Abstract
The redox status of permafrost soils is a decisive factor for their nutrient cycling, organic matter decomposition, and greenhouse gas emissions. Although being associated with a variety of processes, data availability of continuous redox measurements in permafrost soils is scarce. Here, we provide a unique dataset covering three years of soil redox potential measurements, obtained from a monitoring approach at three research sites near Fairbanks, Alaska. Redox potential pattern in the permafrost soil active layer showed large seasonal differences, with reducing conditions in the short summer/autumn to largely oxidizing conditions in winter and spring. However, conditions for methane production were at no time recorded in the three years. Especially the freezing and thawing had substantial impact on the redox status, highlighting that assessment of redox conditions in permafrost soils should be extended beyond the typical summer observation periods.
Similar content being viewed by others
Introduction
Permafrost is thawing. For years and decades, numerous studies reported observations about extent and characteristics of degrading permafrost landscapes in the Pan-Arctic and provided estimates about the regional to global consequences of climate change (e.g. reviewed by Smith et al1.). In Alaska, records of permafrost thaw from the past 40 years suggest that this process accelerated in the last 2 decades2,3,4. Alongside the thawing process, changes in the landscape as well as in the emerging soils occur. Among the most evident changes are alterations in the cover and species composition of aboveground vegetation, which are partly shaped by the evolving landscape morphology5. Within soil, a deepening of the active layer enhances soil development, which, in turn, strongly hinges on the hydrologic properties of the degrading permafrost soil. On well-drained slope positions permafrost degradation may promote transition to dry degraded soils, whereas permafrost degradation in depressions will likely cause accumulation of water, thus resulting in water-saturated soils and/or wetlands6,7. But also in rather flat terrain, thawing of the ice-rich ground results in surface subsidence, favoring water redistribution towards the center (i.e., thermokarst development), while the higher situated surroundings become drier3.
The amount of soil ice and the hydrology of the active layer are a strong control on the rate of soil organic matter (SOM) turnover. While SOM in intact permafrost soils can be stabilized for millennia8,9, permafrost degradation generally enhances mobilization and transformation of SOM10,11,12. Availability of oxygen as the most favorable electron acceptor governs the microbial aerobic/anaerobic pathways of SOM transformations. A key soil parameter, which links microbial demand for and availability of oxygen in the soil, is the redox potential. By describing oxidizing and reducing conditions in soil, the soil’s redox status directly affects and regulates SOM decomposition processes13,14,15, and ultimately determine the production of greenhouse gases like CO2, CH4, and N2O. In addition, redox potentials provide information about dynamics of redox sensitive elements such as N, Mn, Fe, or S, whose oxidized forms can serve as alternative electron acceptors16,17,18. These elements are also indirectly affecting SOM cycling, e.g. by controlling nutrient availability19,20 or by formation or dissolution of stabile associations with organic compounds linked to the redox sensitive dynamics of Fe and Mn oxides/hydroxides21,22,23.
Redox processes in permafrost soils are thus a key focus in climate change research, especially regarding permafrost thaw and greenhouse gas release. However, the discussion about the redox status in these soils is largely held on the basis of indirect evidence such as, e.g., greenhouse gas emissions24,25, redox-sensitive iron phases26,27, microbial community composition10,28, partly investigated in laboratory studies29,30. Only a limited number of studies realized punctual or seasonal redox potential measurements during summer field campaigns16,31,32,33,34. Mostly, investigations are decoupled from annual redox dynamic in these soils, thus neglecting the majority of the year and especially the critical phases of freezing and thawing. With this study we are filling this knowledge gap by providing an extensive dataset from an in-situ continuous redox potential monitoring for a 3-year period in a (Sub-)Arctic discontinuous permafrost region35. Further objectives were (i) to investigate in which season and for what duration conditions in permafrost soils enable key processes like Fe oxyhydroxide dissolution or methane production, and, (ii) to evaluate how permafrost retreat and a corresponding change in soil hydrology is affecting annual redox dynamics in degraded permafrost soils.
In this study we show substantial changes in soil redox potentials (EH) of an intact permafrost active layer between the season, ranging from oxidizing conditions (up to +800 mV) to moderately reducing conditions (about −100 mV). At no point in time, the investigated intact- and degraded permafrost sites were found to be considerable CH4-producing environments.
Results and discussion
Assessment of technical-related artifacts
Studies dealing with field monitoring of soil redox potentials measured in-situ for an entire year or longer are limited, as seasonal or punctual measurements are more common32,36,37. If conducted, long-term monitoring studies are largely done in warm38,39 or temperate regions40,41,42,43. Therefore, it is not well documented if redox potential measurements during freezing and thawing are affected by H2O phase transitions from liquid (water) to solid (ice) and vice versa. For example, Vorenhout et al.44 presented certain changes in topsoil EH in a northern Netherlands salt marsh during winter, but the interpretation of such dynamics were not reported. In order to exclude that potential technical-related artifacts are (mis-) interpreted as microbial decomposition processes in these seasons, we simulated such phase transitions during freeze-thaw cycles (FTC) under controlled laboratory conditions in a climate chamber. At conducting a total of 9 FTC ranging from +10 °C to −5 °C (typical annual soil temperature fluctuations at the studied Interior Alaska sites) with 3 redox electrodes in a soil matrix under water saturated conditions, we observed increases in EH values due to the freezing of about 50–70 mV (Fig. 1). Upon thawing, EH values dropped rapidly again by about the same extent. This freeze-thaw dynamic was repeatedly measured for the 9 cycles and was also reproduced when the FTC started at room temperature (FTC C-4 to C-5; Fig. 1). Such increase after freezing and decrease after thawing is likely attributable to the temperature-dependency of microbial metabolism45,46. This is supported by a second set of FTC tests conducted in a water-saturated abiotic matrix of quartz in silt-size, which revealed rather a decrease of EH after freezing of about 25-30 mV only (Supplementary Fig. S1a-c). Similar pattern were also observed in diurnal soil EH fluctuations as reported by Dorau et al.47 and suggest that technical-related background noise (artifacts) are comparably minor.
A total of 9 FTCs were conducted and are shaded in grey (C-1 to C-9). Temperature thresholds were removed between C-4 and C-5 to investigate if a FTC restart from 20 °C room temperature is having an influence on soil EH measurements. EH values are temperature corrected (see Material and Methods Section and Supplementary Fig. S3). Soil material had a pH of 7.8 (in H2O), electric conductivity of 504 µS cm−1, organic carbon content of 9.01 % and total nitrogen of 0.32 %, resulting in a C/N ratio of 28.4.
Since the logger unit, the battery for energy supply, and all connecting modules for the electrodes were undergoing the same temperature changes inside the climate chamber (Supplementary Fig. S2a), we could further exclude that frequent temperature changes are affecting other parts of the monitoring setup.
