Abstract
Soil extracellular enzymes are critical drivers of carbon (C) and nutrient cycling in terrestrial ecosystems. However, the effects of phosphorus (P) additions on soil enzyme activities and stoichiometries, particularly under varying nitrogen (N) addition regimes worldwide, are not well comprehended. Here, we conducted a meta-analysis based on 155 field studies across various ecosystems (forests, croplands and grasslands), which shows that P enrichment conditions enhances C-acquiring enzymes but has no effect on the enzymatic C:N ratio. P addition alone reduces P-acquiring enzymes by 14% without affecting N-acquiring enzymes. In contrast, P combined with N addition boosts N-acquiring enzymes by 21% while leaving P-acquiring enzymes unchanged. Notably, the combined effect of P and N addition on microbial C limitation (assessed via vector length) and enzymatic stoichiometries is less pronounced than that of P addition alone. Key drivers of these responses include mean annual precipitation, soil microbial biomass, and its stoichiometries. These results suggest that N addition mitigates the stoichiometric imbalance and microbial C limitation induced by P addition, potentially promoting soil organic C accumulation. Our findings emphasize the critical need to account for such interactive effects in models predicting soil biogeochemical cycles under future changes in global exogenous N and P inputs.
Similar content being viewed by others
Introduction
Phosphorus (P) is a key limiting nutrient in terrestrial ecosystems for microbial and plant growth1. Unlike nitrogen (N), which is highly mobile and deposited via multiple pathways, P has fewer sources and lower mobility2,3,4. Consequently, rising N deposition often exacerbates P limitation, influencing soil microbial activities and biogeochemical cycles5,6. Soil extracellular enzymes, primarily of microbial origin, regulate elemental cycling by mediating soil carbon (C) dynamics and nutrient availability7,8,9. These processes can either mitigate or amplify climate change impacts on terrestrial ecosystems. Despite their recognized importance in mediating nutrient cycling, the impacts of soil P availability on soil enzyme activities are not well comprehended.
The enzyme C:N:P stoichiometry provides understanding into how microbes allocate resources for acquiring C, N, and P10, and is potentially linked to C use efficiency under resource constraints11. However, this approach has faced growing critical scrutiny in recent methodological evaluations12,13,14,15. A key concern is the overlapping ecological functions of target enzymes, which undermines the reliability of distinguishing microbial resource limitations; specifically, the enzymatic C:N ratio fails to clearly indicate microbial C or N limitation because both C- and N-acquiring enzymes can contribute to C acquisition15,16. Despite these limitations, the approach remains a valuable heuristic framework for synthesizing patterns across ecosystems, proving useful in meta-analyses or broad-scale investigations7,11,17,18.
More recently, building on this methodological framework, a comprehensive study revealed that soil microorganisms are more limited by P than by N at the global scale19. However, the patterns of microbial C limitation under global environment changes remain poorly understood. Ecosystems experiencing strong microbial C and/or nutrient limitations show reduced capacity to maintain critical functions, particularly soil C stabilization and pool size, under nutrient enrichment conditions20,21. Therefore, exploring the effects of P enrichment on the enzymatic stoichiometries could significantly improve our understand of soil C dynamics.
Resource allocation theory predicts that nutrient additions reduce the investment of nutrient-acquiring enzymes22, thus increasing enzymatic C: nutrient ratios. Yet, experimental studies have reported inconsistent results regarding soil enzyme stoichiometry. For instance, P addition has shown positive23, negative24, or neutral effects25 on enzymatic C:N ratios, and both positive26 and negative effects27 on enzymatic C:P ratios. These discrepancies highlight the need for a comprehensive meta-analysis. Unfortunately, to date, only one synthesis, limited to 21 references and focused solely on enzymatic C:P ratio, suggested greater microbial P demand in P-deficient tropical soils28. Consequently, more evidence is urgently needed to clearly understand the effects of P addition on the enzymatic stoichiometries.
The enzymatic stoichiometries responses to P addition are likely influenced by experimental conditions, ecosystem types, and the climates of the experimental sites. The P addition effects on enzymatic stoichiometries could be more significant with the increased experimental durations and P addition rates given P’s persistence in soil6. Ecosystem traits, including vegetation type and nutrient strategies, could also modulate outcomes29,30. Additionally, the effects of P addition may be regulated by microbial communities, as changes in soil available nutrients may alter the structure of soil microbial communities, thereby affecting enzyme activities and their stoichiometry31,32,33. Furthermore, such as temperature and soil water availability could affect microbial physiology7,34, so the responses of the enzymatic stoichiometries to P addition may also regulate by climatic factors.
The responses of enzymatic stoichiometries to P addition may also mediate by soil N availability. Elevated N addition can intensify microbial P limitation35,36, while P addition might alleviate, potentially shifting constraints to C or N37. Microbes may adjust enzyme production and associated enzyme stoichiometry to alleviate these limitations accordingly38. Studies suggest P addition alone has weaker effects on C-acquiring enzymes and higher enzymatic C:P ratio compared to combined P and N addition23,25,39. So far, several meta-analyses have explored the combined effects of P and N addition on soil processes, focusing on belowground C cycling processes39,40, microbial communities and respiration5,41, and changes in soil P fractions42. However, whether N addition could globally regulate the effect of P addition on the enzymatic stoichiometries remains unaddressed.
