Abstract
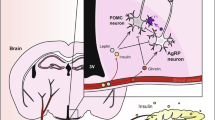
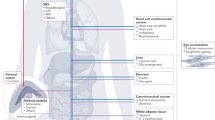
Systemic metabolic abnormalities including insulin resistance, lipid dysregulation, mitochondrial dysfunction and inflammation are highly prevalent in psychiatric illness and may contribute to increased mortality, illness severity and treatment resistance. This Review synthesizes current evidence linking systemic and central metabolic dysfunction with mental health outcomes across disorders. We highlight bidirectional interactions between brain function and metabolic state, and examine how psychotropic medications, stressors and disease mechanisms contribute to metabolic burden. In turn, we discuss how systemic dysfunction may impair brain structure and function. We review emerging metabolism-based interventions used in psychiatry, including pharmacologic agents (metformin, glucagon-like peptide-1 agonists and pioglitazone), lifestyle strategies (intermittent fasting, ketogenic metabolic therapy and exercise) and theoretical models (mitochondrial dysfunction, ‘allostatic load model’ and ‘selfish brain hypothesis’). We summarize the interventions, their observed outcomes, and a ranked assessment of the current level of evidence and class of recommendation. This reflects that some metabolic-based approaches show promising results from clinical studies while other emerging strategies remain too preliminary or inconsistent to draw conclusions, underscoring the need for further trials in humans. We conclude by briefly discussing practical and safety considerations, identifying limitations in the current literature, and propose future directions for building a more integrated model of mental and metabolic health.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$79.00 per year
only $6.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Chesney, E., Goodwin, G. M. & Fazel, S. Risks of all-cause and suicide mortality in mental disorders: a meta-review. World Psychiatry 13, 153–160 (2014).
Nielsen, R. E., Banner, J. & Jensen, S. E. Cardiovascular disease in patients with severe mental illness. Nat. Rev. Cardiol. 18, 136–145 (2021).
Penninx, B. W. J. H. & Lange, S. M. M. Metabolic syndrome in psychiatric patients: overview, mechanisms, and implications. Dialogues Clin. Neurosci. 20, 63–73 (2018).
Blázquez, E. et al. Significance of brain glucose hypometabolism, altered insulin signal transduction, and insulin resistance in several neurological diseases. Front. Endocrinol. 13, 873301 (2022).
Nousen, E. K., Franco, J. G. & Sullivan, E. L. Unraveling the mechanisms responsible for the comorbidity between metabolic syndrome and mental health disorders. Neuroendocrinology 98, 254–266 (2013).
Gao, X. et al. Genetic evidence for the causal relations between metabolic syndrome and psychiatric disorders: a Mendelian randomization study. Transl. Psychiatry 14, 46 (2024).
Berk, M., Forbes, M. & Narayanaswamy, J. The history of biomarkers in psychiatry: lessons learned, lessons forgotten, lessons rediscovered. Br. J. Psychiatry 227, 437–438 (2025).
Henkel, N. D. et al. Schizophrenia: a disorder of broken brain bioenergetics. Mol. Psychiatry 27, 2393–2404 (2022).
Vancampfort, D. et al. Metabolic syndrome and metabolic abnormalities in bipolar disorder: a meta-analysis of prevalence rates and moderators. Am. J. Psychiatry 170, 265–274 (2013).
Goldstein, B. I. et al. The burden of obesity among adults with bipolar disorder in the United States. Bipolar Disord. 13, 387–395 (2011).
Penninx, B. Immuno-metabolic depression: from concept to implementation. Eur. Psychiatry 66, S11 (2023).
Cen, M. et al. Associations between metabolic syndrome and anxiety, and the mediating role of inflammation: findings from the UK Biobank. Brain Behav. Immun. 116, 1–9 (2024).
Michopoulos, V., Vester, A. & Neigh, G. Posttraumatic stress disorder: a metabolic disorder in disguise? Exp. Neurol. 284, 220–229 (2016).
Vancampfort, D. et al. The prevalence of metabolic syndrome in alcohol use disorders: a systematic review and meta-analysis. Alcohol Alcohol. 51, 515–521 (2016).
Yu, Z. & Muehleman, V. Eating disorders and metabolic diseases. Int. J. Environ. Res. Public Health 20, 2446 (2023).
