Abstract
The extinction of woody vegetation in Antarctica remains difficult to constrain due to its fragmented macrofossil record. Despite its long-standing polar position, Antarctica hosted extensive vegetation throughout the Paleogene. This changed near the Eocene-Oligocene Transition (ca. 34 Ma) as glaciation led to vegetation decline. Sparse evidence suggests tundra-like forests persisted until the Pliocene in East Antarctica, but the Neogene record from West Antarctica is largely restricted to palynoflora data. Here, we report early Miocene plant macrofossils from West Antarctica, consisting of Nothofagus leaves. U-Pb zircon geochronology confirms tundra-like vegetation existed in this region during the early Miocene (ca. 22–20 Ma), representing the youngest macrofossil record of West Antarctica. These findings suggest that Nothofagus either persisted through Antarctica’s harsh Late Cenozoic Ice Age conditions or recolonised during intermittent warm periods. This substantially advances our understanding of West Antarctica’s vegetation history and extends the known record of Nothofagus in Antarctic ecosystems.
Similar content being viewed by others
Introduction
One of the most enigmatic aspects of Antarctica’s history is its transition from terrestrial ecosystems dominated by thriving forests in the early Cenozoic to its current configuration, with no tree or shrub layers and terrestrial biota limited to a few invertebrates, lichens, mosses, diatoms and microbial groups1,2,3. The present combination of low temperatures, lack of humidity and winter darkness results in a limited distribution and diversity of plant species4. However, throughout the Mesozoic and early Cenozoic, Antarctica was home to a wide variety of plant life5, despite already being situated at polar latitudes6. Although plant macrofossils are very sparse, it has been inferred that Antarctica may have been capable of supporting woody plants well into the Neogene7,8,9,10,11. A major challenge in assessing the evolution of vegetation dynamics in Antarctica is that nearly 98% of the continent is covered by ice12, resulting in a highly spatially and temporally fragmented fossil record. Consequently, the timing of the extirpation of woody vegetation across Antarctica remains poorly understood and is often interpreted through methods other than direct macrofossil evidence, such as palynological and molecular studies.
Although ephemeral ice sheets appeared in Antarctica during the late Mesozoic13, driven by a gradual decrease in global temperatures14,15,16, a permanent ice sheet did not develop until the Eocene–Oligocene Transition17,18 (EOT; ca. 34 Ma), which marked the initiation of the Late Cenozoic Ice Age19. The plant macrofossil record in Antarctica postdating the EOT suggests that some woody vegetation may have persisted in low-diversity Nothofagus-dominated tundra ecosystems20,21,22,23. Nothofagus is a genus of southern beech trees (family Nothofagaceae) that may have been able to inhabit latitudes up to 85°S5,8,9,11 in East Antarctica until as late as the Pliocene. This implies that areas at lower latitudes were probably able to support this vegetation during the Neogene as well, although direct macrofossil evidence remains elusive. However, this idea remains controversial, as ice-cap models are not consistent with the presence of Antarctic vegetation after the EOT, particularly from the Miocene onwards24,25,26. The study of Nothofagus fossils in Antarctica is therefore fundamental for understanding when and how forests became extinct on the continent.
The study of how woody vegetation disappeared from Antarctica is further complicated by marked differences in the fossil record between East and West Antarctica21. East Antarctica has yielded sporadic macrofossil discoveries that provide a broader basis for reconstructing past vegetation dynamics4,5. In contrast, in West Antarctica, the macrofossil record is far more limited and primarily restricted to deposits dating to shortly after the EOT5,21. Furthermore, the evidence for the presence of tundra-like vegetation in West Antarctica during and after the Oligocene is largely drawn from palynological studies. The nature of pollen evidence is such that there is substantial uncertainty as to whether this vegetation was physically present in the same location as the fossil pollen finds, since (i) anemophilic pollen may be able to travel considerable distances and (ii) tundra-like vegetation was present in proximal southern Patagonia during the mid- and late Cenozoic27,28, providing a potential source.
In this study, we present Nothofagus leaf macrofossils from the Miocene-aged glaciomarine sedimentary rocks of the Cape Melville Formation, exposed on King George Island in the northeastern Antarctic Peninsula (Fig. 1). These represent the youngest macrofossil evidence of woody vegetation on record for West Antarctica. Our results are further supported by U-Pb zircon geochronological data (LA-ICP-MS) collected from an ash-layer intercalated within the Cape Melville Formation, providing robust chronological control for the age of the fossils. Combined, this evidence provides unequivocal proof of tundra-like vegetation in West Antarctica during the Late Cenozoic Ice Age and enhances our understanding of how long these forests managed to persist during Antarctic glaciation.
The locations of the South Shetland Islands (SSI) and the Bransfield Strait (BS) are highlighted. Figure modified from32. Red box in the inset figure shows the location of the South Shetland Islands archipelago with King George Island in red. FZ fault zone.
The geology of Melville Peninsula, King George Island
Melville Peninsula is located at the northeast extremity of King George Island (Fig. 2), which is the largest island in the South Shetland Islands. Although King George Island is mostly covered by an ice cap, the rocks that are exposed host an exceptional Cenozoic stratigraphic record29,30 and therefore hold critical exposures for studies addressing the Cenozoic evolution of Antarctica. The South Shetland archipelago extends parallel to the northern Antarctic Peninsula on the western side (Fig. 1), separated from the peninsula by the Bransfield Strait, which is a relatively young back-arc rift basin formed during the last 4 Myr31. The archipelago of the South Shetland Islands is mainly composed of the products of the active margin developed as a result of the eastward subduction of the Phoenix Plate beneath the continental crust of the Antarctic Peninsula32,33,34,35,36,37. This active margin was tectonically active from at least the late Palaeozoic to early Mesozoic38,39,40, until approximately 20–19 Ma41, when arc magmatism ceased as a result of trench–ridge collisions along the margin.
The archipelago of the South Shetland Islands records tectonic, global sea-level and climate change throughout the Mesozoic and Cenozoic32, which has been divided into three main stages: (i) deep marine sedimentation during the Jurassic and Early Cretaceous33,42; (ii) subaerial arc volcanism and sedimentation with a proliferation of plants and fauna from ca. 140–34 Ma23,33,35,43,44,45; and (iii) glacial and interglacial deposits from ca. 34 Ma29,30,32. While Jurassic and Early Cretaceous rocks are exposed in the southwest of the South Shetland Islands32,36, Cenozoic formations are confined to the northeast of the archipelago46,47. The exposures of King George Island are dominated by Cenozoic volcanic and sedimentary strata32,36,48,49. Notably, in the southwestern region hosts the Moby Dick Group, an Eocene to Miocene volcano-sedimentary succession50,51 (Fig. 2) composed of three formations: (i) Sherratt Bay Formation, (ii) Destruction Bay Formation and (iii) Cape Melville Formation.
The Sherratt Bay Formation consists of an andesitic-basaltic succession which is exposed on the eastern edge of the Melville Peninsula (Fig. 2). While this unit has been interpreted as a terrestrial plateau-basalt sheet at the base of the Moby Dick Group49, it has been argued that it may be instead be a doleritic sill48. Its age remains poorly constrained. A K-Ar date of ca. 18 Ma8, previously attributed to the Sherratt Bay Formation, is likely associated with the early Miocene dike intrusions that are widespread on the Melville Peninsula rather than the Sherratt Bay rocks themselves41.
A stratigraphic hiatus separates the Sherratt Bay Formation from the overlying Oligocene fossiliferous Destruction Bay Formation. Although the presence of fossil wood has been mentioned for the Destruction Bay Formation46, no further details have been presented or documented. This formation consists of a ca. 40–100 m thick succession of volcaniclastic rocks, dominated by reworked basaltic material with horizons (mostly siltstones) rich in marine invertebrates46 (Fig. 2). The sedimentary features of this unit suggest a nearshore depositional environment under non-glacial conditions46. The age of the Destruction Bay Formation is loosely bracketed between 25.3 ± 0.8 Ma (brachiopods, 87Sr/86Sr52) and 23.6 ± 0.7 Ma (basaltic tuff, K-Ar53).
