Abstract
Stable isotopes are widely used to investigate plant-water interactions, yet many studies report differences in deuterium (δ2H) ratios between plants and source waters (termed δ2H offsets). These δ2H offsets challenge the assumption that water uptake and transport in plants do not alter the isotopic composition of water. Here, we develop a conceptual framework that distinguishes three soil water pools and two plant water pools based on water potential. We synthesize data from 110 published studies across 212 field sites worldwide and reanalyze 6333 δ2H offset measurements using possible source water lines. We find that δ2H offsets are absent when using appropriate water pools (e.g., use sap flow water rather than bulk xylem tissue water to represent plant water), with mean offsets not statistically different from zero. We argue that accurately separating correct water pools from bulk water pools in soils and plants is key to reconciling the observed δ2H offsets.
Similar content being viewed by others
Introduction
The global flux of water through terrestrial plants (i.e., transpiration) accounts for about two-thirds of land evapotranspiration and shapes ecosystems, climates, and natural resources1,2,3. Understanding where water comes from and how water moves through the soil-vegetation-atmosphere continuum is important for predicting the impacts of global climate change and anthropogenic activities on critical hydrological processes and ecosystem services4,5. One of the most effective tools for quantifying plant water uptake patterns and linking plant ecology to hydrologic flows is the analysis of stable isotope ratios in environmental water pools and fluxes6,7,8,9,10. Global isotopic databases of hydrogen (δ2H) and oxygen (δ18O) in different environmental water stores accumulated over the past six decades have enabled examinations of local, regional, and global-scale land-atmosphere interactions11,12, evapotranspiration partitioning1,13,14,15, and plant water source identifications16.
Although Lin and Sternberg17 and Ellsworth and Williams18 noted that hydrogen isotope fractionation did occur for halophytic species that relied heavily on the symplastic root water uptake pathways in saline environments, it is generally acknowledged that the biological processes of root water uptake and water transport within terrestrial plants do not result in isotope fractionation19. However, over the three decades following Lin and Sternberg’s work, significant δ2H offsets between plant and source waters have been increasingly documented in non-halophyte species across both natural settings and controlled experiments (hydroponic or soil-potted), with far fewer corresponding reports for δ18O values10,20,21,22,23,24,25,26,27,28. Such δ2H offsets between plant and source waters challenge the reliability of isotope methods in ecohydrological studies based on the assumption of isotopic consistency during plant water uptake and transport and subsequent water extraction processes4,10,20,21,22,27,28,29,30. For instance, the widespread observations of δ2H offsets raise concerns about the accuracy of previously reported global and seasonal patterns of plant water uptake16,31,32,33 and their underlying drivers12,16,34, as these studies often did not account for such offsets. The reporting of δ2H offsets has also renewed the unresolved debates regarding whether the isotope ratios of soil water accessible to plants remain separated from those of water rapidly bypassing soils and recharging open channels (i.e., the “ecohydrological separation” or “ecohydrological connectivity” hypothesis)1,4,35,36. Given the widespread practice of using the isotopic signatures of bulk woody xylem water as proxies for those of plant-available soil water, the observed δ2H separation between soil water accessible to plants and preferential soil water recharging streams could also result from δ2H changes during plant water use processes (i.e., δ2H offsets) in addition to “ecohydrological separation” within soils. Additionally, isotope-based evapotranspiration partitioning methods typically leverage isotopic differences between transpired water vapor, consistent with source waters, and evaporated water vapor, often exhibiting a δ2H-depletion compared to source waters37. This isotopic difference between transpiration and evaporation provides a basis for separating their contributions to total evapotranspiration1,37,38,39. However, deviations of transpired vapor from the isotopic composition of source waters (i.e., δ2H offsets) may result in an increasing or decreasing disparity in isotope values between transpiration and evaporation, consequently leading to inaccurate estimations of transpiration’s contribution to overall evapotranspiration. As such, it is urgently necessary to rigorously examine the fidelity of isotope methods used in ecohydrological investigations.
The δ2H offset between plant and source waters may result from (1) inherent δ2H changes deriving from isotope fractionation during root water uptake and subsequent water transport from roots to leaves, (2) deceptive δ2H changes due to methodological artifacts imparted during the water extraction process, (3) inaccurate δ2H signature measurements due to improper water pool extractions or mixing of distinct water pools within soils and plants, or potentially all three factors combined. A series of greenhouse experiments20,40,41 and a xylem sap bleeding experiment for the field-grown Populus euphratica21 demonstrated that the within-stem isotope heterogeneity, which can be derived from isotope fractionation during plant water use, could be the primary cause for δ2H offsets between plants and source waters20,21,40,41. In contrast, another greenhouse experiment reported that kinetic hydrogen fractionation during cryogenic extraction is the primary cause of δ2H offsets, resulting from dynamic exchange between organically bound δ2H and liquid water during extraction26. An extensive catchment observation27 and a global meta-analysis28 encompassing different plant species, climate zones, and water extraction methods quantified a highly variable δ2H offset between bulk xylem water and its sources, ranging from −57.2‰ to 36.3‰, with the mean value of −5.1‰ (p < 0.05) and −3.0‰ (p < 0.05), respectively. They further identified that such significant δ2H offsets with high variability cannot be attributed solely to methodological artifacts, but also to various environmental factors, such as air temperature and relative humidity, which can affect leaf physiology and, consequently, hydrogen isotope fractionation processes within plants across different climate zones. More recently, a field study quantified contributions of methodological artifacts and within-stem isotope heterogeneities to plant-source water δ2H offsets, accounting for 75.1% and 24.9%, respectively10. Overall, we posit that it is essential to eliminate deceptive δ2H offsets resulting from both methodological artifacts and mixing water pool measurements before questioning the reliability of isotope methods in ecohydrological investigations and undertaking attribution analysis for the inherent δ2H offsets induced by the within-plant isotope fractionation. Failure to do so compromises the reliability of interpretations regarding the causes and fidelity of the observed δ2H offsets.
The goal of this study is to thoroughly evaluate the fidelity and causes of δ2H offsets. Here, we test a key hypothesis that δ2H offsets do not occur if the correct water pools, with accurate δ2H signatures, are collected using appropriate extraction methods in isotope studies. We will first construct a water potential-based framework that accounts for water flow and corresponding isotope heterogeneities within the soil-vegetation-atmosphere continuum (Fig. 1). Plant waters can be separated into two distinct pools with unique isotope signatures: the non-fractionated sap flow water and the fractionated non-conducting xylem tissue water (termed the “two plant water pools” concept, Fig. 1A). It is worth noting that the “two plant water pools” concept simplifies isotopic heterogeneities among various non-conducting xylem tissue water pools, which may arise from isotopic fractionation during processes, such as membrane transport, differences in water residence time, and internal water mixing dynamics23,42,43. Similarly, we identify three distinct soil water pools with unique isotope signatures within the “three soil water pools” concept (Fig. 1B): gravitational soil water, which drains freely through the soil profile; plant-available soil water, retained in mesopores and micropores and accessible to roots; and hygroscopic soil water, which forms thin films tightly bound to soil particles and is unavailable to plants. This framework will guide us to accurately separate water pools within plants and soils for isotope-based ecohydrological investigations (Fig. 1C). To test this framework, a global re-quantification of δ2H offsets between plant water and the “possible-source” water line (PWL) is performed using a linear regression that integrates the isotopic signatures of all possible water sources across different combinations of three soil water pools. Both the proposed conceptual framework and the re-evaluation of the global δ2H offset dataset presented in this study will help comprehensively assess and verify the robustness and reliability of isotope methods in ecohydrological investigations.