Taken together, our pre-tests could show that in-situ measurements of the soil redox potential are feasible and provide reliable results with the chosen equipment under the given environmental conditions in a (Sub-)Arctic region like Interior Alaska.
Depth-resolved redox potential dynamics in a permafrost soil active layer
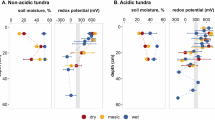
The research area is situated in the discontinuous permafrost region of Fairbanks, Interior Alaska (Supplementary Fig. S4a). Cryosols in this area are having an active layer thickness up to 60 cm48,49. The mineral topsoil, covered by 25-30 cm peat, is showing water contents of about 50%, thus being 10-20 vol.% larger compared to the underlying subsoil (Fig. 2a/b), suggesting that it is not the frozen ground which prevents incoming water from percolating downwards, but rather the horizons above. We thus infer that the major water flux at this site is present as lateral flow in the transition zone between the comparably loose peat layer and the dense mineral topsoil (Fig. 2a, Supplementary Fig. S4d). The 3-year overview of the soil redox dynamics revealed immense shifts from reducing conditions in the summer period to oxidizing conditions in the winter months (Fig. 2a) with a maximum amplitude of about 800 mV. Summer EH values dropped below 100 mV, providing moderately reducing conditions which enable reduction processes of nitrate and manganese oxides (already initiated during weakly reducing conditions at about 300 mV), and iron oxides50. Similar summer EH trends were previously presented by Patzner et al.32 from the upper 10 cm of water-logged soils (bogs and fens) in differently degrading permafrost peatlands in the Abisko region (Stordalen mire). Once reaching the moderately reducing conditions, the stated study and our own measurements during summer 2023 revealed rather constant redox potentials in permafrost soils over the comparably short summer season. A potential explanation could be the initiation of sulfate reduction41 which prevented further EH decrease. Beyond the summer dynamics, our monitoring results provide insights into the so far unconsidered transition phases between summer and winter season.
Panel (a) shows monitoring data in the intact topsoil measured in a depth of about 5 cm in the mineral soil and (b) shows subsoil data measured in a depth of about 35 cm in the mineral soil. The upper graphs represent water content (blue) and soil temperature (pink) data, while the bottom graphs depict the soil redox potential (EH; green). All graphs show individual values measured every 15 minutes by 3 replicate electrodes per site and depth. The blue-grey bar between the graphs illustrates the different seasons and processes, ranging from a thawed period in summer, a freezing period in autumn and early winter, a frozen period in winter, and a thawing period in spring and early summer. Categorization followed soil temperature and water content dynamics. Observation period ranges from 01.09.2021 to 30.09.2024. Please note that the data gap in winter 22/23 resulted from a failure in power supply.
The freezing process of the mineral topsoil took considerable time and lasted for up to 2 months. In this period, the soil temperature remained at the 0°C boundary, demonstrating a distinct zero curtain effect due to the latent heat release during the crystallization of liquid H2O to ice51. During this period of freezing, a rapid increase in soil EH of up to 700 mV was observable (Fig. 2a, details are provided in the “Freezing and thawing” chapter). This trend was reproduced in each of the 3 years and exceeded the comparably small amplitude fluctuations of about 20-50 mV during laboratory FTC (Fig. 1, Supplementary Fig. S1a-c), thus providing evidence for massive changes in soil processes during this time. This includes less microbial O2 consumption and potentially more O2 being translocated into this soil depth by a negative soil water pressure due to freezing of the upper layers, resulting in a upward-directed water flow in the remaining unfrozen soil (advection)51. Ice formation during freezing of the organic layer may further pushes remaining oxygen-containing soil pore air towards the underlying mineral topsoil. Once the mineral topsoil was frozen, there was a period of 4-5 months with oxidizing conditions and EH values fluctuating between 400-700 mV. In this time, field replicate electrodes displayed more heterogeneous EH values compared to thawed periods (Fig. 2a). Reiser et al.43 likewise observed more EH fluctuations in a Swiss Gleyic Cambisol subsoil in 40 cm, which could be related to a low oxygen diffusion rate (ODR) at this depth. Low ODR at the permafrost site may further explain the slow continuous decrease in soil EH during the winter period, when microbial activity is hampered but not completely suppressed. The initiation of the thawing process showed some diffuse effects on topsoil EH, ranging from an immediate gradual decrease detected by one electrode to rapid EH increase by another electrode (Fig. 2a). Such spatial variations might reflect the heterogeneous distribution of ice and/or water in the active layer and thus availability of O2. In any case, increasing temperatures and a concomitant rise in microbial activity and demand for oxygen is highlighted by the constant decrease in soil EH over the summer period, reaching the lower threshold of moderately reducing conditions (-100 mV) at the end of summer in every year (Fig. 2a). The EH level did not drop below -100 mV, which indicates that Fe(III)-containing iron phases serve as electron acceptors throughout the summer months, maintaining EH values at a rather constant level. Since Fe contents in the intact permafrost soil are small49, it seems likely that such Fe phases are frequently delivered from the surrounding upslope area within percolating soil solution. Especially short-range ordered and colloidal Fe can be translocated within sub-surface water flow and are likewise susceptible to reduction52,53.
Subsoil dynamics were surprisingly different, given the fact that the vertical distance between topsoil and subsoil depth at this site was only 15–29 cm (Supplementary Table S1). Water contents were smaller with 30–40% compared to the topsoil. Likewise, the temperature amplitude between summer and winter seasons was less in the subsoil (−1 – 2° C) compared to the topsoil (−3 – 8°C), showing the large heat capacity of the water-rich upper soil54. Smaller temperature amplitude is further explaining the even more prolonged zero curtain effect in the subsoil, which extend the freezing phases but shortens the thawing phases (Fig. 2b). Field observations suggested a reducing environment due to the greyish color and thus only minor amounts of redox-sensitive oxides (Table 1, Supplementary Fig. S4d), accompanied by a limited microbial activity49. However, subsoil EH values showed oxidizing conditions in more than 90% of the time in the 3-year observation period (Fig. 2b, Table 2). Typically, the frozen ground is a natural water percolation barrier7, resulting in long periods of water saturation and consequently strongly reducing conditions. For example, active layer redox dynamics were previously reported by Fiedler et al.16 from permafrost soils in the Lena Delta, with oxidizing conditions in the upper part of the soil (EH about 400 mV) and reducing conditions near the permafrost table (EH about -150 mV). Our monitoring data showed the opposite depth trends for multiple years (Fig. 2a/b), attributable to the pronounced water flux within the organic layer-mineral topsoil transition zone and providing evidence that thawing permafrost subsoils should not generally be considered as a strongly reducing and thus potentially CH4 producing environment14,26.
