Abstract
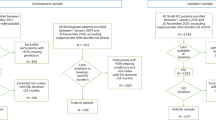
Xanomeline/trospium (Cobenfy) was recently approved by the US Food and Drug Administration for the treatment of adults with schizophrenia. Despite promising findings in placebo-controlled trials, there is limited understanding of its real-world use. Here we perform a post-hoc analysis of medical records following Cobenfy add-on administration in an inpatient population. In an initial cohort of 24 patients, ~40% experienced positive responses. To identify predictive clinical features, we used a combination of hierarchical clustering and linear discriminant analysis, which showed that negative symptoms and stimulant use were the largest predictors of a positive response, while the presence of intellectual delay was negatively predictive. This pattern was independently replicated in 25 patients. Overall, this work supports the notion of biologically distinct psychosis subgroups and invites further research into the underlying biological substrates.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$79.00 per year
only $6.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data related to this Article are available via GitHub at https://github.com/orgs/Halassa-Lab/repositories.
Code availability
Code is available via GitHub at https://github.com/orgs/Halassa-Lab/repositories.
References
Brannan, S. K. et al. Muscarinic cholinergic receptor agonist and peripheral antagonist for schizophrenia. N. Engl. J. Med. 384, 717–726 (2021).
Yohn, S. E. & Conn, P. J. Positive allosteric modulation of M(1) and M(4) muscarinic receptors as potential therapeutic treatments for schizophrenia. Neuropharmacology 136, 438–448 (2018).
Correll, C. U. & Schooler, N. R. Negative symptoms in schizophrenia: a review and clinical guide for recognition, assessment, and treatment. Neuropsychiatr. Dis. Treat. 16, 519–534 (2020).
Marder, S. R. & Umbricht, D. Negative symptoms in schizophrenia: Newly emerging measurements, pathways, and treatments. Schizophr. Res. 258, 71–77 (2023).
Rosenheck, R. et al. Impact of clozapine on negative symptoms and on the deficit syndrome in refractory schizophrenia. Department of Veterans Affairs Cooperative Study Group on Clozapine in Refractory Schizophrenia. Am. J. Psychiatry 156, 88–93 (1999).
Kaul, I. et al. Efficacy and safety of the muscarinic receptor agonist KarXT (xanomeline–trospium) in schizophrenia (EMERGENT-2) in the USA: results from a randomised, double-blind, placebo-controlled, flexible-dose phase 3 trial. Lancet 403, 160–170 (2024).
Horan, W. P. et al. Efficacy of KarXT on negative symptoms in acute schizophrenia: a post hoc analysis of pooled data from 3 trials. Schizophr. Res. https://doi.org/10.1016/j.schres.2024.08.001 (2024).
Kirkpatrick, B., Fenton, W. S., Carpenter, W. T. Jr. & Marder, S. R. The NIMH-MATRICS consensus statement on negative symptoms. Schizophr. Bull. 32, 214–219 (2006).
Tandon, R. et al. The impact on functioning of second-generation antipsychotic medication side effects for patients with schizophrenia: a worldwide, cross-sectional, web-based survey. Ann. Gen. Psychiatry 19, 42 (2020).
Kaboodvand, N., Shabanpour, M. & Guterstam, J. Neural correlates of impulsivity in amphetamine use disorder. Psychiatry Res. Neuroimaging 343, 111860 (2024).
Weikop, P., Jensen, K. L. & Thomsen, M. Effects of muscarinic M(1) receptor stimulation on reinforcing and neurochemical effects of cocaine in rats. Neuropsychopharmacology 45, 1994–2002 (2020).
Cuthbert, B. N. & Insel, T. R. Toward the future of psychiatric diagnosis: the seven pillars of RDoC. BMC Med. 11, 126 (2013).
Halassa, M. M. et al. Developing algorithmic psychiatry via multi-level spanning computational models. Cell Rep. Med. https://doi.org/10.1016/j.xcrm.2025.102094 (2025).
Javitt, D. C. Xanomeline–trospium treatment of cognitive impairments of schizophrenia: hope for some, or hope for all?. Am. J. Psychiatry 182, 237–239 (2025).
Huang, A. S. et al. A prefrontal thalamocortical readout for conflict-related executive dysfunction in schizophrenia. Cell Rep. Med. https://doi.org/10.1016/j.xcrm.2024.101802 (2024).
Acknowledgements
I thank D. Ongur, S. Fitzpatrick, D. Rothman and A. Jaffe for feedback on the data and recommendations for improvement. I thank P. R. Halassa for assistance with my code, and R. D. Wimmer for discussions and help with manuscript preparation and revision.
Author information
Authors and Affiliations
Contributions
M.M.H. analyzed the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The author declares no competing interests.
Peer review
Peer review information
Nature Mental Health thanks Alessandro di Lisi and the other, anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 LDA results with equivocal patients included post classification.
Equivocal patients (n = 5) were grouped with the non-responders when projected onto the LDA principal vector.
Extended Data Fig. 2 LDA results for the combined cohort 1 and cohort 2 dataset.
LDA derived discriminant function revealed clear separation between responsive and non-responsive patient groups (n = 49).
Extended Data Fig. 3 LDA feature weights for the combined dataset ordered by absolute magnitude.
Note that this matches the original datasets with an increase in Intellectual disability negative weights and a slight decrease in stimulant use predictive value. Total n = 49 patients.
Supplementary information
Supplementary Information (download PDF )
Supplementary Table 1.
Supplementary Table 1 (download CSV )
Raw data for all 49 subjects.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Halassa, M.M. Preliminary real-world predictors of response to muscarinic targeting in psychosis. Nat. Mental Health 3, 1512–1518 (2025). https://doi.org/10.1038/s44220-025-00529-w
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44220-025-00529-w