Overlying the Destruction Bay Formation is the Cape Melville Formation (Fig. 2), which is composed of glacio-marine sediments including sandstones, conglomerates, clay-shales and silty shales with occasional iceberg-rafted dropstones46. The dropstones often show glacial striae and glacially polished facets, thus giving primary evidence for the presence of a continental ice-sheet in Antarctica46,47. This glacial event has been correlated with the Mi-1 and Mi-1a glaciations47,48, which are the mostrelevant glaciation events following the establishment of the ice-cap in Antarctica during the EOT54. However, it is possible that West Antarctica developed a comparatively smaller ice cap relative to East Antarctica55. An abundant fossil record is present in the Cape Melville Formation, with (i) a thriving community of marine invertebrates, which includes Bivalvia, Gastropoda, Anthozoa, Decapoda, Scaphopoda, Bryozoa, Brachiopoda, Echinodermata, and Polychaeta56, and (ii) reworked palynomorphs (spores and pollen), which include pollen morphogenus Nothofagidites sp. type fusca and brassi47. The benthic foraminifera of the Cape Melville Formation suggest an early Miocene age57 and an inferred Sr age of 22.6 ± 0.4 Ma was obtained from skeletal carbonate52. Recently, a tuff interbedded in the Cape Melville Formation has been dated (40Ar/39Ar on hornblende) to an age of 21.3 ± 3.1 Ma48. These two geochronological constraints have been used to suggest an early Miocene age for the Cape Melville Formation48.
Methods
Fieldwork and fossil preparation
The fieldwork campaign took place on the Melville Peninsula during January and February 2023 as part of the Chilean Antarctic Institute’s ECA-59 expedition aboard the Betanzos Vessel. Plant macrofossils were found preserved as impressions at two localities, Notho1 (62.019°S, 57.633°W) and Notho2 (62.018°S, 57.632°W) (Fig. 3). These fossils are housed in the Palaeontological Collection of Antarctica and Patagonia at the Chilean Antarctic Institute (INACH) in Punta Arenas, Chile. They were examined under a Zeiss Stemi 2000-C stereo microscope, and photographic records were captured using a Sony Alpha 7 III camera with a macro lens. The systematic description followed standardised guidelines58. For the palynological preparation, the standard acetolysis method was followed (Erdtman, 1960). Thirty grams of rock containing the macrofossil CPAP 9105 were processed. The procedure involved crushing the rock with a mortar, treating it with hydrochloric and hydrofluoric acids, filtering the residue through a microscopic mesh, mounting it on a slide, and examining it under a Karl Zeiss Axioscope microscope.
A Nothofagus sp.1 fossil leaf (CPAP 9105) showing the primary and second veins; B drawing with detail of secondary opposite venation; C Nothofagus sp.2 foliar imprint (CPAP 9106) showing the primary and second veins; D drawing with detail of craspedodromous, evenly spaced venation.
LA-ICP-MS zircon U-Pb geochronology
Zircon grains were separated from an ash-layer (23JB15) collected in the Cape Melville Formation using standard crushing, hydraulic, magnetic and heavy liquid procedures. They were then mounted and imaged by cathodoluminescence (CL) using a scanning electron microscope (SEM) at the ETH Zürich. Laser ablation inductively coupled plasma mass spectrometry (LA-ICPMS) techniques were used to determine trace element abundances and U-Pb ages. A 193 nm Resonetics Resolution S155 laser ablation system was used coupled to a Thermo Element XR, Sector-field single collector ICP-MS59. Laser parameters include a 19 μm spot size, a repetition rate of 5 Hz and an energy density of ca. 2 J cm-2. The ablation aerosol was mixed in the fast washout S-155 ablation cell (Laurin Technic) with carrier gas consisting of helium (ca. 0.25 L min-1) and make-up gas consisting of argon (ca. 1 L min-1) and nitrogen (2 mL min-1). The ablated aerosol was then homogenised by flushing through a squid device before introduction into the plasma torch. The single collector sector-field MS is equipped with a high-capacity (80 m3 h-1) interface pump to improve sensitivity. Before each analytical session the instrument was optimised with NIST SRM612 glass to achieve a detection efficiency in the range of 1% (on Pb, Th, U) while keeping a low oxides production (248ThO+/ 232Th+ ≤ 0.25%) and a U/Th ratio of ca. 1. Intensities were recorded for the following isotopes: 27Al, 29Si, 31P, 89Y, 91Zr, 93Nb, 137Ba, 139La, 140Ce, 141Pr, 146Nd, 147Sm, 153Eu, 157Gd, 159Tb, 163Dy, 165Ho, 167Er, 169Tm, 173Yb, 175Lu, 178Hf, 181Ta, 202Hg, 204Pb, 206Pb, 207Pb, 208Pb, 232Th, 235U, and 238U. The full dataset can be found in the associated supplementary dataset (10.5281/zenodo.16738353), following the community-derived guidelines60.
For U-Pb geochronology, GJ-1 was used as the primary calibration reference material (CRM). Validating reference materials (VRMs) included Plešovice (337 Ma61), Temora (417 Ma62) and 91500 (1062 Ma63) zircons. The VRMs results show the achievable precision and accuracy of the method, which is in the range of 1.0%. The data was reduced using the software Iolite 4.564,65 with VizualAge66. No common-Pb correction was applied but integration intervals were set to exclude inclusions, common Pb and discordant parts of the signal. The long-term external uncertainty is in the range of 1.5% for 206Pb/238U ages and is composed of the uncertainty from the applied corrections, uncertainty of the decay constants, lacking common-Pb correction, the uncertainty on the true 206Pb/238U ratio of the primary standard GJ-1, and possible uncertainty from matrix effects. For trace element quantification we used Si as internal standard at 15.2 wt% SiO2 in zircons, and SRM NIST610 as external CRM. Ti was quantified by zircon 91500 (Ti: 4.73 ± 0.15 mg g-1)67.
Results
Systematic Nothofagus description
Two angiosperm leaf impressions were recovered from in situ siltstone layers at two separate sites, both within the Cape Melville Formation of the early Miocene. These rocks are interpreted as part of a glacio-marine depositional environment, offering a rare and valuable insight into the Neogene vegetation of West Antarctica. The samples provide the basis for the systematic description of Nothofagus.
Class EUDICOTYLEDONEA
Order FAGALES
Family NOTHOFAGACEAE Kuprianova 196268
Genus Nothofagus Blume 185069
Type species Nothofagus antarctica Örsted, 187370
Nothofagus sp.1
Figure Fig. 3A, B.
Material
Palaeontological Collection of Antarctica and Patagonia CPAP 9105.
Locality
Notho1, Cape Melville, King George Island, Antarctica.
Description
Incomplete leaf impression corresponding to a microphyll, measuring 3.0 cm in length and 2.5 cm in width (L/W ratio = 1.2), ovate to suborbicular in shape. The primary vein is straight and prominent. Secondary venation is pinnate, opposite, consists of 5–6 pairs of straight, well-defined craspedodromous veins per side. The divergence angle of the secondary veins ranges from ~50° at the base to ~40° toward the apex, indicating a moderate and gradually decreasing pattern. Tertiary venation is not preserved, but thin, divergent basal agrophic veins are present, extending toward the margin. The margin is partially preserved and lobed, with broad, roundedshallow lobes. The apex and base are not preserved.
Floristic comparisons
The specimen described here is morphologically comparable to fossil leaves assigned to Nothofagus subferruginea, taxon documented from the Oligocene to early Miocene of Chile and Argentina71, as well as from strata in West Antarctica on King George Island72,73,74,75. The similarities are primarily based on its ovate to orbiculate shape, craspedodromous venation, and lobed margin. However, it differs in its smaller overall size, greater number of secondary veins, and distinctly dentate margins. It also exhibits morphological similarities with Nothofagus variabilis, originally described by Dusén from Patagonia and widely recorded from the Eocene to the Miocene in formations such as Río Guillermo, Loreto, Río Turbio, Ñirihuau, Cullen, and Barrancas Carmen Silva28. Nothofagus variabilis exhibits an elliptic to broadly ovate or suborbiculate lamina and 6–10 pairs of craspedodromous secondary veins emerging at a moderately acute angle71. However, in contrast to the present material, Nothofagus variabilis possesses a serrate composite margin, with convex–convex primary teeth and straight–straight secondary teeth. The fossil leaf also bears resemblance to Nothofagus cunninghamii and other species of the subgenus Brassospora from the Miocene of New Zealand and Australia, particularly in its lobed margin and straight craspedodromous venation. Nothofagus cunninghamii typically displays 5–6 pairs of secondary veins arranged at a moderately acute angle relative to a straight, pinnate midvein76. Comparative analysis reveals morphological similarities between Nothofagus sp. 1 and Nothofagus gunnii, the tanglefoot or deciduous beech endemic to the highlands of Tasmania, Australia. Both species fall within the microphyll size class and exhibit pinnate primary venation, with a broadly ovate to suborbicular lamina and a comparable length-to-width ratio (~1.2). Additionally, both taxa display craspedodromous secondary venation, and, despite differences in lobation depth, they share the presence of marginal lobes. In Nothofagus gunnii, the lobes are markedly deep with well-defined sinuses, whereas in Nothofagus sp. 1, the lobes are broader, more rounded, and shallow—possibly reflecting a plesiomorphic or less specialised condition. Notable distinctions include the number of secondary veins: Nothofagus sp. 1 has 5–6 pairs per side, exceeding the typical 3–5 pairs observed in Nothofagus gunnii. Furthermore, basal agrophic veins are present in the fossil specimen from King George Island but are absent in Nothofagus gunnii, indicating further divergence in venation architecture.