A The “two plant water pools” concept; B The “three soil water pools” concept;, C The correct water pools within the soil-plant continuum deriving from diverse flow heterogeneities. The water extraction methods and their corresponding extraction range concerning water potential, pore diameter, and different water pools. The soil water extraction methods, along with their corresponding ranges of matric potential and pore diameter, were adapted from Sprenger et al.60. The “two plant water pools” concept refers to the sap flow water pool within xylem conduits and surrounding non-conducting xylem tissue water. The “three soil water pools” concept refers to gravitational soil water, plant-available soil water, and hygroscopic soil water. The plant-available soil water pool and the sap flow water pool within plant xylem conduits are recognized as appropriate water pools for isotope-based ecohydrological investigations.
Establishing a new framework
A new “three soil water worlds” concept to understand the actual sources of plant water use
An essential prerequisite for obtaining the actual sources of plant water use is to correctly extract all possible source waters. For natural terrestrial plants, all the environmental waters, including precipitation, soil water at different depths, groundwater, river water (for riparian plants), fog, dew, and water vapor, can be possible water sources. However, most recent δ2H offset-related studies have considered only a subset of below-ground water sources (i.e., soil water and groundwater) as possible sources for plant water use10,21,27,44,45,46,47. Precipitation was often combined with surface soil water to represent one water source with highly variable isotope signatures. Water uptake from above-ground plant parts (e.g., leaves and bark) is often overlooked in δ2H offset-related studies1,4,16,20,23,48. Occult moisture, such as fog, dew, and water vapor in the air, has been observed to provide critical water sources for plants across a range of biomes49,50,51,52,53,54,55,56,57. Therefore, the absence of isotopic signatures of these critical water sources may lead to deceptive δ2H offsets27,48.
More importantly, water flow heterogeneity within soils is a product of complex factors, such as tension variations, storage time, and the manner in which water flows through soil pores58,59. Such heterogeneous water flow within soils could result in isotopic heterogeneities on the soil pore scale36. The currently recognized “ecohydrological separation” hypothesis is conceptualized as a classification of soil isotopic heterogeneities between fast-flowing soil water recharging into open channels and slow-flowing soil water accessible for plant transpiration9,59. To tackle the challenge of accurately extracting the plant-available soil water pool, the urgency lies in quantitatively determining the range of soil water potential and pore diameter for the plant-available soil water. It is widely recognized that the field capacity of soil and the permanent wilting point are the upper and lower limits of plant-available soil water, between −0.01 and −1.5 MPa in soil water potential and between 0.2 and 30 µm in soil pore diameter60,61,62,63, though variations in wilting points have been observed across biomes64,65 and plant species66,67,68. Measurements of the sap flow in plant xylem conduits suggest that roots prefer to take up plant-available soil water in fine pores1,9,20,21,60. The gravitational water pool in soil water potential > −0.01 MPa (i.e., gravitational water between maximum water-holding capacity and field capacity) drains rapidly and is typically unavailable for plant uptake. The hygroscopic water pool in soil water potential < −1.5 MPa (i.e., hygroscopic water below the permanent wilting point) exists as thin films tightly bound to soil particles and cannot be accessible for plant roots60,69. Revisiting the physical conceptualization of different water pools in isotope-based ecohydrological studies helps address the inherent limitations of the “two water worlds” hypothesis in soils, which we argue inaccurately characterizes the isotopic dynamics of plant-available soil water. Instead, we advocate for a “three soil water pools” concept that encompasses the gravitational soil water pool, the plant-available soil water pool, and the hygroscopic soil water pool (Fig. 1B). This three-pool concept offers a simplified framework for what is likely a continuous spectrum of water potentials within soils. Nevertheless, the transition from a two-pool to a three-pool soil water concept effectively encapsulates the diversity of soil water stores. Although some previous isotope-based ecohydrological studies reported the heterogeneous isotopic signatures of soil water in terms of the “two water worlds” hypothesis20,23,70, only a few studies have directly identified the isotopic compositions of plant-available soil water pool from the perspective of the “three soil water pools” concept20,26,71. The vague or non-specific soil water pool extraction could also be a primary reason for the observed δ2H offsets between plants and source waters.
Some comparative studies encompassing different extraction methods (e.g., lysimeters, wick samplers, mechanical squeezing, centrifugation, distillation, cryogenic vacuum distillation, CO2/hydrogen equilibration, and direct water vapor equilibration) have systematically summarized sampled pore-size distributions and extracted different water pools at the pore scale60,72,73,74,75. While these methods have greatly advanced our understanding of soil water extraction, growing evidence suggests that many of them may not reliably separate the plant-available soil water pool (Fig. 1B). From the perspective of the “three soil water pools” concept, most of the pore water stable isotope analysis methods, including lysimeters, wick samplers, and mechanical squeezing, were often used to extract the gravitational soil water pool and were not able to collect the entire plant- available soil water pool due to the limitation of their maximum tension of 0.05–0.07 MPa60. Some distillation methods, such as azeotropic distillation and cryogenic vacuum distillation, were initially designed to extract bulk soil water (i.e., all three soil water pools)60,76. When gravitational water is present, these methods may struggle to accurately isolate the plant-available soil water pool. Conversely, when soil is at or below field capacity (i.e., lacking gravitational water), the extracted water may include both plant-available soil water and hygroscopic soil water. It is worth noting that the CO2/hydrogen equilibration method, which typically captures soil water within a potential range of −0.01 to −1 MPa, may effectively exclude hygroscopic water and instead separate the plant-available water pool when gravitational soil water is absent77,78. In addition, to identify isotopic signatures of the plant-available soil water pool, some existing hydroponic- and sandy-soil-potted experiments have established well-mixed water conditions by using hydroponic water or, in soil experiments, achieving near-complete replacement of soil water with irrigation water17,18,20,24,26,41,79,80. They simultaneously prevent evaporation-derived water isotopic heterogeneities within possible water sources for plants. These potted experiments can offer precise and reliable isotopic characteristics of the plant-available soil water pool. However, many soil- and plant-isotope studies conducted under natural conditions remain unresolved. Over the recent decade, in situ measurements based on liquid-vapor isotope equilibrium have been recognized as a consistent, integrated (soils and plants) methodology to address these issues81,82,83. This approach has been used to target the gravitational water fraction held at relatively low tensions, typically within a water potential range of approximately 0 to –10 MPa81,82,84,85. Notably, it can also differentiate the plant-available water pool in the absence of gravitational soil water. Nevertheless, accurately identifying the exact water potential threshold at which vapor–liquid isotopic equilibrium occurs remains a challenge. Centrifuge approaches, with rotation speeds up to 14,000 rpm, have been documented to extract the plant-available water pool within a certain range of soil water potential by controlling centrifugation speed60, indicating that the plant-available water pool can be separated from the bulk soil water20,30,86. However, this requires detailed site-specific soil properties, including soil water potentials at field capacity (the upper limit of plant-available water) and permanent wilting point (the lower limit of plant-available water), for heterogeneous soil profiles. Until now, only a few studies have successfully separated the exact plant-available water pools from bulk soil water using the centrifugation method10,29. Here, we emphasize the importance of moving toward community-accepted standardization, such as the potential use of centrifugation, the CO2/hydrogen equilibration method, and in situ measurements based on the liquid-vapor isotope equilibrium, while acknowledging that many methodological details remain unresolved10,20,29,71.
