Abstract
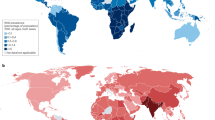
Rheumatic heart disease (RHD) is an acquired chronic inflammatory disease of the heart valves. Regarded as an autoimmune sequela of Streptococcus pyogenes infection, RHD is triggered by the development of carditis during acute rheumatic fever and persists as chronic rheumatic valvulitis in a proportion of patients with acute rheumatic fever. Permanent valve tissue damage ensues, often leading to heart failure. Effective interventions for established RHD are lacking and valve surgery is currently the only treatment option. The limited number of therapeutic targets for heart valve diseases reflects the complexities of studying the mechanisms underlying early valve pathobiology. However, technological advances now enable in-depth profiling of peripheral blood and valve tissue samples from people with RHD, opening new avenues to interrogate human-specific disease processes. In this Review, we revisit established immunological principles of RHD pathogenesis in light of emerging studies. We also explore both systemic and tissue-centric research gaps to advance our understanding of disease mechanisms and to identify human-relevant therapeutic strategies for RHD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Carapetis, J. R., Steer, A. C., Mulholland, E. K. & Weber, M. The global burden of group A streptococcal diseases. Lancet Infect. Dis. 5, 685–694 (2005).
Carapetis, J. R. et al. Acute rheumatic fever and rheumatic heart disease. Nat. Rev. Dis. Primers 2, 15084 (2016).
Ferrari, A. J. et al. Global incidence, prevalence, years lived with disability (YLDs), disability-adjusted life-years (DALYs), and healthy life expectancy (HALE) for 371 diseases and injuries in 204 countries and territories and 811 subnational locations, 1990–2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet 403, 2133–2161 (2024).
Coffey, S. et al. Global epidemiology of valvular heart disease. Nat. Rev. Cardiol. 18, 853–864 (2021).
Ojha, U. et al. Temporal trend analysis of rheumatic heart disease burden in high-income countries between 1990 and 2019. Eur. Heart J. Qual. Care Clin. Outcomes 10, 108–120 (2024).
Zühlke, L. et al. Clinical outcomes in 3343 children and adults with rheumatic heart disease from 14 low- and middle-income countries. Circulation 134, 1456–1466 (2016).
Katzenellenbogen, J. M. et al. The End Rheumatic Heart Disease in Australia Study of Epidemiology (ERASE) Project: data sources, case ascertainment and cohort profile. Clin. Epidemiol. 11, 997–1010 (2019).
Stacey, I. et al. Rheumatic heart disease mortality in Indigenous and non-Indigenous Australians between 2010 and 2017. Heart 109, 1025–1033 (2023).
Gewitz, M. H. et al. Revision of the jones criteria for the diagnosis of acute rheumatic fever in the era of Doppler echocardiography. Circulation 131, 1806–1818 (2015).
Gupta, M., Lent, R. W., Kaplan, E. L. & Zabriskie, J. B. Serum cardiac troponin I in acute rheumatic fever. Am. J. Cardiol. 89, 779–782 (2002).
Hirani, K. et al. Acute rheumatic fever. Lancet 405, 2164–2178 (2025).
Beaton, A. et al. Secondary antibiotic prophylaxis for latent rheumatic heart disease. New Engl. J. Med. 386, 230–240 (2022).
Remenyi, B. et al. Improved long-term survival for rheumatic mitral valve repair compared to replacement in the young. World J. Pediatr. Congenit. Heart Surg. 4, 155–164 (2013).
Karthikeyan, G. et al. Research priorities for the secondary prevention and management of acute rheumatic fever and rheumatic heart disease: a National Heart, Lung, and Blood Institute workshop report. BMJ Glob. Health 8, e012468 (2023).
Zühlke, L. et al. Characteristics, complications, and gaps in evidence-based interventions in rheumatic heart disease: the Global Rheumatic Heart Disease Registry (the REMEDY study). Eur. Heart J. 36, 1115–1122 (2015).
Steer, A. C. & Carapetis, J. R. Acute rheumatic fever and rheumatic heart disease in indigenous populations. Pediatr. Clin. North. Am. 56, 1401–1419 (2009).
Vervoort, D., Antunes, M. J. & Pezzella, A. T. Rheumatic heart disease: the role of global cardiac surgery. J. Card. Surg. 36, 2857–2864 (2021).
