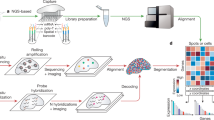
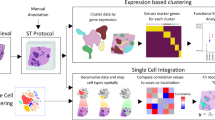
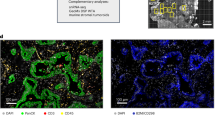
Abstract
Spatial transcriptomics has transformed the mapping of gene expression within intact tissues, yet current sequencing-based platforms are limited by coarse spot-level resolution and sparse sampling that leaves large interspot regions unmeasured. Here we introduce PanoSpace, a computational framework that integrates low-resolution spatial transcriptomics with high-resolution histology and matched single-cell RNA sequencing to reconstruct a continuous, single-cell-level map across entire tissue sections. Originally developed for tumors, PanoSpace accurately reconstructs cellular locations, cell identities and gene expression profiles, enabling detailed characterization of intracell-type heterogeneity and spatially organized cell–cell interactions. Application to breast and prostate cancers reveals complex cellular architectures and tumor microenvironment dynamics mediated by cancer-associated fibroblasts. Thanks to its modular design, PanoSpace can be seamlessly adapted to noncancerous tissues, as demonstrated by precise spatial reconstruction in mouse brain. Together, these results demonstrate that PanoSpace enables comprehensive spatial transcriptomic analysis and facilitates biological discovery.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All datasets used in this study are publicly available. For simulation experiments, the 10x Xenium breast cancer dataset7 was obtained from the 10x Genomics official website (https://www.10xgenomics.com/products/xenium-in-situ/preview-dataset-human-breast), with the paired human breast cancer scRNA-seq ref. 35 available from GEO under accession GSE176078. The 10x Xenium Prime 5K human lung cancer dataset was downloaded from https://www.10xgenomics.com/datasets/xenium-human-lung-cancer-post-xenium-technote, with the matched lung cancer scRNA-seq ref. 54 available at GSE131907. The 10x Xenium Prime 5K fresh-frozen mouse brain hemisphere dataset was obtained from https://www.10xgenomics.com/datasets/xenium-prime-fresh-frozen-mouse-brain, and the paired scRNA-seq ref. 55 can be accessed via the Allen Brain Map. For real data analysis, the 10x Visium human breast cancer dataset was downloaded from https://www.10xgenomics.com/datasets/human-breast-cancer-ductal-carcinoma-in-situ-invasive-carcinoma-ffpe-1-standard-1-3-0, with the same scRNA-seq reference as the Xenium breast cancer dataset. The 10x Visium human prostate cancer dataset was obtained from https://www.10xgenomics.com/datasets/human-prostate-cancer-adenocarcinoma-with-invasive-carcinoma-ffpe-1-standard-1-3-0, and the paired scRNA-seq ref. 38 was downloaded from the European Genome-Phenome Archive (EGA) under accession code EGAS00001005115. The 10x Visium adult mouse olfactory bulb dataset was downloaded from https://www.10xgenomics.com/datasets/adult-mouse-olfactory-bulb-1-standard, and the corresponding scRNA-seq ref. 56 is available at GSE121891. The PanNuke dataset47 can be accessed at https://warwick.ac.uk/fac/cross_fac/tia/data/pannuke. The ISH images from Allen Brain Atlas are available at https://mouse.brain-map.org/. Source data are provided with this paper.
Code availability
The source code of PanoSpace, along with Jupyter notebooks for reproducing the results in this study, is available via GitHub at https://github.com/hehuifeng/PanoSpace and is also available via Zenodo at https://doi.org/10.5281/zenodo.17579559 (ref. 57) under the MIT License.
References
Rao, A., Barkley, D., França, G. S. & Yanai, I. Exploring tissue architecture using spatial transcriptomics. Nature 596, 211–220 (2021).
Liu, Y. et al. High-spatial-resolution multi-omics sequencing via deterministic barcoding in tissue. Cell 183, 1665–1681 (2020).
Stahl, P. L. et al. Visualization and analysis of gene expression in tissue sections by spatial transcriptomics. Science 353, 78–82 (2016).
Larsson, L., Frisén, J. & Lundeberg, J. Spatially resolved transcriptomics adds a new dimension to genomics. Nat. Methods 18, 15–18 (2021).