Overall, EH values in this permafrost soil active layer were at all times above the threshold for notable CH4 production (−150 mV55). Permafrost soils in this area were likewise found to have a low diversity and abundance of methanogens in comparison to methanotroph phylotypes56. These findings are in parts reflected on a larger scale by Eddy flux measurements in the same area, showing very low CH4 emissions but large CO2 emissions from the permafrost soils57. Results could demonstrate for this expanded observation period that neither temporally (0 days with EH values < −100 mV) nor spatially (subsoil showed even oxidizing conditions), the water-rich active layer mineral soil can be considered as a CH4 source area.
Impact of permafrost degradation on soil hydrology and redox status
Thawing of permafrost and the increase in active layer thickness have a multitude of effects on soil properties and the overall landscape development. In order to assess the effects of permafrost degradation on soil redox dynamics, we conducted the soil monitoring also at two degraded sites within 1 km distance to the intact permafrost site (Supplementary Fig. S4b). The two sites were selected to represent different hydrological landscapes and soils, i.e., dry uphill soils vs. wet downhill soils. The wet-degraded topsoil had a dynamic hydrology of water saturation level during spring season and snow-melt (up to 60 vol.%) followed by some drainage and decrease in water contents by 10-20 vol.% during the summer months (Fig. 3a). Interestingly, no prominent zero curtain effect was recorded for the soil temperatures, suggesting that freezing of the topsoil occurred comparably fast, maybe due to beneficial moisture conditions in the organic layer facilitating both, a high temperature conductivity from the surface to the mineral soil due to high moisture, but likewise a certain amount of air-filled pores with a small heat capacity54. A sufficient O2 supply in the topsoil is further reflected by oxidizing conditions (EH values > 300 mV) throughout the summer seasons. Aboveground freezing and clogging of soil pores by ice reduces the ODR from the atmosphere to the mineral topsoil, resulting in a gradual decrease in EH values during the winter months (up to 400 mV within 6 months), similar to the intact topsoil (Fig. 2a) but with a smaller magnitude.
Panel (a) shows monitoring data in the wet topsoil measured in a depth of about 5 cm in the mineral soil and (b) shows subsoil data measured in a depth of about 50 cm in the mineral soil. The upper graphs represent water content (blue) and soil temperature (pink) data, while the bottom graphs depict the soil redox potential (EH; green). All graphs show individual values measured every 15 minutes by 3 replicate electrodes per site and depth. The blue-grey bar between the graphs illustrates the different seasons and processes, ranging from a thawed period in summer, a freezing period in autumn and early winter, a frozen period in winter, and a thawing period in spring and early summer. Categorization followed soil temperature and water content dynamics. Observation period ranges from 01.09.2021 to 30.09.2024. Please note that one subsoil water content was not recorded properly since 02/21, as well as one subsoil EH between 03/23 to 09/23 (see Method section for more details).
The mineral subsoil, about 45 cm deeper than the topsoil, revealed a completely different seasonal pattern. The site exploration and first monitoring data from 2021 suggested a water-rich soil profile with similar water contents in the top- and subsoil. However, drier and warmer years from 2022 to 2024, visible by the increasing summer temperatures in both soil depths and decreasing snow cover at the soil surface (Fig. 3a/b; Material and Methods section Fig. 7), resulted in less water supply in the subsoil. This may be due to the ongoing deepening of the active layer along with the water-accumulating permafrost table in addition to enhanced evapotranspiration. A very characteristic feature of the wet-degraded subsoil was the most pronounced zero curtain for all investigated sites (Fig. 3b). The subsoil EH values were strongly dependent on the water content, i.e., large water contents in the installation year 2021 resulted in weakly reducing conditions, whereas the drier years 2022-2024 had largely oxidizing conditions throughout the seasons (Fig. 3b). These fluctuating, but generally weakly reducing to oxidizing conditions also explain why in a previous study, we did find considerable amounts of crystalline Fe phases in samples taken in 202149 (Table 1). It further provides in-situ evidence that this wet (but not saturated) degraded permafrost landscape likely does not constitute a relevant CH4 source area, regardless of the season.
Development of well-drained soils following permafrost degradation showed the smallest water contents in the mineral top- and subsoil of all 3 sites, respectively (Fig. 4a/b). After a short phase of high water contents due to snow melt in late spring, topsoil moisture levels remained at 20 vol.% or below, thus being less than half of the summer water contents of the wet and intact sites (Figs. 2a, and 3a). The subsoil was largely unaffected by precipitation or snow melt events, likely due to lateral flow, i.e., limited percolation from topsoil to subsoil, and uptake by the vegetation (birch trees dominate). Small water contents are further reflected by the reduction of the zero curtain to just a few days (topsoil) or complete absence (subsoil), compared to the pronounced phenomenon up to several months for the other two sites. Generally, soil temperatures (positive and negative ones) at the dry site are about twice as high compared to the other sites, for the topsoil as well as for the subsoil. Corresponding to the unsaturated conditions in this soil, EH values are largely > 600 mV and thus suggesting well-oxidizing conditions in both, top- and subsoil (Fig. 4a/b). Only 1 electrode in topsoil and 1 electrode in the subsoil detected temporal weakly reducing conditions. In case of the topsoil, this was measured in late winter 2022 and is likely related to a comparably wet soil in autumn 2021, and a clogging of pores due to ice formation upon freezing decreased ODR. One subsoil EH value dropped in summer 2023 to about 300 mV, which may be a consequence of a higher microbial activity and a higher demand for O2 than can diffuse from the topsoil down to the subsoil and may illustrate the presence of spatiotemporal anoxic microsites in this soil58.A potential bottleneck in sufficient ODR to the deeper subsoil may be the higher bulk density of the soil horizons between the investigated top- and subsoil depths49. Soil redox conditions at the dry site support findings from Voigt et al25. that arctic soils can turn to CH4 sinks upon drying.
Panel (a) shows monitoring data in the dry topsoil measured in a depth of about 5 cm in the mineral soil and (b) shows subsoil data measured in a depth of about 50 cm in the mineral soil. The upper graphs represent water content (blue) and soil temperature (pink) data, while the bottom graphs depict the soil redox potential (EH; green). All graphs show individual values measured every 15 minutes by 3 replicate electrodes per site and depth. The blue-grey bar between the graphs illustrates the different seasons and processes, ranging from a thawed period in summer, a freezing period in autumn and early winter, a frozen period in winter, and a thawing period in spring and early summer. Categorization followed soil temperature and water content dynamics. Observation period ranges from 01.09.2021 to 30.09.2024. Please note that the data gap in winter 22/23 resulted from a failure in power supply.