A new “two plant water worlds” concept within plants
Evidence of heterogeneous water flows within plants dates back to 201021,38,87,88. These pioneering studies showed that the isotopic signatures of sap flow water within conductive xylem conduits and the steady-state transpiration vapor remain consistent with the designed water sources of plants, whereas bulk xylem water does not show such consistency10,20,21,26,79,80. Upward sap flows from roots to leaves are driven by the passive transpiration process, which is tightly controlled by the energy difference between soil water with a higher potential and water in the atmosphere with a lower potential89,90,91,92. These sap flows that support transpiration loss have been documented to account for more than 95% of total plant water uptake93,94,95,96 and are able to stably reflect the isotopic composition of source water due to the well-mixed, homogeneous water pool within xylem conduits10,20,21. In addition to the xylem sap flow water, three other non-conducting xylem tissue water pools might also exist within woody stems, including intracellular water within living cells (such as growing xylem cells, phloem, and parenchyma), capillary water within apoplastic intercellular spaces between xylem cells, and fiber water within cell walls97,98. Both biological processes, including downward transport of enriched leaf water, radial water transport across different membrane boundaries, and internal water residence time, and environmental cues, have been recognized as critical factors in isotopic heterogeneities within woody stems10,20,69,99,100. Nevertheless, the sap flow within xylem conduits for transpiration would not displace or dilute non-conducting xylem tissue waters20,21. To ensure isotopic consistency during plant water uptake and transport in future isotope-based ecohydrological investigations, a simplified “two plant water pools” concept is proposed in this study. This concept highlights the isotopic heterogeneities between non-fractionated sap flow water directly used for transpiration and fractionated non-conducting xylem tissue water used for plant growth and metabolism. In the meantime, this concept simplifies the isotopic heterogeneities between various non-conducting xylem tissue water pools, integrating them into a unified fractionated non-conducting xylem tissue water pool. This framework enables a clearer distinction between sap flow water and bulk xylem water in isotope-based ecohydrological studies.
Thus far, the concept of sap flow water within plant xylem conduits has not yet been widely adopted as a standard water pool in isotope-based ecohydrological studies10,101. Most previous studies have relied on bulk woody xylem water extractions, which can obscure isotopic heterogeneities between sap flow water and surrounding xylem tissue water. While these pools may equilibrate over longer time scales102, treating them as a single isotopic reservoir under dynamic conditions may compromise the integrity of δ²H interpretations related to root water uptake and internal transport, potentially leading to confounded or misleading conclusions. The absence of a standardized plant water extraction method may hamper robust, spatiotemporal examinations of δ2H offsets between plants and source waters20,41,103. We emphasize this not to discredit previous approaches, but to highlight a promising direction for improving the accuracy and interpretability of isotope-based ecohydrological studies. The biological water potential across plant organs varies widely in response to factors, such as soil water availability, atmospheric water status, plant species, light intensity, and physiological characteristics20,104,105,106,107. The upper and lower thresholds of the sap flow water pool vary dynamically across temporal and spatial scales (Fig. 1A), making it difficult to establish a universal extraction criterion. Therefore, accurately and quantitatively separating the “two plant water pools”—sap flow water within xylem conduits versus surrounding non-conducting xylem tissue water—through the plant water potential method remains a significant technical challenge108,109. For example, distillation-based methods may often extract from both water stores, including sap flow water pool within plant xylem conduits and non-conducting xylem tissue water10,20,26,86,110. Recently, some studies applied a constant empirical value in plant water potential to separate sap flow water pool from bulk xylem water using a centrifuge or pressure chamber20,41,71,86. While this approach offers a practical approximation, it does not fully capture the dynamic nature of plant water potentials, which vary over time, across species, and in response to environmental conditions111. As a result, using a fixed threshold may not accurately isolate the entire sap flow water pool, potentially leading to partial or inconsistent extractions across different temporal and spatial contexts. Our previous study10 proposed that the most mobile part of the sap flow water pool, which is preferentially released from bulk xylem water pools during water extraction, can represent the entire sap flow water pool in isotope-based ecohydrological investigations due to the well-mixed, isotopically homogeneous water pool within xylem conduits. It is worth noting that as little as 0.5 mL of sap flow water is sufficient for stable isotope analysis. Consequently, the most mobile fraction of xylem sap flow water, amounting to just 0.5 mL and under the lowest tension, could serve as a representative proxy for the entire sap flow water pool. However, its small volume makes it more susceptible to evaporative enrichment and more difficult to isolate accurately. Direct water vapor equilibration and in situ measurements based on liquid-vapor isotope equilibrium were applied to collect sap flow water at lower tension within a water potential range of approximately 0 to –10 MPa81,82,84,85. However, it remains uncertain whether the water potential threshold at which vapor–liquid isotopic equilibrium occurs aligns precisely with, or falls within, the threshold for xylem sap flow, given the dynamic nature of plant water potentials. This raises an important question: to what extent can vapor equilibration-based methods reliably isolate the true sap flow water pool without inadvertently integrating woody non-conducting xylem tissue water? Additionally, flow-through chambers have been widely recognized as a direct method for quantifying the stable isotope composition of plant transpired water vapor, which is a powerful indicator of the whole sap flow water pool within the xylem conduit38,87.
Midday plant water potential reflects the peak tension exerted by plants to access water under maximum transpiration demand and thus serves as a physiologically meaningful upper limit for identifying the portion of soil water accessible to plants112. However, operationalizing this conceptual threshold requires detailed site-specific information, including soil water retention curves to accurately link plant water potentials to corresponding soil matric potentials and pore size distributions. In heterogeneous field conditions, particularly in soils with layered textures, high gravel content, or variable structures, such information is often incomplete or uncertain. Additionally, midday plant water potential exhibits substantial variability across species, ecosystems, and climatic conditions, complicating the development of a universally applicable protocol. Despite these challenges, we emphasize that aligning soil water extraction thresholds with the measured plant water potential represents a critical advancement toward integrating plant physiological function with hydrological process understanding. As sensor technologies, in situ monitoring techniques, and modeling frameworks improve, future research will enable the development of standardized yet adaptable methodologies for isolating physiologically meaningful water pools in both soils and plants. Such approaches could significantly improve the accuracy and comparability of isotope-based ecohydrological studies.
Examining δ2H offsets using the new framework
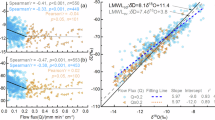
To address the conflicting δ2H offset observations and test our key hypothesis—that δ2H offsets do not occur when correct water pools are used in isotope studies—we conducted a global synthesis to quantify the magnitude of isotope offsets between plants and possible water sources (Fig. 2A). We also examined the extent to which these offsets are caused by both methodological artifacts and improper water pool extractions, which are supposed to be deceptive and eliminable. Our data synthesis shows that the isotope signatures of groundwater, four combinations of the three soil water pools, and two combinations of the two plant water pools have been documented (Supplementary Information Fig. 1). Specifically, the global isotopic datasets consist of hydrogen (2H/1H) and oxygen (18O/16O) isotope ratios for bulk soil water (n = 6430), gravitational soil water (n = 539), the combined plant-available and hygroscopic soil water (n = 2034), groundwater (n = 695), plant-available soil water (n = 13), xylem sap flow water (n = 20), and bulk xylem water (n = 5759) across 212 globally distributed field sites (Supplementary Information Fig. 1). Globally, gravitational soil water and groundwater plot approximately along the global meteoric water line (GMWL) (Supplementary Information Fig. 1B, D). While the bulk soil water, the combined plant-available and hygroscopic soil water, and bulk xylem water plot both along and below the GMWL (Supplementary Fig. 1A, C, F). The plant-available soil water matched well with the xylem sap flow water (Supplementary Information Fig. 1E). Ultimately, a dataset comprising 6,333 PWL-based δ2H offsets was recalculated and categorized according to distinct water pools extracted from bulk soil and plant xylem waters, based on the proposed framework of “three soil water pools” and “two plant water pools” (Fig. 2 and Table 1).