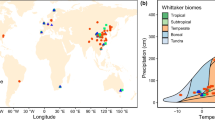
Here, we compiled a global database of 629 paired observations from 155 field studies across diverse ecosystems to assess how P addition, alone or combined with N addition, affects soil enzyme activities and their stoichiometry (Fig. 1). The following questions were addressed: (1) Whether enzymatic stoichiometries responses to P addition vary with soil N availability, and (2) How local climatic, ecosystem type, P addition duration and magnitude, and microbial community composition regulate these effects. We hypothesized that: (1) Enzyme activities and their stoichiometry differ between P addition alone and combined with N, due to the divergent responses of soil microbial communities to P additions under different levels of N addition5,43,44; and (2) Abiotic (e.g., climate) and biotic (e.g., microbial properties) factors co-regulate P addition effects on enzymatic stoichiometries. Our meta-analysis provides a comprehensive synthesis of the effects of P addition, both alone and in combination with N addition, to consolidate disparate findings and advance our ability to model and predict soil biogeochemical cycles.
Map lines delineate study areas and do not necessarily depict accepted national boundaries.
Results
The overall effects of P addition on enzymatic stoichiometries
We found that the lnRRs of C-, N-, and P-acquiring enzyme activities to P addition were regulated by N addition (all p < 0.05. Supplementary Table 1). Specifically, P addition and combined with N addition, increased C-acquiring enzyme activities by 12% and 19%, respectively (Fig. 2a). Meanwhile, P addition alone decreased P-acquiring enzyme activities by 14%, while did not affect N-acquiring enzyme activities (Fig. 2b, c). In contrast, combined P with N addition increased N-acquiring enzyme activities by 21%, while it did not affect P-acquiring enzyme activities (Fig. 2b, c). Further, P addition alone increased the vector length by 8%, but did not affect it combined with N addition (Fig. 2d). P addition, either alone or combined with N addition, increased the enzymatic C:P and N:P ratios, while neither affected the enzymatic C:N ratio (Fig. 2e–g).
a Effect size for C-acquiring enzyme activities; b Effect size for N-acquiring enzyme activities; c Effect size for P-acquiring enzyme activities; d Effect size for vector length; e Effect size for enzyme C:N ratio; f Effect size for enzyme C:P ratio; g Effect size for enzyme N:P ratio. Values are expressed as means ± 95% confidence intervals (CIs). The scatters indicate the effect value of the original data. If 95% CIs did not overlap zero, the nutrient addition effect was considered significant. Ns indicates nutrient addition had no significant effect on enzyme activities, enzymatic stoichiometries, and vector length.
Divergent responses of enzymatic stoichiometries across ecosystems
The lnRRs of enzyme activities and their stoichiometry to P addition were affected by ecosystem types (Supplementary Table 2). More specifically, in forests, P addition, either alone or combined with N addition, decreased P-acquiring enzyme activities while increased C-acquiring enzyme activities (Fig. 3), enzymatic C:P and N:P ratios, whereas did not affect the enzymatic C:N ratio (Fig. 4). More importantly, P addition alone resulted in a 7% increase in vector length (Fig. 4). In croplands, P addition alone increased C-acquiring enzyme activities by 31%, vector length by 21%, enzymatic C:N by 38% and C:P ratio by 37%, whereas P addition combined with N addition had no effect on enzyme activities and their stoichiometry (Figs. 3 and 4). In grasslands, P addition, either alone or combined with N addition, had no effect on P-acquiring enzyme activities, enzymatic C:N ratio and vector length, whereas increased the enzymatic N:P ratio. P addition alone increased the enzymatic C:P ratio by 38% (Fig. 4), whereas combined P with N addition increased C- and N-acquiring enzyme activities by 32% and 22%, respectively (Fig. 3).
Values are expressed as means ± 95% confidence intervals (CIs). The scatters indicate the effect value of the original data. If 95% CIs did not overlap zero, the nutrient addition effect was considered significant. Ns indicates nutrient addition had no significant effect on enzyme activities.
Values are expressed as means ± 95% confidence intervals (CIs). The scatters indicate the effect value of the original data. If 95% CIs did not overlap zero, the nutrient addition effect was considered significant. Ns indicates nutrient addition had no significant effect on enzymatic stoichiometries and vector length. EE C:N: enzyme C:N ratio; EE C:P: enzyme C:P ratio; EE N:P: enzyme N:P ratio.
Multiple factors affecting the enzymatic stoichiometries responses to P addition
The lnRRs of enzyme activities and their stoichiometry to P addition were controlled by mean annual precipitation (MAP) rather than mean annual temperature (MAT), experimental durations and magnitudes (Supplementary Tables 2 and 3). With increasing MAP, the lnRR of P-acquiring enzyme activities to P addition changed from positive to negative (p < 0.001. Fig. 5a), while the enzymatic C:P ratio showed an opposite pattern (p = 0.018. Fig. 5b).
a P-acquiring enzyme activities; b Enzyme C:P ratio. Open circles are observations, with sizes representing weights. Gray shadow around the fitted line represents the 95% confidence intervals. Dashed lines represent the average lnRR of the tested enzyme activities and enzymatic stoichiometries.
