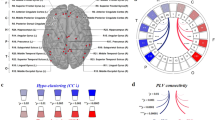
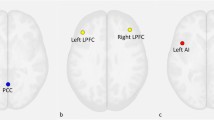
Abstract
Depressive and anxiety disorders are characterized by altered connectivity within and between the default mode network (DMN) and salience network. Basal forebrain subdivisions, critical for regulating network activity, remain understudied across these conditions. To address this gap, we analyzed 7-Tesla resting-state functional magnetic resonance imaging data from a transdiagnostic sample (n = 70), primarily with depressive and anxiety disorders, and healthy controls (n = 77). We used spectral dynamic causal modeling to assess effective connectivity between the medial septum/diagonal band (Ch1–3), nucleus basalis of Meynert (Ch4), ventral pallidum, and DMN and salience network. Healthy participants showed excitatory connectivity from Ch1–3 to the DMN and from Ch4 to the anterior insula. By contrast, clinical participants exhibited greater inhibitory Ch4 to DMN connectivity and increased excitatory connectivity from Ch4 to the anterior insula. Widespread Ch4 connectivity dysfunction may implicate the cholinergic system as a mechanistic and therapeutic target for depressive and anxiety disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$79.00 per year
only $6.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Individual participant and group-level effective connectivity data for this study are available at https://github.com/alecJamieson/basal_forebrain_EC/tree/main/Data.
Code availability
MATLAB and R scripts used to generate the second-level results and associated figures of this study are also available at https://github.com/alecJamieson/basal_forebrain_EC.
References
Dutta, A., McKie, S. & Deakin, J. F. Resting state networks in major depressive disorder. Psychiatry Res. 224, 139–151 (2014).
Bertocci, M. A. et al. Altered patterns of central executive, default mode and salience network activity and connectivity are associated with current and future depression risk in two independent young adult samples. Mol. Psychiatry 28, 1046–1056 (2023).
Zhang, X. et al. Large-scale brain functional network abnormalities in social anxiety disorder. Psychol. Med. 53, 6194–6204 (2023).
Mulders, P. C., van Eijndhoven, P. F., Schene, A. H., Beckmann, C. F. & Tendolkar, I. Resting-state functional connectivity in major depressive disorder: a review. Neurosci. Biobehav. Rev. 56, 330–344 (2015).
Tse, N. Y. et al. A mega-analysis of functional connectivity and network abnormalities in youth depression. Nat. Ment. Health 2, 1169–1182 (2024).
Mohan, A. et al. The significance of the default mode network (DMN) in neurological and neuropsychiatric disorders: a review. Yale J. Biol. Med. 89, 49–57 (2016).
Rabany, L. et al. Resting-state functional connectivity in generalized anxiety disorder and social anxiety disorder: evidence for a dimensional approach. Brain Connect. 7, 289–298 (2017).
Li, R. et al. Dissociable salience and default mode network modulation in generalized anxiety disorder: a connectome-wide association study. Cereb. Cortex 33, 6354–6365 (2023).
Jamieson, A. J., Harrison, B. J., Razi, A. & Davey, C. G. Rostral anterior cingulate network effective connectivity in depressed adolescents and associations with treatment response in a randomized controlled trial. Neuropsychopharmacology 47, 1240–1248 (2022).
Jamieson, A. J. et al. A brain model of altered self-appraisal in social anxiety disorder. Transl. Psychiatry 13, 344 (2023).
Xu, L., Xu, H., Ding, H., Li, J. & Wang, C. Intrinsic network brain dysfunction correlates with temporal complexity in generalized anxiety disorder and panic disorder. Front. Hum. Neurosci. 15, 647518 (2021).
Deif, R. & Salama, M. Depression from a precision mental health perspective: utilizing personalized conceptualizations to guide personalized treatments. Front. Psychiatry 12, 650318 (2021).
Dalgleish, T., Black, M., Johnston, D. & Bevan, A. Transdiagnostic approaches to mental health problems: current status and future directions. J. Consult. Clin. Psychol. 88, 179–195 (2020).
Aguilar, D. D. & McNally, J. M. Subcortical control of the default mode network: role of the basal forebrain and implications for neuropsychiatric disorders. Brain Res. Bull. 185, 129–139 (2022).
Nair, J. et al. Basal forebrain contributes to default mode network regulation. Proc. Natl Acad. Sci. USA 115, 1352–1357 (2018).
Alves, P. N. et al. An improved neuroanatomical model of the default-mode network reconciles previous neuroimaging and neuropathological findings. Commun. Biol. 2, 370 (2019).