Another morphotype displaying similar morphology is Nothofagus australis, synonymised with Nothofagus simplicidens71. N. australis shares a similar geographic and stratigraphic range with Nothofagus variabilis in Patagonia and is characterised by small, leptophyllous to microphyllous ovate leaves with craspedodromous venation and 5–6 straight secondary vein pairs entering the tooth apex medially. However, Nothofagus australis differs in its rounded apex, cuneate to obtuse base, and dentate margin, which contrasts with the lobed margin and specific venation patterns observed in the morphotype described here.
Although the absence of a preserved apex or tertiary venation limits definitive assignment, the overall morphological and venational structure shows the strongest resemblance to southern temperate rainforest species within Nothofagaceae. The finding of Nothofagidites pollen grains with affinities to subgenus Fuscospora, with an equatorial diameter of 23.26 microns, a circular outline, five colpi, a curved mesocolp, an average colpus aperture length of eight microns, ornamentation was not visible under optical microscope, and the exine thickness measured 0.62 µm. in the same beds, would help confirm the taxonomic placement. Furthermore, the late Oligocene–early Miocene was apparently the time of maximum spatial distribution of Nothofagaceae worldwide77.
Class EUDICOTYLEDONEA
Order FAGALES
Family NOTHOFAGACEAE Kuprianova 196268
Genus Nothofagus Blume 185069
Type species Nothofagus antarctica Örsted, 1873
Nothofagus sp. 2
Figure Fig. 3 C-D.
Material
Palaeontological Collection of Antarctica and Patagonia - CPAP 9106.
Locality
Notho2, Cape Melville, King George Island, Antarctica.
Description
The specimen represents an incomplete, simple leaf, estimated to be microphyllous in size. The approximate leaf width 2.5 cm, estimated length ~3.0–3.5 cm (based on contour extrapolation) yielding a length-to-width ratio of ~1.2–1.4. The preserved lamina exhibits an overall broadly ovate to suborbicular shape, with a weakly lobed margin. The base and apex are not preserved, limiting detailed characterisation of these regions. The primary venation is pinnate, marked by a robust, straight midvein. No clear evidence of secondary or tertiary venation is observed, likely due to preservation constraints. The leaf margin is moderately undulate, with shallow sinuses and rounded projections, potentially indicative of broad lobes. The overall contour of the leaf is consistent with morphotypes attributed to Nothofagus sensu lato, particularly members of the subgenus Brassospora, based on the broad lamina, lobed margins, and absence of pronounced dentition. While the absence of visible venation limits a full taxonomic comparison, the gross morphology and marginal patterning bear resemblance to fossil species such as Nothofagus subferruginea and Nothofagus australis as described in Nothofagus sp1.
Geochronology
The ash layer 23JB15, of andesitic composition, was sampled within a section of the sequence dominated by sandstones, approximately 70 m above the formation boundary with the Destruction Bay Formation (Fig. 4a). CL imaging of zircon grains revealed either (i) oscillatory zonation with distinct cores, or (ii) patchy and homogeneous zoning, with an absence of rim-core relationship, both textural varieties are typical of zircons crystallised in magmatic environment37,40 (Fig. 4b). Ablation of zircons yielded concordant analyses spanning 206Pb/238U ages from 19.3 ± 1.0 to 68.1 ± 3.1 Ma. From these, two main populations are observed: an older cluster that yielded ages between 63.1 ± 2.6 and 68.1 ± 3.1 Ma, and a younger group with concordant dates between 19.3 ± 1.0 and 22.3 ± 1.3 Ma (Fig. 4c). The latter yields a weighted mean age of 20.97 ± 0.22 Ma (n = 27, MSWD = 1.2, Fig. 4d). Late Cretaceous magmatism is present in several parts of the Antarctic Peninsula78 and may represent part of the basement underlying King George Island79, making it the most likely source of the 68–63 Ma inherited zircon grains. The younger zircon ages are closely associated with the late-stage arc magmatic pulses, which are particularly well documented in this sector of King George Island41. The full dataset is available in associated supplementary dataet (https://doi.org/10.5281/zenodo.16738353).
A Composite stratigraphic section indicating the position of the sample analysed for U-Pb geochronology (23JB15) and the level where the leaf fossils were found. B Weighted mean 206Pb/238U dates of the volcanic ash-layer, sample 23JB15. Bars represent single ablation spots and represent 2σ uncertainty. C Wetherill concordia plot of zircon U–Pb data for the sample 23JB15. D Representative SEM-based cathodoluminescence images of the dated zircons with location of analytical spots, including 206Pb/238U ages.
Discussion
Age of the Nothofagus leaves
The Nothofagus leaves discovered in the early Miocene Cape Melville Formation are stratigraphically closely associated with the ash layer 23JB15 (Fig. 4). The latter contains zircon grains exhibiting internal oscillatory zoning and homogeneous U-Pb ages, indicating direct magmatic sourcing with minimal, if any, reworking or sedimentary input. Thus, the weighted mean age of 20.97 ± 0.22 Ma provides the best estimate for the depositional age of the ash layer and constrains the age of the Cape Melville Formation. No substantial stratigraphic discordance or hiatus was identified between the ash layer and the fossiliferous horizon, nor within the entire Cape Melville Formation. Unfortunately, no ash bed has been found above the fossilised leaves to further bracket their age. However, the available evidence suggests that the leaves were deposited between 20 and 22 Ma, making this the youngest plant macrofossil record from West Antarctica.
Post-EOT vegetation evolution of Antarctica: east versus west
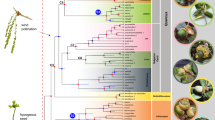
Despite being located in a polar position since at least the Cretaceous34,80, East Antarctica maintained vegetation dominated by floristically rich forests prior to the EOT21 (Fig. 5). This vegetation was diverse in both composition and stature81, with forests dominated by Nothofagus, Araucaria and podocarps, including large trees82. This suggests that the climate was not extreme, and vegetation resembled the forests currently found in Patagonia, known vernacularly as ‘Valdivian Forest’83,84,85. Temperatures fell at the end of the Eocene, leading to the collapse of these forests as a viable ecosystem86. The Oligocene pollen records suggest that southern beech forests grew locally, with minor components of podocarps, Proteaceae and other shrubby angiosperms21. This vegetation type is similar to the present-day Patagonian steppe of bunchgrasses and shrubs or the Nothofagus-podocarp forests of New Zealand. These Oligocene forests were dominated by Nothofagus87, which had become reduced in both stature and diversity compared to the pre-EOT vegetation88,89. Among these Oligocene vegetational assemblages, the extensive fossil record of Nothofagus subgenus Fuscospora suggests that it may have been the only species21,90. The fossil leaves described herein, characterised by their microphyllous size, lobed margin, and craspedodromous secondary venation, display a suite of morphological features typically associated with mesic temperate forest environments. The combination of a straight, prominent midvein; 5–6 pairs of opposite, craspedodromous secondary veins; and broad, shallow lobes suggests an affinity with deciduous or semi-deciduous taxa inhabiting humid, temperate regions with seasonal climatic variability. Comparisons with extant and extinct members of Nothofagus, particularly species within the subgenera Fuscospora and Brassospora, also support this interpretation. Morphological affinities with Nothofagus gunnii, a relict Tasmanian species adapted to cool temperate montane forests, and with fossil taxa such as N. variabilis, N. australis, and N. subferruginea—widely distributed in Oligocene to Miocene strata of Patagonia, Antarctica, and Australasia—reinforce the hypothesis of a Nothofagaceae-dominated paleoflora in the studied assemblage. The presence of basal agrophic veins further supports assignment within Nothofagaceae and may indicate phylogenetic proximity to plesiomorphic lineages91. Although incomplete preservation precludes definitive taxonomic placement, the assemblage provides strong evidence for a paleovegetation type resembling modern southern temperate rainforests.