A The global distribution of past research sites in dryland and non-dryland ecosystems is evaluated here. The aridity index (AI), defined as the ratio of precipitation to potential evapotranspiration, was used to classify climate zones as follows: hyper-arid (AI < 0.03), arid (0.03 ≤ AI < 0.2), semi-arid (0.2 ≤ AI < 0.5), dry sub-humid (0.5 ≤ AI ≤ 0.65), and humid (AI > 0.65). The AI data and base map were derived from the Version 3 product of the global aridity index and potential evapotranspiration (ET0) Database116. Sites overlap due to multiple samples or water extraction methods in a small area. B Schematic diagram of the quantification method of PW-excess, SW-excess, and LC-excess. The line-condition excess (LC-excess) describes the δ2H offset of the water samples from the local meteoric water line (LMWL)117. The SW-excess is defined as the δ2H offset between plant water and the soil water line (SWL)22. The PW-excess is proposed to quantify the δ2H offsets between plant water and the PWL48. PW-excess could provide more accurate information on isotope offsets between plants and source waters because PWL considers more possible water sources that plants use than both LMWL and SWL. C Violin plots of the distribution of five-scenario δ2H offsets considering different water pool combinations within soils and plants. Violin widths represent kernel density distributions. Box plots within violins show means (centre lines), medians (two square dots), interquartile ranges (boxes) and whiskers extending to 1.5 times the interquartile range, and outliers (points). Values above each violin indicate sample size (N) and mean ± standard deviation. “**” above each violin represents there is significant difference between δ2H offsets and zero (p > 0.05), while “NS” indicates offsets not statistically different from zero (p > 0.05). δ2H offsets in the “bulk soil vs. bulk xylem” scenario represent δ2H offsets between the PWL based on bulk soil water (i.e., three soil water pools) and bulk woody xylem water (i.e., two plant water pools). δ2H offsets in the “gravitational vs. bulk xylem” scenario represent δ2H offsets between the PWL based on gravitational soil water and bulk woody xylem water. δ2H offsets in the “plant-available + hygroscopic vs. bulk xylem” scenario represent δ2H offsets between the PWL based on the combination of plant-available and hygroscopic soil water (i.e., two water pools including plant-available soil water and hygroscopic soil water) and bulk xylem water. δ2H offsets in the “plant-available + hygroscopic vs. sap flow” scenario represent δ2H offsets between the PWL based on the combination of plant-available and hygroscopic soil water and sap flow water. δ2H offsets in the “plant-available vs. sap flow” scenario represent δ2H offsets between the PWL based on plant-available soil water and sap flow water.
Based on our global data synthesis, a total of five δ2H offset scenarios of different water pool combinations within soils and plants have been categorized to quantify the magnitude of δ2H offsets (Fig. 2): (1) bulk soil water vs. bulk xylem water, (2) gravitational soil water vs. bulk xylem water, (3) combined plant-available and hygroscopic soil water vs. bulk xylem water, (4) combined plant-available and hygroscopic soil water vs. sap flow water, and (5) plant-available soil water vs. sap flow water. Our findings showed that the δ2H offsets between bulk xylem water and the PWL based on bulk soil water (−4.9 ± 8.2‰), between bulk xylem water and the PWL based on gravitational soil water (−10.3 ± 8.2‰), and between bulk xylem water and the PWL based on the combination of plant-available and hygroscopic soil water (1.1 ± 10.1‰) were significantly different from zero (p < 0.01, Fig. 2C). In contrast, the mean δ2H offsets between sap flow water and the PWL based on the combination of plant-available and hygroscopic soil water (0.8 ± 0.8‰, p = 0.14), as well as between sap flow water and the PWL based on plant-available soil water (0.1 ± 0.7‰, p = 0.62) are close to zero (Fig. 2C and Table 2). The observed differences in δ²H offsets across the five scenarios with distinct water pools suggest that improper water-pool extractions or mixing water-pool measurements can result in significant, highly variable δ²H offsets (Fig. 2C). It is worth noting that δ2H fractionation induced by methodological artifacts during cryogenic vacuum distillation extraction processes could also contribute to δ²H offsets, potentially amplifying the effects of mixed water-pool measurements. Nevertheless, our results reveal a more pronounced negative δ2H offset associated with gravitational soil water pools compared to both bulk soil water pools and the combined plant-available and hygroscopic soil water pools (p < 0.05, Fig. 2C). This suggests that improper collection of gravitational soil water pools likely play a more significant role in driving substantial δ²H offsets, compared to δ²H fractionation induced by methodological artifacts. In contrast, the inappropriate collection of the hygroscopic soil water pool has a smaller effect on δ2H offsets, which may be attributed to the smaller volume of hygroscopic water within the soil profile113. The average δ2H offset between sap flow water and the PWL, based on the plant-available soil water, exhibited the minimal value (0.1 ± 0.7‰) with the least deviation among all five scenarios and showed no statistically significant difference from zero20,26. This result supports our key hypothesis that no δ2H offset arises when appropriate extraction methods and accurate water pools are used. We argue that accurate separation of the correct water pool from bulk water pools within soils or plants, using appropriate extraction methods, is key to reconciling the observed contradictory δ2H offsets.
The results of two potted experimental studies20,26 that reported similar hydrogen isotope signatures for the plant-available soil water pool and the sap flow water pool further support the hypothesis of the absence of inherent δ2H offsets. In addition, a field-grown riparian Populus euphratica21, reliant solely on groundwater, provides evidence that its sap flow water aligned consistently with its exclusive water source-groundwater. Despite these encouraging findings, the vast majority of published studies to date—98.3% of the 110 studies we reviewed (see Table 1)—have employed water-sampling methods unlikely to reliably isolate the water pools most relevant to transpiration. These legacy approaches, while foundational to the development of ecohydrological investigations, often relied on extraction techniques that do not discriminate against different water pools in either soil or plant tissues. As a result, our global data synthesis and δ2H offset re-quantifications cast doubt on the reliability of prior interpretations of the global and seasonal patterns of critical ecohydrological pathways. For instance, many recent ecohydrology studies used isotopic differences between gravitational soil water (or stream water) and bulk woody xylem water in dual-isotope space (δ2H vs. δ18O) to confirm the “ecohydrological separation” hypothesis between gravitational soil water recharging to open channels and plant-available soil water. Indeed, water flow heterogeneities (i.e., our “two plant water pools” concept) within plants can lead to substantial isotopic differences between bulk xylem water and unfractionated sap flow water. Consequently, inferences drawn from the “ecohydrological separation” hypothesis are generally confounded. Therefore, there is an urgent need for a framework that integrates recent technical innovations and physiological evidence to advance community-accepted standardization extraction approaches and more accurately characterize plant water-use patterns. Our work will help guide the ecohydrological community toward more comparable isotope-based investigations by highlighting the limitations of past approaches and identifying best practices for future sampling.
A key limitation of our analysis is the limited number of studies that meet the ideal sampling criteria necessary to distinguish the proper water pools in both soils and plants. Among the existing literature, only a few studies—primarily those involving potted plants—have accurately captured the isotopic signatures of the relevant water pools. However, this scarcity of ideal datasets is not a shortcoming of the conceptual framework we propose, but rather a reflection of the current state of ecohydrological research. This lack of suitable data underscores the urgent need for more physiologically and hydrologically rigorous sampling approaches. Our intention is not to overstate conclusions based on limited data, but to highlight and promote emerging best practices that are foundational for improving future ecohydrological investigations. In addition, we recognize that our reliance on publicly available datasets, originally collected under heterogeneous methodological assumptions and technical constraints, introduces inherent limitations and variabilities. Nonetheless, this data-driven synthesis serves as a critical first step in advancing the field toward a more mechanistic and ecologically meaningful understanding of plant–soil water interactions. By identifying methodological gaps and proposing pathways for improved sampling, we aim to conduct further research that can validate and refine this framework under field-relevant conditions.