Wang, X. et al. Three-dimensional intact-tissue sequencing of single-cell transcriptional states. Science 361, eaat5691 (2018).
Shah, S. et al. Dynamics and spatial genomics of the nascent transcriptome by intron seqFISH. Cell 174, 363–376 (2018).
Janesick, A. et al. High resolution mapping of the tumor microenvironment using integrated single-cell, spatial and in situ analysis. Nat. Commun. 14, 8353 (2023).
Moffitt, J. R. et al. Molecular, spatial, and functional single-cell profiling of the hypothalamic preoptic region. Science 362, eaau5324 (2018).
Hu, J. et al. Deciphering tumor ecosystems at super resolution from spatial transcriptomics with TESLA. Cell Syst. 14, 404–417 (2023).
Tu, J. J., Yan, H., Zhang, X. F. & Lin, Z. Precise gene expression deconvolution in spatial transcriptomics with STged. Nucleic Acids Res. 53, gkaf087 (2025).
Tu, J. J., Li, H. S., Yan, H. & Zhang, X. F. EnDecon: cell type deconvolution of spatially resolved transcriptomics data via ensemble learning. Bioinformatics 39, btac825 (2023).
Wang, M. G., Chen, L. & Zhang, X. F. Dual decoding of cell types and gene expression in spatial transcriptomics with PANDA. Nucleic Acids Res. 52, 12173–12190 (2024).
Biancalani, T. et al. Deep learning and alignment of spatially resolved single-cell transcriptomes with Tangram. Nat. Methods 18, 1352–1362 (2021).
Vahid, M. R. et al. High-resolution alignment of single-cell and spatial transcriptomes with CytoSPACE. Nat. Biotechnol. 41, 1543–1548 (2023).
Cable, D. M. et al. Robust decomposition of cell type mixtures in spatial transcriptomics. Nat. Biotechnol. 40, 517–526 (2022).
Dong, R. & Yuan, G. C. SpatialDWLS: accurate deconvolution of spatial transcriptomic data. Genome Biol. 22, 145 (2021).
Kleshchevnikov, V. et al. Cell2location maps fine-grained cell types in spatial transcriptomics. Nat. Biotechnol. 40, 661–671 (2022).
Lopez, R. et al. DestVI identifies continuums of cell types in spatial transcriptomics data. Nat. Biotechnol. 40, 1360–1369 (2022).
Elosua-Bayes, M., Nieto, P., Mereu, E., Gut, I. & Heyn, H. SPOTlight: seeded NMF regression to deconvolute spatial transcriptomics spots with single-cell transcriptomes. Nucleic Acids Res. 49, e50 (2021).
Andersson, A. et al. Single-cell and spatial transcriptomics enables probabilistic inference of cell type topography. Commun. Biol. 3, 565 (2020).
Sun, D., Liu, Z., Li, T., Wu, Q. & Wang, C. STRIDE: accurately decomposing and integrating spatial transcriptomics using single-cell RNA sequencing. Nucleic Acids Res. 50, e42 (2022).
Ma, Y. & Zhou, X. Spatially informed cell-type deconvolution for spatial transcriptomics. Nat. Biotechnol. 40, 1349–1359 (2022).
Liu, Z., Wu, D., Zhai, W. & Ma, L. SONAR enables cell type deconvolution with spatially weighted Poisson-Gamma model for spatial transcriptomics. Nat. Commun. 14, 4727 (2023).
Chen, J. et al. Cell composition inference and identification of layer-specific spatial transcriptional profiles with POLARIS. Sci. Adv. 9, eadd9818 (2023).
Zhang, D. et al. Inferring super-resolution tissue architecture by integrating spatial transcriptomics with histology. Nat. Biotechnol. 42, 1372–1377 (2024).
Bergenstrahle, L. et al. Super-resolved spatial transcriptomics by deep data fusion. Nat. Biotechnol. 40, 476–479 (2022).
He, B. et al. Integrating spatial gene expression and breast tumour morphology via deep learning. Nat. Biomed. Eng. 4, 827–834 (2020).
Wu, Y. et al. STASCAN deciphers fine-resolution cell distribution maps in spatial transcriptomics by deep learning. Genome Biol. 25, 278 (2024).