Despite no fully water-saturated soil (like peat-rich fens or bogs) being included in this study, we nonetheless show that water-rich soils and well-drained soils, as two possible emerging soils during and after permafrost thaw, undergo drastic changes in their active layer redox potential dynamics compared to non-degraded permafrost. The consecutive monitoring for 3 years further revealed that EH values are less fluctuating throughout the seasons compared to their intact counterpart. In addition to findings from a previous study that the degraded soils harbor only ¼ of the SOC stored in the intact permafrost soil49, this study points out that soil conditions for this enhanced organic matter mineralization are facilitating aerobic decomposition in both, topsoil and subsoil horizons.
Freezing and thawing—known processes but unknown consequences for redox potential in permafrost soil
The year-round monitoring included 4 measurements per hour, yielding 96 data points per day. In order to first identify daily variability or longer phases with large redox potential fluctuations within the annual redox dynamic, daily mean values (n = 96) and their standard deviations (SD) were calculated and visualized in Fig. 5. Sharp increase in soil EH caused data variations of >200 mV per day and were more prevalent in the intact permafrost topsoil (Fig. 5a) for a limited number of days, which were concentrated shortly before winter in 2021, 2022, and 2023. Freezing of the soil coincided with pronounced fluctuations in soil redox potential. These shifts may reflect rapid changes in microbial metabolism caused by freezing59,60,61, but could also result from abiotic processes such as oxygen redistribution, pore ice formation, or altered water flow paths which caused changes in ion concentrations in the remaining liquid phase62. Thawing on the other side showed an overall decrease in EH values and appeared to be more gradual with less deviations in daily EH values, but in return over a longer period. This likely illustrates the steady adaptation of the microbial activity to the changing environmental conditions in spring seasons63 as well as a higher availability of substrate like root exudates or dissolved organic matter64. Soil EH values in summer and winter showed only minor changes in daily EH, thus highlighting that freezing and thawing appear to be the most critical phases in annual soil EH dynamics. The intact permafrost subsoil however showed less variations in daily EH values between seasonal shifts, which corresponds to the smaller amplitude in annual soil water content changes compared to the topsoil (Fig. 2). In addition, the subsoil is less susceptible to changes in surface temperature or precipitation than the topsoil. While freeze–thaw cycles are known to restructure the soil microbiome and influence respiration dynamics13,61,63, our dataset does not allow us to disentangle biotic from abiotic drivers of the observed redox responses.
Figure a-f shows daily mean redox potentials (n = 96) measured with the 3 replicate electrodes per site and depth in the upper graphs, together with their standard deviations (SD; n = 96) in the bottom graphs. Observation period ranges from 01.09.2021 to 30.09.2024.
The degraded soils did not show any notable rapid changes in EH values upon soil freezing (Fig. 5c-f). If any considerable variations (SD > 50 mV) were present at the wet or dry topsoil, such phases occurred only during thawing of the soils around May to June. The observable increase in soil EH upon thawing can be assigned to the melting of snow and ice at the soil surface or within the pore space of the organic layer, thus facilitating more oxygen diffusion into the mineral topsoil (Figs. 3a, and 4a). While the subsoil at the Dry site had only minor seasonal variations in water content and EH values (Fig. 5f), the Wet subsoil experienced large daily EH fluctuations (Fig. 5d). However, rapid changes in soil EH were not induced by freezing or thawing, but appeared during the summer months. Even though the measured water contents in the wet subsoil were not exceptionally large (Fig. 3b), the ongoing permafrost thaw at the foot at this small slope, where the wet site is situated, does not provide steady saturated conditions but rather promotes a dynamic and inconsistent soil environment in terms of water contents, oxygen availability, and thus redox potential. The frequent change in soil EH from oxidizing to reducing conditions and vice versa further explains the much higher share of non-crystalline Fe phases in the dynamic wet subsoil compared to the dry subsoils49.
A more detailed look into the water content, temperature, and redox pattern during freezing and thawing events at the intact site revealed that the initiation of topsoil freezing resulted in an immediate response of topsoil EH increase (Fig. 6a). Because the sensors were installed in a defined grid in the field with about 1 m distance between replicate sensors (Supplementary Fig. S5), we were able to examine how the data are impacted across the spatial scale at each site. There was a temporal offset in topsoil EH changes of several days between the three pairs of electrodes and sensors during both freezing and thawing, highlighting the heterogeneity of the area over a total distance of 5 to 6 m, a typical feature of permafrost landscapes7,65,66. Contrasting, the respective pairs of electrodes and sensors showed a close correlation of water content and redox potential trends, thus indicating strong spatial dependencies on the decimeter-scale in the mineral topsoil ( <1 m distance). Freezing of the intact permafrost subsoil in 2021 showed within a 2-week timeframe first an EH decrease followed by an EH increase towards the end of the freezing process (Fig. 6b). In a soil monolith incubation experiment from a coastal Calcaric Gleysol, similarly fast reactions of soil EH on changes from high water table to soil aeration were observed47. We consider it unlikely that such large and rapid soil EH changes are solely a consequence of microbial O2 consumption67. Oxygen depletion in intact soils is constrained by oxygen diffusion, so while microbial respiration contributes to redox decline, achieving a ~300 mV shift typically requires multiple days under sustained water saturation. Rapid 300–400 mV changes within a day are physically implausible from biotic activity alone, pointing to abiotic mechanisms. Presumably, a translocation of O2-depleted and O2-containing soil pore air by soil ice expansion and a frequent change in flow paths of remaining liquid pore water from different areas of the soil during the freezing process may have caused this pattern. However, this phenomenon was not reproduced in the following years. Thawing of the subsoil appeared to be more gradual with no immediate response of the EH values, which corresponds to the overall small temperature amplitude in the subsoil.
Panel (a) and (b) show the topsoil and subsoil monitoring of the intact permafrost site, respectively. Within each panel, the left-sided graphs of temperature (T), water content (WC), and redox potential (EH) represent a freezing event, while the three graphs on the right side represents a thawing event. For a better attribution of T, WC, and EH measured in the same direction of the field set-up, field replicate measurements 1−3 are differentiated by shading of the respective color. Please note that similar detailed graphs of the wet- and dry degraded sites can be found in the Supplementary Information (Supplementary Fig. S6a-d).
The wet and dry degraded soils showed no fast EH response during freezing such as the intact site (Supplementary Fig. S6a-d). Thawing, however, induced a rapid increase in topsoil EH within 5 days from weakly reducing to oxidizing conditions (Supplementary Fig. S6a/c), likely due to reestablishment of pathways from atmosphere to the soil pore network68. The data revealed that even though soil water contents in the degraded sites temporarily rise due to snow melt in spring, degraded permafrost soils in this area constitute a non-reducing environment.