Cunningham, M. W. Molecular mimicry, autoimmunity, and infection: the cross-reactive antigens of group A Streptococci and their sequelae. Microbiol. Spectr. 7, 1128 (2019).
Guilherme, L., Kalil, J. & Cunningham, M. Molecular mimicry in the autoimmune pathogenesis of rheumatic heart disease. Autoimmunity 39, 31–39 (2006).
Kalil, J. & Guilherme, L. Rheumatic fever: a model of autoimmune disease due to molecular mimicry between human and pathogen proteins. Crit. Rev. Immunol. 40, 419–422 (2020).
Kaplan, M. H. & Suchy, M. L. Immunologic relation of streptococcal and tissue antigens: II. cross-reaction of antisera to mammalian heart tissue with a cell wall constituent of certain strains of group A Streptococci. J. Exp. Med. 119, 643–650 (1964).
Kaplan, M. H. & Svec, K. H. Immunologic relation of streptococcal and tissue antigens. J. Exp. Med. 119, 651–666 (1964).
Dale, J. B. & Beachey, E. H. Epitopes of streptococcal M proteins shared with cardiac myosin. J. Exp. Med. 162, 583–591 (1985).
Cunningham, M. W. et al. Human and murine antibodies cross-reactive with streptococcal M protein and myosin recognize the sequence GLN-LYS-SER-LYS-GLN in M protein. J. Immunol. 143, 2677–2683 (1989).
Gulizia, J. M., Cunningham, M. W. & McManus, B. M. Immunoreactivity of anti-streptococcal monoclonal antibodies to human heart valves. Evidence for multiple cross-reactive epitopes. Am. J. Pathol. 138, 285–301 (1991).
Van De Rijn, I., Sabriskie, J. & McCarty, M. Group A streptococcal antigens cross-reactive with myocardium. Purification of heart-reactive antibody and isolation and characterization of the streptococcal antigen. J. Exp. Med. 146, 579–599 (1977).
Shikhman, A. R., Greenspan, N. S. & Cunningham, M. W. A subset of mouse monoclonal antibodies cross-reactive with cytoskeletal proteins and group A streptococcal M proteins recognizes N-acetyl-β-D-glucosamine. J. Immunol. 151, 3902–3913 (1993).
Galvin, J. E., Hemric, M. E., Ward, K. & Cunningham, M. W. Cytotoxic mAb from rheumatic carditis recognizes heart valves and laminin. J. Clin. Invest. 106, 217–224 (2000).
Guilherme, L. et al. Human heart–infiltrating t-cell clones from rheumatic heart disease patients recognize both streptococcal and cardiac proteins. Circulation 92, 415–420 (1995).
Martins, T. B. et al. Comprehensive analysis of antibody responses to streptococcal and tissue antigens in patients with acute rheumatic fever. Int. Immunol. 20, 445–452 (2008).
Faé, K. C. et al. PDIA3, HSPA5 and vimentin, proteins identified by 2-DE in the valvular tissue, are the target antigens of peripheral and heart infiltrating T cells from chronic rheumatic heart disease patients. J. Autoimmunity 31, 136–141 (2008).
Karthikeyan, G., Fung, E. & Foo, R. S. Y. Alternative hypothesis to explain disease progression in rheumatic heart disease. Circulation 142, 2091–2094 (2020). This commentary proposed that the preferential involvement of the mitral valve in RHD might be explained by the interplay between increased hemodynamic stress and TGFβ-mediated fibrotic remodeling.
Bright, P. D., Mayosi, B. M. & Martin, W. J. An immunological perspective on rheumatic heart disease pathogenesis: more questions than answers. Heart 102, 1527–1532 (2016).
McGregor, R. et al. Mapping autoantibodies in children with acute rheumatic fever. Front. Immunol. 12, 702877 (2021).
McGregor, R. et al. PhIP-seq uncovers marked heterogeneity in acute rheumatic fever autoantibodies. JCI Insight 11, e196619 (2025).
Chung, A. W. et al. Systems immunology reveals a linked IgG3–C4 response in patients with acute rheumatic fever. Immunol. Cell Biol. 98, 12–21 (2020).
