Abstract
Determining microbial necromass carbon and plant detritus carbon is essential for accurate global soil organic carbon modeling. Yet soil organic carbon precursors are usually ignored in alpine assessments. We examined 363 samples from dominant Himalayan ecosystems and determined the distribution of microbial necromass carbon and plant detritus carbon in 0-30 cm soil and their responses to land use changes. Plant detritus carbon was 6.75 times greater than microbial necromass carbon and dominant across soil depths and ecosystems. Microbial biomass carbon strongly influenced soil organic carbon precursors. Plant detritus carbon exhibited greater sensitivity to land use change than microbial necromass carbon and decomposed preferentially, thus, buffering against fluctuations of the stable soil organic carbon fraction. Our results highlight plant detritus carbon’s key role in maintaining soil organic carbon persistence and suggest that incorporating both residual carbons and land use responses into Earth system models could improve future soil organic carbon projections.
Similar content being viewed by others
Introduction
Soils of alpine ecosystems store the largest carbon pool in terrestrial ecosystems1,2, but the pool is unstable and highly sensitive to climate change and human activities3,4,5. It is predicted that the regional environment and food security will be threatened severely by unsustainable land use in the 22nd century6, particularly in impoverished and remote alpine regions, where local residents depend mainly on subsistence-level forest resource use and livestock grazing for livelihoods7. Soil organic carbon (SOC) has a pivotal role in regulating the global carbon cycle8, but the precursors of SOC, that is, microbial necromass carbon (MNC) and plant debris carbon (PDC), are keys to its stability and cycling9,10,11. These precursors have long been ignored in alpine regions due to the large costs involved in their measurements and historical research preferences for temperate and tropical ecosystems11,12,13. Consequently, our understanding of SOC precursors in alpine regions is poor, which limits the accuracy of carbon cycle models and predictions.
By biodegradation, microbial and plant residues are transformed into MNC and PDC14,15, and can be traced by amino sugars and lignin phenols, respectively16,17,18. Mineral-protected MNC stabilizes and accumulates SOC by slow decomposition and forms chemically diverse SOC19,20, while microbial death pathways modulate nutrient recycling and SOC cycling21. In contrast, PDC represents mainly residual energy in stable SOC that resists microbial utilization, thereby contributing to long-term SOC persistence10. MNC and PDC respond differently to land use changes. For example, replacing maize-soybean with mulberry and sugarcane had no effect on MNC22, conversion of rice paddies to agricultural uplands decreased PDC but increased MNC23, while grassland degradation led to an accumulation of lignin phenols24. However, accurate quantification of the effect of land use changes on MNC and PDC in alpine ecosystems is still lacking8.
Prior research has explored factors influencing the accumulation of MNC and PDC across various ecosystems25,26,27. For example, in grasslands, aridity caused an accumulation of amino sugars and decomposition of lignin phenols16, while at an aridity index above 0.77, MNC decreased sharply28. In addition, plant biomass and soil properties exerted strong effects on MNC and PDC in alpine grasslands and wetlands11,29. In temperate forests, MNC was influenced by climate, microbial traits and soil properties, and played a pivotal role in SOC accumulation12; while climate-smart agriculture promoted MNC accumulation by increasing living microbial biomass30. Nevertheless, the environmental drivers of MNC and PDC in the heterogeneous alpine ecosystems remain unknown.
The Himalayan mountains, the largest mountainous region in the world, with the poorest people7,31, have a long history of frequent land use changes in attempts to improve their livelihoods based on farming and grazing livestock32. This has resulted mainly in the conversion of undisturbed forest to burned/logged forest to cropland and wetland to grassland to desert33. In the present study, we determined the distribution and key environmental factors affecting soil MNC and PDC in the top 30 cm in intervals of 10 cm in alpine ecosystems by analyzing amino sugars and lignin phenols in 363 samples collected from forest, wetland, grassland, cropland and desert ecosystems. We measured the distribution of MNC and PDC in the five ecosystems and determined whether the distribution patterns varied across soil depths. Then we assessed the effect of the geo-environment, plant input, and soil physical, chemical, mineral and microbial properties and quantified the impacts of land use change (conversion of forest-to-cropland and wetland-to-desert) on MNC and PDC contents. We hypothesized that: (1) MNC dominates over PDC across depths and ecosystems; (2) the environmental drivers influencing the accumulation of MNC and PDC differ, with microbial properties dominating MNC accumulation and plant input dominating PDC accumulation; and (3) PDC is more sensitive to land use change than MNC.