Concluding remarks
Perennial monitoring of redox dynamics in (Sub-)Arctic permafrost soils is an intricate venture and available data are scarce. With this dataset covering 3 consecutive years, we tackled this knowledge gap and could show that a water-rich and non-degraded permafrost soil in Interior Alaska is having surprisingly large temporal and spatial variations of soil redox potential. The upper parts of the soil did show reducing conditions during the short summer, but eventually, neither mineral top- nor subsoil of the active layer reached redox conditions which would promote notable production of CH4. Freezing and thawing of soils are two of the most prominent processes in permafrost landscapes, however their impact on the redox dynamic was largely overlooked in previous studies. These processes were found to have major effects on soil EH values with shifts of several hundred mV within a few days, likely due to changes in microbial activity, water movements, pore blocking by ice formation, or the establishment of connections between the atmosphere and the soil pore network. Permafrost degradation and the re-shaping of soil hydrology due to context specific geomorphological features of the area and advancing soil development resulted in largely oxidizing conditions that facilitate microbial aerobic decomposition of the resident soil organic matter, regardless of the season and soil depth. Nonetheless, redox state and its temporal fluctuations reflected the respective site conditions very well and were found to be an important tool for assessing the soil conditions relevant for element cycling in permafrost landscapes.
To improve our process understanding, there is a demand for more data from in-situ investigations of annual redox potential dynamics in soils of such heterogeneous landscapes like permafrost-affected regions.
Material and methods
Study sites and soils
Soil monitoring stations were implemented at three sites in Interior Alaska, near Fairbanks. They are situated in a discontinuous permafrost region and cover a non-degraded permafrost soil (depth of permafrost varied between 45-60 cm; hereone, intact site), and two contrasting soils from degraded permafrost landscapes. One dry-degraded soil is located on an upslope position (permafrost level well below 200 cm depth; hereone, dry site) and one wet-degraded soil on a small elevation in a downslope position (permafrost level between 100 and 200 cm depth; hereone, wet site). A location map, landscape and soil profile pictures are provided in Supplementary Fig. S4a-h. Selected physico-chemical parameters of the topsoil and subsoil depths are provided in Table 1 and Supplementary Table S1. More details on the research sites can be found in Liebmann et al.49.
Experimental field setup
Soil monitoring equipment was obtained from ecoTech Umwelt Meßsysteme GmbH (Bonn, Germany) and each set included a weatherproof shelf equipped with a data logging station (enviLog Midi data logger), 2 connection modules for sensors and electrodes, and a 12 V and 27 Ah rechargeable battery as energy supply. 6 redox electrodes according to Mansfeldt40,41 for measuring soil redox potentials (EH) were used, consisting of a 1 mm in diameter and 5 mm long platinum tip attached to a carbon fiber shaft of varying length. An Ag/AgCl reference electrode in 3 M KCl was needed in order to measure voltage differences in mV. To prevent frost damage of the 40 cm long reference electrode, it was placed in a 60 cm deep installed salt bridge, a perforated PVC pipe filled with 3 M KCl-Agar gel, which ensures permanent contact between the electrode and surrounding soil. Soil temperature (T) and volumetric water content (WC) were measured by ratiometric coaxial impedance dielectric reflectometers, short Hydra probes (Stevens, Portland, USA). All electrodes and sensors were tested in the field for functionality and accuracy prior to installation. Redox electrodes were checked with a 470 ± 10 mV ORP test solution for Pt electrodes (Hanna Instruments, Woonsocket, USA) and yielded on average 465 ± 2 mV, Hydra probes were tested in a water saturated sand matrix and yielded a precision of ± 2 vol.%.
All sites were equipped with a similar setup of sensors and electrodes in August 2021. The setup was designed to monitor the development of soil T, WC and EH in the mineral topsoil and subsoil at three positions per site to account for small scale spatial heterogeneities of the soils. A schematic top view scheme of installation arrangement is provided in Supplementary Fig. S5. Redox electrodes were installed within a 2-meter radius around the logger station and Hydra probes within a 3-meter radius. The mineral topsoil and subsoil installation depths were defined as 5 cm and 50 cm below the organic layer. Since the intact site had a shallower active layer, the mineral subsoil depth increment was defined as the 5 cm above the frozen ground (on average corresponding to a mineral soil depth of 35 cm). Due to pronounced heterogeneities in organic layer thickness and the shallow active layer, individual installations depths were documented and are provided in Supplementary Table S1.
Installation of sensors was done on the condition that soil disturbance shall to be minimized. To achieve this, we avoided the excavation of large soil pits and the typical horizontal installation into a soil profile wall. Alternatively, we installed the sensors vertically (redox electrodes) or in a 45° angle (Hydra probes) from the soil surface into the required measuring depth. Holes for the soil redox electrodes were pre-drilled by pushing a metal stick with the same diameter as the actual electrodes into the soil. To prevent preferential water flow at the contact area between the soil and the electrode shaft, a small rubber patch was placed around the shaft and fixed within the organic layer (Supplementary Fig. S7). Holes for the Hydra probe were drilled in a 45 °C angle by using a hand corer on a wooden triangular framework with a 45 ° angle to ensure a correct and uniform drilling orientation. The 4 metal pins on the tips of the Hydra probes were pushed in the undisturbed soil at the bottom of the drilling holes and the holes were backfilled by a slurry made out of the drilled soil material and water. Next to avoiding a horizontal installation, we also decided against a vertical installation for the Hydra probe, because of the disturbance of the natural soil structure due to the drilling and the possible effects on water percolation and its consequences for WC measurements at the end of a vertical drill hole. To further prevent preferential water flow in the backfilled drilling hole, we inserted a layer of swelling bentonite powder for better clogging of the backfilling.
Soil data were continuously recorded every 15 min. The first week after installation was considered as equilibration time and data were not included in the evaluation. Winter season 2022/2023 resulted in an energy failure at the intact and dry site and data were not recorded for about 4 months. The wet site was not affected by this, however, one Hydra probe got damaged in winter 2021/2022 and a broken wire of one redox electrode resulted in occasional malfunction of data recording within the redox module in 2023, an issue which unfortunately intensified in summer 2024.
Weather conditions in the research area are routinely recorded by a weather station on the University of Alaska Fairbanks (UAF) campus, operated and publicly accessible through the Alaska Climate Research Center (ACRC) from the University of Alaska Fairbanks. The recording weather station is located on the UAF campus (64°51'36“N 147°50'54“W) and has a distance of about 300 m to the dry site, 700 m to the intact site, and about 850 m to the wet site, which we consider as a representative location for the sites in this study (Supplementary Fig. S4b). The parameters of air temperature, rainfall, snowfall, and snow depth in the measuring period from 01.09.2021 until 30.09.2024 were accessed and processed in this study (Fig. 7).
All parameters show daily values, with mean air temperature (red line), total rainfall per day (blue bar) total snowfall per day (grey bar) and snow depth (black line).