The co-occurrence of Nothofagidites pollen grains with affinities to Fuscospora or Lophozonia within the same stratigraphic context would provide additional support for this taxonomic interpretation. Taken together, these observations are consistent with established global biogeographic patterns. The transition from the late Oligocene to the early Miocene is marked by cooler climatic conditions compared to the earlier Miocene92,93. Nonetheless, the palaeobotanical record from this interval, primarily composed of pollen and spores, with only limited occurrences of fossil wood and leaf impressions, suggests that tundra-like, low-diversity vegetation may have persisted beyond the EOT, likely restricted to climatically favourable or refugial microenvironments21,71,79,80.
It is uncertain whether this vegetation persisted into the early Miocene, as most of the record is based on pollen evidence21,89,90. The cold, glacial climate of the Miocene was briefly interrupted by a warm period ca. 17-15 Ma (the Middle Miocene Climatic Optimum; MMCO)94. This warming resulted in a temporary increase in both the diversity and stature of East Antarctic vegetation22,95,96, which facilitated the return of several Nothofagus taxa21. However, this was immediately followed by abrupt cooling97, accompanied by the gradual disappearance of woody plants, although the continued presence of Nothofagus pollen suggests that they may have persisted in glacial refugia in a manner analogous to the glacial refugia detected through Pleistocene glaciations in southern Patagonia98,99, which explain the degree of endemism in this part of South America and the speed of subsequent recolonisation96,100. After the MMCO, the ice sheet expanded in Antarctica throughout the Neogene and Quaternary, preventing the establishment of woody plants, with the exception of one notable site with tundra-like vegetation of mid-Pliocene age. The latter is found at less than 500 km from the South Pole5,21,101, and had an assemblage similar to that in the modern-day southern Patagonia (Cape Horn)21,45. This vegetation consisted of cushion-forming mosses and vascular plants with deciduous Nothofagus and rare podocarps9,21,102. Notably, there is a lack of consensus on the age of the rocks that host these fossils7,21 and on how these tundra-like forests may have persisted in East Antarctica during the middle Pliocene.
To the west of the Transantarctic Mountains lies the West Antarctic region which, like East Antarctica, was already located in a polar position during the late Mesozoic38,40,80,103. Our understanding of the Cenozoic vegetation evolution of the Antarctic Peninsula has been mostly constrained from the outcrops exposed in King George Island104 and Seymour Island75,105,106,107. These outcrops also hold most of the evidence for woody vegetation in West Antarctica4,5. Middle Eocene petrified forests are found in the centre-east of the island, revealing a once diverse ecosystem of Nothofagus and Araucaria conifers, among other species81,108. This vegetation survived until the late Eocene4, when King George Island was on the locus of the arc axis in the Antarctic Peninsula32,36. After the EOT, as the climate turned colder and drier109, the woody vegetation transitioned to tundra-like forests dominated by Nothofagus21. Although it has been suggested that from the Oligocene onwards there may have been a relatively extensive ice cap in the Antarctic Peninsula, the lateral extent of such an ice cap remains unclear30. Nevertheless, the development of an ice-cap during the Oligocene may have caused the tundra-like vegetation to succumb, at least temporarily4.
Evidence for woody vegetation is absent in West Antarctica after the early Oligocene, except for evidence from palynological studies on the early Miocene glacio-marine Cape Melville Formation on King George Island30,47, which suggests the presence of a monotypic Nothofagus assemblage. Prior to our study, it was not possible to determine whether the source of the Nothofagus pollen was autochthonous or allochthonous, given the anemophilous nature of its grains. Because Nothofagus pollen is so widely wind-dispersed, it is difficult to use palynological evidence alone to reconstruct possible species co-occurrences within local vegetation. However, the macrofossil record does not suffer from this problem, since it is likely that most Nothofagus macrofossils are deposited very close to their source plants100. While the Nothofagus leaves were found in the shallow marine Cape Melville Formation, suggesting transport before deposition, this does not preclude the presence of local vegetation, as they were recovered from a land-proximal marine environment56.
Miocene tectonic models of the Antarctic Peninsula and the proximity of other Nothofagus communities
The Miocene tectonic history of the Antarctic Peninsula provides context for assessing the persistence of Nothofagus communities in West Antarctica during the early Miocene. The geological history of the Antarctic Peninsula is dominated by the development of a continental arc throughout the Mesozoic and part of the Cenozoic39,40, which was the result of the eastward subduction of the oceanic lithosphere of the Phoenix Plate41. This subduction system waned during the Cenozoic to eventually cease at ca. 20 Ma34,41,110. The sea floor remnants of the region and paleomagnetic studies have been used to reconstruct this sector of Antarctica and South America and suggest that during the Jurassic and Cretaceous, Patagonia was in juxtaposition to the north of the Antarctic Peninsula103,110. Therefore, during this period, there would have been a strong floral and faunal connection between these two regions83. However, during the Cenozoic the Antarctic Peninsula drifted to the south with respect to South America as a response to the formation of the Scotia Plate, a sliver of oceanic lithosphere located between the Antarctic and the South American plates111 (Fig. 6). This extension led to the opening of the Drake Passage, which effectively disconnected Antarctica from South America, causing the isolation of the Antarctic continent from any other landmass and setting up the Antarctic Circumpolar Current112. This event, which occurred near to the EOT, marks the onset of the Late Cenozoic Ice Age111 and is considered, at least in part, to have contributed to the initiation of this Earth period113. Throughout the Palaeogene, the Antarctic Peninsula experienced counterclockwise rotation, which caused it to progressively further separate from Patagonia110. During the early Miocene, when the Cape Melville Formation was deposited, it is estimated that the Antarctic Peninsula, and thus King George Island as well, was more than 500 km to the south with respect to Patagonia41,110 (Fig. 6). While anemophilic pollen may be able to travel considerable distances114 and tundra-like vegetation was present in southern Patagonia during the mid- and late Cenozoic27,28, it is unlikely that leaves could have been transported and preserved more than 500 km south. This supports our argument that the Nothofagus leaf fossils originated from an autochthonous tundra-like forest.
Redrawn from reference110. ASSSZ Ancestral South Sandwich Subduction zone.
Survival or Recolonisation?
The presence of Nothofagus-dominated tundra-like forests as recently as the early Miocene in West Antarctica (this work) and the mid-Pliocene in East Antarctica raises the question of whether these species were resilient and adapted to survive in glacial refugia to changing climatic conditions throughout the Neogene, or briefly recolonised Antarctica under particularly favourable conditions during warm intervals. From an autoecological perspective, Nothofagus seed dispersal is by gravity and wind (anemochory), while germination is generally epigeal and occurs not far from the seed-producing tree crown115. Although some authors argue that Nothofagus seeds can disperse over long transoceanic distances115, there is still a considerable body of research that claims that their seeds are intolerant to seawater116,117,118, and so would have been unable to recolonise the Antarctic Peninsula from the southern tip of South America after the Drake Passage opened during the Paleogene119.
The base of the Cape Melville Formation is marked by a glacial diamictite, suggesting that the ice grounding line extended into the marine realm. This glacial advance is generally correlated with the Mi-1 and Mi-1a events, which occurred close to the Oligocene-Miocene boundary32,48. Following this, the marine sedimentary succession evolved to ice-proximal and then ice-distal conditions prior to the deposition of the rocks hosting the Nothofagus leaf macrofossils9,12,30. The presence of tundra-like forest thus coincides with evidence for a climate amelioration in this part of Antarctica. The evidence for extensive ice cover at the Oligocene-Miocene boundary indicates that this site is itself unlikely to have been a persistent glacial refugium.