Results
Dominance patterns of PDC and MNC in alpine ecosystems
The variability of residual carbon at the same soil depth in alpine ecosystems was consistent with the variability of the biomarkers. The greatest variability in amino sugars occurred in the top 10 cm of soil across the five ecosystems (Supplementary Fig. 1); significant differences in microbial necromass carbon (MNC) across alpine ecosystems were observed only in the first 10 cm of soil, with no significant difference at the 10–30 cm depth (Supplementary Fig. 2). In contrast, lignin phenols and plant detritus carbon (PDC) differed consistently across ecosystems at 0–30 cm depths, with greater concentrations in forests and croplands than in wetlands, grasslands, and deserts (Supplementary Figs. 1 and 2). Comparing the two residual carbons, PDC dominated in all alpine ecosystems and soil depths (Fig. 1). The average concentration of PDC (median: 3647 mg kg−1; interquartile range: 4292) was 6.75 times greater than MNC (median: 540 mg kg−1; interquartile range: 2055) (Fig. 1). The difference was smallest in desert ecosystems among the five ecosystems, but PDC (median: 1683 mg kg−1; interquartile range: 2602) was still 2.18 times greater than MNC (median: 771 mg kg−1; interquartile range: 2445). Overall, PDC contributed 17.9% and MNC contributed 2.4% to SOC, with their relative contributions varying across soil depths and ecosystems (Fig. 1). Both residual carbons increased with an increase in SOC (Fig. 2), but the slope was steeper for PDC (slope: 0.78; P < 0.001) than for MNC (slope: 0.30; P < 0.01). As a consequence, PDC became more dominant relative to MNC as the concentration of SOC increased.
Microbial necromass carbon (MNC) and plant detritus carbon (PDC) distribution in forest, wetland, grassland, cropland and desert ecosystems in 0–30 cm soil depth (n = 102, 54, 63, 90 and 54, respectively). The horizontal black bold lines inside the boxplots represent the median, and the black whiskers represent the interquartile range from the first and third quartiles. Results from paired Wilcoxon signed-rank tests, P values for two-tailed tests.
All data were transformed into natural-log for linear regressions, points of the same color correspond to the same ecosystem, and shadings represent 95% CIs. The overall linear regressions are: LN MNC = 0.3 × LN SOC + 5.25, n = 363; LN PDC = 0.78 × LN SOC + 5.83, n = 363.
Environmental drivers of residue carbon
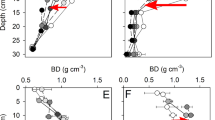
MNC was correlated positively with plant input (plant C, N, P), soil nitrogen and phosphorus contents, and microbial biomass carbon (MBC) (P < 0.05; Supplementary Fig. 3), and a structural equation model (SEM) supported these positive correlations (Fig. 3). Plant input and MBC were the most important predictors of MNC accumulation in soil, with standardized pathway coefficients of 0.20 (P < 0.001) and 0.29 (P < 0.001), respectively. Soil moisture content (SMC) was correlated negatively with MNC accumulation in soil, with a standardized pathway coefficient of −0.14 (P < 0.05). MBC was correlated positively with PDC (r = 0.58; P < 0.001; Supplementary Fig. 3) and, based on the SEM, MBC was the dominant predictor of PDC with a standardized pathway coefficient of 0.43 (P < 0.001; Fig. 3). Furthermore, total nitrogen (TN), total phosphorus (TP), silt, and SMC were correlated positively with PDC (Supplementary Fig. 3), with correlation coefficients of 0.71 (P < 0.001), 0.31 (P < 0.001), 0.51 (P < 0.001), and 0.41 (P < 0.001), respectively. Soil bulk density (BD), sand, and soil pH were correlated negatively with PDC, with correlation coefficients of −0.44 (P < 0.001), −0.33 (P < 0.001), and −0.27 (P < 0.001), respectively.
Based on an a priori model (Fig. S6), the SEM identified environmental drivers of MNC and PDC (n = 363). The black and red lines represent positive and negative standardized pathway coefficients, respectively. Standardized pathway coefficients are next to each path, and the widths of the paths are scaled to the standardized path coefficients. The dashed lines indicate that the standardized path coefficient is below 0.1.
Sensitivity of residue carbons to land use change
Land use changes driven by farming and grazing activities, that is, conversion of undisturbed forest to burned/logged forest to cropland and wetland to grassland to desert, modified residual carbons relationships, independent of SOC (Fig. 4). Forest conversion to cropland reduced both MNC and PDC, with a greater reduction in PDC than MNC (effect size: −0.418, CI: −0.202 to −0.633, P < 0.001); similarly, wetland degradation to desert also reduced both residual carbons, with a greater reduction in PDC than MNC (effect size: −1.55, CI: −1.20 to −1.90, P < 0.001) (Fig. 5). The acid-to-aldehyde ratios of vanillyl (V) and syringyl (S) phenols reflect lignin phenol oxidation, with higher ratios implying greater PDC decomposition11,16. Across land use changes, (Ad/Al)s and (Ad/Al)v were correlated positively with MNC, suggesting preferential PDC decomposition to MNC, whereas plant carbon input, MBC, and AWCD were correlated positively with PDC. Compared to native ecosystems (legacy cropland and natural desert), secondary ecosystems (converted cropland and converted desert) contained a lesser concentration of residual carbons (Fig. 6). Specifically, PDC in converted cropland was lesser than in legacy cropland (effect size of PDC: −0.499, CI: −0.190 to −0.808, P < 0.01). Burned forests contained less PDC (−0.429, CI: −0.215 to −0.643, P < 0.001) and MNC (−0.554, CI: −1.005 to −0.102, P < 0.05) than logged forests (Supplementary Fig. 4).