Laboratory freeze-thaw cycle experiment
Laboratory experiments were designed to test the limits of electrodes and logger units under extreme environmental conditions. Redox monitoring trends over the years showed some distinct increases and decreases during freezing and thawing events, irrespective of study site and soil depth. In order to exclude that such sharp changes in measured redox values is a consequence of material and/or technical artifacts, we simulated under laboratory conditions freeze-thaw cycles (FTC) in a freezing chamber. For this, a temperature range of −5°C to +10°C was chosen as it reflects annual top- and subsoil temperature changes at the study sites. A logger unit similar to the field installations (see Supplementary Fig. S2a) was placed inside the freezing chamber to simulate similar field conditions also for the logger and energy supply.
In a first test series, we investigated whether rapid changes in temperature and a transition of aggregate status from liquid water to solid ice is having impacts on redox potential measurements. For this, we used an inert matrix consisting of quartz grains (in silt size) to avoid changes in EH values due to changes in microbial activities. A 5-liter beaker was filled with the quartz substrate and ultra-pure water (18.2 MΩ) was added until saturation was reached. Three soil redox electrodes, one reference electrode, and two temperature sensors for regulating the freeze-thaw cycles and logging temperature data in the substrate were installed. Considering that freezing of the substrate is taking place from outside towards inside, three setups of changing positions of redox and reference electrodes tested whether different H2O aggregate statuses at redox and reference electrodes influence the measurements. Tests revealed minor changes in EH values of about 20-30 mV during FTC, while a change in electrode positions within the matrix had no influence on EH (Supplementary Fig. S1 a-c; Fig. S2b-d). Based on this, another FTC experiment was carried out with the same technical setup but soil material from a water saturated alluvial soil was chosen as the test substrate (Supplementary Fig. S2a). Soil pH (H2O) was 7.8, carbon and nitrogen contents amounted to 9.01 % and 0.32 % respectively, and the electric conductivity was at 504 µS cm−1 (Fig. 1). After electrode installation, the setup was allowed to equilibrate under freeze-thaw cycling conditions for 12 days, respectively 8 FTCs. Afterwards, a total of 9 FTC were conducted, with a break between cycle 4 and 5 to investigate whether a higher starting temperature (20 °C room temperature) has an effect on redox potential trends.
Data processing
The measured voltage of soil EH was corrected for the deviation against the standard hydrogen electrode. Typically, a correction value of 207 mV is used and refers to an offset at 25°C. Since the deviation against the standard hydrogen electrode is correlated with temperature (Supplementary Fig. S3), individual temperature-dependent correction values were calculated. By using the respective soil temperature data measured by the Hydra probes, we were able to account for temperature-induced alterations of the electrode potential from the Ag/AgCl reference electrode47 for about 1.9 million individual redox potential values. A further adjustment of EH values to pH 7 was not considered47, pH values of the respective soil depths are provided in Table 1. We decided to present single data from each electrode/sensor without averaging the 3 replicates per site and depth in order to better visualize small scale spatial heterogeneities in the soils. If not stated differently, soil monitoring data are presented as individual data per 15 min recording interval. Data from the FTC experiment were processed accordingly.
Definition of the soil redox status categories and thresholds (at pH 7) along the redox ladder in soil69 were adapted from Reddy and DeLaune50, Dorau and Mansfeldt70, and Mansfeldt71. Oxidizing soil conditions with free O2 consumption was set to EH values > 300 mV, weakly reducing conditions with beginning NO3- and MnO2 reduction was defined between 300 to 100 mV, moderately reducing soil conditions with beginning FeOOH reduction between 100 to −100 mV, and below −100 mV are strongly reducing conditions including SO42- and CO2 reduction.
For a general overview of soil redox data during the 3-year observation period, daily mean values and standard deviations were calculated from the 96 single values per day and means were assigned to the defined soil redox categories. Since no EH values below −100 mV were recorded in the observation period, this category is not included in the overview (Table 2).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Laboratory redox experimental data72 and soil monitoring data73 are available through the BonaRes repository and can be accessed by the following links: https://doi.org/10.4228/zalf-vxtq-fk90; https://doi.org/10.4228/zalf-j9xq-q235. Weather data are publicly available from the Alaska Climate Research Center (ACRC)74, Station ID USC00502107.
References
Smith, S. L., O’Neill, H. B., Isaksen, K., Noetzli, J. & Romanovsky, V. E. The changing thermal state of permafrost. Nat. Rev. Earth Environ. 3, 10–23 (2022).
Osterkamp, T. E. Characteristics of the recent warming of permafrost in Alaska. J. Geophys. Res. 112, F02S02 (2007).
Osterkamp, T. E. et al. Physical and Ecological Changes Associated with Warming Permafrostand Thermokarst in Interior Alaska. Permafr. Periglac Process 20, 235–256 (2009).
Yi, Y. et al. Characterizing permafrost active layer dynamics and sensitivity to landscape spatial heterogeneity in Alaska. The Cryosphere 12, 145–161 (2018).
Jorgenson, M. T. & Osterkamp, T. E. Response of boreal ecosystems to varying modes of permafrost degradation. Can. J. For. Res. 35, 2100–2111 (2005).
O’Donnell, J. A. et al. The Effects of Permafrost Thaw on Soil Hydrologic, Thermal, and Carbon Dynamics in an Alaskan Peatland. Ecosystems 17 (2012).
Ping, C. L., Jastrow, J. D., Jorgenson, M. T., Michaelson, G. J. & Shur, Y. L. Permafrost soils and carbon cycling. SOIL 1, 147–171 (2015).
Vonk, J. E. et al. High biolability of ancient permafrost carbon upon thaw. Geophys. Res. Lett. 40, 2689–2693 (2013).
Estop-Aragonés, C. et al. Assessing the Potential for Mobilization of Old Soil Carbon After Permafrost Thaw: A Synthesis of 14C Measurements From the Northern Permafrost Region. Glob. Biogeochem. Cycles 34, e2020GB006672 (2020).
Feng, J. et al. Warming-induced permafrost thaw exacerbates tundra soil carbon decomposition mediated by microbial community. Microbiome 8, 3 (2020).
Turetsky, M. R. et al. Carbon release through abrupt permafrost thaw. Nat. Geosci. 13, 138–143 (2020).
Miner, K. R. et al. Permafrost carbon emissions in a changing Arctic. Nat. Rev. Earth Environ. 3, 55–67 (2022).
Ernakovich, J. G., Lynch, L. M., Brewer, P. E., Calderon, F. J. & Wallenstein, M. D. Redox and temperature-sensitive changes in microbial communities and soil chemistry dictate greenhouse gas loss from thawed permafrost. Biogeochemistry 134, 183–200 (2017).
Knoblauch, C., Beer, C., Liebner, S., Grigoriev, M. N. & Pfeiffer, E.-M. Methane production as key to the greenhouse gas budget of thawing permafrost. Nat. Clim. Change 8, 309–312 (2018).