Perry, B. I., McIntosh, G., Weich, S., Singh, S. & Rees, K. The association between first-episode psychosis and abnormal glycaemic control: systematic review and meta-analysis. Lancet Psychiatry 3, 1049–1058 (2016).
Pereira, A. C. P. et al. Genetic association and sequencing of the insulin-like growth factor 1 gene in bipolar affective disorder. Am. J. Med. Genet. 156, 177–187 (2011).
Perry, B. I. et al. Longitudinal trends in childhood insulin levels and body mass index and associations with risks of psychosis and depression in young adults. JAMA Psychiatry 78, 416–425 (2021).
Kamintsky, L. et al. Blood–brain barrier imaging as a potential biomarker for bipolar disorder progression. Neuroimage Clin. 26, 102049 (2020).
Hajek, T. et al. Insulin resistance, diabetes mellitus, and brain structure in bipolar disorders. Neuropsychopharmacology 39, 2910–2918 (2014).
Fernandes, B. S. et al. Insulin resistance in depression: a large meta-analysis of metabolic parameters and variation. Neurosci. Biobehav. Rev. 139, 104758 (2022).
Toalson, P., Ahmed, S., Hardy, T. & Kabinoff, G. The metabolic syndrome in patients with severe mental illnesses. Prim. Care Companion J. Clin. Psychiatry 6, 152–158 (2004).
Arnold, S. E. et al. Brain insulin resistance in type 2 diabetes and Alzheimer disease: concepts and conundrums. Nat. Rev. Neurol. 14, 168–181 (2018).
Kleinridders, A., Ferris, H. A., Cai, W. & Kahn, C. R. Insulin action in brain regulates systemic metabolism and brain function. Diabetes 63, 2232–2243 (2014).
Mehdi, S., Wani, S. U. D., Krishna, K. L., Kinattingal, N. & Roohi, T. F. A review on linking stress, depression, and insulin resistance via low-grade chronic inflammation. Biochem. Biophys. Rep. 36, 101571 (2023).
Nasca, C. et al. Insulin receptor substrate in brain-enriched exosomes in subjects with major depression: on the path of creation of biosignatures of central insulin resistance. Mol. Psychiatry 26, 5140–5149 (2021).
Sapolsky, R. M. The possibility of neurotoxicity in the hippocampus in major depression: a primer on neuron death. Biol. Psychiatry 48, 755–765 (2000).
Silver, D. et al. Glucose biomarkers and antidepressant response: a scoping review of interventions targeting major depressive disorder and metabolic dysfunction. J. Affect. Disord. 395, 120606 (2025).
Fentie, D. & Yibabie, S. Magnitude and associated factors of dyslipidemia among patients with severe mental illness in dire Dawa, Ethiopia: neglected public health concern. BMC Cardiovasc. Disord. 23, 298 (2023).
Watson, K. T. et al. Association of insulin resistance with depression severity and remission status: defining a metabolic endophenotype of depression. JAMA Psychiatry 78, 439 (2021).
Watson, K. T. et al. Incident major depressive disorder predicted by three measures of insulin resistance: a Dutch cohort study. Am. J. Psychiatry 178, 914–920 (2021).
Rosmond, R., Dallman, M. F. & Björntorp, P. Stress-related cortisol secretion in men: relationships with abdominal obesity and endocrine, metabolic and hemodynamic abnormalities. J. Clin. Endocrinol. Metab. 83, 1853–1859 (1998).
Miura, H. et al. A link between stress and depression: shifts in the balance between the kynurenine and serotonin pathways of tryptophan metabolism and the etiology and pathophysiology of depression. Stress 11, 198–209 (2008).
Lawson, E. A. et al. Leptin levels are associated with decreased depressive symptoms in women across the weight spectrum, independent of body fat. Clin. Endocrinol. 76, 520–525 (2012).
Zou, X. et al. Role of leptin in mood disorder and neurodegenerative disease. Front. Neurosci. 13, 378 (2019).
Harris, J. J., Jolivet, R. & Attwell, D. Synaptic energy use and supply. Neuron 75, 762–777 (2012).
Li, C. T. et al. Prefrontal glucose metabolism in medication-resistant major depression. Br. J. Psychiatry 206, 316–323 (2015).