Fu, Y. C., Das, A., Wang, D., Braun, R. & Yi, R. scHolography: a computational method for single-cell spatial neighborhood reconstruction and analysis. Genome Biol. 25, 164 (2024).
Zhang, P. et al. Systematic inference of super-resolution cell spatial profiles from histology images. Nat. Commun. 16, 1838 (2025).
Wan, X. et al. Integrating spatial and single-cell transcriptomics data using deep generative models with SpatialScope. Nat. Commun. 14, 7848 (2023).
Hörst, F. et al. CellViT: vision transformers for precise cell segmentation and classification. Med. Image Anal. 94, 103143 (2024).
Grant Application Resources for Visium Spatial Gene Expression Document Number LIT000086 Rev H (10x Genomics, 2024).
Wolock, S. L., Lopez, R. & Klein, A. M. Scrublet: computational identification of cell doublets in single-cell transcriptomic data. Cell Syst. 8, 281–291 (2019).
Wu, S. Z. et al. A single-cell and spatially resolved atlas of human breast cancers. Nat. Genet. 53, 1334–1347 (2021).
Xu, C. et al. Probabilistic harmonization and annotation of single-cell transcriptomics data with deep generative models. Mol. Syst. Biol. 17, e9620 (2021).
Itoh, G. et al. Cancer-associated fibroblasts induce cancer cell apoptosis that regulates invasion mode of tumours. Oncogene 36, 4434–4444 (2017).
Wu, S. Z. et al. Cryopreservation of human cancers conserves tumour heterogeneity for single-cell multi-omics analysis. Genome Med. 13, 81 (2021).
Graham, S. et al. Hover-Net: simultaneous segmentation and classification of nuclei in multi-tissue histology images. Med. Image Anal. 58, 101563 (2019).
Lein, E. et al. Genome-wide atlas of gene expression in the adult mouse brain. Nature 445, 168–176 (2007).
Nagendran, M. et al. 1457 Visium HD enables spatially resolved, single-cell scale resolution mapping of FFPE human breast cancer tissue. J. Immunother. Cancer 11, A1620 (2023).
Chen, A. et al. Spatiotemporal transcriptomic atlas of mouse organogenesis using DNA nanoball-patterned arrays. Cell 185, 1777–1792 (2022).
Oquab, M. et al. DINOv2: learning robust visual features without supervision. Preprint at https://arxiv.org/abs/2304.07193 (2024).
Simonyan, K. & Zisserman, A. Very deep convolutional networks for large-scale image recognition. Preprint at https://arxiv.org/abs/1409.1556 (2014).
Lu, M. Y. et al. A visual-language foundation model for computational pathology. Nat. Med. 30, 863–874 (2024).
Maurer, K. et al. Coordinated immune networks in leukemia bone marrow microenvironments distinguish response to cellular therapy. Sci. Immunol. 10, eadr0782 (2025).
Gamper, J. et al. in Digital Pathology (eds Reyes-Aldasoro, C. et al.) 11–19 (Springer, 2019).
Flamary, R. et al. POT: Python optimal transport. J. Mach. Learn. Res. 22, 3571–3578 (2021).
Gurobi Optimizer Reference Manual (Gurobi Optimization, LLC, 2024).
Chapelle, O., Schölkopf, B. & Zien, A. Semi-Supervised Learning (MIT, 2006).
Belkin, M., Niyogi, P. & Sindhwani, V. Manifold regularization: a geometric framework for learning from labeled and unlabeled examples. J. Mach. Learn. Res. 7, 2399–2434 (2006).
Weigert, M. & Schmidt, U. Nuclei instance segmentation and classification in histopathology images with StarDist. In The IEEE International Symposium on Biomedical Imaging Challenges (ISBIC, 2022); https://doi.org/10.1109/ISBIC56247.2022.9854534
Yan, L., Cheng, J., Nie, Q. & Sun, X. Dissecting multilayer cell–cell communications with signaling feedback loops from spatial transcriptomics data. Genome Res. 35, 1400–1414 (2025).
Kim, N. et al. Single-cell RNA sequencing demonstrates the molecular and cellular reprogramming of metastatic lung adenocarcinoma. Nat. Commun. 11, 2285 (2020).