The sparse occurrence of Neogene woody macrofossils in Antarctica may indicate that Nothofagus recolonised Antarctica during warmer intervals in suitable locations via long-distance dispersal120,121, rather than persisting without interruption. This is based on the premise that continuous persistence would have yielded, albeit locally, a continuous record of woody plant fossils. Episodic recolonisation from tundra-like vegetational relicts would result in a more fragmented fossil record, which better fits the observed macrofossil data. Tundra-like forests existed in regions subjected to glacial conditions during this period, including Patagonia122, Falkland/Malvinas Islands123 and Oceania124,125, and potentially could have acted as seed sources for the resurgence of these forests in Antarctica. However, the absence of fossils does not necessarily disprove the presence of glacial refugia. Both the lack of appropriate sedimentation conditions and the inaccessibility of many sedimentary strata could produce a discontinuous and incomplete fossil record. In addition, the mechanism by which long-distance dispersal may have occurred remains unclear.
The record of tundra-like vegetation in East Antarctica after the early Miocene is limited to two episodes: the MMCO22 and the mid-Pliocene9,12 (Fig. 5). The fossiliferous site dated to the latter is located less than 500 km from the South Pole. If such latitudes were populated by Nothofagus-dominated forest relicts during these periods, it is reasonable to assume that this vegetation was also present at lower latitudes of Antarctica. However, direct macrofossil evidence is absent in West Antarctica after the early Miocene. Although the evidence presented here represents a substantial advance in our understanding of the Neogene vegetation history of West Antarctica, the question of persistence or recolonisation remains unresolved. Nevertheless, evidence presented herein supports refugia.
Stability of the Neogene ice-cap
The presence of low-diversity tundra-like vegetation either temporarily recolonising Antarctica during favourable conditions or persisting through the Late Cenozoic Ice Age has critical implications for understanding the stability of the Antarctic ice sheet. Although consecutive glaciations followed the EOT, during which the ice cap is conservatively estimated to have reached 80% to 110% of its modern volume126, the presence of tundra-like forests suggests that at least some sectors remained ice-free during warm periods or possibly throughout the Neogene, lasting as late as the Pliocene. These findings challenge the assumption of a stable Antarctic ice sheet since the EOT. Furthermore, evidence indicates that ice sheets could have melted rapidly during brief warm periods127,128,129,130, demonstrating their sensitivity to climate warming. This highlights the potential for rapid changes in the Antarctic ice cap under current global warming scenarios, where the return of woody vegetation could occur relatively quickly.
Conclusions
The Nothofagus leaf imprints from the Cape Melville Formation on King George Island presented here (Fig. 5), combined with our geochronological results (Fig. 4), provide the first robust evidence for the presence of tundra-like vegetation in West Antarctica during the early Miocene, suggesting that tundra-like forests were present in the region at least between 22 and 20 Ma and potentially even before this period, establishing it as the youngest woody macrofossil record from West Antarctica. This finding advances the debate surrounding palynoflora-based interpretations of Neogene tundra-like forests in West Antarctica. Furthermore, it fills critical gaps in our understanding of how woody vegetation became extinct in Antarctica and underscores the remarkable adaptability of Nothofagus within Antarctic ecosystems.
Localised refugia with tundra-like vegetation in West Antarctica may have either survived numerous glaciations since the EOT or recolonised during warm interglacial periods. King George Island likely served as such an enclave during the early Miocene, suggesting a more complex climatic history than previously thought. This implies that Antarctic conditions may not have been uniformly harsh for woody vegetation, at least episodically. The biology of Nothofagus and its syndrome of reproduction argue against recolonisation from nearby areas (e.g. Patagonia). On the other hand, repopulation of tundra-like forests in Antarctica from glacial refugia is also uncertain, as the ice sheet should have reached volumes during the Late Cenozoic Ice Age that were similar to or greater than those of the present day. Therefore, whether tundra-like vegetation recolonised Antarctica or persisted under the harsh conditions of the Late Cenozoic Ice Age remains a challenging and open question, which should be the focus of future studies.
The presence of early Miocene tundra-like vegetation in West Antarctica suggests that the vegetation dynamics may have been more synchronised between East and West Antarctica than previously assumed, highlighting the complex stability of Antarctica’s ice sheet.
Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its supplementary information files, and are also accessible via the following repository: https://zenodo.org/records/16738354.
References
Green, T. G. A., Schroeter, B. & Sancho, L. G. Plant Life in Antarctica. In Functional Plant Ecology (CRC Press, 2007).
Allegrucci, G., Carchini, G., Todisco, V., Convey, P. & Sbordoni, V. A molecular phylogeny of Antarctic Chironimidae and its implications for biogeographical history. Polar Biol 29, 320–326 (2006).
Convey, P. et al. Antarctic terrestrial life – challenging the history of the frozen continent?. Biol. Rev. 83, 103–117 (2008).
Cantrill, D. J. & Poole, I. The Vegetation of Antarctica through Geological Time. (Cambridge University Press, 2012).
Francis, J. E. et al. 100 Million Years of Antarctic Climate Evolution: Evidence from Fossil Plants. Open-File Report 19–28 (2007).
Scotese, C. R., Gahagan, L. M. & Larson, R. L. Plate tectonic reconstructions of the Cretaceous and Cenozoic ocean basins. Tectonophysics 155, 27–48 (1988).
Ashworth, A. C. & Cantrill, D. J. Neogene vegetation of the Meyer Desert Formation (Sirius Group) Transantarctic Mountains, Antarctica. Palaeogeogr. Palaeoclimatol. Palaeoecol. 213, 65–82 (2004).
Fleming, R. F. & Barron, J. A. Evidence of Pliocene Nothofagus in Antarctica from Pliocene marine sedimentary deposits (DSDP Site 274). Mar. Micropaleontol. 27, 227–236 (1996).
Francis, J. E. & Hill, R. S. Fossil plants from the Pliocene Sirius Group, Transantarctic Mountains; evidence for climate from growth rings and fossil leaves. PALAIOS 11, 389–396 (1996).
Roberts, A. P., Wilson, G. S., Harwood, D. M. & Verosub, K. L. Glaciation across the Oligocene–Miocene boundary in southern McMurdo Sound, Antarctica: new chronology from the CIROS-1 drill hole. Palaeogeogr. Palaeoclimatol. Palaeoecol. 198, 113–130 (2003).
Webb, P.-N. & Harwood, D. M. Pliocene Fossil Nothofagus (Southern Beech) from Antarctica: Phytogeography, Dispersal Strategies, and Survival in High Latitude Glacial-Deglacial Environments. In Forest Development in Cold Climates (eds Alden, J. N., Mastrantonio, J. L. & Ødum, S.) 135–165 (Springer US, Boston, MA, https://doi.org/10.1007/978-1-4899-1600-6_10. 1993).
Rees-Owen, R. L. et al. The last forests on Antarctica: reconstructing flora and temperature from the Neogene Sirius Group, Transantarctic Mountains. Org. Geochem. 118, 4–14 (2018).
Barr, I. D. et al. 60 million years of glaciation in the Transantarctic Mountains. Nat. Commun. 13, 5526 (2022).
Davies, A. et al. Assessing the impact of aquifer-eustasy on short-term Cretaceous sea-level. Cretac. Res. 112, 104445 (2020).
Li, L., Keller, G. & Stinnesbeck, W. The Late Campanian and Maastrichtian in northwestern Tunisia: palaeoenvironmental inferences from lithology, macrofauna and benthic foraminifera. Cretac. Res. 20, 231–252 (1999).
Linnert, C. et al. Evidence for global cooling in the Late Cretaceous. Nat. Commun. 5, 4194 (2014).
Evans, D., Wade, B. S., Henehan, M., Erez, J. & Müller, W. Revisiting carbonate chemistry controls on planktic foraminifera Mg / Ca: implications for sea surface temperature and hydrology shifts over the Paleocene–Eocene Thermal Maximum and Eocene–Oligocene transition. Clim. Past 12, 819–835 (2016).
Liu, Z. et al. Global Cooling During the Eocene-Oligocene Climate Transition. Science 323, 1187–1190 (2009).
Lear, C. H., Bailey, T. R., Pearson, P. N., Coxall, H. K. & Rosenthal, Y. Cooling and ice growth across the Eocene-Oligocene transition. Geology 36, 251–254 (2008).
Birkenmajer, K. & Zastawniak, E. Plant remains of the Dufayel Island Group (Early Tertiary?), King George Island, South Shetland Islands (West Antarctica). Acta Palaeobot. 1–2, (1986).
Cantrill, D. J. & Poole, I. After the heat: late Eocene to Pliocene climatic cooling and modification of the Antarctic vegetation. in The Vegetation of Antarctica through Geological Time (eds Cantrill, D. J. & Poole, I.) 390–457 (Cambridge University Press, Cambridge, https://doi.org/10.1017/CBO9781139024990.009. 2012).