Goyal, M. S. & Raichle, M. E. Glucose requirements of the developing human brain. J. Pediatr. Gastroenterol. Nutr. 66, S46–S49 (2018).
Beltrán, F. A., Acuña, A. I., Miró, M. P. & Castro, M. A. in Neuroscience—Dealing with Frontiers (ed. Contreras, C. M.) Ch. 14 (InTechOpen, 2012).
Magistretti, P. J. & Pellerin, L. Cellular mechanisms of brain energy metabolism and their relevance to functional brain imaging. Phil. Trans. R. Soc. Lond. B 354, 1155–1163 (1999).
Kann, O., Papageorgiou, I. E. & Draguhn, A. Highly energized inhibitory interneurons are a central element for information processing in cortical networks. J. Cereb. Blood Flow Metab. 34, 1270–1282 (2014).
Peng, P., Wang, Q., Lang, X. E., Liu, T. & Zhang, X.-Y. Association between thyroid dysfunction, metabolic disturbances, and clinical symptoms in first-episode, untreated Chinese patients with major depressive disorder: undirected and Bayesian network analyses. Front. Endocrinol. 14, 1138233 (2023).
Andreazza, A. C. et al. Brain and body energy metabolism and potential for treatment of psychiatric disorders. Nat. Ment. Health 3, 763–771 (2025).
Wertheim-Tysarowska, K., Gos, M., Sykut-Cegielska, J. & Bal, J. Genetic analysis in inherited metabolic disorders—from diagnosis to treatment. Own experience, current state of knowledge and perspectives. Dev. Period Med. 19, 413–431 (2015).
Henchcliffe, C. & Beal, M. F. Mitochondrial biology and oxidative stress in Parkinson disease pathogenesis. Nat. Rev. Neurol. 4, 600–609 (2008).
Zuccoli, G. S., Saia-Cereda, V. M., Nascimento, J. M. & Martins-de-Souza, D. The energy metabolism dysfunction in psychiatric disorders postmortem brains: focus on proteomic evidence. Front. Neurosci. 11, 493 (2017).
McPherson, P. A. C. & McEneny, J. The biochemistry of ketogenesis and its role in weight management, neurological disease and oxidative stress. J. Physiol. Biochem. 68, 141–151 (2012).
Rog, J. et al. The potential role of the ketogenic diet in serious mental illness: current evidence, safety, and practical advice. J. Clin. Med. 13, 2819 (2024).
Chaturvedi, S. et al. Human metabolic enzymes deficiency: a genetic mutation based approach. Scientifica 2016, 9828672 (2016).
Patel, K. P. et al. The spectrum of pyruvate dehydrogenase complex deficiency: clinical, biochemical and genetic features in 371 patients. Mol. Genet. Metab. 106, 385–394 (2012).
Du, F. et al. In vivo evidence for cerebral bioenergetic abnormalities in schizophrenia measured using 31 P magnetization transfer spectroscopy. JAMA Psychiatry 71, 19 (2014).
Altar, C. A. et al. Deficient hippocampal neuron expression of proteasome, ubiquitin, and mitochondrial genes in multiple schizophrenia cohorts. Biol. Psychiatry 58, 85–96 (2005).
Rowland, L. M. et al. Elevated brain lactate in schizophrenia: a 7 T magnetic resonance spectroscopy study. Transl. Psychiatry 6, e967 (2016).
Sullivan, C. R. et al. Measurement of lactate levels in postmortem brain, iPSCs, and animal models of schizophrenia. Sci. Rep. 9, 5087 (2019).
Campbell, I. & Campbell, H. A pyruvate dehydrogenase complex disorder hypothesis for bipolar disorder. Med. Hypotheses 130, 109263 (2019).
Sullivan, C. R., O’Donovan, S. M., McCullumsmith, R. E. & Ramsey, A. Defects in bioenergetic coupling in schizophrenia. Biol. Psychiatry 83, 739–750 (2018).
Morris, G. et al. A model of the mitochondrial basis of bipolar disorder. Neurosci. Biobehav. Rev. 74, 1–20 (2017).
Campbell, I. H. & Campbell, H. The metabolic overdrive hypothesis: hyperglycolysis and glutaminolysis in bipolar mania. Mol. Psychiatry (2024).