Harrison, B. J. et al. Dynamic subcortical modulators of human default mode network function. Cereb. Cortex 32, 4345–4355 (2022).
Peeters, L. M. et al. Cholinergic modulation of the default mode like network in rats. iScience 23, 101455 (2020).
Mesulam, M. M., Mufson, E. J., Levey, A. I. & Wainer, B. H. Cholinergic innervation of cortex by the basal forebrain: cytochemistry and cortical connections of the septal area, diagonal band nuclei, nucleus basalis (substantia innominata), and hypothalamus in the rhesus monkey. J. Comp. Neurol. 214, 170–197 (1983).
Fritz, H. C. J. et al. The corticotopic organization of the human basal forebrain as revealed by regionally selective functional connectivity profiles. Hum, Brain Mapp. 40, 868–878 (2019).
Yuan, R., Biswal, B. B. & Zaborszky, L. Functional subdivisions of magnocellular cell groups in human basal forebrain: test–retest resting-state study at ultra-high field, and meta-analysis. Cereb. Cortex 29, 2844–2858 (2019).
Li, C. S. et al. Resting state functional connectivity of the basal nucleus of Meynert in humans: in comparison to the ventral striatum and the effects of age. Neuroimage 97, 321–332 (2014).
Root, D. H., Melendez, R. I., Zaborszky, L. & Napier, T. C. The ventral pallidum: subregion-specific functional anatomy and roles in motivated behaviors. Prog. Neurobiol. 130, 29–70 (2015).
Zhao, Y., Kirschenhofer, T., Harvey, M. & Rainer, G. Mediodorsal thalamus and ventral pallidum contribute to subcortical regulation of the default mode network. Commun. Biol. 7, 891 (2024).
Klaassen, A.-L., Heiniger, A., Sánchez, P. V., Harvey, M. A. & Rainer, G. Ventral pallidum regulates the default mode network, controlling transitions between internally and externally guided behavior. Proc. Natl Acad. Sci. USA 118, e2103642118 (2021).
Yamakawa, G. R. et al. The cholinergic forebrain arousal system acts directly on the circadian pacemaker. Proc. Natl Acad. Sci. USA 113, 13498–13503 (2016).
Cabrera, S. M., Chavez, C. M., Corley, S. R., Kitto, M. R. & Butt, A. E. Selective lesions of the nucleus basalis magnocellularis impair cognitive flexibility. Behav. Neurosci. 120, 298–306 (2006).
Herman, A. M. et al. A cholinergic basal forebrain feeding circuit modulates appetite suppression. Nature 538, 253–256 (2016).
Howkers, M. B. J., Goodman, E. & Sim, V. M. Some behavioral changes in man following anticholinesterase administration. J. Nerv. Ment. Dis. 138, 383–389 (1964).
Janowsky, D. S., el-Yousef, M. K., Davis, J. M., Hubbard, B. & Sekerke, H. J. Cholinergic reversal of manic symptoms. Lancet 1, 1236–1237 (1972).
Dulawa, S. C. & Janowsky, D. S. Cholinergic regulation of mood: from basic and clinical studies to emerging therapeutics. Mol. Psychiatry 24, 694–709 (2019).
Mineur, Y. S. et al. Cholinergic signaling in the hippocampus regulates social stress resilience and anxiety—and depression-like behavior. Proc. Natl Acad. Sci. USA 110, 3573–3578 (2013).
Chen, L. et al. Fluoxetine and ketamine reverse the depressive but not anxiety behavior induced by lesion of cholinergic neurons in the horizontal limb of the diagonal band of Broca in male rat. Front. Behav. Neurosci. 15, 602708 (2021).
Knowland, D. et al. Distinct ventral pallidal neural populations mediate separate symptoms of depression. Cell 170, 284–297 (2017).
Kim, R., Ananth, M. R., Desai, N. S., Role, L. W. & Talmage, D. A. Distinct subpopulations of ventral pallidal cholinergic projection neurons encode valence of olfactory stimuli. Cell Rep. 43, 114009 (2024).
Keuken, M. C., Isaacs, B. R., Trampel, R., van der Zwaag, W. & Forstmann, B. U. Visualizing the human subcortex using ultra-high field magnetic resonance imaging. Brain Topogr. 31, 513–545 (2018).
Razi, A., Kahan, J., Rees, G. & Friston, K. J. Construct validation of a DCM for resting state fMRI. Neuroimage 106, 1–14 (2015).
Friston, K. J., Kahan, J., Biswal, B. & Razi, A. A DCM for resting state fMRI. Neuroimage 94, 396–407 (2014).
