Abstract
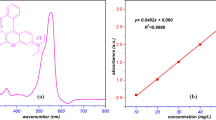
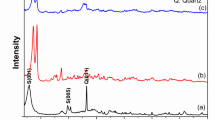
The efficient separation and simultaneous detection of thorium from complex aqueous environments remain a critical challenge in radioactive waste management, where minimizing secondary waste and reducing adsorbent consumption are paramount. Here we report a fluorochromic metal–organic framework (MOF), Eu-NDC, that functions as a self-indicating adsorbent for ultraselective Th(IV) sensing and separation. Eu-NDC exhibits a distinctive red-to-blue emission transition upon Th(IV) binding, achieving an ultralow detection limit of 9.2 nM while selectively distinguishing Th(IV) from other tetravalent cations. Furthermore, the material combines high Th(IV) adsorption capacity (504.3 mg g−1) with superior selectivity, indicated by a distribution coefficient of 2.8 × 106 ml g−1 and a Th(IV)/U(VI) separation factor of 1,806. Mechanistic studies reveal that Th(IV) uptake occurs via a dissolution–recrystallization process. Importantly, the fluorochromic response of Eu-NDC provides a self-indicating capability, whereby changes in emission colour directly reflect adsorption progress. This dual-functionality, integrating ultraselective adsorption with direct optical feedback, establishes MOFs as a multifunctional platform for smart, self-indicating radionuclide separation and environmental remediation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available in the article and its Supplementary Information. The X-ray crystallographic coordinates for structures reported in this study have been deposited at the Cambridge Crystallographic Data Centre (CCDC), under deposition numbers 2485502-2485503. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif. Source data are provided with this paper.
References
Katz, J. J., Morss, L. R., Edelstein, N. M. & Fuger J. The Chemistry of the Actinide and Transactinide Elements (Springer, 2011).
Maes, S., Zhuang, W.-Q., Rabaey, K., Alvarez-Cohen, L. & Hennebel, T. Concomitant leaching and electrochemical extraction of rare earth elements from monazite. Environ. Sci. Technol. 51, 1654–1661 (2017).
Zhu, Z. W., Pranolo, Y. & Cheng, C. Y. Separation of uranium and thorium from rare earths for rare earth production—a review. Miner. Eng. 77, 185–196 (2015).
Guidelines for Drinking-Water Quality 4th edn (World Health Organization, 2022).
Toxicological Profile for Thorium (Agency for Toxic Substances and Disease Registry, 2019).
Kumar, R. et al. Historical overview and recent progress on supramolecular sensors for thorium recognition. TrAC, Trends Anal. Chem. 172, 117551 (2024).
Mei, S. et al. Assembling a heterobimetallic actinide metal–organic framework by a reaction-induced preorganization strategy. Angew. Chem. Int. Ed. 62, e202306360 (2023).
Hu, Q. et al. AIE and ICT synergistic lysosome-targeted ratiometric fluorescence sensor for the detection and imaging of Th4+ in the liver of zebrafish and mice. Anal. Chem. 97, 6101–6110 (2025).
Song, A.-M. et al. Rational designed metal–organic framework with nanocavity traps for selectively recognizing and separating of radioactive thorium in rare earth wastewater. Adv. Funct. Mater. 34, 2406932 (2024).
Liu, W. et al. Ratiometric monitoring of thorium contamination in natural water using a dual-emission luminescent europium organic framework. Environ. Sci. Technol. 53, 332–341 (2019).
Roozbahani, G. M. et al. Computation-assisted nanopore detection of thorium ions. Anal. Chem. 90, 5938–5944 (2018).
Kumar, S. R. et al. Development of the smartphone-assisted colorimetric detection of thorium by using new Schiff’s base and its applications to real time samples. Inorg. Chem. 57, 15270–15279 (2018).
Xie, F., Zhang, T. A., Dreisinger, D. & Doyle, F. A critical review on solvent extraction of rare earths from aqueous solutions. Miner. Eng. 56, 10–28 (2014).
Xu, L. et al. Selective capture mechanism of radioactive thorium from highly acidic solution by a layered metal sulfide. ACS Appl. Mater. Interfaces 13, 37308–37315 (2021).
Yuan, L.-Y. et al. Introduction of bifunctional groups into mesoporous silica for enhancing uptake of thorium(IV) from aqueous solution. ACS Appl. Mater. Interfaces 6, 4786–4796 (2014).
Liu, X. et al. Efficient and selective capture of thorium ions by a covalent organic framework. Nat. Commun. 14, 5097 (2023).
Song, A.-M. et al. N,O-interlocked cavity-structured functionalized metal-organic frameworks for one-step efficient removal of highly toxic thorium from tailings wastewater. Chem. Eng J. 516, 164093 (2025).