Lorenz, N. et al. An acute rheumatic fever immune signature comprising inflammatory markers, IgG3, and Streptococcus pyogenes-specific antibodies. iScience 27, 110558 (2024).
Vanderlugt, C. L. & Miller, S. D. Epitope spreading in immune-mediated diseases: implications for immunotherapy. Nat. Rev. Immunol. 2, 85–95 (2002).
Middleton, F. M., McGregor, R., Webb, R. H., Wilson, N. J. & Moreland, N. J. Cytokine imbalance in acute rheumatic fever and rheumatic heart disease: mechanisms and therapeutic implications. Autoimmun. Rev. 21, 103209 (2022). High-content serology profiling of ARF autoantibodies demonstrated vast repertoire heterogeneity across patients and enabled identification of new cardiac targets of ARF autoantibodies through orthogonal validation.
Kim, M. L. et al. Dysregulated IL-1β-GM-CSF axis in acute rheumatic fever that is limited by hydroxychloroquine. Circulation 138, 2648–2661 (2018). TH1 cell-skewed inflammation in ARF (through IL-1β) promotes expansion of a potentially disease-driving T cell subset (GMCSF+, CXCR3+) that promotes transition from systemic inflammation to tissue-specific autoimmunity.
Muhamed, B., Parks, T. & Sliwa, K. Genetics of rheumatic fever and rheumatic heart disease. Nat. Rev. Cardiol. 17, 145–154 (2020).
Muhamed, B., Shaboodien, G. & Engel, M. E. Genetic variants in rheumatic fever and rheumatic heart disease. Am. J. Med. Genet. C Semin. Med. Genet. 184, 159–177 (2020). Meta-analysis evaluating 66 candidate gene studies of RHD, shortlisted single-nucleotide polymorphisms in TGFB1 and IL1B associated with RHD susceptibility and highlighted the need for large-scale genome-wide association studies for RHD.
Gray, L. -A. et al. Genome-wide analysis of genetic risk factors for rheumatic heart disease in aboriginal australians provides support for pathogenic molecular mimicry. J. Infect. Dis. 216, 1460–1470 (2017).
Parks, T. et al. Association between a common immunoglobulin heavy chain allele and rheumatic heart disease risk in Oceania. Nat. Commun. 8, 14946 (2017).
Machipisa, T. et al. Association of novel locus with rheumatic heart disease in Black African individuals. JAMA Cardiol. 6, 1000–1011 (2021).
Auckland, K. et al. The human leukocyte antigen locus and rheumatic heart disease susceptibility in South Asians and Europeans. Sci. Rep. 10, 9004 (2020).
Kirvan, C. A. et al. IgG2 rules: N-acetyl-β-D-glucosamine-specific IgG2 and Th17/Th1 cooperation may promote the pathogenesis of acute rheumatic heart disease and be a biomarker of the autoimmune sequelae of Streptococcus pyogenes. Front. Cardiovasc. Med. 9, 919700 (2022).
Krisher, K. & Cunningham, M. W. Myosin: a link between streptococci and heart. Science 227, 413–415 (1985).
Kaplan, M. H., Bolande, R., Rakita, L. & Blair, J. Presence of bound immunoglobulins and complement in the myocardium in acute rheumatic fever. New Engl. J. Med. 271, 637–645 (1964).
Lotfi, N. et al. Roles of GM-CSF in the pathogenesis of autoimmune diseases: an update. Front. Immunol. 10, 1265 (2019).
Middleton, F. et al. Perturbation of the T cell compartment underlies rheumatic fever pathogenesis. Preprint at SSRN https://doi.org/10.2139/ssrn.5145933 (2025).
Faé, K. C. et al. CXCL9/Mig mediates T cells recruitment to valvular tissue lesions of chronic rheumatic heart disease patients. Inflammation 36, 800–811 (2013).
Godfrey, D. I., Koay, H. -F., McCluskey, J. & Gherardin, N. A. The biology and functional importance of MAIT cells. Nat. Immunol. 20, 1110–1128 (2019).
Anderson, J. et al. Immune signature of acute pharyngitis in a Streptococcus pyogenes human challenge trial. Nat. Commun. 13, 769 (2022).
Ozkaya, M. et al. The number and activity of CD3+TCR Vα7.2+CD161+ cells are increased in children with acute rheumatic fever. Int. J. Cardiol. 333, 174–183 (2021).