Sullivan, C. R. et al. Neuron-specific deficits of bioenergetic processes in the dorsolateral prefrontal cortex in schizophrenia. Mol. Psychiatry 24, 1319–1328 (2019).
Middleton, F. A., Mirnics, K., Pierri, J. N., Lewis, D. A. & Levitt, P. Gene expression profiling reveals alterations of specific metabolic pathways in schizophrenia. J. Neurosci. 22, 2718–2729 (2002).
Bubber, P., Hartounian, V., Gibson, G. E. & Blass, J. P. Abnormalities in the tricarboxylic acid (TCA) cycle in the brains of schizophrenia patients. Eur. Neuropsychopharmacol. 21, 254–260 (2011).
Allen, J. et al. Mitochondria and mood: mitochondrial dysfunction as a key player in the manifestation of depression. Front. Neurosci. 12, 386 (2018).
Ait Tayeb, A. E. K. et al. Plasma acetyl-L-carnitine and L-carnitine in major depressive episodes: a case–control study before and after treatment. Psychol. Med. 53, 2307–2316 (2023).
Kirkbride, J. B. et al. The social determinants of mental health and disorder: evidence, prevention and recommendations. World Psychiatry 23, 58–90 (2024).
National Academies of Sciences, Engineering, and Medicine; Division of Behavioral and Social Sciences and Education; Board on Children, Youth, and Families; Committee on Fostering Healthy Mental, Emotional, and Behavioral Development Among Children and Youth. Fostering Healthy Mental, Emotional, and Behavioral Development in Children and Youth: A National Agenda (National Academies Press, 2019).
Correll, C. U. et al. Prevalence, incidence and mortality from cardiovascular disease in patients with pooled and specific severe mental illness: a large-scale meta-analysis of 3,211,768 patients and 113,383,368 controls. World Psychiatry 16, 163–180 (2017).
Baron, K. G. & Reid, K. J. Circadian misalignment and health. Int. Rev. Psychiatry 26, 139–154 (2014).
Suvarna, B. et al. Health risk behaviours and allostatic load: a systematic review. Neurosci. Biobehav. Rev. 108, 694–711 (2020).
Shen, Q. et al. Psychiatric disorders and subsequent risk of cardiovascular disease: a longitudinal matched cohort study across three countries. EClinicalMedicine 61, 102063 (2023).
Sarnyai, Z. & Ben-Shachar, D. Schizophrenia, a disease of impaired dynamic metabolic flexibility: a new mechanistic framework. Psychiatry Res. 342, 116220 (2024).
Seeman, T. E. et al. Price of adaptation–allostatic load and its health consequences. MacArthur studies of successful aging. Arch. Intern. Med. 157, 2259–2268 (1997).
Finlay, S., Rudd, D., McDermott, B. & Sarnyai, Z. Allostatic load and systemic comorbidities in psychiatric disorders. Psychoneuroendocrinology 140, 105726 (2022).
Kelly, D. P. et al. The allostatic triage model of psychopathology (ATP Model): how reallocation of brain energetic resources under stress elicits psychiatric symptoms. Neurosci. Biobehav. Rev. 179, 106419 (2025).
Peters, A. et al. The selfish brain: competition for energy resources. Neurosci. Biobehavi. Rev. 28, 143–180 (2004).
Peters, A., McEwen, B. S. & Friston, K. Uncertainty and stress: why it causes diseases and how it is mastered by the brain. Prog. Neurobiol. 156, 164–188 (2017).
Peters, A. & McEwen, B. S. Editorial introduction. Physiol. Behav. 106, 1–4 (2012).
Peters, A. & McEwen, B. S. Stress habituation, body shape and cardiovascular mortality. Neurosci. Biobehav. Rev. 56, 139–150 (2015).
Pillinger, T., McCutcheon, R., Siskind, D. & Srivastava, V. Holistic prevention and management of physical health side-effects of psychotropic medication: second report of the Lancet Psychiatry Physical Health Commission. Lancet Psychiatry 12, 673–699 (2025).
Ballon, J. S. et al. Molecular pathophysiology of metabolic effects of antipsychotic medications. Trends Endocrinol. Metabol. 25, 593–600 (2014).
Chang, H. H. et al. The role of valproate in metabolic disturbances in bipolar disorder patients. J. Affect. Disord. 124, 319–323 (2010).