A linear mixed-effects model was employed to examine the relationships between changes in soil enzymatic C:N:P stoichiometry and soil available nutrients and their ratios, as well as microbial properties. The lnRR of enzymatic C:N ratio was negatively associated with the lnRR of microbial biomass N (MBN) but positively associated with the lnRRs of microbial biomass C (MBC) and MBC/N ratio (all p < 0.05. Fig. 6a). The lnRRs of enzymatic C:P ratio and C-acquiring enzyme activities showed similar relationships with these microbial properties as enzymatic C:N ratio, both of which were negatively associated with the lnRR of MBN but positively associated with the lnRR of MBC/N ratio (all p < 0.05. Fig. 6b, d). Additionally, the lnRR of enzymatic N:P ratio was negatively associated with the lnRRs of MBC and microbial biomass P (MBP) (Fig. 6c), and the lnRR of N-acquiring enzyme activities was negatively associated with the lnRR of MBC/N ratio (Fig. 6e). Finally, the lnRR of P-acquiring enzyme activities was not affected by any microbial properties. In contrast, it was positively affected by soil available N (AN), the ratio of soil dissolved organic C to available P (DOC/AP), and the ratio of soil AN to AP (AN/AP), while negatively affected by AP (all p < 0.05. Fig. 6f).
a Enzyme C:N ratio; b Enzyme C:P ratio; c Enzyme N:P ratio; d C-acquiring enzyme activities; e N-acquiring enzyme activities; f P-acquiring enzyme activities. The relationship was considered significant when the 95% confidence intervals did not overlap zero. Values in parentheses represent the number of observations. Solid and empty circles indicate significant and non-significant effects, respectively. DOC dissolved organic carbon, AN soil available nitrogen, AP soil available phosphorus, MBC microbial biomass carbon, MBN microbial biomass nitrogen, MBP microbial biomass phosphorus.
Discussion
Given the importance of soil extracellular enzymes in mediating C and nutrient cycling for terrestrial ecosystems, we conducted a global synthesis to explore the impacts of P addition, either alone or combined with N addition, on soil enzyme activities and their stoichiometry. Moving beyond studies that focus on single nutrient additions, our meta-analysis demonstrates that (1) P enrichment conditions significantly increased the enzymatic C:P and N:P ratios, while maintaining a stable enzymatic C:N ratio globally, (2) the responses of soil enzyme activities and their stoichiometry to P addition were primarily driven by MAP and were associated with microbial biomass and their stoichiometry, rather than by experimental durations, magnitudes, or MAT, and (3) N addition mitigates the stoichiometric imbalance and microbial C limitation induced by P addition, which was beneficial for soil organic C (SOC) accumulation. These findings provide compelling evidence that the impacts of anthropogenic P additions on soil enzyme activities and their stoichiometry could be modulated by soil N availability on a global scale. This offers a scientific basis for the targeted regulation of soil C-N-P cycling and has the potential to enhance SOC sequestration.
Responses of soil enzyme stoichiometries to P addition
P addition alone was found to significantly decrease P-acquiring enzyme activities (Fig. 2c). While resource allocation theory predicts coordinated up-regulation of both C- and N-acquiring enzymes after P addition22, we observed a selective response limited to C-acquisition pathways (Fig. 2b). This finding aligns with previous reports of decoupled nutrient acquisition responses to P enrichment45,46,47. Such a phenomenon may result from microbial down-regulation of N-rich P-acquiring enzymes production following P addition, which alleviates cellular N limitation and subsequently reduces investment in N-acquiring enzymes48. However, N-acquiring enzyme activities were significantly enhanced under combined P and N addition, rather than P addition alone (Fig. 2b). We attribute this to a shift in plant-microbe competition for N. As N is a primary limiting nutrient in terrestrial ecosystems1, its deficiency can inhibit the positive effects of P additions on the growth of both plants (e.g., enhanced aboveground production) and microbes39,49. Relative to P addition alone, combined P with N addition elicited a stronger growth response in vegetation, as increased N availability better satisfies plant nutritional requirements39. This intensified plant competition for N likely exacerbates N scarcity for soil microorganisms. In response, microbes strategically up-regulate N-acquiring enzyme production to mobilize organic N from the soil, thereby compensating for the increased competition from plants. Furthermore, different patterns of soil enzyme activity responses to P addition, either alone or combined with N addition, may also imply a shift in microbial resource acquisition strategies, which is related to changes in microbial physiology and community composition, such as in the fungi/ bacteria ratio7. Our observation of a declining MBC/P ratio under P enrichment conditions (Supplementary Fig. 1) supports this interpretation, suggesting a shift toward bacterial-dominated communities, as bacteria exhibit a lower C/P ratio relative to fungi6,30,50.
Evidence suggests that P addition, either alone or combined with N addition, increased the enzymatic C:P and N:P ratios but had no effect on the enzymatic C:N ratio (Fig. 2e–g), which reflects the adaptive strategy of soil microbes in response to altered nutrient availability. A plausible mechanism underlying this response involves P-induced improvements in plant litter quality (e.g., lower C:N and C:P) and quantity1,51. The decomposition of such nutrient-rich litter requires a relatively lower microbial investment in N- and P-acquiring enzymes compared to C-acquiring enzymes30, thereby elevating the enzymatic C:P and N:P ratios. This also explains why the microbial C limitation, reflected by vector length, of combined P with N addition was lower than that of P addition alone (Fig. 2d). Another plausible mechanism is that P addition directly alleviates soil P limitation, as evidenced by the significant decreased soil AN/AP ratio (Supplementary Fig. 1), triggering a strategic down-regulation of P-acquiring enzymes to save metabolic costs10,22. Finally, soil pH is an important factor that influences soil enzyme activities and their stoichiometry18,52. In this study, soil pH was significantly decreased under combined P with N addition, rather than P addition alone (Supplementary Fig. 1). Changes in soil pH can directly alter enzyme activities by shifting conditions away from their optimal pH range53, and can indirectly influence microbial community composition54, ultimately increasing the enzymatic C:P and N:P ratios.