Tang, J. et al. Efficient separation of radium from natural thorium using a mesoporous silica-supported composite resin with sulfonic acid groups for the acquisition of targeted α-nuclides 212Pb. Chem. Eng J. 485, 150022 (2024).
Zhang, Y. et al. Selective separation of thorium from rare earth ions using bisphosphonate-functionalized ionic single crystals co-self-assembled via π–π and ionic interactions. Nano Lett. 25, 7665–7672 (2025).
Taylor, R. Reprocessing and Recycling of Spent Nuclear Fuel (Woodhead Publishing, 2015).
Ye, Y. et al. Spontaneous electrochemical uranium extraction from wastewater with net electrical energy production. Nat. Water 1, 887–898 (2023).
Yuan, Y. et al. High-capacity uranium extraction from seawater through constructing synergistic multiple dynamic bonds. Nat. Water 3, 89–98 (2025).
Chen, S. et al. Electron-buffering rechargeable microelectrode adsorbents for rapid environmental remediation of uranium-containing wastewater. Nat. Water 3, 937–948 (2025).
Xie, Y. et al. Recent progress of radionuclides separation by porous materials. Sci. China Chem. 67, 3515–3577 (2024).
Sun, Q., Aguila, B. & Ma, S. Opportunities of porous organic polymers for radionuclide sequestration. Trends Chem. 1, 292–303 (2019).
Bao, W. et al. Cutting-edge characterization techniques to decipher adsorption mechanisms of radionuclides and heavy metals. Coord. Chem. Rev. 539, 216748 (2025).
Bayat, M., Mardani, H., Roghani-Mamaqani, H. & Hoogenboom, R. Self-indicating polymers: a pathway to intelligent materials. Chem. Soc. Rev. 53, 4045–4085 (2024).
Liu, T. et al. Recyclable robust plastic scintillation resin achieving the exceptional separation and detection of technetium-99. Adv. Sci. 12, 2411523 (2024).
Wang, J. et al. Miniaturized separation-sensing tandem enabled by fluorescent monoliths. Angew. Chem. Int. Ed. 64, e202502020 (2025).
Li, L. et al. Seamless integration of rapid separation and ultrasensitive detection for complex biological samples using multistage annular functionalized carbon nanotube arrays. Adv. Mater. 36, 2312518 (2024).
Chen, M. et al. Nanoscale metal–organic frameworks that are both fluorescent and hollow for self-indicating drug delivery. ACS Appl. Mater. Interfaces 13, 18554–18562 (2021).
Sharma, M. et al. Breaking new ground: innovative adsorbents for uranium and thorium ions removal and environmental cleanup. Coord. Chem. Rev. 517, 216008 (2024).
Delgado Friedrichs, O., O’Keeffe, M. & Yaghi, O. M. Three-periodic nets and tilings: regular and quasiregular nets. Acta Crystallogr A 59, 22–27 (2003).
Xue, D.-X. et al. Tunable rare earth fcu-MOF platform: access to adsorption kinetics driven gas/vapor separations via pore size contraction. J. Am. Chem. Soc. 137, 5034–5040 (2015).
Howarth, A. J. et al. Chemical, thermal and mechanical stabilities of metal–organic frameworks. Nat. Rev. Mater. 1, 15018 (2016).
Dong, Y. et al. Ultrastable ceramic-based metal–organic framework membranes with missing linkers for robust desalination. Nat. Water 2, 464–474 (2024).
Liang, R.-R. et al. A robust pyrazolate metal–organic framework for integrated perfluorooctanoic acid concentration and degradation. Nat. Water 2, 1218–1225 (2024).
Wang, X., Jiang, Y., Tissot, A. & Serre, C. Luminescent sensing platforms based on lanthanide metal-organic frameworks: current strategies and perspectives. Coord. Chem. Rev. 497, 215454 (2023).
Cui, Y., Yue, Y., Qian, G. & Chen, B. Luminescent functional metal–organic frameworks. Chem. Rev. 112, 1126–1162 (2012).
Bünzli, J.-C. G. Lanthanide luminescence for biomedical analyses and imaging. Chem. Rev. 110, 2729–2755 (2010).
Du, X., Xie, H., Qin, T., Yuan, Y. & Wang, N. Ultrasensitive optical detection of strontium ions by specific nanosensor with ultrahigh binding affinity. Nat. Commun. 15, 6530 (2024).
Cui, Y. et al. Dissolution–recrystallization: a novel mechanism for fluorochromic detection of Th4+ using color-tunable luminescent metal–organic frameworks. Angew. Chem. Int. Ed. 63, e202410453 (2024).
Pearson, R. G. Hard and soft acids and bases—the evolution of a chemical concept. Coord. Chem. Rev. 100, 403–425 (1990).
Rönfeldt, P. et al. Water-based synthesis and properties of a scandium 1,4-naphthalenedicarboxylate. Z. Anorg. Allg. Chem. 646, 1373–1379 (2020).