Lewis, A. R. et al. Mid-Miocene cooling and the extinction of tundra in continental Antarctica. Proc. Natl. Acad. Sci. USA 105, 10676–10680 (2008).
Poole, I., Hunt, R. J. & Cantrill, D. J. A Fossil wood flora from king george island: ecological implications for an Antarctic Eocene vegetation. Ann. Bot. 88, 33–54 (2001).
DeConto, R. M. & Pollard, D. Rapid Cenozoic glaciation of Antarctica induced by declining atmospheric CO2. Nature 421, 245–249 (2003).
DeConto, R. M. & Pollard, D. A coupled climate–ice sheet modeling approach to the Early Cenozoic history of the Antarctic ice sheet. Palaeogeogr. Palaeoclimatol. Palaeoecol. 198, 39–52 (2003).
Kennett, J.P. & Hodell, D.A. Evidence for relative climatic stability of Antarctica during the early Pliocene: a marine perspective. Geogr. Ann. Ser. Phys. Geogr. 75, 205–220 (1993).
Pujana, R. R., Panti, C., Cuitiño, J. I., Massini, J. L. G. & Mirabelli, S. L. A new megaflora (fossil woods and leaves) from the Miocene of southwestern Patagonia. Ameghiniana 52, 350–366 (2015).
Sandoval, C. A., Yabe, A., Nishida, H. & Hinojosa, L. F. Climate and vegetation of the Miocene of Tierra del Fuego: Filaret formation. Paleoceanogr. Paleoclimatol. 39, e2023PA004770 (2024).
Troedson, A. L. & Riding, J. B. Upper Oligocene to Lowermost Miocene Strata of King George Island, South Shetland Islands, Antarctica: stratigraphy, facies analysis, and implications for the glacial history of the Antarctic peninsula. J. Sediment. Res. 72, 510–523 (2002).
Troedson, A. L. & Smellie, J. L. The Polonez Cove Formation of King George Island, Antarctica: stratigraphy, facies and implications for mid-Cenozoic cryosphere development. Sedimentology 49, 277–301 (2002).
Lawver, L. A., Keller, R. A., Fisk, M. R. & Strelin, J. A. Bransfield Strait, Antarctic Peninsula Active Extension behind a Dead Arc. In Backarc Basins: Tectonics and Magmatism (ed. Taylor, B.) 315–342 (Springer US, Boston, MA, https://doi.org/10.1007/978-1-4615-1843-3_8. 1995).
Bastias, J. et al. The South Shetland Islands, Antarctica: Lithostratigraphy and geological map. Front. Earth Sci. 10, 2760 (2023).
Bastias, J. et al. The Byers Basin: Jurassic-Cretaceous tectonic and depositional evolution of the forearc deposits of the South Shetland Islands and its implications for the northern Antarctic Peninsula. Int. Geol. Rev. 62, 1467–1484 (2019).
Bastias-Silva, J. et al. A temporal control on the isotopic compositions of the Antarctic Peninsula arc. Commun. Earth Environ. 5, 1–11 (2024).
Haase, K. M., Beier, C., Fretzdorff, S., Smellie, J. L. & Garbe-Schönberg, D. Magmatic evolution of the South Shetland Islands, Antarctica, and implications for continental crust formation. Contrib. Mineral. Petrol. 163, 1103–1119 (2012).
Smellie, J. L., Pankhurst, R., Thomson, M. R. A. & Davies, R. E. S. The Geology of the South Shetland Islands: VI. Stratigraphy, Geochemistry and Evolution. vol. 87 (British Antarctic Survey, 1984).
Bastias, J. et al. A revised interpretation of the Chon Aike magmatic province: Active margin origin and implications for the opening of the Weddell Sea. Lithos 386–387, 106013 (2021).
Riley, T. R. et al. Palaeozoic – Early Mesozoic geological history of the Antarctic Peninsula and correlations with Patagonia: kinematic reconstructions of the proto-Pacific margin of Gondwana. Earth-Sci. Rev. 236, 104265 (2023).
Burton-Johnson, A. & Riley, T. R. Autochthonous v. accreted terrane development of continental margins: a revised in situ tectonic history of the Antarctic Peninsula. J. Geol. Soc. 172, 822–835 (2015).
Bastias, J. et al. The Gondwanan margin in West Antarctica: Insights from Late Triassic magmatism of the Antarctic Peninsula. Gondwana Res. 81, 1–20 (2020).
Burton-Johnson, A., Bastias, J. & Kraus, S. Breaking the ring of fire: how ridge collision, slab age, and convergence rate narrowed and terminated the Antarctic Continental Arc. Tectonics 42, e2022TC007634 (2023).
Hathway, B. & Lomas, S. A. The Jurassic–Lower Cretaceous Byers Group, South Shetland Islands, Antarctica: revised stratigraphy and regional correlations. Cretac. Res. 19, 43–67 (1998).
Hunt, R. J. & Poole, I. Paleogene West Antarctic climate and vegetation history in light of new data from King George Island. https://doi.org/10.1130/0-8137-2369-8.395 (2003).
Manfroi, J. et al. “Antarctic on fire”: Paleo-wildfire events associated with volcanic deposits in the Antarctic Peninsula during the Late Cretaceous. Front. Earth Sci. 11, 1045784 (2023).
Poole, I. & Cantrill, D. J. Cretaceous and Cenozoic vegetation of Antarctica integrating the fossil wood record. Geol. Soc. Lond. Spec. Publ. 258, 63–81 (2006).
Birkenmajer, K., Andrzej, G., Kreuzer, H. & Muller, P. K-Ar dating of the Melville Glaciation (Early Miocene) in West Antarctica. Bull Pol. Acad. Sci. Earth Sci. 33, 1523 (1985).
Warny, S., Kymes, C. M., Askin, R. A., Krajewski, K. P. & Bart, P. J. Remnants of Antarctic vegetation on King George Island during the early Miocene Melville Glaciation. Palynology 40, 66–82 (2016).
Smellie, J. L., McIntosh, W. C., Whittle, R., Troedson, A. & Hunt, R. J. A lithostratigraphical and chronological study of Oligocene-Miocene sequences on eastern King George Island, South Shetland Islands (Antarctica), and correlation of glacial episodes with global isotope events. Antarct. Sci. 33, 502–532 (2021).
Birkenmajer, K. Oligocene-Miocene glacio-marine sequences of King George Island (South Shetland Islands), Antarctica. Palaeontol. Pol. 49, 9–36 (1987).
Birkenmajer, K. Pliocene tillite-bearing succession of King George Island (South Shetland Islands, Antártica). (Springer, 1982).
Birkenmajer, K. Discovery Of Pliocene Glaciation On King-George-Island, South Shetland Islands (West Antarctica). Bull. Acad. Pol. Sci. -Ser. Sci. TERRE 27, 59–67 (1979).
Dingle, R. & Lavelle, M. Antarctic Peninsular cryosphere: Early Oligocene (c. 30 Ma) initiation and a revised glacial chronology. J. Geol. Soc. 155, 433–437 (1998).
Birkenmajer, K., Soliani Junior, E. & Kawashita, K. Early Miocene k-ar age of volcanic basement of the Melville glaciation deposits, King George island, west Antartica. Bull. Pol. Acad. Sci. Earth Sci. 36, 25–34 (1988).
Liebrand, D. et al. Antarctic ice sheet and oceanographic response to eccentricity forcing during the early Miocene. Clim. Past 7, 869–880 (2011).
Klages, J. P. et al. Ice sheet–free West Antarctica during peak early Oligocene glaciation. Science 385, 322–327 (2024).
Whittle, R. J., Quaglio, F., Griffiths, H. J., Linse, K. & Crame, J. A. The Early Miocene Cape Melville Formation fossil assemblage and the evolution of modern Antarctic marine communities. Naturwissenschaften 101, 47–59 (2014).
Birkenmajer, K. & Luczkowska, E. Foraminiferal evidence for a Lower Miocene age of glaciomarine and related strata, Moby Dick Group, King George Island (South Shetland Islands, Antarctica). (Springer, 1987).
Ellis, B. et al. Manual of Leaf Architecture (Cornell University Press, 2009).
Guillong, M., Quadt, A. von, Sakata, S., Peytcheva, I. & Bachmann, O. LA-ICP-MS Pb–U dating of young zircons from the Kos–Nisyros volcanic centre, SE Aegean arc. J. Anal. At. Spectrom. 29, 963–970 (2014).