Green and blue lines indicate positive and negative correlations with microbial necromass carbon and plant detritus carbon; R is the correlation coefficient. MNC (mg kg−1 soil) = microbial necromass carbon concentration in soil; PDC (mg kg−1 soil) = plant detritus carbon concentration in soil. (Ad/Al)s and (Ad/Al)v = acid-to-aldehyde ratios of syringyl phenols and cinnamyl phenols; Plant carbon input = the total carbon in plants aboveground, litter, and 0–30 cm roots; MBC microbial biomass carbon, AWCD microbial carbohydrate utilization efficiency.
A Depicts land use change driven by farming activity (conversion of undisturbed forest to burned/logged forest to cropland); B is land use change driven by grazing activity (conversion of wetland to grassland to desert). In calculating the effect size of forest conversion to cropland, undisturbed forest was the control group (n = 12), and converted cropland was the treatment group (n = 27). Similarly, in calculating the effect size of the conversion of wetland to desert, healthy wetland was the control group (n = 9), and converted desert was the treatment group (n = 27). PDC plant detritus carbon, V vanillyl phenols, S syringyl phenols, C cinnamyl phenols, (Ad/Al)v = acid-to-aldehyde ratios of vanillyl phenols, (Ad/Al)s = acid-to-aldehyde ratios of syringyl phenols. MNC microbial necromass carbon, BNC bacterial necromass carbon, FNC fungal necromass carbon, B/F = ratio of bacterial necromass carbon to fungal necromass carbon. The horizontal lines represent the confidence interval (95% CIs) of the effect size (log response ratios), from the lower confidence limit to the upper confidence limit. Asterisks indicate statistical significance, where ***P < 0.001; **<P < 0.01; and *<P < 0.05.
A Croplands; B deserts. Legacy croplands and natural deserts are classified as native ecosystems; converted croplands and converted deserts are classified as secondary ecosystems. In calculating the effect size, legacy cropland was the control group (n = 54), and converted cropland was the treatment group (n = 27). Similarly, natural desert was the control group (n = 27), and converted desert was the treatment group (n = 27). PDC plant detritus carbon, V vanillyl phenols, S syringyl phenols, C cinnamyl phenols, (Ad/Al)v = acid-to-aldehyde ratios of vanillyl phenols, (Ad/Al)s = acid-to-aldehyde ratios of syringyl phenols. MNC microbial necromass carbon, BNC bacterial necromass carbon, FNC fungal necromass carbon, B/F = ratio of bacterial necromass carbon to fungal necromass carbon. The horizontal lines represent the confidence interval (95% CIs) of the effect size (log response ratios), from the lower confidence limit to the upper confidence limit. Asterisks indicate statistical significance, where ***P < 0.001; **<P < 0.01; and *<P < 0.05.
Discussion
In previous studies, MNC was recognized as the primary stable fraction of SOC influencing its long-term persistence15,19,34. However, in the present study, SOC was consistently dominated by PDC and not MNC in the first 30 cm of soil in the alpine ecosystems. PDC was more sensitive than MNC to land use change in alpine regions, given the large PDC losses induced by the changes. Due to the dominant role of PDC in alpine SOC and its greater sensitivity to land use change, it should be a key focus of future carbon management in global alpine regions.
Preferential accumulation of PDC in alpine regions
Microbes are central to SOC formation in temperate ecosystems, with microbial necromass contributing 30–80% to SOC content18,35. However, in the present study, PDC dominated consistently across alpine ecosystems, with MNC contributing only 2.4% to SOC content. These differences among ecosystems may be due to the low temperature36 and limited soil nitrogen and phosphorus in the alpine regions35,37,38, which reduce the decomposition of plant detritus and production of microbial necromass. Soil organic carbon (SOC) accumulation in this region is mainly a result of PDC accumulation through the “ex vivo microbial modification pathway”39. In addition, plants in the harsh environments of alpine regions are generally rich in lignin, as plant cell walls and secondary walls are strengthened in response to environmental stresses40.
Microbial properties and plant input regulating residue carbon
Concentrations of MNC and PDC in alpine ecosystems were driven by distinct and overlapping environmental factors, with microbial properties being the main influence on carbon accumulation in both residue carbons. Microbial properties regulate the conversion of plant-derived carbon into soil carbon, either into PDC or MNC12,33, and also mediate the transformation of PDC into MNC23,41,42. The accumulation of microbial necromass depends on the production, degradation, and reuse of microbial metabolites21,43, while high microbial biomass directly enhances the large-scale production of microbial necromass25. Lignin decomposition is related to temperature and microbial communities44, and low oxygen conditions are not conducive to lignin decomposition23. Consequently, low microbial metabolism in alpine regions reduces microbial turnover11,45 even with high levels of both microbial biomass carbon and nitrogen, thus favoring PDC accumulation.
Plant input provides essential energy for microbial growth46, leading to the formation and retention of microbial necromass47. The diversity and quantity of plant input also favor the retention of PDC in the soil48. However, in the present study, plant input promoted MNC accumulation and did not affect PDC accumulation. This could be the reason why plant input stimulates microbial growth and metabolism49, leading to the decomposition of PDC50 and even mineral-bound carbon51. Moreover, increased nutrient demand by rapid microbial growth, in turn, accelerates PDC decomposition. For example, Tamura et al.48 reported that the addition of Pueraria lobata litter resulted in the retention of a small amount of lignin in the soil. Microbial metabolic activity and the selective utilization of carbon sources were suggested to drive the conversion of PDC to MNC52; however, the relationships between AWCD, DI and MNC were weak in the present study, indicating a minimal effect on MNC.