Chaudhry, N., Patidar, Y. & Puri, V. Mitochondrial myopathy, encephalopathy, lactic acidosis, and stroke-like episodes unveiled by valproate. J. Pediatr. Neurosci. 8, 135 (2013).
Campbell, I. H., Campbell, H. & Smith, D. J. Insulin signaling as a therapeutic mechanism of lithium in bipolar disorder. Transl. Psychiatry 12, 350 (2022).
Teasdale, S. B. et al. Implementing lifestyle interventions in mental health care: third report of the Lancet Psychiatry Physical Health Commission. Lancet. Psychiatry 12, 700–722 (2025).
Calkin, C. V. et al. Treating insulin resistance with metformin as a strategy to improve clinical outcomes in treatment-resistant bipolar depression (the TRIO-BD Study): a randomized, quadruple-masked, placebo-controlled clinical trial. J. Clin. Psychiatry 83, (2022).
Hamal, C. et al. Metformin for the improvement of comorbid depression symptoms in diabetic patients: a systematic review. Cureus (2022).
Karnewar, S. et al. Metformin regulates mitochondrial biogenesis and senescence through AMPK mediated H3K79 methylation: relevance in age-associated vascular dysfunction. Biochim. Biophys. Acta 1864, 1115–1128 (2018).
Vizuete, A. F. K. et al. A mechanism of action of metformin in the brain: prevention of methylglyoxal-induced glutamatergic impairment in acute hippocampal slices. Mol. Neurobiol. 61, 3223–3239 (2024).
Correll, C. U. et al. Metformin add-on vs. antipsychotic switch vs. continued antipsychotic treatment plus healthy lifestyle education in overweight or obese youth with severe mental illness: results from the IMPACT trial. World Psychiatry 19, 69–80 (2020).
Athauda, D. & Foltynie, T. The glucagon-like peptide 1 (GLP) receptor as a therapeutic target in Parkinson’s disease: mechanisms of action. Drug Discov. Today 21, 802–818 (2016).
Reich, N. & Hölscher, C. The neuroprotective effects of glucagon-like peptide 1 in Alzheimer’s and Parkinson’s disease: an in-depth review. Front. Neurosci. 16, 970925 (2022).
De Giorgi, R. et al. An analysis on the role of glucagon-like peptide-1 receptor agonists in cognitive and mental health disorders. Nat. Mental Health 3, 354–373 (2025).
Norgaard, C. H. et al. Treatment with glucagon-like peptide-1 receptor agonists and incidence of dementia: data from pooled double-blind randomized controlled trials and nationwide disease and prescription registers. Alzheimers Dement. 8, e12268 (2022).
Klausen, M. K. et al. Exenatide once weekly for alcohol use disorder investigated in a randomized, placebo-controlled clinical trial. JCI Insight 7, e159863 (2022).
Khaity, A. et al. Glucagon-like peptide-1 receptor-agonists treatment for cardio-metabolic parameters in schizophrenia patients: a systematic review and meta-analysis. Front. Psychiatry 14, 1153648 (2023).
Mansur, R. B. et al. Liraglutide promotes improvements in objective measures of cognitive dysfunction in individuals with mood disorders: a pilot, open-label study. J. Affect. Disord. 207, 114–120 (2017).
Mansur, R. B. et al. Treatment with a GLP-1R agonist over four weeks promotes weight loss-moderated changes in frontal-striatal brain structures in individuals with mood disorders. Eur. Neuropsychopharmacol. 27, 1153–1162 (2017).
McElroy, S. L. et al. Liraglutide in obese or overweight individuals with stable bipolar disorder. J. Clin. Psychopharmacol. 44, 89–95 (2024).
Richards, J. et al. Successful treatment of binge eating disorder with the GLP-1 agonist semaglutide: a retrospective cohort study. Obes. Pillars 7, 100080 (2023).
Smith, R. C. et al. Effects of pioglitazone on metabolic abnormalities, psychopathology, and cognitive function in schizophrenic patients treated with antipsychotic medication: a randomized double-blind study. Schizophr. Res. 143, 18–24 (2013).
Kemp, D. E. et al. Use of insulin sensitizers for the treatment of major depressive disorder: a pilot study of pioglitazone for major depression accompanied by abdominal obesity. J. Affect. Disord. 136, 1164–1173 (2012).