Conclusions
Recent observations revealed significant differences in δ2H values between plants and source waters (termed δ2H offsets), with a large range of −57.2‰ to 36.3‰. These challenge the fundamental assumption that no isotope fractionation occurs during plant water uptake and transport or during subsequent water-extraction processes. In addition, it is unclear what causes substantial δ2H offsets and whether they can be eliminated. To account for the isotopic heterogeneities of soils and plants, we propose a water potential-derived framework of the “three soil water pools” concept and the “two plant water pools” concept. This proposed framework will aid in accurately separating the plant-available water pool from bulk soil water, and the sap flow water pool from bulk woody xylem water, which is key to eliminating the “deceptive” δ2H offsets. We hypothesize that observed δ2H offsets are likely due to inaccurate δ2H signature collection caused by methodological artifacts or improper water-pool extraction, rather than δ2H fractionation during plant water use. To test this, we used 110 published studies across 212 globally distributed field sites to re-quantify 6,333 measurements of δ2H offsets between plant water and the “possible-source” water line, a linear regression line that integrates isotopic characteristics of all water sources for different combinations of three soil water pools. The δ2H offsets between sap flow water and “possible-source” water line amount to 0.1 ± 0.7‰ and show no statistical difference from zero (p > 0.05). Therefore, our global literature synthesis and δ2H offset re-quantifications showed that δ2H offsets would be absent when appropriate extraction methods and accurate water pools were used, supporting our hypothesis. Accurately separating the correct water pools from bulk water pools in soils and plants using appropriate extraction methods is key to reconciling the observed contradictory δ2H offsets. This framework, which provides a critical lens for interpreting plant-soil water heterogeneity, will help guide the ecohydrological community toward more reliable and comparable isotope-based investigations by highlighting the limitations of past approaches and identifying best practices for future sampling. Our findings are crucial for enhancing our capacity to understand and predict the vegetation responses and feedback to future global water fluxes.
Methods
We built a database from peer-reviewed journal articles published in English during the last four decades (i.e., from 1985 to 2024) using the search engines Web of Science and Google Scholar using “(water AND isotop*) AND (plant OR tree OR vegetat*) AND (dual OR (hydrogen AND oxygen))” as the search query, to ensure that we include all studies using isotopes to determine plant water sources. We selected studies that report (1) dual-isotope values for both plant and source waters, (2) sufficient source water data (n ≥ 3) to fit a PWL, and (3) soil water status to diagnose different water worlds within soils. Halophytic species living in saline environments were excluded from the dataset to avoid the influence of δ2H fractionation induced by the symplastic root water uptake pathway associated with salt tolerance17,18. Data were derived directly from tables or figures using the GetData Graph Digitizer software v2.26 (http://getdata-graph-digitizer.com/download.php). The data collection benefited from recent isotope data synthesis efforts to investigate the plant groundwater use and isotope separation between plant transpiration and groundwater/streamwater4,114,115. It is noted that the measurement precision for δ18O and δ2H has not changed significantly since the 1980s, even though measurement speeds are much faster, so earlier publications will not affect the synthesis results.
A total of 110 published studies across 212 globally distributed field sites have been synthesized in this study (Supplementary Information References). The site-specific information, such as latitude, longitude, mean annual precipitation, mean annual temperature, potential evapotranspiration, ecosystem type, climate zone, groundwater level, the range of soil water content, soil bulk density, soil texture, soil water potential, soil water extraction methods, soil depths, plant species, plant water extraction methods, and stable isotopes of various source waters and plant waters was recorded if they are available. The re-classification of three soil water pools and two plant water pools was carefully examined in this study based on the following categorical variables derived from the literature: (i) climatic conditions (e.g., drylands and non-drylands based on the aridity index, the ratio between potential evapotranspiration and precipitation), (ii) groundwater levels, (iii) soil water conditions (e.g., soil depths, soil water contents, soil water potentials, and soil textures), (iv) water extraction methods, and (v) environmental conditions (e.g., hydroponic-potted or sandy soil-potted experiments and natural field experiments). Gravitational soil water would be considered to be available in the site if one of the below situations occurs in the literature: (a) the maximum soil water content within soil profiles is more than the field capacity; (b) irrigation or preferential flow exists; (c) soil water potential exceeds the field capacity threshold; (d) groundwater is regarded as one of the potential water sources for plants; (e) in riparian zones; and (f) gravitational soil water have been extracted and their corresponding isotopic compositions are available. We classified the soil water isotope dataset at the site as representing the “bulk soil water pool” when they were obtained using distillation- or equilibrium-based extraction methods, and when evidence of gravitational soil water was concurrently reported at the same site. Accordingly, we acknowledge that some datasets categorized as representing “bulk soil water pool” in our global synthesis, particularly those collected from different soil depths at the same site, may reflect mixtures of the three conceptual soil water pools (gravitational, plant-available, and hygroscopic soil water), or subset thereof (the combination of plant-available and hygroscopic soil water pools, or the combination of gravitational and plant-available soil water pools).
The “possible-source water excess” (PW-excess), introduced in our previous studies5,10,48, is utilized in this study to quantify the δ2H offsets between plant water and the PWL (Fig. 2B). It is defined as PW-excess = δ2H–apδ18O−bp, where ap and bp are the slope and intercept of the PWL determined by a linear regression fit through all possible source waters for different combinations of three soil water pools. We used all available source water data from each study during one growing season to construct the PWL. In short, PW-excess (i.e., δ2H offsets) is calculated as the vertical deviation of the plant water’s isotope value from the PWL (i.e., how far the plant’s δ²H falls above or below the source-water line). PW-excess offers more accurate quantifications of δ2H offsets between plants and source waters, as the PWL accounts for all possible water sources utilized by plants compared to the local meteoric water line and soil water line typically employed in previous studies20,22,27,48 (Fig. 2B). We consider different combinations of soil water pools at varying depths as potential sources of water for plant uptake. Additionally, alternative sources such as groundwater, spring water, fog, and dew are considered possible sources, provided their use by plants has been documented in previous studies. In this study, 9.4% of the potential source water isotope datasets had a coefficient of determination (R2) below 0.5 in the linear regression analysis. To improve the model fit for these datasets, non-linear regression models, specifically, quadratic and cubic equations, were subsequently applied. We compared the performance of linear and non-linear models by examining R2 values; when R2 values were comparable, model selection was further guided by the Akaike information criterion (AIC), a widely used metric that balances model fit and complexity. In cases where non-linear models showed a markedly improved R2 or lower AIC value, we ultimately evaluated differences in the estimated δ2H offsets by comparing their means and standard deviations between linear and non-linear regression models. This comparison revealed that non-linear regressions often produced higher average δ2H offsets and greater variability, suggesting that these models may overfit the data in certain cases, particularly when driven by outliers or noise rather than true curvature in the isotopic relationship. Consequently, while non-linear models may capture some complex dynamics, caution is warranted in their application, and linear models may offer more robust and interpretable estimates in δ2H offset datasets with limited sample sizes or high variance. The underlying compiled dataset is publicly available via Figshare (see Data availability). The one-way analysis of variance (ANOVA) was applied to examine differences between PWL-based δ2H offsets and zero. All statistical analyses were conducted using Microsoft Excel (v2016) and R (3.4.1).
Data availability
The datasets supporting the findings of this study are publicly available in the Figshare repository at https://doi.org/10.6084/m9.figshare.31076299. Supplementary Information references provide a complete list of the 110 published studies synthesized in this study.
References
Good, S. P., Noone, D. & Bowen, G. Hydrologic connectivity constrains partitioning of global terrestrial water fluxes. Science 349, 175–177 (2015).
Wang, K. & Dickinson, R. E. A review of global terrestrial evapotranspiration: observation, modeling, climatology, and climatic variability. Rev. Geophys. 50, 1–54 (2012).