PDC as a “vanguard” of SOC response to land use change
The greater sensitivity of PDC than MNC to land use change, and the decreased C/V (Cinnamyl/Vanillyl) and increased (Ad/Al)v (acid-to-aldehyde ratios of Vanillyl phenols) supported the results in the present study53, and indicated that PDC was decomposed to a greater extent than MNC with land use change23,50. Land use change drives the transformation of PDC to MNC, which is more stable and advantageous for the long-term protection and accumulation of SOC11. Furthermore, PDC can stabilize the soil to withstand external disturbances11,41. These findings have important implications for soil carbon sequestration under global land use change.
MNC and PDC were lesser in secondary ecosystems than in native ecosystems mainly due to differences in vegetation. Converted croplands for perennial cash crops, such as rice, banana and tea trees result in fewer and more easily decomposable plant input and slower carbon cycling than the annual harvesting of barley while leaving stubble in legacy croplands. Similarly, natural deserts are characterized predominantly by shrubs, which are rich in lignin, whereas deserts due to degradation of grassland are characterized mainly by easily decomposable herbs, which enhance the positive effect of S/V (Syringyl/Vanillyl)53.
Conclusions
This study provides the first large-scale data on the distribution, environmental drivers, and responses to land use change of microbial necromass carbon (MNC) and plant detritus carbon (PDC) in alpine ecosystems. Our first hypothesis was rejected, as PDC and not MNC dominated across ecosystems and soil depths. However, our second and third hypotheses were supported, as both SOC precursors were influenced primarily by microbial properties, with plant input also contributing to MNC accumulation. PDC was more sensitive to land use change, as it was more vulnerable to disturbance. These findings fill a critical knowledge gap on SOC precursors in alpine regions and advance our mechanistic understanding of how MNC and PDC are impacted by different ecosystems and anthropogenic pressures. By clarifying their relative importance and drivers, this study provides an empirical foundation for refining carbon cycle predictions in alpine regions. Integrating these findings into Earth system models will improve projections of soil carbon sequestration potential under global land use change.
Materials and methods
Study area
The study region, located in the Himalayan mountains within China and referred to as China-Himalayan, is characterized by a large range in altitude (760–4860 m), mean annual precipitation (245–1397 mm) and mean annual temperature (−11.5–6.9 °C) (WorldClim-Global Climate Data: http://www.worldclim.org), and supports an array of ecosystem types. Land use in this region is diverse, supporting livelihoods, which include livestock and crop production and forest industries. Based on archeological, palynological, and agricultural research in this region, farming and grazing are the primary drivers of forest conversion to cropland, and wetland degradation to desert54,55,56. Based on the regional ecosystems and land use history, we selected 41 sites in the China-Himalayan region that represent five dominant alpine ecosystems: forest, wetland, grassland, cropland and desert. These sites included secondary ecosystems formed by long-term land use changes, resulting mainly from two activities, farming and livestock grazing54,56. Background information for each site was obtained by interviews with local herders and cross-validated with regional land use records where available; more detailed site information is provided in Supplementary Table 1 and Supplementary Fig. 5.
Field investigation and sampling
According to the AusPlots Rangelands Survey Protocols Manual57, each site was surveyed following a series of procedures: plot selection and layout; photo-panoramas; plant tissue sampling for chemical analysis; and soil sampling for microbial and chemical analyses. Specifically, a 100 × 100 m permanent plot was established in a homogenous patch of terrestrial vegetation, and five 1 × 1 m quadrats, located at the four corners and the center of the plot, were used for detailed surveys and sampling.
We recorded plant species, vegetation cover, height, biomass and litter in the five quadrats in each plot to study short-term stabilizing carbon changes. Forest plant carbon input was measured in only the herbaceous layer and in all the litter. Soil samples and roots were collected from a depth of 0–30 cm, at 10 cm intervals, using a soil sampling auger, and aboveground plant samples, including litter, were collected in each quadrat and brought back to the laboratory at Lanzhou University. Soil bulk density (BD) was measured using a 100 cm3 cylindrical soil sample, and soil moisture content (SMC) was determined by oven-drying the cylindrical soil core at 105 °C for 48 h58. The five soil cores from each 10 cm soil depth in each plot were combined into one sample. After air-drying, three of the five replicate samples from each 10 cm soil depth in each plot were selected randomly for the determination of amino sugars, lignin phenols and soil properties16,59. Six samples were lost from the forest plots, resulting in a total of 363 data sets.