Major factors regulating soil enzyme stoichiometries responses to P addition
Variability in plant physiological traits, vegetation longevity, nutrient uptake efficiency, and litter characteristics (including quality and quantity) can potentially lead to variation in enzymatic stoichiometries across ecosystems17. Our observation of P effects on the enzymatic C:N:P vary across different ecosystem types supports this interpretation (Figs. 3, 4 and Supplementary Table 2). This is inconsistent with the findings of terrestrial ecosystem stoichiometries to elevated CO255, drought56, and N addition57. The soil N:P ratio of forests (19.0, average mean) was higher than that of croplands (4.4) and grasslands (10.7) globally58, indicating that forest soils are more limited by P. Therefore, P-acquiring enzyme activities decreased, and the enzymatic C:P and N:P ratios increased in forests after P addition, either alone or combined with N addition (Fig. 3). In contrast, croplands exhibited marked increases in C-acquiring enzyme activities and elevated the enzymatic C:N and C:P ratios following P addition alone, reflecting a relative microbial C limitation. This is because agricultural management has resulted in lower C inputs (e.g., lower litter return) to croplands compared to forests and grasslands59. Meanwhile, vector length revealed that grasslands had less microbial C limitation than croplands or forests after P addition. This pattern likely stems from accelerated litter decomposition rates in grassland ecosystems, which promote more efficient C and nutrient cycling11,60.
MAP was a more critical climate driver of extracellular enzyme activities and their stoichiometry than MAT (Fig. 5 and Supplementary Table 2). Under elevated MAP regimes, enhanced soil moisture facilitates greater P mobility by improving diffusion of applied P fertilizers, as well as desorption and dissolution of soil inorganic P4,45,61. Concurrently, elevated MAP regimes likely promote plant growth and litter mass56, thereby improving the return of nutrients (e.g., P) to soil. All these changes may inhibit microbial investment in P-acquiring enzyme production, consequently increasing the enzymatic C:P ratio (Fig. 5). Our results, together with a previous study4, strongly suggest a positive interaction between P fertilization and water availability.
Importantly, our results showed that the responses of enzymatic stoichiometries were related to the responses of microbial biomass and its stoichiometries (Fig. 6), which was in line with our second hypothesis. These findings also align with established literature demonstrating that alterations in soil P availability can fundamentally reshape microbial community structure and metabolic function31,32,33, thereby affecting enzyme activities and their stoichiometry17,43. Distinct microbial taxa (e.g., bacteria vs fungi) exhibit specialized enzymatic production capabilities, generating complementary substrate degradation networks that drive microbial community co-metabolism7. Crucially, given the observed tight coupling between enzymatic stoichiometries and microbial properties, we propose that shifts in community composition constitute the primary mechanistic link connecting soil nutrient availability to extracellular enzymatic stoichiometries.
N addition alleviates P-induced imbalances in soil enzyme stoichiometry
The response of vector length to P addition alone was significantly increased by 8% (Fig. 1). Moreover, the log-transformed enzymatic C:N:P ratio under P addition alone (0.84:0.89:1) exhibited a deviation from the global scale (1:1:1)8, which is consistent with a prior report of P-induced stoichiometric disruption in terrestrial ecosystems6. Conversely, the response of vector length was insignificant, and the responses of enzymatic C:N and C:P ratios to combined P with N addition were relatively lower than those to P addition alone (Fig. 2d–f). Meanwhile, the enzymatic ratio restored near-equilibrium ratios (0.89:1.02:1) under combined P with N addition. These findings indicate that N addition could mitigate P-induced stoichiometric imbalances globally. The primary reason lies in the fact that soil nutrients (mainly N and P) and stoichiometric becomes more balanced following combined P with N addition62,63. In contrast, single nutrient additions generally induce soil nutrient and stoichiometric imbalances37,38, which potentially mismatches soil resource supply with the intrinsic demands of soil microbial biomass10,64,65. This leads microbes to disproportionately invest in enzymes to acquire the limiting nutrient, thereby disrupting enzyme stoichiometry. In conclusion, our results underscore the critical role of coupled N and P inputs in maintaining biogeochemical equilibrium in terrestrial ecosystems.
Implications for SOC accumulation
In this study, SOC and MBC were strongly linked to soil total N and MBN, respectively (Supplementary Fig. 2a, b), suggesting a coupling of C and N even in P-rich environments. The response of SOC was also positively linked to the responses of soil enzymes following P addition (Supplementary Fig. 2c, d), indicating increased soil enzyme activities in C cycling40,66. However, P addition alone did not alter SOC, while combined P with N addition significantly promoted it accumulation (Supplementary Fig. 1). These three potential mechanisms may explain why P addition exerts a greater impact on SOC with increasing soil N availability. Firstly, the contribution of plant C inputs to soils is greater under combined P with N addition than under P addition alone because of sufficient N for plant growth39. Secondly, a transition in microbial community occurs, with bacteria dominating fungi under P enrichment6, which may reduce the utilization of recalcitrant C compounds and accelerate SOC decomposition67. However, N-induced soil acidification could inhibit microbial activity37, thereby counteracting SOC decomposition. Finally, aggravated microbial C limitation may trigger a metabolic shift from biomass production to maintenance respiration and/or metabolism, concomitant with increased extracellular enzyme synthesis during decomposition64. This physiological response may accelerate SOC mineralization, potentially reduce terrestrial C storage capacity, and provide positive feedback to climate warming through enhanced CO2 efflux.
