal elements. Coord. Chem. Rev. 253, 606–624 (2009).
Vanysek, P. Electrochemical series. CRC Handb. Chem. Phys. 8, 8–33 (2000).
Milazzo, G., Caroli, S. & Braun, R. D. Tables of standard electrode potentials. J. Electrochem. Soc. 125, 261C–261C (1978).
Peng, J., Wang, X., Li, H., Chen, L. & Wu, F. High-capacity, long-life iron fluoride all-solid-state lithium battery with sulfide solid electrolyte. Adv. Energy Mater. 13, 2300706 (2023).
Meng, Y. et al. Synergistic Lewis acid and hydrogen bonding strategy to enable polymerized and wide-temperature Li-Fe-F conversion solid-state batteries. Adv. Energy Mater. 15, e02645 (2025).
Wu, H., Hu, J., Yu, S. & Li, C. Heterostructure conductive interface and melt-penetration-bonding process to afford all-solid-state Li-FeF3 garnet batteries with high cathode loading. Energy Environ. Sci. 18, 923–936 (2025).
Furness, J. W., Kaplan, A. D., Ning, J., Perdew, J. P. & Sun, J. Accurate and numerically efficient R2 scan meta-generalized gradient approximation. J. Phys. Chem. Lett. 11, 8208–8215 (2020).
Kirklin, S., Meredig, B. & Wolverton, C. High-throughput computational screening of new Li-ion battery anode materials. Adv. Energy Mater. 3, 252–262 (2013).
Lee, D. H., Carroll, K. J., Calvin, S., Jin, S. & Meng, Y. S. Conversion mechanism of nickel fluoride and NiO-doped nickel fluoride in li ion batteries. Electrochim. Acta 59, 213–221 (2012).
Wang, F. et al. Conversion reaction mechanisms in lithium ion batteries: study of the binary metal fluoride electrodes. J. Am. Chem. Soc. 133, 18828–18836 (2011).
Hua, X. et al. Revisiting metal fluorides as lithium-ion battery cathodes. Nat. Mater. 20, 841–850 (2021).
Maier, J. Thermodynamics of electrochemical lithium storage. Angew. Chem. Int. Ed. 52, 4998–5026 (2013).
Balaya, P., Li, H., Kienle, L. & Maier, J. Fully reversible homogeneous and heterogeneous Li storage in RuO2 with high capacity. Adv. Funct. Mater. 13, 621–625 (2003).
Pearson, D. H., Ahn, C. C. & Fultz, B. White lines and d-electron occupancies for the 3D and 4D transition metals. Phys. Rev. B 47, 8471–8478 (1993).
Daulton, T. L. & Little, B. J. Determination of chromium valence over the range cr(0)-cr(vi) by electron energy loss spectroscopy. Ultramicroscopy 106, 561–573 (2006).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for liquid metals. Phys. Rev. B 47, 558–561 (1993).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Döbeli, M., Kottler, C., Glaus, F. & Suter, M. Erda at the low energy limit. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. At. 241, 428–435 (2005).
Kahl, F. et al. Test and characterization of a new post-column imaging energy filter. Adv. Imaging Electron Phys. 212, 35–70 (2019).
Caridad, A. R., Erni, R., Vogel, A. & Rossell, M. D. Applications of a novel electron energy filter combined with a hybrid-pixel direct electron detector for the analysis of functional oxides by STEM/EELS and energy-filtered imaging. Micron 160, 103331 (2022).
Plotkin-Swing, B. et al. Hybrid pixel direct detector for electron energy loss spectroscopy. Ultramicroscopy 217, 113067 (2020).
Richards, A. Technical Note on ARC Facility and Service Deployment for Publication Reference (University of Oxford Advanced Research Computing, 2015).
Zagorac, D., Müller, H., Ruehl, S., Zagorac, J. & Rehme, S. Recent developments in the inorganic crystal structure database: theoretical crystal structure data and related features. J. Appl. Crystallogr. 52, 918–925 (2019).
Acknowledgements
J.M. is supported by the European Union’s Horizon 2020 research and innovation program (grant no. 95817) and the Swiss Federal Office of Energy (SFOE, grant no. SI/502460-01). V.M. and M.D.R. acknowledge support by the Swiss National Science Foundation (SNSF) under Project No. 200021_219706. F.C.M. and S.I. are grateful to the Faraday Institution CATMAT project (EP/S003053/1, FIRG016) for financial support. M.H.F. is supported by a Rubicon Fellowship from the Netherlands Organization for Scientific Research (NWO). The authors would like to acknowledge the use of the University of Oxford Advanced Research Computing (ARC) facility in carrying out this work47. We thank the HEC Materials Chemistry Consortium (EP/R029431/1) for access and time on the Archer2 supercomputer facilities.
Author information
Authors and Affiliations
Contributions
J.C.: conceptualization, methodology, validation, formal analysis, investigation, data curation, writing—original draft, writing—review and draft, visualization. J.M.: conceptualization, methodology, investigation, writing—review and editing, visualization. V.M.: investigation, writing—review and editing, visualization. F.C.M.: investigation, writing—review and editing, visualization. A.M.: investigation. M.H.F.: writing – review and editing. M.D.R: funding acquisition. M.S.I.: writing—review and editing, visualization, funding acquisition. M.Y.: writing—review and editing, project administration. Y.E.R.: conceptualization, resources, writing – review and editing, visualization, supervision, project administration, funding acquisition.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Materials thanks Chilin Li and Tengrui Wang for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Casella, J., Morzy, J., Montanelli, V. et al. Cr-LiF as a high energy density conversion-type cathode for Li-ion solid-state batteries. Commun Mater (2026). https://doi.org/10.1038/s43246-026-01121-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s43246-026-01121-0