Plant input
Aboveground plants, litter, and roots were oven-dried at 65 °C to constant weight to determine their biomasses. The dried materials, used for chemical analyses, were ground into fine powder using a ball mill (MM400 ball mill, Retsch, Germany). Carbon (C) and nitrogen (N) contents were determined by combustion using an elemental analyzer (Elementar vario MACRO cube, Elementar, Germany), after the powdered samples were wrapped in tin capsules free of carbon and nitrogen. Phosphorus (P) content was measured using a discrete autoanalyzer (Smartchem 200, Italy) following high-temperature digestion with sulfuric acid and hydrogen peroxide59. Plant biomass and plant C/N/P input were calculated following Chen et al.60 as:
Soil bulk properties
Air-dried soil was sieved through a 2 mm screen. Soil pH was measured using a pH meter (PB-10, Sartorlus, Göttingen, Germany) at a soil: deionized water ratio of 1:5 (w:v), and SOC content was measured by the acid-dichromate ferrous sulfate titration method61. Total carbon and nitrogen contents of soil were measured by an elemental analyzer (Vario EL III, Elementar, Hanau, Germany) following combustion, while total phosphorus was measured by a flow analyzer (Smartchem 200, Bergamo, Italy) following high-temperature digestion with sulfuric acid and and perchloric acid digestion62. The reactive Fe and Al contents were extracted using the citrate-bicarbonate-dithionite (CBD) method63 and quantified using inductively coupled plasma optical emission spectrometry (ICP-OES; PerkinElmer, Waltham, MA, USA). Soil texture was determined using a particle size analyser (Malvern Masterizer 2000, Malvern, Worcestershire, UK), and clay was defined as particles smaller than 2 µm. NH4+-N and NO3−-N were extracted with 2 M KCl solution64 and measured using a flow analyzer (Smartchem200, Bergamo, Italy). Microbial biomass carbon (MBC) and microbial carbon utilization efficiency were determined in fresh soil stored at −4 °C. MBC was determined by the chloroform fumigation solution (0.5 mol/L K2SO4) extraction method65,66, and the microbial stationary carbon source utilization efficiency and microbial diversity index were calculated using a BIOLOG EcoPlate67 (Supplementary Information).
Measurements of soil amino sugars and lignin phenols
Two groups of widely accepted biomarkers, namely, amino sugars and lignin phenols, were used to track and calculate MNC18,68 and PDC16,23, respectively. The concentrations of soil amino sugars were measured following Zhang and Amelung69. The soil was hydrolyzed by 6 M HCl at 105 °C, and impurities were removed by evaporation to dryness and freeze-drying. Then, the amino sugars were converted to aldononitrile derivatives using hydroxylamine hydrochloride and 4-(dimethylamino)pyridine. The copper (II) oxide (CuO) oxidation method, modified by Hedges and Ertel70, was used to measure the concentration of lignin phenols. Lignin was converted to lignin oxidation products (LOPs) in high-temperature sealed teflon-lined bombs16, and were then derivatized to produce trimethylsilyl derivatives. Detailed information is presented in Supplementary Information.
Microbial necromass carbon (MNC) was calculated as68:
where GluN and MurA = the concentrations of glucosamine and muramic acid (µg g−1) in the soil, respectively.
Plant detritus carbon (PDC) was calculated as23:
where V, S, and C represent the content of carbon in the Vanillyl phenols, Syringyl phenols and Cinnamyl phenols (µg g−1), respectively.
Data and statistical analyses
The MNC and PDC concentrations in alpine ecosystems and in the 0–30 cm soil depth were not distributed normally based on the Shapiro-Wilk test. Therefore, the non-parametric paired Wilcoxon signed-rank test compared MNC and PDC concentrations, while the non-parametric Kruskal-Wallis test compared MNC and PDC concentrations among the alpine ecosystems and soil depths (IBM, SPSS Statistics 25). Following natural-log transformation, all data conformed to a normal distribution. Ordinary least squares regressions tested the relationships between MNC, PDC and SOC, while Pearson correlations tested the relationships between MNC or PDC and environmental parameters.
A structural equation model (SEM) was constructed using all samples across soil depths and ecosystem types as a single dataset to identify the regulatory pathways of environmental factors driving MNC and PDC. The environmental factors influencing MNC and PDC were classified into six categories: (1) geo-environmental factors: longitude, latitude, mean annual temperature, mean annual precipitation, aridity index, and elevation; (2) plant input: plant biomass, plant carbon input, plant nitrogen input, plant phosphorus input, ratio of plant carbon input to plant nitrogen input, ratio of plant carbon input to plant phosphorus input, and ratio of plant nitrogen input to plant phosphorus input; (3) soil physical properties: soil bulk density (BD), soil moisture content (SMC), and soil pH; (4) mineral properties: clay, silt, sand, free-form iron ion (Fed), and free-form aluminum ion (Ald); (5) soil chemical properties: ammonium nitrogen (NH4+), nitrate nitrogen (NO3−), total nitrogen (TN), total phosphorus (TP), ratio of soil organic carbon to total nitrogen (SOC/TN), ratio of soil organic carbon to total phosphorus (SOC/TP), and ratio of total nitrogen to total phosphorus (TN/TP); and (6) microbial properties: microbial biomass carbon (MBC), microbial carbohydrate utilization efficiency (AWCD), and microbial richness index (DI). An a priori model was generated (Supplementary Fig. 6). Highly correlated variables were screened out, and principal component analysis (PCA) identified closely related multivariate variables of the same type, with the first component PC1 replacing independent variables. Before PCA, the variance inflation factors between the retained variables selected by the model were determined to avoid multi-collinearity, and a value of less than 2 was accepted71. The generation of the model used the maximum likelihood technique, and fit statistics included the ratio of Chi-square to degrees of freedom (χ²/df), goodness-of-fit index (GFI), and the root mean square error (RMSE). SEM used AMOS version 21.0 (Amos Development Corporation, IBM SPSS, Chicago, IL, USA).
