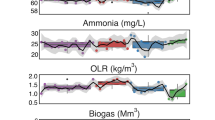
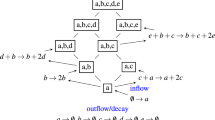
Abstract
Microbial consortia hold immense promise for wastewater treatment by harnessing metabolite exchange-based interspecies interactions to drive energy and matter flow. Yet, challenges in the coordinated modulation of transmembrane transport proteins and extracellular metabolite distribution limit cross-species interactions within consortia. Here we propose a mesospace-domain regulation strategy that leverages hydrogel-assembled mesoscale habitats to precisely modulate β-barrel membrane porins and locally enrich cross-fed molecules, thus remodelling interspecific cooperative metabolism for efficient wastewater treatment. Confining a carefully designed microbiota within mesospace enhances organic wastewater treatment for hexanoate production, achieving a 307.2% higher yield than unconfined systems. This improvement is attributed to mesospace-governed porin regulation and exometabolite enrichment, which reprogram interbacterial interactions from unidirectional electron transfer to bidirectional multimetabolite cross-feeding. This regulatory strategy is also applicable to other wastewater treatment systems, markedly enhancing succinic acid production, denitrification of low carbon-to-nitrogen ratio wastewater and removal of emerging contaminants. These findings illuminate how the mesospace domain orchestrates microbiota metabolism to boost bioconversion efficiency and selectivity for sustainable wastewater management.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data generated in this study are included in the Article and its Supplementary Information. Proteomic raw data have been deposited to the ProteomeXchange Consortium through the iProX partner repository with the dataset identifier PXD057916. The 16S rRNA sequencing data have been deposited to the National Center for Biotechnology Information short-read archive under accession no. PRJNA1377884. Source data are provided with this paper.
References
Flemming, H.-C. & Wuertz, S. Bacteria and archaea on Earth and their abundance in biofilms. Nat. Rev. Microbiol. 17, 247–260 (2019).
Kost, C., Patil, K. R., Friedman, J., Garcia, S. L. & Ralser, M. Metabolic exchanges are ubiquitous in natural microbial communities. Nat. Microbiol. 8, 2244–2252 (2023).
Wang, M. et al. Enhanced anaerobic wastewater treatment by a binary electroactive material: pseudocapacitance/conductance-mediated microbial interspecies electron transfer. Environ. Sci. Technol. 57, 12072–12082 (2023).
Raghoebarsing, A. A. et al. A microbial consortium couples anaerobic methane oxidation to denitrification. Nature 440, 918–921 (2006).
Zhao, C. et al. Metabolite cross-feeding promoting NADH production and electron transfer during efficient SMX biodegradation by a denitrifier and S. oneidensis MR-1 in the presence of nitrate. Environ. Sci. Technol. 57, 18306–18316 (2023).
D’Souza, G. et al. Ecology and evolution of metabolic cross-feeding interactions in bacteria. Nat. Prod. Rep. 35, 455–488 (2018).
Liu, X. et al. Syntrophic interspecies electron transfer drives carbon fixation and growth by Rhodopseudomonas palustris under dark, anoxic conditions. Sci. Adv. 7, eabh1852 (2021).
Tsoi, R. et al. Metabolic division of labor in microbial systems. Proc. Natl Acad. Sci. USA 115, 2526–2531 (2018).
Ren, S. et al. Hydrochar-facilitated anaerobic digestion: Evidence for direct interspecies electron transfer mediated through surface oxygen-containing functional groups. Environ. Sci. Technol. 54, 5755–5766 (2020).
Zhang, P., Zhang, J., Zhang, T., Zhang, L. & He, Y. Zero-valent iron enhanced methane production of anaerobic digestion by reinforcing microbial electron bifurcation coupled with direct inter-species electron transfer. Water Res. 255, 121428 (2024).
Sokolovskaya, O. M., Shelton, A. N. & Taga, M. E. Sharing vitamins: cobamides unveil microbial interactions. Science 369, eaba0165 (2020).
Nikaido, H. & Saier, M. H. Jr Transport proteins in bacteria: common themes in their design. Science 258, 936–942 (1992).
Nikaido, H. Molecular basis of bacterial outer membrane permeability revisited. Microbiol. Mol. Biol. Rev. 67, 593–656 (2003).
Cowan, S. et al. The structure of OmpF porin in a tetragonal crystal form. Structure 3, 1041–1050 (1995).