Toor, D. & Sharma, N. T cell subsets: an integral component in pathogenesis of rheumatic heart disease. Immunol. Res. 66, 18–30 (2018).
Surve, N. Z. et al. A longitudinal study of antibody responses to selected host antigens in rheumatic fever and rheumatic heart disease. J. Med. Microbiol. 70, 5 (2021).
Mukherjee, S. et al. Proteomic analysis of human plasma in chronic rheumatic mitral stenosis reveals proteins involved in the complement and coagulation cascade. Clin. Proteomics 11, 35 (2014).
Guttapadu, R. et al. Profiling system-wide variations and similarities between rheumatic heart disease and acute rheumatic fever–a pilot analysis. PLoS Negl.Trop. Dis. 17, e0011263 (2023).
Gao, G. et al. Identification of altered plasma proteins by proteomic study in valvular heart diseases and the potential clinical significance. PLoS ONE 8, e72111 (2013).
Salie, M. T. et al. Data-independent acquisition mass spectrometry in severe rheumatic heart disease (RHD) identifies a proteomic signature showing ongoing inflammation and effectively classifying RHD cases. Clin. Proteomics 19, 7 (2022). Large proteomics study using data-independent acquisition and machine learning approaches that presents an unbiased characterization of the RHD inflammatory state, supplying candidate biomarkers of RHD that might provide insights into pathogenesis.
Diamantino Soares, A. C. et al. Circulating cytokines predict severity of rheumatic heart disease. Int. J. Cardiol. 289, 107–109 (2019).
Tormin, J. P. A. S. et al. Cytokine gene functional polymorphisms and phenotypic expression as predictors of evolution from latent to clinical rheumatic heart disease. Cytokine 138, 155370 (2021). This study used a multiplex quantification assay to provide a comprehensive characterization of cytokine profiles in people with RHD, then correlated specific cytokine markers and gene single-nucleotide polymorphisms to disease prognosis in these individuals.
Silva, V. R. et al. Decreased cytokine plasma levels and changes in T-cell activation are associated with hemodynamic improvement and clinical outcomes after percutaneous mitral commissurotomy in patients with rheumatic mitral stenosis. Front. Cardiovasc. Med. 7, 604826 (2021).
Bilik, M. Z. et al. Serum Levels of IL-17 and IL-23 in patients with rheumatic mitral stenosis. Medicine 95, e3562 (2016).
Neves, E. G. A. et al. Systemic cytokines, chemokines and growth factors reveal specific and shared immunological characteristics in infectious cardiomyopathies. Cytokine 148, 155711 (2021).
Ueno, H. Human circulating T follicular helper cell subsets in health and disease. J. Clin. Immunol. 36, 34–39 (2016).
Liu, Z. et al. Circulating follicular T helper cells and humoral reactivity in rheumatic heart disease. Life Sci. 245, 117390 (2020).
Wen, Y., Zeng, Z., Gui, C., Li, L. & Li, W. Changes in the expression of Th17 cell-associated cytokines in the development of rheumatic heart disease. Cardiovas. Pathol. 24, 382–387 (2015).
Xiao, F. et al. Sex-dependent aortic valve pathology in patients with rheumatic heart disease. PLoS ONE 12, e0180230 (2017).
Zhang, C., Xiao, Z. & Yang, D. Association between the expression of T helper type 17 cell-related cytokines and valve damage in rheumatic heart disease. Mol. Biotechnol. 67, 4257–4270 (2024).
Yeh, C. -Y. et al. Activated human valvular interstitial cells sustain interleukin-17 production to recruit neutrophils in infective endocarditis. Infect. Immun. 83, 2202–2212 (2015).
Yang, Z. et al. IL-17A induces valvular endothelial inflammation and aggravates calcific aortic valve disease. Biochem. Biophys. Res. Commun. 672, 145–153 (2023).
Morris, K., Mohan, C., Wahi, P. L., Anand, I. S. & Ganguly, N. K. Increase in activated T cells and reduction in suppressor/cytotoxic T cells in acute rheumatic fever and active rheumatic heart disease: a longitudinal study. J. Infect. Dis. 167, 979–983 (1993).