We examined whether anthropogenically-induced land use changes affected MNC, PDC and their proportions using the log response ratio (RR) as the measure of effect size72.
where XW and XC are the MNC, PDC and their proportions before and after land use change, respectively. A positive or negative RR value indicates reinforcing or limiting land use change impacts. Similarly, we examined the differences between burned and logged forests, with undisturbed forests as a control; deserts due to degradation of grassland and natural deserts (control); and converted croplands and legacy cropland (control).
Data availability
The data that support the findings of this study are openly available in figshare at https://doi.org/10.6084/m9.figshare.30170548.v173.
References
Ding, J. et al. Decadal soil carbon accumulation across Tibetan permafrost regions. Nat. Geosci. 10, 420–424 (2017).
Crowther, T. W. et al. The global soil community and its influence on biogeochemistry. Science 365, eaav0550 (2019).
Koven, C. D. et al. Higher climatological temperature sensitivity of soil carbon in cold than warm climates. Nat. Clim. Change 7, 817–822 (2017).
Landrum, L. & Holland, M. M. Extremes become routine in an emerging new Arctic. Nat. Clim. Change 10, 1108–1115 (2020).
Ma, L. & Zuo, H. Quantifying net carbon fixation by Tibetan alpine ecosystems should consider multiple anthropogenic activities. Proc. Natl. Acad. Sci. USA 119, e2115676119 (2022).
Amundson, R. et al. Soil and human security in the 21st century. Science 348, 1261071 (2015).
Gioli, G., et al. The Hindu Kush Himalaya Assessment: Mountains, Climate Change, Sustainability and People. In: Understanding and Tackling Poverty and Vulnerability in Mountain Livelihoods in the Hindu Kush Himalaya (Springer Nature, 2019).
Schmidt, M. W. I. et al. Persistence of soil organic matter as an ecosystem property. Nature 478, 49–56 (2011).
Zhang, Q. et al. Tibetan Plateau grasslands might increase sequestration of microbial necromass carbon under future warming. Commun. Biol. 7, 686 (2024).
Gunina, A. & Kuzyakov, Y. From energy to (soil organic) matter. Glob. Change Biol. 28, 2169–2182 (2022).
Xia, S. et al. Patterns and determinants of plant-derived lignin phenols in coastal wetlands: implications for organic C accumulation. Funct. Ecol. 37, 1067–1081 (2023).
He, J. et al. Latitudinal patterns and drivers of plant lignin and microbial necromass accumulation in forest soils: disentangling microbial and abiotic controls. Soil Biol. Biochem. 194, 109438 (2024).
Lin, Q. et al. Persistence of soil microbial residuals and lignin phenols in forest ecosystems along the latitude gradient. J. Soils Sediments 24, 2425–2436 (2024).
Frimmel, F. H. & Christman, R. F. Humic Substances and their Role in the Environ. (Wiley, 1988).
Cotrufo, M. F. et al. The microbial efficiency-matrix stabilization (MEMS) framework integrates plant litter decomposition with soil organic matter stabilization: do labile plant inputs form stable soil organic matter? Glob. Change Biol. 19, 988–995 (2013).
Ma, T. et al. Divergent accumulation of microbial necromass and plant lignin components in grassland soils. Nat. Commun. 9, 3480 (2018).
Crow, S. et al. Sources of plant-derived carbon and stability of organic matter in soil: implications for global change. Glob. Change Biol. 15, 2003–2019 (2009).
Liang, C. et al. Quantitative assessment of microbial necromass contribution to soil organic matter. Glob. Change Biol. 25, 3578–3590 (2019).
Kallenbach, C., Frey, S. D. & Grandy, A. S. Direct evidence for microbial-derived soil organic matter formation and its ecophysiological controls. Nat. Commun. 7, 13630 (2016).
Zhu, X. et al. The soil microbial carbon pump: from conceptual insights to empirical assessments. Glob. Change Biol. 26, 6032–6039 (2020).
Camenzind, T. et al. Formation of necromass-derived soil organic carbon determined by microbial death pathways. Nat. Geosci. 16, 115–122 (2023).
Huang, Y. et al. Variation of microbial residue contribution to soil organic carbon sequestration following land use change in a subtropical karst region. Geoderma 353, 340–346 (2019).
Chen, X. et al. Contrasting pathways of carbon sequestration in paddy and upland soils. Glob. Change Biol. 27, 2478–2490 (2021).
Breidenbach, A. et al. Microbial functional changes mark irreversible course of Tibetan grassland degradation. Nat. Commun. 13, 2681 (2022).
Han, B. et al. Microbial traits dictate soil necromass accumulation coefficient: a global synthesis. Glob. Ecol. Biogeogr. 33, 151–161 (2024).