Fritts, R. K., McCully, A. L. & McKinlay, J. B. Extracellular metabolism sets the table for microbial cross-feeding. Microbiol. Mol. Bio. Rev. 85, e00135–20 (2021).
Kong, L. et al. Cross-feeding between filamentous cyanobacteria and symbiotic bacteria favors rapid photogranulation. Environ. Sci. Technol. 57, 16953–16963 (2023).
Pronk, M. et al. Full scale performance of the aerobic granular sludge process for sewage treatment. Water Res. 84, 207–217 (2015).
Zhou, Y. et al. Mass flow and metabolic pathway of nonaeration greywater treatment in an oxygenic microalgal–bacterial biofilm. Environ. Sci. Technol. 58, 534–544 (2023).
Wang, Y., Gao, H. & Wells, G. F. Integrated omics analyses reveal differential gene expression and potential for cooperation between denitrifying polyphosphate and glycogen accumulating organisms. Environ. Microbiol. 23, 3274–3293 (2021).
Cheng, C. et al. Electricity-enhanced anaerobic, non-photosynthetic mixotrophy by Clostridium carboxidivorans with increased carbon efficiency and alcohol production. Energy Convers. Manage. 252, 115118 (2022).
Li, F. et al. Modular engineering to increase intracellular NAD(H/+) promotes rate of extracellular electron transfer of Shewanella oneidensis. Nat. Commun. 9, 3637 (2018).
Wirth, S. & Dürre, P. Investigation of putative genes for the production of medium-chained acids and alcohols in autotrophic acetogenic bacteria. Metab. Eng. 66, 296–307 (2021).
Shen, S. et al. Effect of temperature and surfactant on biomass growth and higher-alcohol production during syngas fermentation by Clostridium carboxidivorans P7. Bioresour. Bioprocess. 7, 1–13 (2020).
Marsili, E. et al. Shewanella secretes flavins that mediate extracellular electron transfer. Proc. Natl Acad. Sci. USA 105, 3968–3973 (2008).
Waldrop, G. L., Holden, H. M. & Maurice, M. S. The enzymes of biotin dependent CO2 metabolism: what structures reveal about their reaction mechanisms. Protein Sci. 21, 1597–1619 (2012).
Appel, A. M. et al. Frontiers, opportunities, and challenges in biochemical and chemical catalysis of CO2 fixation. Chem. Rev. 113, 6621–6658 (2013).
Liu, Z., Wang, K., Chen, Y., Tan, T. & Nielsen, J. Third-generation biorefineries as the means to produce fuels and chemicals from CO2. Nat. Catal. 3, 274–288 (2020).
Waters, C. M. & Bassler, B. L. Quorum sensing: cell-to-cell communication in bacteria. Annu. Rev. Cell Dev. Biol. 21, 319–346 (2005).
Mukherjee, S. & Bassler, B. L. Bacterial quorum sensing in complex and dynamically changing environments. Nat. Rev. Microbiol. 17, 371–382 (2019).
Abraham, M. J. et al. GROMACS: High performance molecular simulations through multi-level parallelism from laptops to supercomputers. SoftwareX 1–2, 19–25 (2015).
Tombola, R., Buttiglieri, G., Auset, M. & Gonzalez-Olmos, R. Recycled corrugated wire hose cover as biological carriers for greywater treatment in a sequential batch biofilm reactor. J. Environ. Manage. 240, 475–484 (2019).
Yang, X., Zhou, Y., Zhang, L., Benally, C. & Liu, Y. Enhanced biofilm formation and municipal wastewater treatment efficiency using granular activated carbon modified bio-ball carriers in moving bed biofilm reactor. Bioresour. Technol. 435, 132947 (2025).
Li, Y. et al. Microbial engineering for the production of C2–C6 organic acids. Nat. Prod. Rep. 38, 1518–1546 (2021).
Hoff, B. et al. Unlocking nature’s biosynthetic power—metabolic engineering for the fermentative production of chemicals. Angew. Chem. Int. Ed. 60, 2258–2278 (2020).
Dueholm, M. K. D. et al. MiDAS 4: A global catalogue of full-length 16S rRNA gene sequences and taxonomy for studies of bacterial communities in wastewater treatment plants. Nat. Commun. 13, 1908 (2022).
Ye, J. et al. Wastewater denitrification driven by mechanical energy through cellular piezo-sensitization. Nat. Water 2, 531–540 (2024).