Horstwood, M. S. A. et al. Community-derived standards for LA-ICP-MS U-(Th-)Pb geochronology – uncertainty propagation, age interpretation and data reporting. Geostand. Geoanalytical Res. 40, 311–332 (2016).
Sláma, J. et al. Plešovice zircon — A new natural reference material for U–Pb and Hf isotopic microanalysis. Chem. Geol. 249, 1–35 (2008).
Black, L. P. et al. TEMORA 1: a new zircon standard for Phanerozoic U–Pb geochronology. Chem. Geol. 200, 155–170 (2003).
Wiedenbeck, M. et al. Three natural zircon standards for U-Th-Pb, Lu-Hf, Trace Element and REE analyses. Geostand. Newsl. 19, 1–23 (1995).
Paton, C., Hellstrom, J., Paul, B., Woodhead, J. & Hergt, J. Iolite: Freeware for the visualisation and processing of mass spectrometric data. J. Anal. At. Spectrom. 26, 2508–2518 (2011).
Paton, C. et al. Improved laser ablation U-Pb zircon geochronology through robust downhole fractionation correction. Geochem. Geophys. Geosyst.s 11, 2618 (2010).
Petrus, J. A. & Kamber, B. S. VizualAge: a novel approach to laser ablation ICP-MS U-Pb geochronology data reduction. Geostand. Geoanal. Res. 36, 247–270 (2012).
Szymanowski, D. et al. Isotope-dilution anchoring of zircon reference materials for accurate Ti-in-zircon thermometry. Chem. Geol. 481, 146–154 (2018).
Kuprianova, L. Pollen morphology and phylogeny of Monochlamydeae. In 24 (Alexander Doweld, 1962).
Blume, C. L. Museum Botanicum Lugduno-Batavum. vol. 1 (EJ Brill, 1851).
Oersted, A. Beitrag til kindskab om engefamilies K. Dan. Videnenskabers Selsk. Skr 5, 351–354 (1873).
Tanai, T. Phytogeographic and phylogenetic history of the genus Nothofagus Bl.(Fagaceae) in the southern hemisphere. 21, 505–582 (1986).
Barton, C. Significance of the Tertiary fossil floras of King George Island, South Shetland Islands. (Springer, 1964).
Zastawniak, E., Wrona, R., Gazdzicki, A. & Birkenmajer, K. Plant remains from the top part of the point Hennequin group (Upper Oligocene), King George island (South Shetland islands, Antarctica). (Springer, 1985).
Torres, T. & Méon, H. Nothofagidites Erdtman ex Potoniédans le Paléogène de l’île Roi Georges. Antarctique. Geobios 31, 419–435 (1998).
Tosolini, A.-M. P., Cantrill, D. J., Korasidis, V. A. & Francis, J. E. Palaeocene high-latitude leaf flora of Antarctica Part 2: Tooth-margined angiosperms. Rev. Palaeobot. Palynol. 314, 104895 (2023).
Aguirre, A. & Romero, E. Arquitectura foliar de las especies Australianas y Neocelandesas de Nothofagus (Fagaceae). Bol Soc Argent Bot 20, 227–240 (1982).
Hill, R. S. Terrestrial Palaeobiogeography. In Palaeobiology II 454–459 (John Wiley & Sons, Ltd, https://doi.org/10.1002/9780470999295.ch111 2001).
Bastias, J. et al. Cretaceous magmatism in the Antarctic Peninsula and its tectonic implications. J. Geol. Soc 180, jgs2022–jgs2067 (2022).
Xing, H. et al. A Precambrian basement beneath the King George Island (Antarctica Peninsula) revealed by zircon xenocrystals from Eocene to Miocene volcanic rocks. Lithos 494–495, 107899 (2025).
Grunow, A. M., Kent, D. V. & Dalziel, I. W. D. Mesozoic evolution of West Antarctica and the Weddell Sea Basin: new paleomagnetic constraints. Earth Planet. Sci. Lett. 86, 16–26 (1987).
Askin, R. A. Late Cretaceous–Early Tertiary Antarctic Outcrop Evidence for Past Vegetation and Climates. In The Antarctic Paleoenvironment: A Perspective on Global Change: Part One 61–74 (American Geophysical Union (AGU), https://doi.org/10.1029/AR056p0061 1992).
Francis, J. E. Fossil Wood from Eocene High Latitude Forests: Mcmurdo Sound, Antarctica. In Paleobiology and Paleoenvironments of Eocene Rocks: McMurdo Sound, East Antarctica 253–260 (American Geophysical Union (AGU), https://doi.org/10.1029/AR076p0253 2000).
Leppe, M. et al. Evolution of the Austral-Antarctic flora during the Cretaceous: New insights from a paleobiogeographic perspective. Rev. Chil. Hist. Nat. 85, 369–392 (2012).
Manríquez, L. M. E., Lavina, E. L. C., Fernández, R. A., Trevisan, C. & Leppe, M. A. Campanian-Maastrichtian and Eocene stratigraphic architecture, facies analysis, and paleoenvironmental evolution of the northern Magallanes Basin (Chilean Patagonia). J. South Am. Earth Sci. 93, 102–118 (2019).
Pole, M., Hill, B. & Harwood, D. Eocene Plant Macrofossils from Erratics, Mcmurdo Sound, Antarctica. In Paleobiology and Paleoenvironments of Eocene Rocks: McMurdo Sound, East Antarctica 243–251 (American Geophysical Union (AGU), https://doi.org/10.1029/AR076p0243. 2000).
Truswell, E. M. & Macphail, M. K. Polar forests on the edge of extinction: what does the fossil spore and pollen evidence from East Antarctica say? Aust. Syst. Bot. 22, 57–106 (2009).
Prebble, J. G., Raine, J. I., Barrett, P. J. & Hannah, M. J. Vegetation and climate from two Oligocene glacioeustatic sedimentary cycles (31 and 24 Ma) cored by the Cape Roberts Project, Victoria Land Basin, Antarctica. Palaeogeogr. Palaeoclimatol. Palaeoecol. 231, 41–57 (2006).
Barrett, P. Cenozoic climate and sea level history from glacimarine strata off the Victoria Land coast, Cape Roberts Project, Antarctica. Glacial Sediment. Process. Prod. 259–287 (2007).
Raine, J. Terrestrial palynomorphs from Cape Roberts Project drillhole CRP-1, Ross Sea, Antarctica. Terra Antartica 5, 539–548 (1998).
Askin, R. A. Spores and Pollen from the Mcmurdo Sound Erratics, Antarctica. In Paleobiology and Paleoenvironments of Eocene Rocks: McMurdo Sound, East Antarctica 161–181 (American Geophysical Union (AGU), https://doi.org/10.1029/AR076p0161. 2000).
Vento, B., Agrain, F., Puebla, G. & Pinzon, D. Phylogenetic relationships in genus Nothofagus: the role of Antarctic fossil leaves. Acta Palaeontol. Pol. 68, 1029 (2023).
Paul, H. A., Zachos, J. C., Flower, B. P. & Tripati, A. Orbitally induced climate and geochemical variability across the Oligocene/Miocene boundary. Paleoceanography 15, 471–485 (2000).
Westerhold, T. et al. An astronomically dated record of Earth’s climate and its predictability over the last 66 million years. Science 369, 1383–1387 (2020).
De Schepper, S., Gibbard, P. L., Salzmann, U. & Ehlers, J. A global synthesis of the marine and terrestrial evidence for glaciation during the Pliocene Epoch. Earth-Sci. Rev. 135, 83–102 (2014).
Ashworth, A. et al. The Neogene biota of the Transantarctic Mountains. ANDRILL Relat. Publ. Affil. (2007).
Warny, S. et al. Palynomorphs from a sediment core reveal a sudden remarkably warm Antarctica during the middle Miocene. Geology 37, 955–958 (2009).
Shevenell, A. E., Kennett, J. P. & Lea, D. W. Middle Miocene ice sheet dynamics, deep-sea temperatures, and carbon cycling: a Southern Ocean perspective. Geochem. Geophys. Geosyst. 9, 1736 (2008).
Segovia, R. A., Pérez, M. F. & Hinojosa, L. F. Genetic evidence for glacial refugia of the temperate tree Eucryphia cordifolia (Cunoniaceae) in southern South America. Am. J. Bot. 99, 121–129 (2012).