Wang, B. et al. Microbial necromass as the source of soil organic carbon in global ecosystems. Soil Biol. Biochem. 162, 108422 (2021).
Buckeridge, K. M. et al. Environmental and microbial controls on microbial necromass recycling, an important precursor for soil carbon stabilization. Commun. Earth Environ. 1, 36 (2020).
Hao, Z. et al. Thresholds in aridity and soil carbon-to-nitrogen ratio govern the accumulation of soil microbial residues. Commun. Earth Environ. 2, 236 (2021).
He, M. et al. Depth-dependent drivers of soil microbial necromass carbon across Tibetan alpine grasslands. Glob. Change Biol. 28, 936–949 (2022).
Li, Y. et al. Global synthesis on the response of soil microbial necromass carbon to climate-smart agriculture. Glob. Change Biol. 30, e17302 (2024).
Shang, Z. et al. Carbon Management for Promoting Local Livelihood in the Hindu Kush Himalayan (HKH) Region (Springer Nature, 2020).
Grêt-Regamey, A., Brunner, S. H. & Kienast, F. Mountain ecosystem services: who cares? Mt. Res. Dev. 32, S23–S34 (2012).
Yang, X. et al. Lake-centred sedentary lifestyle of early Tibetan Plateau Indigenous populations at high elevation 4400 years ago. Nat. Ecol. Evol. 8, 2297–2308 (2024).
Cotrufo, M. F. et al. Formation of soil organic matter via biochemical and physical pathways of litter mass loss. Nat. Geosci. 8, 776–779 (2015).
Liang, C. & Balser, T. Microbial production of recalcitrant organic matter in global soils: implications for productivity and climate policy. Nat. Rev. Microbiol. 9, 75 (2011).
Bradford, M. et al. Managing uncertainty in soil carbon feedbacks to climate change. Nat. Clim. Change. 6, 751–758 (2016).
Hu, J. et al. Nitrogen addition increases microbial necromass in croplands and bacterial necromass in forests: a global meta-analysis. Soil Biol. Biochem. 165, 108500 (2022).
Luo, R. et al. Phosphorus addition decreases plant lignin but increases microbial necromass contribution to soil organic carbon in a subalpine forest. Glob. Chang Biol. 28, 4194–4210 (2022).
Liang, C., Schimel, J. P. & Jastrow, J. D. The importance of anabolism in microbial control over soil carbon storage. Nat. Microbiol. 2, 1–6 (2017).
Molina, A. et al. Arabidopsis cell wall composition determines disease resistance specificity and fitness. Proc. Natl. Acad. Sci. USA 118, e2010243118 (2021).
Angst, G. et al. Earthworms act as biochemical reactors to convert labile plant compounds into stabilized soil microbial necromass. Commun. Biol. 2, 441 (2019).
Qin, S. et al. Dual roles of microbes in mediating soil carbon dynamics in response to warming. Nat. Commun. 15, 6439 (2024).
Joergensen, R. G. Amino sugars as specific indices for fungal and bacterial residues in soil. Biol. Fertil. Soils 54, 559–568 (2018).
Duboc, O. et al. Lignin decomposition along an Alpine elevation gradient in relation to physicochemical and soil microbial parameters. Glob. Change Biol. 20, 2272–2285 (2014).
Kabiri, V., Raiesi, F. & Ghazavi, M. A. Tillage effects on soil microbial biomass, SOM mineralization and enzyme activity in a semi-arid Calcixerepts. Agr. Ecosyst. Environ. 232, 73–84 (2016).
Liu, C. et al. Effects of straw carbon input on carbon dynamics in agricultural soils: a meta-analysis. Glob. Change Biol. 20, 1366–1381 (2014).
Zhang, X. et al. Aridity and NPP constrain contribution of microbial necromass to soil organic carbon in the Qinghai-Tibet alpine grasslands. Soil Biol. Biochem. 156, 108213 (2021).
Tamura, M. et al. Plant litter chemistry alters the content and composition of organic carbon associated with soil mineral and aggregate fractions in invaded ecosystems. Glob. Change Biol. 23, 4002–4018 (2017).
Kogel-Knabner, I. The macromolecular organic composition of plant and microbial residues as inputs to soil organic matter: fourteen years on. Soil Biol. Biochem. 34, 139–162 (2017).
Li, J. et al. Differential accumulation of microbial necromass and plant lignin in synthetic versus organic fertilizer-amended soil. Soil Biol. Biochem. 149, 107967 (2020).
Keiluweit, M. et al. Mineral protection of soil carbon counteracted by root exudates. Nat. Clim. Change. 5, 588–595 (2015).
Huang, W. et al. Drivers of microbially and plant-derived carbon in topsoil and subsoil. Glob. Change Biol. 29, 6188–6200 (2023).
Thevenot, M., Dignac, M.-F. & Rumpel, C. Fate of lignins in soils: a review. Soil Biol. Biochem. 42, 1200–1211 (2010).
Liu, S. et al. Sedimentary ancient DNA reveals a threat of warming-induced alpine habitat loss to Tibetan Plateau plant diversity. Nat. Commun. 12, 2995 (2021).