Methodological considerations and limitations
A recognized caveat of our study is its reliance on enzymatic stoichiometry, a method whose underlying assumptions are currently under active debate in the field12,13,14. Studies have noted that the disconnect between enzyme activity and microbial growth13, along with factors like enzyme multifunctionality, means enzyme ratios may not accurately reflect in-situ microbial nutrient limitation15,16. Furthermore, the methodology of using log-transformed enzymatic ratios and their global 1:1:1 benchmark should be cautiously applied in future research, due to the statistical properties of log-transformed ratios and the multifunctionality of soil enzymes68,69. Thus, our observation that N addition alleviates P-induced imbalances in enzyme stoichiometry should be interpreted as a shift in the potential nutrient demand and overall biogeochemical cycling, not as definitive proof of changed microbial nutrient limitation. We strongly advise that future research pair enzymatic assays with direct measures of microbial growth and nutrient limitation to validate these patterns. Another consideration in interpreting our results is that the “P addition” treatments in the underlying studies were almost conducted using calcium (Ca)-based phosphate fertilizers (e.g., superphosphate). Consequently, the observed effects are a composite of both P and Ca inputs. Ca is known to influence soil pH, microbial community structure, and soil organic matter stabilization through its role in aggregation. Therefore, while the primary intended effect is an increase in P availability, the concomitant increase in Ca and its associated effects may have contributed to the patterns we observed70. Disentangling the specific effects of P from those of Ca in field-scale nutrient addition studies remains a challenge.
Methods
Data selection
Relevant published studies that assessed how P addition affected enzyme activities and their stoichiometry were selected by using Web of Science and the China National Knowledge Infrastructure from 1995 to 2024. Our searching keywords were: (phosphorus addition OR phosphorus fertilization OR phosphorus enrichment) AND (enzyme OR “enzymatic stoichiometry” OR “C-acquiring enzyme” OR “N-acquiring enzyme” OR “P-acquiring enzyme” OR “C-, N-, and P-acquiring enzyme”) AND (terrestrial OR soil OR land). For comparison purposes, our study collected most of the extensively measured activities of soil C, N, and P-acquiring enzymes (Supplementary Table 4).
Our database only included field studies that met these four specific criteria: (1) the studies must include both control and P addition alone or both control and combined P with N addition treatments. (2) P application rates and experimental duration were clearly reported. (3) When enzyme activities data were presented graphically, mean values could be extracted using WebPlotDigitizer71. (4) If multiple experiments were conducted within a year, we only used data from the most recent sampling period. To maximize data resolution, we treated different P application rates, experimental durations, and ecosystem types within individual studies as independent observations17. Additionally, soil data were collected from surface layers (0–40 cm in this study), with values recorded as depth-interval midpoints17. Finally, 629 observations were obtained from 155 articles (Fig. 1 and Supplementary Fig. 3). Specifically, 427 observations for P addition alone and 202 observations for combined P with N addition.
Data extraction
Information was extracted from each study site on soil and microbial properties, including SOC, soil total N, AP, AN (sum of ammonium and nitrate concentrations), MBC, MBN, and MBP. Meanwhile, we also recorded the MAT, MAP, and ecosystem types (forest, grassland, and cropland). We used latitude and longitude to derived the MAT and MAP values from WorldClim if these metrics were not reported72.
Notably, the nutrient-acquiring enzymes activities attached to the same functional group were normalized73. For example, some studies in our dataset measured alkaline phosphatase, acid phosphatase, and neutral phosphatase activities simultaneously; thus, the P-acquiring enzyme activities were assessed as follows73:
Estimation of microbial C limitation
Microbial C limitation, reflected by vector length, was assessed using the revised enzyme vector model with the following equation21:
Data analysis
We employed the natural log-transformed response ratio (lnRR) to quantify the responses of variables to P addition, which were calculated as follows:
where Xt and Xc represent the mean values of treatment (P addition alone or combined with N addition) and control, respectively. The weight (Wi) was calculated as follows30:
where Nc and Nt are the replication numbers in control and treatment (P addition alone or combined with N addition), respectively. To account for the variability in soil enzyme activities and their stoichiometry caused by variations in the soil sampling depth, we included the soil depth as a random effect30,74. We used the following model to determine the effects of P addition, either alone or combined with N addition, on soil enzyme variables:
the Eq. 5 included: β0 (model coefficients), πstudy (study-level random effects accounting for within-study autocorrelation), (πdepth) (sampled soil depth), and ε (sampling error). Meta-regression models, which included fixed effects, were employed to test the effects of climates, experimental durations, and experimental rates on the lnRRs of enzyme activities and their stoichiometry by using the lme4 package75. The treatment effects (P addition alone or combined with N addition) were deemed significant when 95% confidence intervals excluded zero. The lnRRs were converted to percentage value calculated as follows:
Publication bias was evaluated via Egger’s regression, and no significant publication bias was found (p > 0.05. Supplementary Table 5). We also examined interactions between nutrient additions and assessed the interaction effect sizes of soil enzyme activities and their stoichiometry using Hedges’ d. The interactions of N and P addition were additive for most soil enzyme activities and their stoichiometry, while synergistic interactions observed for P-acquiring enzymes (Supplementary Fig. 4). More detailed calculations were provided in the Supplementary method. We conducted all analyses in R76.