Roberts, S. et al. Pathogenic mechanisms in rheumatic carditis: focus on valvular endothelium. J. Infect. Dis. 183, 507–511 (2001).
Bas, H. D. et al. A shift in the balance of regulatory T and T helper 17 cells in rheumatic heart disease. J. Invest. Med. 62, 78–83 (2014).
Mukhopadhyay, S. et al. Circulating level of regulatory T cells in rheumatic heart disease: an observational study. Indian Heart J. 68, 342–348 (2016).
Rafeek, R. A. M., Ketheesan, N., Good, M. F., Pandey, M. & Lepletier, A. Low-dose interleukin 2 therapy halts the progression of post-streptococcal autoimmune complications in a rat model of rheumatic heart disease. mBio 16, e0382324 (2025).
Zhao, Z. et al. CD4+ T cells and TGFβ1/MAPK signal pathway involved in the valvular hyperblastosis and fibrosis in patients with rheumatic heart disease. Exp. Mol. Pathol. 114, 104402 (2020).
Li, M. O. & Flavell, R. A. TGF-β: a master of all T cell trades. Cell 134, 392–404 (2008).
Liu, A. C. & Gotlieb, A. I. Transforming growth factor-β regulates in vitro heart valve repair by activated valve interstitial cells. Am. J. Pathol. 173, 1275–1285 (2008).
Kim, L. et al. Overexpression of transforming growth factor-β1 in the valvular fibrosis of chronic rheumatic heart disease. J. Korean Med. Sci. 23, 41–48 (2008).
Passos, L. S. A. et al. Prothymosin alpha: a novel contributor to estradiol receptor alpha-mediated CD8+ T-cell pathogenic responses and recognition of type 1 collagen in rheumatic heart valve disease. Circulation 145, 531–548 (2022). This study compared proteomic profiles of mitral valve tissue from healthy individuals and people with RHD, uncovering new mechanisms of CD8⁺ T cell cytotoxicity linked to estrogen receptor signaling, and demonstrated that incorporating the valve tissue microenvironment enhances the resolution of disease mechanisms.
Rojas, M. et al. Molecular mimicry and autoimmunity. J. Autoimmunity 95, 100–123 (2018).
Carapetis, J. et al. A plasma protein biomarker signature that differentiates acute rheumatic fever from related clinical presentations. Preprint at Res. Sq. https://doi.org/10.21203/rs.3.rs-4345781/v1 (2025).
Resende, A. L., da, S., Neves, E. G. A., Cavalcante, B. M. & Dutra, W. O. Systemic soluble and cellular immune response in acute rheumatic fever and rheumatic heart disease: a systematic review of human studies. Pathogens 14, 1185 (2025).
Eyi̇ol, A. et al. Relationship of serum HLA-B alleles and TNF-α with rheumatic heart disease. Turk. J. Med. Sci. 48, 724–729 (2018).
Amoils, B. et al. Aberrant expression of HLA-DR antigen on valvular fibroblasts from patients with active rheumatic carditis. Clin. Exp. Immunol. 66, 88–94 (1986).
Arts, R. J. W., Joosten, L. A. B. & Netea, M. G. The potential role of trained immunity in autoimmune and autoinflammatory disorders. Front. Immunol. 9, 298 (2018).
Bennett, J. et al. Understanding group A streptococcal pharyngitis and skin infections as causes of rheumatic fever: protocol for a prospective disease incidence study. BMC Infect. Dis. 19, 633 (2019).
Lorenz, N. et al. Serological profiling of group A Streptococcus infections in acute rheumatic fever. Clin. Infect. Dis. 73, 2322–2325 (2021).
Raynes, J. M. et al. Serological evidence of immune priming by group A Streptococci in patients with acute rheumatic fever. Front. Microbiol. 7, 1119 (2016).
Meira, Z. M. A., Goulart, E. M. A., Colosimo, E. A. & Mota, C. C. C. Long term follow up of rheumatic fever and predictors of severe rheumatic valvar disease in Brazilian children and adolescents. Heart 91, 1019–1022 (2005).
Reményi, B. et al. World Heart Federation criteria for echocardiographic diagnosis of rheumatic heart disease—an evidence-based guideline. Nat. Rev. Cardiol. 9, 297–309 (2012).