Wang, L., Good, S. P. & Caylor, K. K. Global synthesis of vegetation control on evapotranspiration partitioning. Geophys. Res. Lett. 41, 6753–6757 (2014).
Evaristo, J., Jasechko, S. & McDonnell, J. J. Global separation of plant transpiration from groundwater and streamflow. Nature 525, 91–107 (2015).
Li, Y. et al. Contrasting water use characteristics of riparian trees under different water tables along a losing river. J. Hydrol. 611, 128017 (2022).
Dawson, T. E. & Ehleringer, J. R. Streamside trees that do not use stream water. Nature 350, 335–337 (1991).
Dawson, T. E., Mambelli, S., Plamboeck, A. H., Templer, P. H. & Tu, K. P. Stable isotopes in plant ecology. Annu. Rev. Ecol. Syst. 33, 507–559 (2002).
Ehleringer, J. R. & Dawson, T. E. Water-uptake by plants-perspectives from stable isotope composition. Plant Cell Environ. 15, 1073–1082 (1992).
Brooks, J. R., Barnard, H. R., Coulombe, R. & McDonnell, J. J. Ecohydrologic separation of water between trees and streams in a mediterranean climate. Nat. Geosci. 3, 100–104 (2010).
Li, Y., Song, X., Wang, L., Sprenger, M. & Ma, Y. Quantitative contribution of cryogenic vacuum extraction and radial water transport to xylem-source water deuterium offsets. Agric. For. Meteorol. 345, 109837 (2024).
Yakir, D. & Wang, X.-F. Fluxes of CO2 and water between terrestrial vegetation and the atmosphere estimated from isotope measurements. Nature 380, 515–517 (1996).
Dubbert, M. & Werner, C. Water fluxes mediated by vegetation: emerging isotopic insights at the soil and atmosphere interfaces. N. Phytol. 221, 1754–1763 (2019).
Dubbert, M., Cuntz, M., Piayda, A. & Werner, C. Oxygen isotope signatures of transpired water vapor: the role of isotopic non-steady-state transpiration under natural conditions. N. Phytol. 203, 1242–1252 (2014).
Sutanto, S. J. et al. A perspective on isotope versus non-isotope approaches to determine the contribution of transpiration to total evaporation. Hydrol. Earth Syst. Sci. 18, 2815–2827 (2014).
Sutanto, S. J., Wenninger, J., Coenders-Gerrits, A. M. J. & Uhlenbrook, S. Partitioning of evaporation into transpiration, soil evaporation and interception: a comparison between isotope measurements and a HYDRUS-1D model. Hydrol. Earth Syst. Sci. 16, 2605–2616 (2012).
Miguez-Macho, G. & Fan, Y. Spatiotemporal origin of soil water taken up by vegetation. Nature 598, https://doi.org/10.1038/s41586-021-03958-6 (2021).
Lin, G. & da S. L. Sternberg, L. Stable Isotopes and Plant Carbon-water Relations (eds Ehleringer, J.R., Hall, A.E. & Farquhar, G.D.) 497–510 (Academic Press, 1993).
Ellsworth, P. Z. & Williams, D. G. Hydrogen isotope fractionation during water uptake by woody xerophytes. Plant Soil 291, 93–107 (2007).
Jasechko, S. et al. Terrestrial water fluxes dominated by transpiration. Nature 496, 347–352 (2013).
Barbeta, A. et al. Evidence for distinct isotopic compositions of sap and tissue water in tree stems: consequences for plant water source identification. N. Phytol. 233, 1121–1132 (2022).
Zhao, L. J. et al. Significant difference in hydrogen isotope composition between xylem and tissue water in Populus Euphratica. Plant Cell Environ. 39, 1848–1857 (2016).
Barbeta, A. et al. Unexplained hydrogen isotope offsets complicate the identification and quantification of tree water sources in a riparian forest. Hydrol. Earth Syst. Sci. 23, 2129–2146 (2019).
Barbeta, A. et al. An explanation for the isotopic offset between soil and stem water in a temperate tree species. N. Phytol. 227, 766–779 (2020).
Roden, J. S. & Ehleringer, J. R. Hydrogen and oxygen isotope ratios of tree-ring cellulose for riparian trees grown long-term under hydroponically controlled environments. Oecologia 121, 467–477 (1999).
Vargas, A. I., Schaffer, B., Li, Y. & Sternberg, L. D. S. L. Testing plant use of mobile vs immobile soil water sources using stable isotope experiments. N. Phytol. 215, 582–594 (2017).
Chen, Y. et al. Stem water cryogenic extraction biases estimation in deuterium isotope composition of plant source water. Proc. Natl. Acad. Sci. USA 117, 33345–33350 (2020).
Zhao, L. et al. The determining factors of hydrogen isotope offsets between plants and their source waters. N. Phytol. https://doi.org/10.1111/nph.19492 (2024).
De la Casa, J. et al. Isotopic offsets between bulk plant water and its sources are larger in cool and wet environments. Hydrol. Earth Syst. Sci. 26, 4125–4146 (2022).
McDonnell, J. J. The two water worlds hypothesis: ecohydrological separation of water between streams and trees? Wiley Interdiscip. Rev. Water 1, 323–329 (2014).
Wen, M. et al. Causes and factors of cryogenic extraction biases on isotopes of xylem water. Water Resour. Res. 58, https://doi.org/10.1029/2022wr032182 (2022).
Allen, S. T., Kirchner, J. W., Braun, S., Siegwolf, R. T. W. & Goldsmith, G. R. Seasonal origins of soil water used by trees. Hydrol. Earth Syst. Sci. 23, 1199–1210 (2019).
Barbeta, A. & Penuelas, J. Relative contribution of groundwater to plant transpiration estimated with stable isotopes. Sci. Rep. 7, https://doi.org/10.1038/s41598-017-09643-x (2017).
Geris, J., Tetzlaff, D., McDonnell, J. J. & Soulsby, C. Spatial and temporal patterns of soil water storage and vegetation water use in humid northern catchments. Sci. Total Environ. 595, 486–493 (2017).
De Deurwaerder, H. P. T. et al. Causes and consequences of pronounced variation in the isotope composition of plant xylem water. Biogeosciences 17, 4853–4870 (2020).
Maxwell, R. M. & Condon, L. E. Connections between groundwater flow and transpiration partitioning. Science 353, 377–380 (2016).
Finkenbiner, C. E., Good, S. P., Brooks, J. R., Allen, S. T. & Sasidharan, S. The extent to which soil hydraulics can explain ecohydrological separation. Nat. Commun. 13, https://doi.org/10.1038/s41467-022-34215-7 (2022).
Xiao, W., Wei, Z. & Wen, X. Evapotranspiration partitioning at the ecosystem scale using the stable isotope method—A review. Agric. For. Meteorol. 263, 346–361 (2018).
Wang, L., Good, S. P., Caylor, K. K. & Cernusak, L. A. Direct quantification of leaf transpiration isotopic composition. Agric. For. Meteorol. 154, 127–135 (2012).
Dubbert, M., Cuntz, M., Piayda, A., Maguas, C. & Werner, C. Partitioning evapotranspiration-testing the Craig and Gordon model with field measurements of oxygen isotope ratios of evaporative fluxes. J. Hydrol. 496, 142–153 (2013).
Geißler, K., Heblack, J., Uugulu, S. & Wanke, H. Partitioning of water between differently sized shrubs and potential groundwater recharge in a semiarid savanna in Namibia. Front. Plant Sci. 10, 462149 (2019).
Zuecco, G. et al. A comparative study of plant water extraction methods for isotopic analyses: Scholander-type pressure chamber vs. cryogenic vacuum distillation. Hydrol. Earth Syst. Sci. 26, 3673–3689 (2022).