Zeidman, P. et al. A guide to group effective connectivity analysis, part 2: second level analysis with PEB. Neuroimage 200, 12–25 (2019).
McLean, C. P., Asnaani, A., Litz, B. T. & Hofmann, S. G. Gender differences in anxiety disorders: prevalence, course of illness, comorbidity and burden of illness. J. Psychiatr. Res. 45, 1027–1035 (2011).
Kuehner, C. Gender differences in unipolar depression: an update of epidemiological findings and possible explanations. Acta Psychiatr. Scand. 108, 163–174 (2003).
Zaborszky, L., Carlsen, J., Brashear, H. R. & Heimer, L. Cholinergic and GABAergic afferents to the olfactory bulb in the rat with special emphasis on the projection neurons in the nucleus of the horizontal limb of the diagonal band. J. Comp. Neurol. 243, 488–509 (1986).
Walker, L. C. et al. Peptidergic neurons in the basal forebrain magnocellular complex of the rhesus monkey. J. Comp. Neurol. 280, 272–282 (1989).
Zaborszky, L., Pang, K., Somogyi, J., Nadasdy, Z. & Kallo, I. The basal forebrain corticopetal system revisited. Ann. N. Y. Acad. Sci. 877, 339–367 (1999).
Zaborszky, L. & Duque, A. Local synaptic connections of basal forebrain neurons. Behav. Brain Res. 115, 143–158 (2000).
Zou, Y. et al. Cell-type-specific optogenetic fMRI on basal forebrain reveals functional network basis of behavioral preference. Neuron https://doi.org/10.1016/j.neuron.2024.01.017 (2024).
Kim, T. et al. Cortically projecting basal forebrain parvalbumin neurons regulate cortical gamma band oscillations. Proc. Natl Acad. Sci. USA 112, 3535–3540 (2015).
Lozano-Montes, L. et al. Optogenetic stimulation of basal forebrain parvalbumin neurons activates the default mode network and associated behaviors. Cell Rep. 33, 108359 (2020).
Do, J. P. et al. Cell type-specific long-range connections of basal forebrain circuit. Elife https://doi.org/10.7554/eLife.13214 (2016).
McLaughlin, K. A. & Nolen-Hoeksema, S. Rumination as a transdiagnostic factor in depression and anxiety. Behav. Res. Ther. 49, 186–193 (2011).
Espinosa, N. et al. Basal forebrain gating by somatostatin neurons drives prefrontal cortical activity. Cereb. Cortex 29, 42–53 (2019).
Sanda, P. et al. Cholinergic modulation supports dynamic switching of resting state networks through selective DMN suppression. PLoS Comput. Biol. 20, e1012099 (2024).
Liu, C. et al. Trait- and state-like co-activation pattern dynamics in current and remitted major depressive disorder. J. Affect. Disord. 337, 159–168 (2023).
Dong, D. et al. Sex-specific resting state brain network dynamics in patients with major depressive disorder. Neuropsychopharmacology 49, 806–813 (2024).
Wang, X., Ongur, D., Auerbach, R. P. & Yao, S. Q. Cognitive vulnerability to major depression: view from the intrinsic network and cross-network interactions. Harvard Rev. Psychiatry 24, 188–201 (2016).
Berman, M. G. et al. Neural and behavioral effects of interference resolution in depression and rumination. Cogn. Affect. Behav. Neurosci. 11, 85–96 (2011).
Shine, J. M. Neuromodulatory influences on integration and segregation in the brain. Trends Cogn. Sci. 23, 572–583 (2019).
Taylor, N. L. et al. Causal evidence for cholinergic stabilization of attractor landscape dynamics. Cell Rep. 43, 114359 (2024).
Munn, B. R., Muller, E. J., Wainstein, G. & Shine, J. M. The ascending arousal system shapes neural dynamics to mediate awareness of cognitive states. Nat. Commun. 12, 6016 (2021).
Shackman, A. J. et al. The neurobiology of dispositional negativity and attentional biases to threat: implications for understanding anxiety disorders in adults and youth. J. Exp. Psychopathol. 7, 311–342 (2016).
Dagyte, G., Den Boer, J. A. & Trentani, A. The cholinergic system and depression. Behav. Brain Res. 221, 574–582 (2011).
Jaffe, R. J., Novakovic, V. & Peselow, E. D. Scopolamine as an antidepressant: a systematic review. Clin. Neuropharmacol. 36, 24–26 (2013).
Furey, M. L. & Drevets, W. C. Antidepressant efficacy of the antimuscarinic drug scopolamine: a randomized, placebo-controlled clinical trial. Arch. Gen. Psychiatry 63, 1121–1129 (2006).