Ravishankara, A., Daniel, J. S. & Portmann, R. W. Nitrous oxide (N2O): the dominant ozone-depleting substance emitted in the 21st century. Science 326, 123–125 (2009).
Canfield, D. E., Glazer, A. N. & Falkowski, P. G. The evolution and future of Earth’s nitrogen cycle. Science 330, 192–196 (2010).
Zhang, W., Guan, A., Peng, Q., Qi, W. & Qu, J. Microbe-mediated simultaneous nitrogen reduction and sulfamethoxazole/N-acetylsulfamethoxazole removal in lab-scale constructed wetlands. Water Res. 242, 120233 (2023).
Choe, D. et al. Adaptive laboratory evolution of a genome-reduced Escherichia coli. Nat. Commun. 10, 935 (2019).
Moon, E. C. et al. Physiological cost of antibiotic resistance: insights from a ribosome variant in bacteria. Sci. Adv. 10, eadq5249 (2024).
Hu, G. et al. Light-driven CO2 sequestration in Escherichia coli to achieve theoretical yield of chemicals. Nat. Catal. 4, 395–406 (2021).
Cui, W. et al. Gut microbial metabolite facilitates colorectal cancer development via ferroptosis inhibition. Nat. Cell Biol. 26, 124–137 (2024).
Cooper, R. E. et al. Iron is not everything: Unexpected complex metabolic responses between iron–cycling microorganisms. ISME J 14, 2675–2690 (2020).
Lindorff-Larsen, K. et al. Improved side-chain torsion potentials for the Amber ff99SB protein force field. J. Comput. Chem. 78, 1950–1958 (2010).
Wang, J., Wolf, R. M., Caldwell, J. W., Kollman, P. A. & Case, D. A. Development and testing of a General Amber Force Field. J. Comput. Chem. 25, 1157–1174 (2004).
Eberhardt, J., Santos-Martins, D., Tillack, A. F. & Forli, S. AutoDock Vina 1.2. 0: new docking methods, expanded force field, and Python bindings. J. Comput. Chem. 61, 3891–3898 (2021).
Jo, S., Kim, T., Iyer, V. G. & Im, W. CHARMM-GUI: a web-based graphical user interface for CHARMM. J. Comput. Chem. 29, 1859–1865 (2008).
Kumar, S., Rosenberg, J. M., Bouzida, D., Swendsen, R. H. & Kollman, P. A. The weighted histogram analysis method for free-energy calculations on biomolecules. I. The method. J. Comput. Chem. 13, 1011–1021 (1992).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant nos. 52300172 to C.L. and 52470053 to Y.C.), the National Key R&D Program of China (grant no. 2025YFE0208000 to Y.C.), the Shanghai Rising-Star Program (grant no. 23YF1448900 to C.L.) and the China Postdoctoral Science Foundation Program (grant nos. 2024T170665 and 2022M722396 to C.L.).
Author information
Authors and Affiliations
Contributions
C.L. and Y.C. developed the research concept, designed experiments, discussed the results and wrote the manuscript; C.L., Y.Y., X.Z. and M.Z. performed the experiments and data analyses; B.P., Q.Z. and J.W. provided constructive advice on experiments and result interpretation; W.-M.W., H.R. and H.-Q.Y. assisted in research design and manuscript editing. All authors discussed, commented on and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Water thanks Liangyan Rong, Qidong Yin and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Texts 1–28, Figs. 1–30, Tables 1–15 and References.
Supplementary Video 1 (download MP4 )
The SMD simulation of a serine molecule being pulled through the OmpF.
Supplementary Video 2 (download MP4 )
The SMD simulation of a serine molecule being pulled through the AI-2-docked OmpF.
Supplementary Data 1 (download XLSX )
Source data for Supplementary Figs. 1, 2, 4–9, 11–19, 21–23 and 25–30.
Source data
Source Data Fig. 2 (download XLSX )
Source data for Fig. 2.
Source Data Fig. 3 (download XLSX )
Source data for Fig. 3.
Source Data Fig. 4 (download XLSX )
Source data for Fig. 4.
Source Data Fig. 5 (download XLSX )
Source data for Fig. 5.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, C., Yin, Y., Zhang, X. et al. Mesospace domain orchestrates microbial consortia by β-barrel porin modulation and local molecule enrichment for wastewater treatment. Nat Water 4, 294–306 (2026). https://doi.org/10.1038/s44221-025-00579-5
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44221-025-00579-5