Rwebembera, J. et al. 2023 World Heart Federation guidelines for the echocardiographic diagnosis of rheumatic heart disease. Nat. Rev. Cardiol. 21, 250–263 (2024).
Williamson, J. M. et al. The impact of acute rheumatic fever diagnosis on rheumatic heart disease severity. Glob. Heart 20, 72 (2025).
Aschoff, L. The rheumatic nodules in the heart. Ann. Rheum. Dis. 1, 161–166 (1939).
Hinton, R. B. et al. Extracellular matrix remodeling and organization in developing and diseased aortic valves. Circ. Res. 98, 1431–1438 (2006).
Shapero, K., Wylie-Sears, J., Levine, R. A., Mayer, J. E. & Bischoff, J. Reciprocal interactions between mitral valve endothelial and interstitial cells reduce endothelial-to-mesenchymal transition and myofibroblastic activation. J. Mol. Cell. Cardiol. 80, 175–185 (2015).
Gee, T. W., Richards, J. M., Mahmut, A. & Butcher, J. T. Valve endothelial-interstitial interactions drive emergent complex calcific lesion formation in vitro. Biomaterials 269, 120669 (2021).
Pinto, A. R. et al. Revisiting cardiac cellular composition. Circ. Res. 118, 400–409 (2016).
Wang, H., Leinwand, L. A. & Anseth, K. S. Cardiac valve cells and their microenvironment—insights from in vitro studies. Nat. Rev. Cardiol. 11, 715–727 (2014).
Shu, S. et al. Cellular landscapes of nondiseased human cardiac valves from end-stage heart failure–explanted heart. Arter. Thromb. Vasc. Biol. 42, 1429–1446 (2022).
Wang, X. et al. Single-cell analysis reveals the loss of FABP4-positive proliferating valvular endothelial cells relates to functional mitral regurgitation. BMC Med. 22, 595 (2024).
Bartoli-Leonard, F., Zimmer, J. & Aikawa, E. Innate and adaptive immunity: the understudied driving force of heart valve disease. Cardiovasc. Res. 117, 2506–2524 (2021).
Guilherme, L. et al. Rheumatic heart disease: proinflammatory cytokines play a role in the progression and maintenance of valvular lesions. Am. J. Pathol. 165, 1583–1591 (2004).
Scalzi, V. et al. Anti-endothelial cell antibodies in rheumatic heart disease. Clin. Exp. Immunol. 161, 570–575 (2010).
Rastogi, M. et al. Anti-endothelial cell antibody rich sera from rheumatic heart disease patients induces proinflammatory phenotype and methylation alteration in endothelial cells. Genes Dis. 5, 275–289 (2018). This study demonstrated an interaction between autoantibodies and endothelial cells that triggers upregulation of inflammatory genes, contributing to the maintenance of a localized inflammatory state.
Kanitkar, M. & Denton, C. P. The role of endothelial cells in autoimmune rheumatic disease. Curr. Opin. Physiol. 37, 100732 (2024).
Delunardo, F. et al. Streptococcal-vimentin cross-reactive antibodies induce microvascular cardiac endothelial proinflammatory phenotype in rheumatic heart disease. Clin. Exp. Immunol. 173, 419–429 (2013).
Meier, L. A. et al. CD301b/MGL2+ mononuclear phagocytes orchestrate autoimmune cardiac valve inflammation and fibrosis. Circulation 137, 2478–2493 (2018).
Faragher, J. L. et al. Autoimmune valvular carditis requires endothelial cell TNFR1 expression. Arter. Thromb. Vasc. Biol. 43, 943–957 (2023).
Xian, S. et al. Activation of activin/Smad2 and 3 signaling pathway and the potential involvement of endothelial‑mesenchymal transition in the valvular damage due to rheumatic heart disease. Mol. Med. Rep. 23, 10 (2020).
Wylie-Sears, J., Aikawa, E., Levine, R. A., Yang, J. -H. & Bischoff, J. Mitral valve endothelial cells with osteogenic differentiation potential. Arter. Thromb. Vasc. Biol. 31, 598–607 (2011).
Bai, L. et al. Anti-IL-17 inhibits PINK1/parkin autophagy and M1 macrophage polarization in rheumatic heart disease. Inflammation 48, 870–884 (2024).