Ethics approval and consent to participate
This presented research did not involve human subjects or animals that would require ethics approval. All methods were carried out in compliance with local and national regulations.
Data availability
The data used can be found in Figshare (https://doi.org/10.6084/m9.figshare.30737228).
Code availability
R code used can be found in Figshare (https://doi.org/10.6084/m9.figshare.30370738).
References
Hou, E. et al. Global meta-analysis shows pervasive phosphorus limitation of aboveground plant production in natural terrestrial ecosystems. Nat. Commun. 11, 637 (2020).
Mahowald, N. et al. Global distribution of atmospheric phosphorus sources, concentrations and deposition rates, and anthropogenic impacts. Glob. Biogeochem. Cycle. 22, GB4026 (2008).
Penuelas, J. et al. Human-induced nitrogen-phosphorus imbalances alter natural and managed ecosystems across the globe. Nat. Commun. 4, 2934 (2013).
Margalef, O. et al. The effect of global change on soil phosphatase activity. Glob. Change Biol. 27, 5989–6003 (2021).
Wu, W. et al. Meta-analysis of the impacts of phosphorus addition on soil microbes. Agric. Ecosyst. Environ. 340, 108180 (2022).
Sun, Y. et al. Phosphorus additions imbalance terrestrial ecosystem C:N:P stoichiometry. Glob. Change Biol. 28, 7353–7365 (2022).
Li, J. et al. Soil enzyme activity and stoichiometry in response to precipitation changes in terrestrial ecosystems. Soil Biol. Biochem. 191, 109321 (2024).
Sinsabaugh, R. L. et al. Stoichiometry of soil enzyme activity at global scale. Ecol. Lett. 11, 1252–1264 (2008).
Fatemi, F. R., Fernandez, I. J., Simon, K. S. & Dail, D. B. Nitrogen and phosphorus regulation of soil enzyme activities in acid forest soils. Soil Biol. Biochem. 98, 171–179 (2016).
Sinsabaugh, R. L., Hill, B. H. & Shah, J. J. F. Ecoenzymatic stoichiometry of microbial organic nutrient acquisition in soil and sediment. Nature 462, 795–U117 (2009).
He, P., Zhang, Y., Shen, Q., Ling, N. & Nan, Z. Microbial carbon use efficiency in different ecosystems: a meta-analysis based on a biogeochemical equilibrium model. Glob. Change Biol. 29, 4758–4774 (2023).
Mori, T. Does ecoenzymatic stoichiometry really determine microbial nutrient limitations?. Soil Biol. Biochem. 146, 107816 (2020).
Rosinger, C., Rousk, J. & Sandén, H. Can enzymatic stoichiometry be used to determine growth-limiting nutrients for microorganisms? A critical assessment in two subtropical soils. Soil Biol. Biochem. 128, 115–126 (2019).
Mori, T., Rosinger, C. & Margenot, A. J. Enzymatic C:N:P stoichiometry: questionable assumptions and inconsistencies to infer soil microbial nutrient limitation. Geoderma 429, 116242 (2023).
Mori, T., Aoyagi, R., Kitayama, K. & Mo, J. Does the ratio of β-1,4-glucosidase to β-1,4-N-acetylglucosaminidase indicate the relative resource allocation of soil microbes to C and N acquisition?. Soil Biol. Biochem. 160, 108363 (2021).
Kunito, T. et al. Ecoenzymatic stoichiometry as a temporally integrated indicator of nutrient availability in soils. Soil Sci. Plant Nutr. 70, 246–269 (2024).
Liu, H. et al. Nitrogen addition induces microbial phosphorus limitations in bulk soil but not in rhizospheric soil: a global analysis. Soil Res. 252, 106609 (2025).
Cui, Y. et al. Ecoenzymatic stoichiometry reveals widespread soil phosphorus limitation to microbial metabolism across Chinese forests. Commun. Earth Environ. 3, 184 (2022).
Cui, Y. et al. Global patterns of nutrient limitation in soil microorganisms. Proc. Natl. Acad. Sci. USA 122, e2424552122 (2025).
Moorhead, D., Cui, Y., Sinsabaugh, R. & Schimel, J. Interpreting patterns of ecoenzymatic stoichiometry. Soil Biol. Biochem. 180, 108997 (2023).
Cui, Y., Moorhead, D. L., Peng, S., Sinsabaugh, R. L. & Peñuelas, J. Predicting microbial nutrient limitations from a stoichiometry-based threshold framework. Innov. Geosci. 2, 100048 (2024).
Allison, S. D. & Vitousek, P. M. Responses of extracellular enzymes to simple and complex nutrient inputs. Soil Biol. Biochem. 37, 937–944 (2005).
Zhou, Z. H., Wang, C. K. & Jin, Y. Stoichiometric responses of soil microflora to nutrient additions for two temperate forest soils. Biol. Fertil. Soils 53, 397–406 (2017).
Liu, M. H., Gan, B. P., Li, Q., Xiao, W. F. & Song, X. Z. Effects of nitrogen and phosphorus addition on soil extracellular enzyme activity and stoichiometry in Chinese fir (Cunninghamia lanceolata) forests. Front. Plant Sci. 13, 834184 (2022).
Wang, F. C. et al. Divergent responses of soil microbial community to long-term nitrogen and phosphorus additions in a subtropical Chinese fir plantation. Catena 242, 108132 (2024).
Keane, J. B. et al. Soil C, N and P cycling enzyme responses to nutrient limitation under elevated CO2. Biogeochemistry 151, 221–235 (2020).