Herndon, E., Kinsman-Costello, L. & Godsey, S. Biogeochemical Cycling of Redox-Sensitive Elements in Permafrost-Affected Ecosystems. in Biogeochemical Cycles 245–265 (American Geophysical Union (AGU), https://doi.org/10.1002/9781119413332.ch12.(2020)
Fiedler, S., Wagner, D., Kutzbach, L. & Pfeiffer, E.-M. Element Redistribution along Hydraulic and Redox Gradients of Low-Centered Polygons, Lena Delta, Northern Siberia. Soil Sci. Soc. Am. J. 68, 1002–1011 (2004).
Sulman, B. N. et al. Simulated Hydrological Dynamics and Coupled Iron Redox Cycling Impact Methane Production in an Arctic Soil. J. Geophys. Res. Biogeosciences 127, e2021JG006662 (2022).
Romanowicz, K. J., Crump, B. C. & Kling, G. W. Genomic evidence that microbial carbon degradation is dominated by iron redox metabolism in thawing permafrost. ISME Commun 3, 124 (2023).
Herndon, E. M. et al. Iron (Oxyhydr)Oxides Serve as Phosphate Traps in Tundra and Boreal Peat Soils. J. Geophys. Res. Biogeosciences 124, 227–246 (2019).
Kappler, A. et al. An evolving view on biogeochemical cycling of iron. Nat. Rev. Microbiol. 19, 360–374 (2021).
Gentsch, N. et al. Properties and bioavailability of particulate and mineral-associated organic matter in Arctic permafrost soils, Lower Kolyma Region, Russia. Eur. J. Soil Sci. 66, 722–734 (2015).
Chen, C., Hall, S. J., Coward, E. & Thompson, A. Iron-mediated organic matter decomposition in humid soils can counteract protection. Nat. Commun. 11, 2255 (2020).
Monhonval, A. et al. Mineral organic carbon interactions in dry versus wet tundra soils. Geoderma 436, 116552 (2023).
Elberling, B. et al. High-Arctic Soil CO2 and CH4 Production Controlled by Temperature, Water, Freezing and Snow. in Advances in Ecological Research 40, 441–472 (2008). Academic Press.
Voigt, C. et al. Arctic soil methane sink increases with drier conditions and higher ecosystem respiration. Nat. Clim. Change 13, 1095–1104 (2023).
Patzner, M. S. et al. Iron mineral dissolution releases iron and associated organic carbon during permafrost thaw. Nat. Commun. 11, 6329 (2020).
Barker, A. J. et al. Iron Oxidation–Reduction Processes in Warming Permafrost Soils and Surface Waters Expose a Seasonally Rusting Arctic Watershed. ACS Earth Space Chem 7, 1479–1495 (2023).
Lipson, D. A. et al. Changes in microbial communities along redox gradients in polygonized Arctic wet tundra soils. Environ. Microbiol. Rep. 7, 649–657 (2015).
Treat, C. C. et al. A pan-Arctic synthesis of CH4 and CO2 production from anoxic soil incubations. Glob. Change Biol. 21, 2787–2803 (2015).
Carlsen, E. C. L. et al. Redox determines greenhouse gas production kinetics and metabolic traits in water-saturated thawing permafrost peat. ISME Commun 5, ycaf009 (2025).
Street, L. E. et al. Redox dynamics in the active layer of an Arctic headwater catchment; examining the potential for transfer of dissolved methane from soils to stream water. J. Geophys. Res. Biogeosciences 121, 2776–2792 (2016).
Patzner, M. S. et al. Seasonal Fluctuations in Iron Cycling in Thawing Permafrost Peatlands. Environ. Sci. Technol. 56, 4620–4631 (2022).
Barczok, M. et al. Iron transformation mediates phosphate retention across a permafrost thaw gradient. Commun. Earth Environ. 5, 635 (2024).
Rooney, E. C. et al. Decoupling of redox processes from soil saturation in Arctic tundra. Commun. Earth Environ. 5, 746 (2024).
Obu, J. et al. Northern Hemisphere permafrost map based on TTOP modelling for 2000–2016 at 1 km2 scale. Earth-Sci. Rev. 193, 299–316 (2019).
van Bochove, E., Beauchemin, S. & Thériault, G. Continuous Multiple Measurement of Soil Redox Potential Using Platinum Microelectrodes. Soil Sci. Soc. Am. J. 66, 1813–1820 (2002).
Hodges, C. et al. Using fixed-potential electrodes to quantify iron and manganese redox cycling in upland soils. Biogeochemistry 162, 25–42 (2023).
Thomas, C. R., Miao, S. & Sindhoj, E. Environmental factors affecting temporal and spatial patterns of soil redox potential in Florida Everglades wetlands. Wetlands 29, 1133–1145 (2009).
Rodríguez-Escales, P., Barba, C., Sanchez-Vila, X., Jacques, D. & Folch, A. Coupling Flow, Heat, and Reactive Transport Modeling to Reproduce In Situ Redox Potential Evolution: Application to an Infiltration Pond. Environ. Sci. Technol. 54, 12092–12101 (2020).
Mansfeldt, T. In situ long-term redox potential measurements in a dyked marsh soil. J. Plant Nutr. Soil Sci. 166, 210–219 (2003).
Mansfeldt, T. Redox potential of bulk soil and soil solution concentration of nitrate, manganese, iron, and sulfate in two Gleysols. J. Plant Nutr. Soil Sci. 167, 7–16 (2004).
Dorau, K. & Mansfeldt, T. Comparison of redox potential dynamics in a diked marsh soil: 1990 to 1993 versus 2011 to 2014. J. Plant Nutr. Soil Sci. 179, 641–651 (2016).
Reiser, R., Stadelmann, V., Weisskopf, P., Grahm, L. & Keller, T. System for quasi-continuous simultaneous measurement of oxygen diffusion rate and redox potential in soil. J. Plant Nutr. Soil Sci. 183, 316–326 (2020).
Vorenhout, M., van der Geest, H. G., van Marum, D., Wattel, K. & Eijsackers, H. J. P. Automated and Continuous Redox Potential Measurements in Soil. J. Environ. Qual. 33, 1562–1567 (2004).
Fierer, N., Colman, B. P., Schimel, J. P. & Jackson, R. B. Predicting the temperature dependence of microbial respiration in soil: A continental-scale analysis. Glob. Biogeochem. Cycles 20, (2006).
Wang, C. et al. The temperature sensitivity of soil: microbial biodiversity, growth, and carbon mineralization. ISME J 15, 2738–2747 (2021).
Dorau, K., Bohn, B., Weihermüller, L. & Mansfeldt, T. Temperature-induced diurnal redox potential in soil. Environ. Sci. Process. Impacts 23, 1782–1790 (2021).