Kullmann, S. et al. Brain insulin resistance at the crossroads of metabolic and cognitive disorders in humans. Physiol. Rev. 96, 1169–1209 (2016).
Brocchi, A. et al. Effects of intermittent fasting on brain metabolism. Nutrients 14, 1275 (2022).
Paoli, A. et al. Common and divergent molecular mechanisms of fasting and ketogenic diets. Trends Endocrinol. Metab. 35, 125–141 (2024).
Mattson, M. P. The cyclic metabolic switching theory of intermittent fasting. Nat. Metab. 7, 665–678 (2025).
Kapogiannis, D. et al. Brain responses to intermittent fasting and the healthy living diet in older adults. Cell Metab. 36, 1668–1678.e5 (2024).
Yannakoulia, M. & Scarmeas, N. Diets. N. Engl. J. Med. 390, 2098–2106 (2024).
Miller, V. J., Villamena, F. A. & Volek, J. S. Nutritional ketosis and mitohormesis: potential implications for mitochondrial function and human health. J. Nutr. Metab. 2018, 5157645 (2018).
Dhillon, K. K. & Gupta, S. Biochemistry, Ketogenesis (StatPearls Publishing, 2025).
Jensen, N. J., Wodschow, H. Z., Nilsson, M. & Rungby, J. Effects of ketone bodies on brain metabolism and function in neurodegenerative diseases. Int. J. Mol. Sci. 21, 8767 (2020).
Bough, K. J. et al. Mitochondrial biogenesis in the anticonvulsant mechanism of the ketogenic diet. Ann. Neurol. 60, 223–235 (2006).
Düking, T. et al. Ketogenic diet uncovers differential metabolic plasticity of brain cells. Sci. Adv. 8, 7639 (2022).
Janssen-Aguilar, R. et al. Ketogenic diets and depression and anxiety: a systematic review and meta-analysis. JAMA Psychiatry https://doi.org/10.1001/jamapsychiatry.2025.3261 (2025).
Ene, H. M., Karry, R., Farfara, D. & Ben-Shachar, D. Mitochondria play an essential role in the trajectory of adolescent neurodevelopment and behavior in adulthood: evidence from a schizophrenia rat model. Mol. Psychiatry 28, 1170–1181 (2023).
Castro Monteiro, F. et al. Physical activity and sedentary behavior levels among individuals with mental illness: a cross-sectional study from 23 countries. PLoS ONE 19, e0301583 (2024).
Chekroud, S. R. et al. Association between physical exercise and mental health in 1.2 million individuals in the USA between 2011 and 2015: a cross-sectional study. Lancet Psychiatry 5, 739–746 (2018).
Vancampfort, D. et al. The efficacy, mechanisms and implementation of physical activity as an adjunctive treatment in mental disorders: a meta-review of outcomes, neurobiology and key determinants. World Psychiatry 24, 227–239 (2025).
Marx, W. et al. Clinical guidelines for the use of lifestyle-based mental health care in major depressive disorder: World Federation of Societies for Biological Psychiatry (WFSBP) and Australasian Society of Lifestyle Medicine (ASLM) taskforce. World J. Biol. Psychiatry 24, 333–386 (2023).
Maurus, I. et al. EPA guidance on lifestyle interventions for adults with severe mental illness: a meta-review of the evidence. Eur. Psychiatry 67, e80 (2024).
Firth, J. et al. A meta-review of “lifestyle psychiatry”: the role of exercise, smoking, diet and sleep in the prevention and treatment of mental disorders. World Psychiatry 19, 360–380 (2020).
Halperin, J. L. et al. Further evolution of the ACC/AHA clinical practice guideline recommendation classification system: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 67, 1572–1574 (2016).
Qin, Z. J. et al. Metformin for neurocognitive dysfunction in schizophrenia: a systematic review. Front. Psychiatry 15, 1540153 (2025).
Zeinoddini, A. et al. Pioglitazone adjunctive therapy for depressive episode of bipolar disorder: a randomized, double-blind, placebo-controlled trial. Depress. Anxiety 32, 167–173 (2015).
Teicholz, N. et al. Myths and facts regarding low-carbohydrate diets. Nutrients 17, 1047 (2025).