Furey, M. L., Khanna, A., Hoffman, E. M. & Drevets, W. C. Scopolamine produces larger antidepressant and antianxiety effects in women than in men. Neuropsychopharmacology 35, 2479–2488 (2010).
Mocko, P., Sladowska, K., Kawalec, P., Babii, Y. & Pilc, A. The potential of scopolamine as an antidepressant in major depressive disorder: a systematic review of randomized controlled trials. Biomedicines https://doi.org/10.3390/biomedicines11102636 (2023).
Drevets, W. C., Bhattacharya, A. & Furey, M. L. in Advances in Pharmacology Vol. 89 (eds Duman, R. S. & Krystal, J. H.) 357–386 (Academic Press, 2020).
Fogaça, M. V. et al. Inhibition of GABA interneurons in the mPFC is sufficient and necessary for rapid antidepressant responses. Mol. Psychiatry 26, 3277–3291 (2021).
Beisteiner, R. et al. Clinical fMRI: evidence for a 7T benefit over 3T. Neuroimage 57, 1015–1021 (2011).
Larsen, R. S. & Waters, J. Neuromodulatory correlates of pupil dilation. Front. Neural Circuits 12, 21 (2018).
Russchen, F. T., Amaral, D. G. & Price, J. L. The afferent connections of the substantia innominata in the monkey, Macaca fascicularis. J. Comp. Neurol. 242, 1–27 (1985).
Mesulam, M.-M. & Mufson, E. J. Neural inputs into the nucleus basalis of the substantia innominata (Ch4) in the rhesus monkey. Brain 107, 253–274 (1984).
Chang, C.-h & Grace, A. A. Amygdala–ventral pallidum pathway decreases dopamine activity after chronic mild stress in rats. Biol. Psychiatry 76, 223–230 (2014).
Guo, H., Guo, J., Gao, Z., Luo, F. & Zhang, E. The role of amygdala‐ventral pallidum pathway in depression‐like behaviors in male mice. J. Neurosci. Res. 102, e25258 (2024).
Tolin, D. et al. Diagnostic Interview for anxiety, mood, and OCD and related neuropsychiatric disorders (DIAMOND) (The Institute of Living, Hartford Healthcare Corporation, 2016).
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders 5th edn (American Psychiatric Association, 2013).
Swinbourne, J. et al. The comorbidity between eating disorders and anxiety disorders: prevalence in an eating disorder sample and anxiety disorder sample. Aust. N. Z. J. Psychiatry 46, 118–131 (2012).
Spinhoven, P., Penninx, B. W., van Hemert, A. M., de Rooij, M. & Elzinga, B. M. Comorbidity of PTSD in anxiety and depressive disorders: prevalence and shared risk factors. Child Abuse Negl. 38, 1320–1330 (2014).
Sheehan, D. V. et al. The Mini-International Neuropsychiatric Interview (MINI): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J. Clin. Psychiatry 59, 22–33 (1998).
Lovibond, S. H. Manual for the Depression Anxiety Stress Scales (Sydney psychology foundation, 1995).
Moeller, S. et al. Multiband multislice GE-EPI at 7 tesla, with 16-fold acceleration using partial parallel imaging with application to high spatial and temporal whole-brain fMRI. Magn. Reson. Med. 63, 1144–1153 (2010).
Marques, J. P. et al. MP2RAGE, a self bias-field corrected sequence for improved segmentation and T1-mapping at high field. Neuroimage 49, 1271–1281 (2010).
Lafayette, L., Sauter, G., Vu, L. & Meade, B. Spartan performance and flexibility: an HPC-cloud chimera. Paper presented at OpenStack Summit, Barcelona, 27 October 2016; https://levlafayette.com/files/2016spartanopenstack.pdf
Wilke, M. An alternative approach towards assessing and accounting for individual motion in fMRI timeseries. Neuroimage 59, 2062–2072 (2012).
O’Brien, K. R. et al. Robust T1-weighted structural brain imaging and morphometry at 7T using MP2RAGE. PLoS ONE 9, e99676 (2014).
Ashburner, J. A fast diffeomorphic image registration algorithm. Neuroimage 38, 95–113 (2007).
Kasper, L. et al. The PhysIO toolbox for modeling physiological noise in fMRI data. J. Neurosci. Meth. 276, 56–72 (2017).
Glover, G. H., Li, T. Q. & Ress, D. Image-based method for retrospective correction of physiological motion effects in fMRI: RETROICOR. Magn. Reson. Med. 44, 162–167 (2000).