Yao, Z. et al. A taxonomy of transcriptomic cell types across the isocortex and hippocampal formation. Cell 184, 3222–3241 (2021).
Tepe, B. et al. Single-cell RNA-seq of mouse olfactory bulb reveals cellular heterogeneity and activity-dependent molecular census of adult-born neurons. Cell Rep. 25, 2689–2703 (2018).
He, H. F. PanoSpace: a framework for whole-slide single-cell spatial reconstruction in spatial transcriptomics. Zenodo https://doi.org/10.5281/zenodo.17579559 (2025).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant nos. 12271198 and 11871026 to X.-F.Z.; 62377019, 12131020, 31930022, T2350003, T2341007, 42450084, 42450135, 42450192, 12326614 and 1202660 to L.C.; and 12426672 to P.P.), the self-determined research funds of CCNU from the colleges’ basic research and operation of MOE (grant nos. CCNU25AI001, CCNU24JC004 and CCNU25JCPT029 to X.-F.Z.), the National Key R&D Program of China (grant nos. 2022YFA1004800 and 2025YFF1207900 to L.C.), the Zhejiang Province Vanguard Goose-Leading Initiative (grant no. 2025C01114 to L.C.), the Science and Technology Commission of Shanghai Municipality (grant no. 23JS1401300 to L.C.) and JST Moonshot R&D (grant no. JPMJMS2021 to L.C.).
Author information
Authors and Affiliations
Contributions
This study was conceived and led by X.-F.Z. and L.C. H.-F.H. developed and performed data analyses. H.-F.H. and X.-F.Z. wrote the paper. P.P., S.-T.Y. and M.-G.W. assisted with data analyses and paper writing. X.-F.Z. and L.C. supervised the entire project. All authors read and approved the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Computational Science thanks Min Wu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available. Primary Handling Editor: Ananya Rastogi, in collaboration with the Nature Computational Science team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Notes 1–6, Figs. 1–69 and Tables 1–20.
Source data
Source Data Fig. 2 (download XLSX )
Source data underlying Fig. 2b–f, including per-cell spatial expression values and cell-type annotations for Xenium and PanoSpace (Fig. 2b–e), as well as statistical data (Jensen–Shannon divergence values) for cell-type composition and gene-level predictions in breast and lung cancer samples (Fig. 2f). Each sheet corresponds to the dataset used for the respective panel.
Source Data Fig. 3 (download XLSX )
Source data underlying Fig. 3a–f, including per-cell or spot-level values used to generate cell-type composition maps and gene-expression comparisons across computational frameworks (PanoSpace, HisToCell, iStar, TESLA, Tangram, SpatialScope, CytoSPACE and EndoCon). The file also contains data for CAF–cancer epithelial interaction analyses, including ligand–receptor–target network information and importance scores quantifying the contribution of each ligand–receptor pair to downstream targets (Fig. 3d–f). Each sheet corresponds to the dataset used for the respective panel.
Source Data Fig. 4 (download XLSX )
Source data underlying Fig. 4a–f, including per-cell or spot-level values used to generate cell-type composition maps and gene-expression comparisons across computational frameworks (PanoSpace, HisToCell, iStar, TESLA, Tangram, SpatialScope, CytoSPACE and EndoCon). The file also contains data for CAF–cancer epithelial interaction analyses, including ligand–receptor–target network information and importance scores quantifying the contribution of each ligand–receptor pair to downstream targets (Fig. 4d–f). Each sheet corresponds to the dataset used for the respective panel.
Source Data Fig. 5 (download XLSX )
Source data underlying Fig. 5a–e, including per-cell or spot-level values used to generate cell-type distribution maps and gene-expression visualizations across computational frameworks (PanoSpace, Tangram, SpatialScope, CytoSPACE, iStar, TESLA and EnDecon) in mouse brain and olfactory bulb sections. The file also contains statistical data for cell-type and gene-level Jensen–Shannon divergence comparisons among methods (Fig. 5c). Each sheet corresponds to the dataset used for the respective panel.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
He, HF., Peng, P., Yang, ST. et al. Unlocking single-cell level and continuous whole-slide insights in spatial transcriptomics with PanoSpace. Nat Comput Sci (2026). https://doi.org/10.1038/s43588-025-00938-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43588-025-00938-y