Boltri, M. et al. Keto therapy—unveiling the potential of ketogenic diet in psychiatric care: a scoping review. Nutrition 134, 112710 (2025).
Hurtado, I., Robles, C., Peiró, S., García-Sempere, A. & Sanfélix-Gimeno, G. Association of glucagon-like peptide-1 receptor agonists with suicidal ideation and self-injury in individuals with diabetes and obesity: a propensity-weighted, population-based cohort study. Diabetologia 67, 2471–2480 (2024).
Strumila, R. et al. GLP-1 agonists and risk of suicidal thoughts and behaviours: confound by indication once again? A narrative review. Eur. Neuropsychopharmacol. 87, 29–34 (2024).
Davis, H. A., Smith, Z. R. & Smith, G. T. Longitudinal transactions between negative urgency and fasting predict binge eating. Appetite 192, 107113 (2024).
Heun, R. A systematic review on the effect of Ramadan on mental health: minor effects and no harm in general, but increased risk of relapse in schizophrenia and bipolar disorder. Glob. Psychiatry 1, 7–16 (2018).
Saner, E. et al. TOWARD: a metabolic health intervention that improves food addiction and binge eating symptoms. Front. Psychiatry 16, 1612551 (2025).
Sethi, S. et al. Ketogenic diet intervention on metabolic and psychiatric health in bipolar and schizophrenia: a pilot trial. Psychiatry Res. 335, 115866 (2024).
Carmen, M. et al. Treating binge eating and food addiction symptoms with low-carbohydrate ketogenic diets: a case series. J. Eat. Disord. 8, 2 (2020).
Alberti, K. G. et al. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 120, 1640–1645 (2009).
World Health Organization. Obesity: Preventing and Managing the Global Epidemic Technical Report Series (2000).
DeFronzo, R. A. & Tripathy, D. Skeletal muscle insulin resistance is the primary defect in type 2 diabetes. Diabetes Care 32, S157–S163 (2009).
Grundy, S. M. et al. Definition of metabolic syndrome: report of the National Heart, Lung, and Blood Institute/American Heart Association conference on scientific issues related to definition. Circulation 109, 433–438 (2004).
ElSayed, N. A. et al. 2. Classification and diagnosis of diabetes: standards of care in diabetes—2023. Diabetes Care 46, S19–S40 (2023).
Nunnari, J. & Suomalainen, A. Mitochondria: in sickness and in health. Cell 148, 1145–1159 (2012).
Sethi, S. et al. Metabolic psychiatry: a conceptual framework targeting metabolic dysfunction in psychiatric disease. Preprint at OSF https://osf.io/preprints/psyarxiv/wm8g3_v1 (2024).
Acknowledgements
We wish to acknowledge P-Squared Philanthropies, Baszucki Brain Research Fund (BBRF), Dharma Fund, and Sobrano Fund for philanthropically supporting our work at Stanford Metabolic Psychiatry. Thank you to BBRF and Stellate Communications for helping support the design of the manuscript figure.
Author information
Authors and Affiliations
Contributions
S.S. and Z.S. developed the concept and drafted the paper. M.B., A.C.A., L.R.M.-P., I.C., H.C., C.L., N.R., J.V., C.V.C., J.F., R.M., S.C., T.L., D.D’A. and M.F. critically revised the paper for important intellectual content.
Corresponding authors
Ethics declarations
Competing interests
S.S. has served as a scientific advisor for Found Health and is a co-founder of Metabolic Psychiatry Labs. Z.S. is chief scientist at Ally Sciences Ltd. M.B. has served as a consultant to AstraZeneca, Otsuka Pharmaceutical, Glaxosmithkline, Janssen Cilag, Lundbeck Merck and Servier. L.R.M.-P. is a cofounder of Neuroblox. S.C. has served as an adviser to Abbott Laboratories, Cargill and holds a patent with Nestle Health Sciences. The other authors declare no competing interests.
Peer review
Peer review information
Nature Mental Health thanks Riccardo De Giorgi, Luigi Saccaro and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Fig. 1 and Supplementary Tables 1–3.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sethi, S., Berk, M., Andreazza, A.C. et al. Metabolic psychiatry targeting metabolic dysregulation in mental health. Nat. Mental Health (2026). https://doi.org/10.1038/s44220-026-00609-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44220-026-00609-5