Deng, H. Y. et al. Soil ecosystem multifunctionality is strongly linked with crop yield after four decades chemical fertilization in black soil. Agric. Ecosyst. Environ. 368, 109007 (2024).
Mori, T. The ratio of β−1,4-glucosidase activity to phosphomonoesterase activity remains low in phosphorus-fertilized tropical soils: a meta-analysis. Appl. Soil Ecol. 180, 104635 (2022).
Forrester, D. I. & Bauhus, J. A review of processes behind diversity-productivity relationships in forests. Curr. Rep. 2, 45–61 (2016).
Chen, X. & Chen, H. Y. H. Plant mixture balances terrestrial ecosystem C:N:P stoichiometry. Nat. Commun. 12, 4562 (2021).
Castle, S. C. et al. Nutrient limitation of soil microbial activity during the earliest stages of ecosystem development. Oecologia 185, 513–524 (2017).
DeForest, J. L., Dorkoski, R., Freedman, Z. B. & Smemo, K. A. Multi-year soil microbial and extracellular phosphorus enzyme response to lime and phosphate addition in temperate hardwood forests. Plant Soil 464, 391–404 (2021).
Ma, S. H. et al. Inconsistent responses of soil microbial community structure and enzyme activity to nitrogen and phosphorus additions in two tropical forests. Plant Soil 460, 453–468 (2021).
Dove, N. C., Torn, M. S., Hart, S. C. & Tas, N. Metabolic capabilities mute positive response to direct and indirect impacts of warming throughout the soil profile. Nat. Commun. 12, 2089 (2021).
Moorhead, D. L., Sinsabaugh, R. L., Hill, B. H. & Weintraub, M. N. Vector analysis of ecoenzyme activities reveal constraints on coupled C, N and P dynamics. Soil Biol. Biochem. 93, 1–7 (2016).
Zuccarini, P. et al. Effects of nitrogen deposition on soil enzymatic activity and soil microbial community in a Mediterranean holm oak forest. Geoderma 430, 116354 (2023).
Huang, X. et al. Nitrogen deposition mitigates long-term phosphorus input-induced stimulative effects on soil respiration in a tropical forest. Geoderma 453, 117142 (2025).
Yuan, X. et al. Linkages of stoichiometric imbalances to soil microbial respiration with increasing nitrogen addition: Evidence from a long-term grassland experiment. Soil Biol. Biochem. 138, 107580 (2019).
Luo, X., Zhang, L., Lin, Y., Wen, D. & Hou, E. Nitrogen availability mediates soil organic carbon cycling in response to phosphorus supply: a global meta-analysis. Soil Biol. Biochem. 185, 109158 (2023).
Jiang, J. et al. Antagonistic and additive interactions dominate the responses of belowground carbon-cycling processes to nitrogen and phosphorus additions. Soil Biol. Biochem. 156, 108216 (2021).
Zheng, X. et al. Interactions between nitrogen and phosphorus in modulating soil respiration: a meta-analysis. Sci. Total Environ. 905, 167346 (2023).
Yu, Q. et al. Differential responses of soil phosphorus fractions to nitrogen and phosphorus fertilization: a global meta-analysis. Glob. Biogeochem. Cycle 38, e2023GB008064 (2024).
Wang, J., Wu, Y., Li, J., He, Q. & Bing, H. Soil enzyme stoichiometry is tightly linked to microbial community composition in successional ecosystems after glacier retreat. Soil Biol. Biochem. 162, 108429 (2021).
Fang, X. et al. Phosphorus addition alters the response of soil organic carbon decomposition to nitrogen deposition in a subtropical forest. Soil Biol. Biochem. 133, 119–128 (2019).
Zhang, H. et al. Phosphorus addition stimulates overall carbon acquisition enzymes but suppresses overall phosphorus acquisition enzymes: a global meta-analysis. Agric. Ecosyst. Environ. 375, 109219 (2024).
Jing, X. et al. Neutral effect of nitrogen addition and negative effect of phosphorus addition on topsoil extracellular enzymatic activities in an alpine grassland ecosystem. Appl. Soil Ecol. 107, 205–213 (2016).
Xiao, W., Chen, X., Jing, X. & Zhu, B. A meta-analysis of soil extracellular enzyme activities in response to global change. Soil Biol. Biochem. 123, 21–32 (2018).
Turner, B. L. & Wright, S. J. The response of microbial biomass and hydrolytic enzymes to a decade of nitrogen, phosphorus, and potassium addition in a lowland tropical rain forest. Biogeochemistry 117, 115–130 (2014).
Prescott, C. E. et al. Surplus carbon drives allocation and plant-soil interactions. Trends Ecol. Evol. 35, 1110–1118 (2020).
Mouginot, C. et al. Elemental stoichiometry of fungi and bacteria strains from grassland leaf litter. Soil Biol. Biochem. 76, 278–285 (2014).
Gong, J. R. et al. Phosphorus fertilization affects litter quality and enzyme activity in a semiarid grassland. Plant Soil 492, 91–108 (2023).
Nannipieri, P., Trasar-Cepeda, C. & Dick, R. P. Soil enzyme activity: a brief history and biochemistry as a basis for appropriate interpretations and meta-analysis. Biol. Fertil. Soils 54, 11–19 (2018).
Puissant, J. et al. The pH optimum of soil exoenzymes adapt to long term changes in soil pH. Soil Biol. Biochem. 138, 107601 (2019).