Gomes, N. F. A. et al. Histopathological characterization of mitral valvular lesions from patients with rheumatic heart disease. Arq. Bras. Cardiol. 116, 404–412 (2021).
Rajamannan, N. M. et al. Calcified rheumatic valve neoangiogenesis is associated with vascular endothelial growth factor expression and osteoblast-like bone formation. Circulation 111, 3296–3301 (2005).
Yoshioka, M. et al. Chondromodulin-I maintains cardiac valvular function by preventing angiogenesis. Nat. Med. 12, 1151–1159 (2006).
Mariscalco, G. et al. Imbalance between pro-angiogenic and anti-angiogenic factors in rheumatic and mixomatous mitral valves. Int. J. Cardiol. 152, 337–344 (2011).
Osinski, V. et al. Profibrotic VEGFR3-dependent lymphatic vessel growth in autoimmune valvular carditis. Arterioscler. Thromb. Vasc. Biol. 44, 807–821 (2024). Initial evidence emerging from animal models of autoimmune valvulitis suggests a role for VEGFR3-mediated lymphangiogenesis, with this phenomenon being identified in the mitral valves of people with RHD.
Liu, A. C., Joag, V. R. & Gotlieb, A. I. The emerging role of valve interstitial cell phenotypes in regulating heart valve pathobiology. Am. J. Pathol. 171, 1407–1418 (2007).
Shun, C. -T. et al. Activation of human valve interstitial cells by a viridians Streptococci modulin induces chemotaxis of mononuclear cells. J. Infect. Dis. 199, 1488–1496 (2009).
Elhewala, A. A., Sanad, M., Soliman, A. M., Sami, M. M. & Ahmed, A. A. Matrix metalloproteinase-9 in pediatric rheumatic heart disease with and without heart failure. Biomed. Rep. 14, 4 (2020).
Rutkovskiy, A. et al. Valve interstitial cells: the key to understanding the pathophysiology of heart valve calcification. J. Am. Heart Assoc. 6, e006339 (2017).
De Oliveira Martins, C. et al. Distinct mitral valve proteomic profiles in rheumatic heart disease and myxomatous degeneration. Clin. Med. Insights Cardiol. 8, 79–86 (2014). The fibrotic pathology observed in RHD valves is uniquely associated with an imbalance and disorganization of extracellular matrix components, driven in part by TNF and MMP-25 signaling networks.
Martins, C. D. O. et al. Rheumatic heart disease and myxomatous degeneration: differences and similarities of valve damage resulting from autoimmune reactions and matrix disorganization. PLoS ONE 12, e0170191 (2017).
Päll, T. et al. Soluble CD44 interacts with intermediate filament protein vimentin on endothelial cell surface. PLoS ONE 6, e29305 (2011).
Zhong, J. et al. Vitronectin-binding PAI-1 protects against the development of cardiac fibrosis through interaction with fibroblasts. Lab Invest. 94, 633–644 (2014).
Tincani, A., Rebaioli, C. B., Taglietti, M. & Shoenfeld, Y. Heart involvement in systemic lupus erythematosus, anti-phospholipid syndrome and neonatal lupus. Rheumatology 45, iv8–iv13 (2006).
Voskuyl, A. E. The heart and cardiovascular manifestations in rheumatoid arthritis. Rheumatology 45, iv4–iv7 (2006).
Pan, S. -Y. et al. Cardiac damage in autoimmune diseases: target organ involvement that cannot be ignored. Front. Immunol. 13, 1056400 (2022).
Pandian, N. G. et al. Recommendations for the use of echocardiography in the evaluation of rheumatic heart disease: a report from the American Society of Echocardiography. J. Am. Soc. Echocardiogr. 36, 3–28 (2023).
Walker, G. A., Masters, K. S., Shah, D. N., Anseth, K. S. & Leinwand, L. A. Valvular myofibroblast activation by transforming growth factor-β. Circ. Res. 95, 253–260 (2004).
Choi, J. -H. et al. Identification of antigen-presenting dendritic cells in mouse aorta and cardiac valves. J. Exp. Med. 206, 497–505 (2009).
Cardeira-da-Silva, J. et al. Antigen presentation plays positive roles in the regenerative response to cardiac injury in zebrafish. Nat. Commun. 15, 3637 (2024).