Wei, Z. et al. Rigidifying fluorescent linkers by metal–organic framework formation for fluorescence blue shift and quantum yield enhancement. J. Am. Chem. Soc. 136, 8269–8276 (2014).
Wang, J. & Guo, X. Adsorption kinetic models: physical meanings, applications, and solving methods. J. Hazard. Mater. 390, 122156 (2020).
Park, K. C. et al. Capture instead of release: defect-modulated radionuclide leaching kinetics in metal–organic frameworks. J. Am. Chem. Soc. 144, 16139–16149 (2022).
Chiron, N., Guilet, R. & Deydier, E. Adsorption of Cu(II) and Pb(II) onto a grafted silica: isotherms and kinetic models. Water Res. 37, 3079–3086 (2003).
Hassan, A. et al. Ultrafast removal of thorium and uranium from radioactive waste and groundwater using highly efficient and radiation-resistant functionalized triptycene-based porous organic polymers. ACS Appl. Mater. Interfaces 16, 24547–24561 (2024).
Xiong, X. H. et al. Selective extraction of thorium from uranium and rare earth elements using sulfonated covalent organic framework and its membrane derivate. Chem. Eng J. 384, 123240 (2020).
Yang, M.-J. et al. One-step open-ring strategy for constructing sandwich-like metal–organic framework with highly selective removal of thorium. Chem. Eng J. 505, 159718 (2025).
Zhong, W. Y. et al. Ultrahigh-efficient N,O-functionalized covalent organic framework towards thorium adsorption from uranium and rare earth elements. Sep. Purif. Technol. 347, 127603 (2024).
Chen, X. et al. β-ray irradiation resistant MOF-based trap for efficient capture of Th(IV) ion. Sep. Purif. Technol. 297, 121517 (2022).
He, N. et al. Constructing ordered and tunable extrinsic porosity in covalent organic frameworks via water-mediated soft-template strategy. Nat. Commun. 15, 3896 (2024).
Lu, H. et al. A new concept of radiation detection based on a fluorochromic and piezochromic nanocluster. J. Am. Chem. Soc. 144, 3449–3457 (2022).
Lu, H. et al. Visible colorimetric dosimetry of UV and ionizing radiations by a dual-module photochromic nanocluster. Nat. Commun. 12, 2798 (2021).
Kong, S. et al. Intrinsic narrowband blue phosphorescent materials and their applications in 3D printed self-monitoring microfluidic chips. Adv. Mater. 36, 2412468 (2024).
Sheldrick, G. M. SHELXT - integrated space-group and crystal-structure determination. Acta Crystallogr. Sect. A 71, 3–8 (2015).
Sheldrick, G. M. Crystal structure refinement with SHELXL. Acta Crystallogr. Sect. C 71, 3–8 (2015).
Dolomanov, O. V., Bourhis, L. J., Gildea, R. J., Howard, J. A. K. & Puschmann, H. OLEX2: a complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 42, 339–341 (2009).
Spek, A. PLATON SQUEEZE: a tool for the calculation of the disordered solvent contribution to the calculated structure factors. Acta Crystallogr. Sect. C 71, 9–18 (2015).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant nos. 22322609, U22B20139 and 22506141) and the New Cornerstone Science Foundation through the New Cornerstone Investigator Program and the XPLORER PRIZE. SCXRD data were collected using a Bruker D8 Quest diffractometer in the State Key Laboratory of Multiphase Flow in Power Engineering. We thank Z. Pan at the National Innovation Platform for Industry-Education Integration of Energy Storage Technology of Xi’an Jiaotong University for assistance with variable-temperature PXRD and SEM-EDS measurements.
Author information
Authors and Affiliations
Contributions
J.L. and S.W. conceived and supervised the project. Y.C. participated in all aspects of the study. Y.B., J.Y. and G.Z. conducted the Th(IV) adsorption experiments. C.Y. performed the Th(IV) detection experiments. J.Q. and Y.W. contributed to the study of the detection mechanism. J.L., Y.C. and S.W. wrote the paper. All authors analysed the results and provided critical feedback on the paper.
Corresponding authors
Ethics declarations
Competing interests
J.L. and Y.C., in conjunction with Xi’an Jiaotong University, have filed a patent (202511992028.2) in China based on the results presented in this study. The other authors declare no competing interests.
Peer review
Peer review information
Nature Water thanks Xiaojun Wang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–27, Tables 1–6, Discussion and References.
Supplementary Data 1 (download XLSX )
Supplementary source data for Supplementary Figs. 1–26.
Source data
Source Data Figs. 1–5 (download XLSX )
Source data for Figs. 1–5.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cui, Y., Bai, Y., Yang, J. et al. A smart self-indicating metal–organic framework with real-time fluorochromic response for ultraselective thorium remediation. Nat Water 4, 521–531 (2026). https://doi.org/10.1038/s44221-026-00612-1
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44221-026-00612-1