Rousk, J., Brookes, P. C. & Bååth, E. The microbial PLFA composition as affected by pH in an arable soil. Soil Biol. Biochem 42, 516–520 (2010).
Wang, C., Sun, Y., Chen, H. Y. H. & Ruan, H. Effects of elevated CO2 on the C:N stoichiometry of plants, soils, and microorganisms in terrestrial ecosystems. Catena 201, 105219 (2021).
Sun Y. et al. Coherent responses of terrestrial C:N stoichiometry to drought across plants, soil, and microorganisms in forests and grasslands. Agric. For. Meteorol. 292–293, 108104 (2020).
Sun, Y., Wang, C., Chen, H. Y. H. & Ruan, H. Responses of C:N stoichiometry in plants, soil, and microorganisms to nitrogen addition. Plant Soil 456, 277–287 (2020).
Xu, X., Thornton, P. E. & Post, W. M. A global analysis of soil microbial biomass carbon, nitrogen and phosphorus in terrestrial ecosystems. Glob. Ecol. Biogeogr. 22, 737–749 (2013).
Zhao, M. Y., Liu, X. Y. & Zhang, X. Y. A review of research advances on carbon sinks in farmland ecosystem. Acta Ecol. Sin. 516, 2710–2710 (2022).
Lange, M. et al. Plant diversity increases soil microbial activity and soil carbon storage. Nat. Commun. 6, 6707 (2015).
Dijkstra, F. A. et al. Climate change alters stoichiometry of phosphorus and nitrogen in a semiarid grassland. N. Phytol. 196, 807–815 (2012).
Li, S. et al. Phosphorus limitation regulates the responses of microbial carbon metabolism to long-term combined additions of nitrogen and phosphorus in a cropland. Soil Biol. Biochem. 200, 109614 (2025).
Gu, X., Zhang, F., Xie, X., Cheng, Y. & Xu, X. Effects of N and P addition on nutrient and stoichiometry of rhizosphere and non-rhizosphere soils of alfalfa in alkaline soil. Sci. Rep. 13, 12119 (2023).
Cui, Y. et al. Extracellular enzyme stoichiometry reveals the carbon and phosphorus limitations of microbial metabolisms in the rhizosphere and bulk soils in alpine ecosystems. Plant Soil 458, 7–20 (2019).
Cleveland, C. C. & Liptzin, D. C. :N:P stoichiometry in soil: is there a “Redfield ratio” for the microbial biomass?. Biogeochemistry 85, 235–252 (2007).
Kuzyakov, Y. & Blagodatskaya, E. Microbial hotspots and hot moments in soil: concept and review. Soil Biol. Biochem. 83, 184–199 (2015).
Ning, Q. et al. Carbon limitation overrides acidification in mediating soil microbial activity to nitrogen enrichment in a temperate grassland. Glob. Change Biol. 27, 5976–5988 (2021).
Puissant, J. Does the C:N:P 1:1:1 ratio hold? Examining log-transformation bias in enzyme stoichiometry. Glob. Change Biol. 31, e70228 (2025).
Mori, T. The problem is not how we calculate enzyme stoichiometry threshold—It is that we calculate it. Glob. Change Biol. 31, e70519 (2025).
Zou, X. Calcium as a confounding variable in phosphorus attribution: a commentary on Hu et al. (2025). Soil Environ. Health 3, 100172 (2025).
Burda, B. U., O’Connor, E. A., Webber, E. M., Redmond, N. & Perdue, L. A. Estimating data from figures with a Web-based program: Considerations for a systematic review. Res. Synth. Methods 8, 258–262 (2017).
Hijmans, R. J., Cameron, S. E., Parra, J. L., Jones, P. G. & Jarvis, A. Very high resolution interpolated climate surfaces for global land areas. Int. J. Climatol. 25, 1965–1978 (2005).
Han, S. et al. Soil aggregate size-dependent relationships between microbial functional diversity and multifunctionality. Soil Biol. Biochem. 154, 10843 (2021).
Liu, M. et al. Unprotected carbon dominates decadal soil carbon increase. Nat. Commun. 16, 2008 (2025).
Bates, D., Mächler, M., Bolker, B. M. & Walker, S. C. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 67, 1–48 (2015).
R Development Core Team. R: a language and environment for statistical computing. R Foundation for Statistical Computing, (Vienna, 2024).
Acknowledgements
This study was supported by the National Key Research and Development Program of China (No. 2023YFD2200404, 2021YFD2200403); the Key Project of the Open Competition in Jiangsu Forestry (LYKJ【2022】01); the Postgraduate Research & Practice Innovation Program of Jiangsu Province (KYCX24_1358); the Natural Science Foundation of Jiangsu Province (BK20250706); and the Natural Science Foundation of the Jiangsu Higher Education Institutions of China (24KJB220005).
Author information
Authors and Affiliations
Contributions
H.L., H.R., H.C., T.R., and X.Z. led the writing of the manuscript and designed the research; H.L. and T.R. collected the data; H.L, J.L., and H.C. analyzed the data. All authors contributed significantly to the drafts and gave final approval for publication.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Earth & Environment thanks the anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editors: Somaparna Ghosh A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, H., Ren, T., Liao, J. et al. Nitrogen deposition alleviates phosphorus-induced imbalances in soil enzyme stoichiometry. Commun Earth Environ 7, 88 (2026). https://doi.org/10.1038/s43247-025-03115-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43247-025-03115-1