Behzadi, Y., Restom, K., Liau, J. & Liu, T. T. A component based noise correction method (CompCor) for BOLD and perfusion based fMRI. Neuroimage 37, 90–101 (2007).
Zaborszky, L. et al. Stereotaxic probabilistic maps of the magnocellular cell groups in human basal forebrain. Neuroimage 42, 1127–1141 (2008).
Pauli, W. M., Nili, A. N. & Tyszka, J. M. A high-resolution probabilistic in vivo atlas of human subcortical brain nuclei. Sci. Data 5, 180063 (2018).
Fair, D. A. et al. Functional brain networks develop from a “local to distributed” organization. PLoS Comput. Biol. https://doi.org/10.1371/journal.pcbi.1000381 (2009).
Yarkoni, T., Poldrack, R. A., Nichols, T. E., Van Essen, D. C. & Wager, T. D. Large-scale automated synthesis of human functional neuroimaging data. Nat. Methods 8, 665–670 (2011).
Dosenbach, N. U. et al. Distinct brain networks for adaptive and stable task control in humans. Proc. Natl Acad. Sci. USA 104, 11073–11078 (2007).
Sridharan, D., Levitin, D. J. & Menon, V. A critical role for the right fronto-insular cortex in switching between central-executive and default-mode networks. Proc. Natl Acad. Sci. USA 105, 12569–12574 (2008).
Ereira, S., Waters, S., Razi, A. & Marshall, C. R. Early detection of dementia with default-mode network effective connectivity. Nat. Ment. Health 2, 787–800 (2024).
Stoliker, D. et al. Neural mechanisms of resting-state networks and the amygdala underlying the cognitive and emotional effects of psilocybin. Biol. Psychiatry 96, 57–66 (2024).
Novelli, L., Friston, K. & Razi, A. Spectral dynamic causal modeling: a didactic introduction and its relationship with functional connectivity. Netw. Neurosci. 8, 178–202 (2024).
Friston, K. J. et al. Bayesian model reduction and empirical Bayes for group (DCM) studies. Neuroimage 128, 413–431 (2016).
Rosa, M. J., Friston, K. J. & Penny, W. Post-hoc selection of dynamic causal models. J. Neurosci. Methods 208, 66–78 (2012).
Friston, K. J. & Penny, W. Post hoc Bayesian model selection. Neuroimage 56, 2089–2099 (2011).
Benjamin, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. B 57, 289–300 (1995).
Edlow, B. L. et al. 7 Tesla MRI of the ex vivo human brain at 100 micron resolution. Sci. Data 6, 244 (2019).
Xia, M., Wang, J. & He, Y. BrainNet Viewer: a network visualization tool for human brain connectomics. PLoS ONE 8, e68910 (2013).
Acknowledgements
The authors thank B. Thai, H. Carey and A. Nielson for their contributions to data collection. The authors also acknowledge the facilities and scientific and technical assistance of the National Imaging Facility, a National Collaborative Research Infrastructure Strategy (NCRIS) capability, at the Melbourne Brain Centre Imaging Unit, University of Melbourne. In addition, the authors are grateful to Siemens for providing the MP2RAGE sequence as a ‘works in progress package’ and CMRR (University of Minnesota) for sharing the multiband EPI sequence. This study was supported by National Health and Medical Research Council of Australia (NHMRC) Project Grants (1161897) to B.J.H. and (1073041) to K.L.F. T.S. is supported by a NHMRC/MRFF Investigator Grant (MRF1193736), a BBRF Young Investigator Grant, and a University of Melbourne McKenzie Fellowship. J.A.A. and S.I. are supported by Australian Government Research Training Program Scholarships.
Author information
Authors and Affiliations
Contributions
A.J.J. conceived the study, performed the formal analyses, drafted the manuscript and prepared the visualizations. T.S. and B.J.H. contributed to conceptualization, secured funding and assisted in reviewing and editing the manuscript. C.G.D. contributed through critical review and editing. B.A.M. and K.L.F. supported the work through funding acquisition and provision of resources, respectively. S.I., J.A.A. and R.K.G. were involved in data collection and assisted in reviewing and editing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interest.
Peer review
Peer review information
Nature Mental Health thanks Mala Ananth, Chun-Hong Liu and Mingrui Xia for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–5, Tables 1–4, Methods and Results.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jamieson, A.J., Steward, T., Davey, C.G. et al. Altered basal forebrain regulation of intrinsic brain networks in depressive and anxiety disorders. Nat. Mental Health 3, 1202–1213 (2025). https://doi.org/10.1038/s44220-025-00496-2
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44220-025-00496-2