Dubbert, M., Caldeira, M. C., Dubbert, D. & Werner, C. A pool-weighted perspective on the two-water-worlds hypothesis. N. Phytol. 222, 1271–1283 (2019).
von Freyberg, J., Allen, S. T., Grossiord, C. & Dawson, T. E. Plant and root-zone water isotopes are difficult to measure, explain, and predict: some practical recommendations for determining plant water sources. Methods Ecol. Evol. 11, 1352–1367 (2020).
Wang, J., Fu, B. J., Lu, N. & Li, Z. Seasonal variation in water uptake patterns of three plant species based on stable isotopes in the semi-arid Loess Plateau. Sci. Total Environ. 609, 27–37 (2017).
Chen, G. et al. Isotopic fractionation induced by a surface effect influences the estimation of the hydrological process of topsoil. Hydrol. Process. 35, e14019 (2021).
Zhang, B. et al. Ecohydrological separation between tree xylem water and groundwater: insights from two types of forests in subtropical China. Plant Soil https://doi.org/10.1007/s11104-022-05607-x (2022).
Zhao, Y. & Wang, L. Insights into the isotopic mismatch between bulk soil water and Salix matsudana Koidz trunk water from root water stable isotope measurements. Hydrol. Earth Syst. Sci. 25, 3975–3989 (2021).
Li, Y., Ma, Y., Song, X., Wang, L. & Han, D. A δ2H offset correction method for quantifying root water uptake of riparian trees. J. Hydrol. 593, 125811 (2021).
Goldsmith, G. R., Lehmann, M. M., Cernusak, L. A., Arend, M. & Siegwolf, R. T. Inferring foliar water uptake using stable isotopes of water. Oecologia 184, 763–766 (2017).
Emery, N. C. Foliar uptake of fog in coastal California shrub species. Oecologia 182, 731–742 (2016).
Burgess, S. & Dawson, T. The contribution of fog to the water relations of Sequoia sempervirens (D. Don): foliar uptake and prevention of dehydration. Plant Cell Environ. 27, 1023–1034 (2004).
Gerlein-Safdi, C. et al. Dew deposition suppresses transpiration and carbon uptake in leaves. Agric. For. Meteorol. 259, 305–316 (2018).
Hill, A. J., Dawson, T. E., Dody, A. & Rachmilevitch, S. Dew water-uptake pathways in Negev desert plants: a study using stable isotope tracers. Oecologia 196, 353–361 (2021).
Agam, N. & Berliner, P. R. Dew formation and water vapor adsorption in semi-arid environments—a review. J. Arid Environ. 65, 572–590 (2006).
Limm, E. B., Simonin, K. A., Bothman, A. G. & Dawson, T. E. Foliar water uptake: a common water acquisition strategy for plants of the redwood forest. Oecologia 161, 449–459 (2009).
Wang, L., Kaseke, K. F. & Seely, M. K. Effects of non-rainfall water inputs on ecosystem functions. Wiley Interdiscip. Rev. Water 4, https://doi.org/10.1002/wat2.1179 (2017).
Wang, L. et al. Convergent vegetation fog and dew water use in the Namib Desert. Ecohydrology 12, https://doi.org/10.1002/eco.2130 (2019).
Good, S. P., Moore, G. W. & Miralles, D. G. A mesic maximum in biological water use demarcates biome sensitivity to aridity shifts. Nat. Ecol. Evol. 1, 1883 (2017).
Sprenger, M. & Allen, S. T. What ecohydrologic separation is and where we can go with it. Water Resour. Res. 56, 1 (2020).
Sprenger, M., Herbstritt, B. & Weiler, M. Established methods and new opportunities for pore water stable isotope analysis. Hydrol. Process. 29, 5174–5192 (2015).
Foth, H. D. Fundamentals of Soil Science. John Wiley and Sons, Inc. (1951).
Schj⊘ nning, P. Size distribution of dispersed and aggregated particles and of soil pores in 12 Danish soils. Acta Agric. Scand. B Plant Soil Sci. 42, 26–33 (1992).
Schachtschabel, P., Blume, H.-P. & Thiele-Bruhn, S. Lehrbuch der Bodenkunde (Enke Stuttgart, 1976).
Bassiouni, M., Good, S. P., Still, C. J. & Higgins, C. W. Plant water uptake thresholds inferred from satellite soil moisture. Geophys. Res. Lett. 47, e2020GL087077 (2020).
Wood, J. D., Gu, L., Hanson, P. J., Frankenberg, C. & Sack, L. The ecosystem wilting point defines drought response and recovery of a Quercus-Carya forest. Glob. Change Biol. 29, 2015–2029 (2023).
Torres, L. et al. Impacts of soil type and crop species on permanent wilting of plants. Geoderma 384, 114798 (2021).
Rai, R. K., Singh, V. P. & Upadhyay, A. Planning and Evaluation of Irrigation Projects (eds Rai, R.K., Singh, V. P. & Upadhyay, A.) 505–523 (Academic Press, 2017).
Wiecheteck, L. H. et al. Comparing the classical permanent wilting point concept of soil (− 15,000 hPa) to biological wilting of wheat and barley plants under contrasting soil textures. Agric. Water Manag. 230, 105965 (2020).
Khan, M. S. et al. Empirical relations for optical attenuation prediction from liquid water content of fog. Radioengineering 21, 911–916 (2012).
Bowling, D. R., Schulze, E. S. & Hall, S. J. Revisiting streamside trees that do not use stream water: can the two water worlds hypothesis and snowpack isotopic effects explain a missing water source? Ecohydrology 10, 1–12 (2017).
Millar, C. et al. On the urgent need for standardization in isotope-based ecohydrological investigations. Hydrol. Process. 36, https://doi.org/10.1002/hyp.14698 (2022).
Orlowski, N. et al. Inter-laboratory comparison of cryogenic water extraction systems for stable isotope analysis of soil water. Hydrol. Earth Syst. Sci. 22, 3619–3637 (2018).
Orlowski, N., Pratt, D. L. & McDonnell, J. J. Intercomparison of soil pore water extraction methods for stable isotope analysis. Hydrol. Process. 30, 3434–3449 (2016).
Oerter, E. J. & Bowen, G. J. Spatio-temporal heterogeneity in soil water stable isotopic composition and its ecohydrologic implications in semiarid ecosystems. Hydrol. Process. 33, 1724–1738 (2019).
West, A. G., Goldsmith, G. R., Brooks, P. D. & Dawson, T. E. Discrepancies between isotope ratio infrared spectroscopy and isotope ratio mass spectrometry for the stable isotope analysis of plant and soil waters. Rapid Commun. Mass Spectrom. 24, 1948–1954 (2010).
Osman, K. T. & Osman, K. T. Soil water, irrigation, and drainage. in Soils: Principles, properties and management, 67–88 https://doi.org/10.2134/agronmonogr14.c6 (2013).
Figueroa-Johnson, M. A., Tindall, J. A. & Friedel, M. A comparison of 18 Oδ composition of water extracted from suction lysimeters, centrifugation, and azeotropic distillation. Water Air Soil Pollut. 184, 63–75 (2007).
West, A. G., Patrickson, S. J. & Ehleringer, J. R. Water extraction times for plant and soil materials used in stable isotope analysis. Rapid Commun. Mass Spectrom. 20, 1317–1321 (2006).
Kuebert, A. et al. Tracing plant source water dynamics during drought by continuous transpiration measurements: an in-situ stable isotope approach. Plant Cell Environ. 46, 133–149 (2023).
Amin, A. et al. No evidence of isotopic fractionation in olive trees (Olea europaea): a stable isotope tracing experiment. Hydrol. Sci. J. 66, 2415–2430 (2021).
Beyer, M., Kühnhammer, K. & Dubbert, M. In situ measurements of soil and plant water isotopes: a review of approaches, practical considerations and a vision for the future. Hydrol. Earth Syst. Sci. 24, 4413–4440 (2020).