Lira-Noriega, A. & Manthey, J. D. Relationship of genetic diversity and niche centrality: a survey and analysis. Evolution 68, 1082–1093 (2014).
Veblen, T. T., Hill, R. S. & Read, J. The Ecology and Biogeography of Nothofagus Forests. (Yale University Press, 1996).
Hill, R. S., Harwood, D. M. & Webb, P.-N. Nothofagus beardmorensis (Nothofagaceae), a new species based on leaves from the Pliocene Sirius Group, Transantarctic Mountains, Antarctica. Rev. Palaeobot. Palynol. 94, 11–24 (1996).
Thompson, R. S. & Fleming, R. F. Middle Pliocene vegetation: reconstructions, paleoclimatic inferences, and boundary conditions for climate modeling. Mar. Micropaleontol. 27, 27–49 (1996).
Poblete, F. et al. Paleomagnetism and tectonics of the South Shetland Islands and the northern Antarctic Peninsula. Earth Planet. Sci. Lett. 302, 299–313 (2011).
Birkenmajer, K., Soliani, E. & Kawashita, K. Geochronology of Tertiary Glaciations on King George Island, West Antarctica. (Springer, 1989).
Tosolini, A.-M. P., Cantrill, D. J. & Francis, J. E. Paleocene high-latitude leaf flora of Antarctica Part 1: entire-margined angiosperms. Rev. Palaeobot. Palynol. 285, 104317 (2021).
Tosolini, A.-M. P., Cantrill, D. J. & Francis, J. E. Paleocene flora from Seymour Island, Antarctica: revision of Dusén’s angiosperm taxa. Alcheringa Australas. J. Palaeontol. 37, 366–391 (2013).
Gandolfo, M. A., Marenssi, S. A. & Santillana, S. N. Flora y paleoclima de la Formación La Meseta (Eoceno medio), isla Marambio (Seymour), Antártida. Paleógeno América Sur Península Antártica 5, 155–162 (1998).
Torres, T. & Lemoigne, Y. Maderas fosiles Terciarias de la formacion caleta Arctowski, Isla Rey Jorge, Antartica. Ser. Científica - Inst. Antártico Chil. 69–107 (1988).
Dingle, R. V., Marenssi, S. A. & Lavelle, M. High latitude Eocene climate deterioration: evidence from the northern Antarctic Peninsula. J. South Am. Earth Sci. 11, 571–579 (1998).
Gao, L. et al. Plate Rotation of the Northern Antarctic Peninsula Since the Late Cretaceous: Implications for the Tectonic Evolution of the Scotia Sea Region. J. Geophys. Res. Solid Earth 128, e2022JB026110 (2023).
Barker, P. F. Scotia Sea regional tectonic evolution: implications for mantle flow and palaeocirculation. Earth-Sci. Rev. 55, 1–39 (2001).
Barker, P. F. & Thomas, E. Origin, signature and palaeoclimatic influence of the Antarctic Circumpolar Current. Earth-Sci. Rev. 66, 143–162 (2004).
Kennett, J. P. Cenozoic evolution of Antarctic glaciation, the circum-Antarctic Ocean, and their impact on global paleoceanography. J. Geophys. Res 82, 3843–3860 (1977). 1896-1977.
Herzschuh, U. Reliability of pollen ratios for environmental reconstructions on the Tibetan Plateau. J. Biogeogr. 34, 1265–1273 (2007).
Knapp, M. et al. Relaxed molecular clock provides evidence for long-distance dispersal of nothofagus (Southern Beech). PLOS Biol 3, e14 (2005).
Sanmartíin, I. & Ronquist, F. Southern hemisphere biogeography inferred by event-based models: plant versus animal patterns. Syst. Biol. 53, 216–243 (2004).
Craw, R. C., Grehan, J. R. & Heads, M. J. Panbiogeography: Tracking the History of Life. (Oxford University Press, 1999).
Linder, H. P. & Crisp, M. D. Nothofagus and Pacific Biogeography. Cladistics 11, 5–32 (1995).
Livermore, R., Nankivell, A., Eagles, G. & Morris, P. Paleogene opening of Drake Passage. Earth Planet. Sci. Lett. 236, 459–470 (2005).
Vento, B., Agrain, F. A., Puebla, G. G. & Pinzón Aceros, D. A. Phylogenetic relationships in Nothofagus: the role of Antarctic fossil leaves. (Taylor and Francis, 2023).
Vento, B., Agrain, F. & Puebla, G. Ancient Antarctica: the early evolutionary history of Nothofagus. Hist. Biol. 36, 136–146 (2024).
Tapia, M. J. et al. Response of the Patagonian floras to climatic cooling during Oligocene−Miocene transition and the expansion of Antarctic glaciation. Evol. Earth 1, 100015 (2023).
Poole, I. & Cantrill, D. The Arboreal Component of The Neogene Forest Bed, West Point Island, Falkland Islands. https://doi.org/10.1163/22941932-90001653. (2007)
Acosta, M. C., Mathiasen, P. & Premoli, A. C. Retracing the evolutionary history of Nothofagus in its geo-climatic context: new developments in the emerging field of phylogeology. Geobiology 12, 497–510 (2014).
Pujana, R. R., Fernández, D. A., Panti, C. & Caviglia, N. The micro- and megafossil record of Nothofagaceae from South America. Bot. J. Linn. Soc. 196, 1–20 (2021).
Liebrand, D. et al. Evolution of the early Antarctic ice ages. Proc. Natl. Acad. Sci. 114, 3867–3872 (2017).
DeConto, R. M. & Pollard, D. Contribution of Antarctica to past and future sea-level rise. Nature 531, 591–597 (2016).
Dolan, A. M. et al. Modelling the enigmatic Late Pliocene Glacial Event — Marine Isotope Stage M2. Glob. Planet. Change 128, 47–60 (2015).
Huang, X. et al. How changing the height of the Antarctic ice sheet affects global climate: a mid-Pliocene case study. Clim. Past 19, 731–745 (2023).
Naish, T. R. et al. Orbitally induced oscillations in the East Antarctic ice sheet at the Oligocene/Miocene boundary. Nature 413, 719–723 (2001).
Truswell, E. M. Cretaceous and Tertiary vegetation of Antarctica: a palynological perspective. In Antarctic paleobiology: its role in the reconstruction of Gondwana 71–88 (Springer, 1990).
Acknowledgements
JBS was funded by the Swiss National Science Foundation (project P5R5PN_217947) and the project RT-01-22 funded by the Chilean Antarctic Institute (INACH). LG was funded by the Natural Science Foundation of China (NSFC) (42322607 and 42076223), ML and CrTr were supported by EVOTEM Millennium Nucleus (ANID MILENIO–NCN2023_025). ClTa was supported by 2023FEQUIP-RB-03. Fieldwork on the Antarctic Peninsula was supported by the Chilean Antarctic Institute. Especial thanks to the team supporting in the field Georgette Mell, Billy Wallace and Ignacio Reyes. The fossils are housed in the Palaeontological Collection of Antarctica and Patagonia at the Chilean Antarctic Institute (INACH). The authors are grateful to three anonymous reviewers for providing constructive criticism that improved the paper.
Author information
Authors and Affiliations
Contributions
J.B.-S.: Conceptualisation, data collection, fieldwork, methodology, data curation, formal analysis, writing—original draft, writing—review and editing. M.L.: Methodology, data curation, formal analysis, writing—original draft, writing—review and editing. L.M.: Data curation, writing—original draft. C.T.: Data curation, writing—original draft. B.R.S.F.: Writing—original draft, writing—review and editing. M.M.: Fieldwork, writing—original draft. G.W.: Writing—original draft, writing—review and editing. L.T.: Data collection, writing—original draft, writing—review and editing. C.C.-M.: Writing—original draft, writing—review and editing. L.G.: Writing—original draft, writing—review and editing. D.S.: Writing—original draft, writing—review and editing. H.M.: Data curation. C.S.: Editing. F.H.: Editing. C.T.: Editing, fieldwork.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Earth and Environment thanks Xin Qian and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editors: Jun Shen, Carolina Ortiz Guerrero, and Mengjie Wang. [A peer review file is available].
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Bastias-Silva, J., Leppe, M., Manriquez, L. et al. Neogene plant macrofossils from West Antarctica reveal persistence of Nothofagaceae forests into the early Miocene. Commun Earth Environ 6, 965 (2025). https://doi.org/10.1038/s43247-025-02921-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43247-025-02921-x