Driscoll, K., Cruz, A. D. & Butcher, J. T. Inflammatory and biomechanical drivers of endothelial-interstitial interactions in calcific aortic valve disease. Circ. Res. 128, 1344–1370 (2021).
Lupieri, A. et al. Rheumatic heart valve disease: navigating the challenges of an overlooked autoimmune disorder. Front. Cardiovasc. Med. 12, 1537104 (2025).
Sikder, S. et al. Group A streptococcal M-protein specific antibodies and T-cells drive the pathology observed in the rat autoimmune valvulitis model. Autoimmunity 52, 78–87 (2019).
Chen, Y. et al. Global burden and trend of rheumatic heart disease among women of childbearing age, 1990–2021, with projection to 2040. BMC Cardiovasc. Disord. 25, 580 (2025).
Afshan, G. et al. Circulating prothymosin alpha and immunoglobulin G3 in acute rheumatic fever and rheumatic heart disease: a case-control study. Am. Heart J. Plus 59, 100630 (2025).
Sarkar, S. et al. Association of rheumatic fever & rheumatic heart disease with plausible early & late-stage disease markers. Indian J. Med. Res. 145, 758–766 (2017).
Liu, Z. et al. Chronic mitral valve fibrosis in rheumatic heart disease: from immune trigger to inflammatory and mechanical progression. J. Am. Heart Assoc. 14, e045169 (2025).
Wilson, N. J. et al. The treatment of acute rheumatic fever: novel use of hydroxychloroquine. Pediatr. Infect. Dis. J. 39, e120–e122 (2020).
Asrial, A. A. et al. Effect of dapagliflozin on patients with rheumatic heart disease mitral stenosis. J. Clin. Med. 12, 5898 (2023).
Ambari, A. M. et al. Angiotensin converting enzyme inhibitors (ACEIs) decrease the progression of cardiac fibrosis in rheumatic heart disease through the inhibition of IL-33/sST2. Front. Cardiovasc. Med. 7, 115 (2020).
Chockalingam, A. et al. Safety and efficacy of enalapril in multivalvular heart disease with significant mitral stenosis—SCOPE-MS. Angiology 56, 151–158 (2005).
Ferguson, J. Valvular disease of the heart, accompanied by rheumatic subcutaneous nodules. BMJ 1, 1150–1150 (1885).
Fraser, W. J., Haffejee, Z., Jankelow, D., Wadee, A. & Cooper, K. Rheumatic Aschoff nodules revisited. II: Cytokine expression corroborates recently proposed sequential stages. Histopathology 31, 460–464 (1997).
Kemeny, E., Grieve, T., Marcus, R., Sareli, P. & Zabriskie, J. B. Identification of mononuclear cells and T cell subsets in rheumatic valvulitis. Clin. Immunol. Immunopathol. 52, 225–237 (1989).
Lannigan, R. & Zaki, S. Location of gamma globulin in the endocardium in rheumatic heart disease by the ferritin-labelled antibody technique. Nature 217, 173–174 (1968).
Acknowledgements
We acknowledge support from the National Health and Medical Research Council of Australia Ideas grant (2039228, to H.K.V.) and the National Heart Foundation of Australia Vanguard grant (107962, to H.K.V. and A.S). We acknowledge support from the Fox Family Foundation. This work was supported by the Novo Nordisk Foundation Center for Stem Cell Medicine, reNEW, which is supported by a Novo Nordisk Foundation grant (NNF21CC0073729, to S.Y., E.R.P. and H.K.V.). E.R.P is supported by a National Health and Medical Research Council Investigator grant. H.K.V. was supported by a Postdoctoral Fellowship (106645) from the National Heart Foundation of Australia and is currently supported by a Future Leader Fellowship (109309) from the National Heart Foundation of Australia.
Author information
Authors and Affiliations
Contributions
S.Y. drafted the manuscript. All authors reviewed and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
E.R.P. is a cofounder, scientific advisor and stockholder in Dynomics. The other authors declare no competing interests.
Peer review
Peer review information
Nature Cardiovascular Research thanks the anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yeow, S., Frost, H., Porrello, E.R. et al. Valve biology and rheumatic heart disease pathogenesis. Nat Cardiovasc Res 5, 204–217 (2026). https://doi.org/10.1038/s44161-026-00792-9
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44161-026-00792-9