Wang, N. et al. Pollen analysis of middle to late Holocene records shows little evidence for grazing disturbance to alpine grassland on the Tibetan Plateau until modern times. Palaeogeogr. Palaeoclimatol. Palaeoecol. 627, 111745 (2023).
Wang, N. et al. Broad-scale valley agriculture reaches back to the Ming Dynasty based on multiproxy records from Guli Lake, northeastern Tibetan Plateau. Quat. Sci. Rev. 352, 109208 (2025).
White, A. et al. AusPlots Rangelands Survey Protocols Manual Version 1.2.9. (The University of Adelaide Press, Adelaide, 2012).
Bai, Y. et al. Long-term active restoration of extremely degraded alpine grassland accelerated turnover and increased stability of soil carbon. Glob. Change Biol. 26, 7217–7228 (2020).
Jia, B. et al. Shrubification decreases soil organic carbon mineralization and its temperature sensitivity in alpine meadow soils. Soil Biol. Biochem. 168, 108651 (2022).
Chen, L. et al. Soil carbon persistence governed by plant input and mineral protection at regional and global scales. Ecol. Lett. 24, 1018–1028 (2021).
Nelson, D. W., Sommers, L. E. Total carbon, organic carbon, and organic matter. In: Methods of Soil Analysis: Part 3 Chemical Methods (eds. Sparks, D. L., Page, A.L., Helmkphenole, P.A., et al.), Ch. 34, 961–1010 (Soil Science of America Book Series, 1996).
AFNOR. Qualité des Sols, vol. 1, recueil de normes, Paris, (1999).
Mehra, O. P. & Jackson, M. L. Iron oxide removal from soils and clays by a dithionite–citrate system buffered with sodium bicarbonate. Clays Clay Miner. 7, 317–327 (2013).
Allen, S. E. Chemical Analysis of Ecological Materials (Blackwell Scientific Publications, Oxford, 1989).
Brookes, P. C. et al. Chloroform fumigation and the release of soil nitrogen: a rapid direct extraction method to measure microbial biomass nitrogen in soil. Soil Biol. Biochem. 17, 837–842 (1985).
Vance, E. D., Brookes, P. C. & Jenkinson, D. S. An extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 19, 703–707 (1987).
Campbell, C. D. et al. A rapid microtiter plate method to measure carbon dioxide evolved from carbon substrate amendments so as to determine the physiological profiles of soil microbial communities by using whole soil. Appl. Environ. Microbiol. 69, 3593–3599 (2003).
Hu, H. et al. Reducing the uncertainty in estimating soil microbial- derived carbon storage. Proc. Natl. Acad. Sci. USA 121, e2401916121 (2024).
Zhang, X. D. & Amelung, W. Gas chromatographic determination of muramic acid, glucosamine, mannosamine, and galactosamine in soils. Soil Biol. Biochem. 28, 1201–1206 (1996).
Hedges, J. I. & Ertel, J. R. Characterization of lignin by gas capillary chromatography of cupric oxide oxidation products. Anal. Chem. 54, 174–178 (1982).
Dormann, C. F. et al. Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography 36, 27–46 (2013).
García-Palacios, P. et al. Dominance of particulate organic carbon in top mineral soils in cold regions. Nat. Geosci. 17, 145–150 (2024).
Peng, Z., Shang, Z. & Ma, T. Data for ‘‘Plant detritus carbon dominates over microbial necromass carbon in topsoil of alpine ecosystems. figshare. Dataset. https://doi.org/10.6084/m9.figshare.30170548.v1 (2025).
Acknowledgements
This study was supported by the Natural Science Foundation of China (31961143012, U21A20183), the Science-based Advisory Program of The Alliance of National and International Science Organizations for the Belt and Road Regions (ANSO-SBA-2023-02), the Second Tibetan Plateau Expedition (2019QZKK0302). We are grateful to Dr. Bin Jia, Han Mao, Yifan Li for laboratory measurements and Haijun Zeng, Xudong Li, Wenyan Li, Xiaochun Wang, Haonan Guo for fieldwork.
Author information
Authors and Affiliations
Contributions
Zhanhuan Shang and Zhen Peng designed the study. Zhen Peng, Binyu Luo, Lingyan Qi, Tianyun Qi, and Dongmei Li did measurements and collected samples. Zhen Peng, Tian Ma, Zhuoma Dengzeng, Chunyan Wang, Di Lu, Yue Qi, Pengwei Zheng, and Xianrong Guan did laboratory measurements. Zhen Peng analyzed the data and wrote the manuscript. Zhanhuan Shang supported the study. Zhen Peng, Tian Ma, Allan Degen, Peipei Liu and Zhanhuan Shang wrote and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Earth and Environment thanks Zhiliang Zhang and the other anonymous reviewer(s) for their contribution to the peer review of this work.Primary Handling Editors: Leiyi Chen and Somaparna Ghosh. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Peng, Z., Ma, T., Degen, A. et al. Plant detritus carbon dominates over microbial necromass carbon in topsoil of alpine ecosystems. Commun Earth Environ 6, 912 (2025). https://doi.org/10.1038/s43247-025-02860-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43247-025-02860-7