Rooney, E. C. et al. Topography and canopy cover influence soil organic carbon composition and distribution across a forested hillslope in the discontinuous permafrost zone. Permafr. Periglac. Process. 34, 331–358 (2023).
Liebmann, P. et al. Permafrost degradation and its consequences for carbon storage in soils of Interior Alaska. Biogeochemistry 167, 199–223 (2024).
Reddy, K. R. & DeLaune, R. D. Biogeochemistry of Wetlands: Science and Applications. (CRC Press, Boca Raton). https://doi.org/10.1201/9780203491454.(2008).
Outcalt, S. I., Nelson, F. E. & Hinkel, K. M. The zero-curtain effect: Heat and mass transfer across an isothermal region in freezing soil. Water Resour. Res. 26, 1509–1516 (1990).
Buettner, S. W., Kramer, M. G., Chadwick, O. A. & Thompson, A. Mobilization of colloidal carbon during iron reduction in basaltic soils. Geoderma 221–222, 139–145 (2014).
ThomasArrigo, L. K. & Kretzschmar, R. Iron speciation changes and mobilization of colloids during redox cycling in Fe-rich, Icelandic peat soils. Geoderma 428, 116217 (2022).
Ochsner, T. E., Horton, R. & Ren, T. A New Perspective on Soil Thermal Properties. Soil Sci. Soc. Am. J. 65, 1641–1647 (2001).
Wang, Z. P., DeLaune, R. D., Patrick, W. H. Jr. & Masscheleyn, P. H. Soil Redox and pH Effects on Methane Production in a Flooded Rice Soil. Soil Sci. Soc. Am. J. 57, 382–385 (1993).
Wang, H. et al. Methane-cycling microbiomes in soils of the pan-Arctic and their response to permafrost degradation. Commun. Earth Environ. 6, 748 (2025).
Ueyama, M., Iwata, H., Endo, R. & Harazono, Y. Methane and carbon dioxide emissions from the forest floor of a black spruce forest on permafrost in interior Alaska. Polar Sci 35, 100921 (2023).
Lacroix, E. M. et al. Consider the Anoxic Microsite: Acknowledging and Appreciating Spatiotemporal Redox Heterogeneity in Soils and Sediments. ACS Earth Space Chem 7, 1592–1609 (2023).
Chen, S. et al. Freeze-thaw strength increases microbial stability to enhance diversity-soil multifunctionality relationship. Commun. Earth Environ. 5, 578 (2024).
Knight, C. G. et al. Soil microbiomes show consistent and predictable responses to extreme events. Nature 636, 690–696 (2024).
Ji, X., Liu, M., Yang, J. & Feng, F. Meta-analysis of the impact of freeze–thaw cycles on soil microbial diversity and C and N dynamics. Soil Biol. Biochem. 168, 108608 (2022).
Lundin, L.-C. & Johnsson, H. Ion dynamics of a freezing soil monitored in situ by time domain reflectometry. Water Resour. Res. 30, 3471–3478 (1994).
Schimel, J. P. & Clein, J. S. Microbial response to freeze-thaw cycles in tundra and taiga soils. Soil Biol. Biochem. 28, 1061 (1996).
Haei, M. et al. Effects of soil frost on growth, composition and respiration of the soil microbial decomposer community. Soil Biol. Biochem. 43, 2069–2077 (2011).
Hugelius, G. et al. Estimated stocks of circumpolar permafrost carbon with quantified uncertainty ranges and identified data gaps. Biogeosciences 11, 6573–6593 (2014).
Gentsch, N. et al. Storage and transformation of organic matter fractions in cryoturbated permafrost soils across the Siberian Arctic. Biogeosciences 12, 4525–4542 (2015).
Hall, S. J., Huang, W., Napieralski, S. A. & Roden, E. Shared Microbial Taxa Respond Predictably to Cyclic Time-Varying Oxygen Limitation in Two Disparate Soils. Front. Microbiol. 13, (2022).
Dorau, K., Uteau, D., Hövels, M. P., Peth, S. & Mansfeldt, T. Soil aeration and redox potential as function of pore connectivity unravelled by X-ray microtomography imaging. Eur. J. Soil Sci. 73, e13165 (2022).
Sapkota, Y., Duball, C., Vaughan, K., Rabenhorst, M. & Berkowitz, J. Indicator of Reduction in Soil (IRIS) devices: A review. Sci. Total Environ. 852, 158419 (2022).
Dorau, K. & Mansfeldt, T. Manganese-Oxide-Coated Redox Bars as an Indicator of Reducing Conditions in Soils. J. Environ. Qual. 44, 696–703 (2015).
Mansfeldt, T. Soil redox potential. in Field Measurement Methods in Soil Science 210 (Gebr. Borntraeger Verlagsbuchhandlung, Stuttgart, 2019).
Liebmann, P. et al. Freeze-thaw cycle experiments [Data set]. Leibniz Centre for Agricultural Landscape Research (ZALF) https://doi.org/10.4228/ZALF-VXTQ-FK90 (2025).
Liebmann, P. et al. Soil monitoring dataset Fairbanks Alaska [Data set]. Leibniz Centre for Agricultural Landscape Research (ZALF) https://doi.org/10.4228/ZALF-J9XQ-Q235 (2025).
Alaska Climate Research Center - Data Portal. https://akclimate.org/data/data-portal/ (2025).
Acknowledgements
Oliver Donnerhack is acknowledged for his help during field work and Tino Peplau for helpful discussions about redox monitoring data. This study was funded by the Deutsche Forschungsgemeinschaft (DFG) and the Czech Science Foundation (GAČR) within the framework of the Joint German-Czech Project “CRYOVULCAN—Vulnerability of carbon in Cryosols”, with the individual grants GU 406/35-1, UR 198/41, VO 2111/6-1, GAČR project n. 20-21259 J. We are grateful for comments from 3 reviewers which helped to improve the manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
Funding was acquired by G.G., T.U., C.V., and J.B. The study and experimental field setup was designed by P.L., G.G., J.B., T.U., O.S., and C.V. Field installations and data collection were done by P.L., C.V., G.G., M.W., M.V., H.W., and A.K. Laboratory experiments were designed and conducted by P.L., S.W.-B., and T.M. Data processing and evaluation was done by P.L. P.L. wrote the first draft of the manuscript with contributions from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Earth and Environment thanks the anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editors: Kalliopi Violaki, Somaparna Ghosh [A peer review file is available.]
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liebmann, P., Vogel, C., Kholodov, A. et al. Perennial redox potential dynamics in Alaskan degraded and non-degraded permafrost soils. Commun Earth Environ 7, 120 (2026). https://doi.org/10.1038/s43247-025-03143-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43247-025-03143-x