Volkmann, T. H. M., Haberer, K., Gessler, A. & Weiler, M. High-resolution isotope measurements resolve rapid ecohydrological dynamics at the soil-plant interface. N. Phytol. 210, 839–849 (2016).
Volkmann, T. H. M., Kuehnhammer, K., Herbstritt, B., Gessler, A. & Weiler, M. A method for in situ monitoring of the isotope composition of tree xylem water using laser spectroscopy. Plant Cell Environ. 39, 2055–2063 (2016).
Marshall, J. D., Cuntz, M., Beyer, M., Dubbert, M. & Kuehnhammer, K. Borehole equilibration: testing a new method to monitor the isotopic composition of tree xylem water in situ. Front. Plant Sci. 11, 2020 (2020).
Magh, R. K. et al. Technical note: conservative storage of water vapour–practical in situ sampling of stable isotopes in tree stems. Hydrol. Earth Syst. Sci. 26, 3573–3587 (2022).
Wen, M. et al. Inter-comparison of extraction methods for plant water isotope analysis and its indicative significance. J. Hydrol. 625, 130015 (2023).
Wang, L. X. et al. Partitioning evapotranspiration across gradients of woody plant cover: assessment of a stable isotope technique. Geophys. Res. Lett. 37, 7 (2010).
Good, S. P. et al. δ2H isotopic flux partitioning of evapotranspiration over a grass field following a water pulse and subsequent dry down. Water Resour. Res. 50, 1410–1432 (2014).
Turner, N. C. Techniques and experimental approaches for the measurement of plant water status. Plant Soil 58, 339–366 (1981).
Breshears, D. D. et al. Tree die-off in response to global change-type drought: mortality insights from a decade of plant water potential measurements. Front. Ecol. Environ. 7, 185–189 (2009).
Kirkham, M. B. Principles of soil and plant water relations (Elsevier, 2023).
Parkash, V. & Singh, S. A review on potential plant-based water stress indicators for vegetable crops. Sustainability 12, 3945 (2020).
Sharma, P. K. & Kumar, S. Soil Physical Environment and Plant Growth: Evaluation and Management 33–71 (Springer International Publishing, 2023).
Kamakura, M., Tsuruta, K., Azuma, W. A. & Kosugi, Y. Hydraulic architecture and internal water storage of Japanese cypress using measurements of sap flow and water potential. Ecohydrology 14, e2325 (2021).
Mahara, Y., Ohta, T., Ohshima, J. & Iizuka, K. Origin and hydrodynamics of xylem sap in tree stems, and relationship to root uptake of soil water. Sci. Rep. 11, 8404 (2021).
Waring, R. H., Whitehead, D. & Jarvis, P. G. The contribution of stored water to transpiration in Scots pine. Plant, Cell Environ. 2, 309–317 (1979).
Tyree, M. T. & Yang, S. Water-storage capacity of thuja, tsuga and acer stems measured by dehydration isotherms: the contribution of capillary water and cavitation. Planta 182, 420–426 (1990).
Jupa, R., Plavcová, L., Gloser, V. & Jansen, S. Linking xylem water storage with anatomical parameters in five temperate tree species. Tree Physiol. 36, 756–769 (2016).
Dawson, T. E. & Ehleringer, J. R. Isotipic enrichment of water in the woody tissues of plants-implications for plant water source, water-uptake, and other studies which use the stable isotopic composition of cellulose. Geochim. Cosmochim. Acta 57, 3487–3492 (1993).
Nehemy, M. F. et al. Phloem water isotopically different to xylem water: potential causes and implications for ecohydrological tracing. Ecohydrology 15, https://doi.org/10.1002/eco.2417 (2022).
Orlowski, N. et al. Challenges in studying water fluxes within the soil-plant-atmosphere continuum: a tracer-based perspective on pathways to progress. Sci. Total Environ. 881, 163510 (2023).
Fabiani, G., Penna, D., Barbeta, A. & Klaus, J. Sapwood and heartwood are not isolated compartments: Consequences for isotope ecohydrology. Ecohydrology 15, https://doi.org/10.1002/eco.2478 (2022).
Ceperley, N. et al. Toward a common methodological framework for the sampling, extraction, and isotopic analysis of water in the critical zone to study vegetation water use. WIREs Water 11, e1727 (2024).
Li, W., Yu, T. F., Li, X. Y. & Zhao, C. Y. Sap flow characteristics and their response to environmental variables in a desert riparian forest along lower Heihe River basin, Northwest China. Environ. Monit. Assess. 188, https://doi.org/10.1007/s10661-016-5570-2 (2016).
Anderegg, W. R. L. & Venturas, M. D. Plant hydraulics play a critical role in Earth system fluxes. N. Phytol. 226, 1535–1538 (2020).
Grossiord, C. et al. Plant responses to rising vapor pressure deficit. N. Phytol. 226, 1550–1566 (2020).
Anderegg, W. R. L. Spatial and temporal variation in plant hydraulic traits and their relevance for climate change impacts on vegetation. N. Phytol. 205, 1008–1014 (2015).
Buckley, T. & Sack, L. Resolving micro-scale water potential gradients within leaves. J. Plant Hydraul. 10, 1 (2024).
Venturas, M. D., Sperry, J. S. & Hacke, U. G. Plant xylem hydraulics: what we understand, current research, and future challenges. J. Integr. Plant Biol. 59, 356–389 (2017).
Wen, M. et al. Correction of cryogenic vacuum extraction biases and potential effects on soil water isotopes application. J. Hydrol. 603, https://doi.org/10.1016/j.jhydrol.2021.127011 (2021).
Papastefanou, P. et al. A dynamic model for strategies and dynamics of plant water-potential regulation under drought conditions. Front. Plant Sci. 11, 373 (2020).
Naor, A. Midday stem water potential as a plant water stress indicator for irrigation scheduling in fruit trees. III International Symposium on Irrigation of Horticultural Crops. 537, 447–454 (1999).
Jaynes, D., Logsdon, S. & Horton, R. Field method for measuring mobile/immobile water content and solute transfer rate coefficient. Soil Sci. Soc. Am. J. 59, 352–356 (1995).
Abassi, M., Lamhamedi, M. S., Hachani, C. & Bejaoui, Z. Interspecific variability and morpho-physiological responses to salinity of Salix main mediterranean species. Can. J. For. Res. 49, 606–615 (2019).
Amin, A. et al. Depth distribution of soil water sourced by plants at the global scale: a new direct inference approach. Ecohydrology 13, e2177 (2020).
Zomer, R. J., Xu, J. & Trabucco, A. Version 3 of the global aridity index and potential evapotranspiration database. Sci. Data 9, 409 (2022).
Landwehr, J. M. & Coplen, T. B. Line-conditioned excess: a new method for characterizing stable hydrogen and oxygen isotope ratios in hydrologic systems. Vol. 92 (IAEA, 2006).
Acknowledgements
This work was supported by the Division of Earth Sciences of the National Science Foundation (EAR-1554894 and EAR-2444867).
Author information
Authors and Affiliations
Contributions
Yue Li and Lixin Wang designed the research, synthesized the database, conducted the analysis, and wrote the first draft. Stephen P. Good contributed ideas and revised the draft. All authors contributed to the writing and reviewing of the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Earth & Environment thanks Matthias Beyer and the other anonymous reviewer(s) for their contribution to the peer review of this work. Primary handling editors: Mengjie Wang. [A peer review file is available].
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, Y., Good, S.P. & Wang, L. Demystifying stable hydrogen isotope offsets between plants and source waters. Commun Earth Environ 7, 213 (2026). https://doi.org/10.1038/s43247-026-03230-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43247-026-03230-7