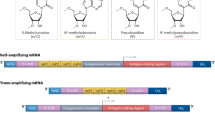
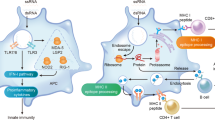
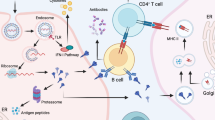
Abstract
mRNA vaccines benefit from rapid design, scalable manufacturing and strong safety profiles, and they are being preclinically and clinically explored for various infectious diseases, cancer and autoimmune disorders. However, global access to mRNA vaccines remains limited by technical, logistical, economic, regulatory and ethical challenges. In this Review, we discuss the current mRNA vaccine landscape, from approved products to emerging candidates, and outline key barriers to equitable access. We highlight engineering strategies to address these barriers, including thermostability improvements, alternative administration routes, new delivery systems and alternative RNA platforms, such as self-amplifying and circular RNA, as well as the integration of artificial intelligence- and machine learning-based design and optimization. We also discuss the regulatory, ethical and social dimensions of vaccine deployment, as well as the importance of building public trust and community engagement. Coordinated technical and policy advances are essential to remove barriers, enhance access and advance global health equity.
Key points
-
The success of COVID-19 mRNA vaccines established a robust platform that is now expanding to other infectious diseases and non-infectious diseases, including cancer and autoimmune disorders.
-
The full potential of mRNA vaccines can only be realized through broad accessibility.
-
Expanding mRNA vaccine access requires integrated engineering strategies, including thermostability improvements, alternative delivery routes, new carriers and RNA platforms, such as self-amplifying and circular constructs.
-
Artificial intelligence-driven and machine learning-driven mRNA vaccine design and optimization could shorten iteration times, accelerate progress from development to scalable manufacturing and support expanded access.
-
Equitable mRNA vaccine access requires coordinated policy, regulatory and community actions that harmonize global regulations, secure sustained global funding, and engagement of communities to address vaccine hesitancy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Chaudhary, N., Weissman, D. & Whitehead, K. A. mRNA vaccines for infectious diseases: principles, delivery and clinical translation. Nat. Rev. Drug Discov. 20, 817–838 (2021).
Leong, K. Y., Tham, S. K. & Poh, C. L. Revolutionizing immunization: a comprehensive review of mRNA vaccine technology and applications. Virol. J. 22, 71 (2025).
Fang, E. et al. Advances in COVID-19 mRNA vaccine development. Signal Transduct. Target. Ther. 7, 94 (2022).
Baden, L. R. et al. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N. Engl. J. Med. 384, 403–416 (2021).
Polack, F. P. et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N. Engl. J. Med. 383, 2603–2615 (2020).
Pardi, N., Hogan, M. J., Porter, F. W. & Weissman, D. mRNA vaccines—a new era in vaccinology. Nat. Rev. Drug Discov. 17, 261–279 (2018).
Sahin, U., Karikó, K. & Türeci, Ö. mRNA-based therapeutics — developing a new class of drugs. Nat. Rev. Drug Discov. 13, 759–780 (2014).
Qin, S. et al. mRNA-based therapeutics: powerful and versatile tools to combat diseases. Signal Transduct. Target. Ther. 7, 166 (2022).
Barbier, A. J., Jiang, A. Y., Zhang, P., Wooster, R. & Anderson, D. G. The clinical progress of mRNA vaccines and immunotherapies. Nat. Biotechnol. 40, 840–854 (2022).
Hou, X., Zaks, T., Langer, R. & Dong, Y. Lipid nanoparticles for mRNA delivery. Nat. Rev. Mater. 6, 1078–1094 (2021).
Li, S., Zheng, L., Zhong, J. & Gao, X. Advancing mRNA vaccines for infectious diseases: key components, innovations, and clinical progress. Essays Biochem. https://doi.org/10.1042/ebc20253009 (2025).
Pardi, N. & Krammer, F. mRNA vaccines for infectious diseases — advances, challenges and opportunities. Nat. Rev. Drug Discov. 23, 838–861 (2024).
Liu, C. et al. mRNA-based cancer therapeutics. Nat. Rev. Cancer 23, 526–543 (2023).
Sayour, E. J., Boczkowski, D., Mitchell, D. A. & Nair, S. K. Cancer mRNA vaccines: clinical advances and future opportunities. Nat. Rev. Clin. Oncol. 21, 489–500 (2024).
Deyhimfar, R. et al. The clinical impact of mRNA therapeutics in the treatment of cancers, infections, genetic disorders, and autoimmune diseases. Heliyon 10, e26971 (2024).
Youssef, M., Hitti, C., Puppin Chaves Fulber, J. & Kamen, A. A. Enabling mRNA therapeutics: current landscape and challenges in manufacturing. Biomolecules 13, 1497 (2023).
Rosa, S. S., Prazeres, D. M. F., Azevedo, A. M. & Marques, M. P. C. mRNA vaccines manufacturing: challenges and bottlenecks. Vaccine 39, 2190–2200 (2021).
Wells, C. R. & Galvani, A. P. The global impact of disproportionate vaccination coverage on COVID-19 mortality. Lancet Infect. Dis. 22, 1254–1255 (2022).
Street, A., Kersaudy Kerhoas, M. & Ndlovu, Z. From equitable access to equitable innovation: rethinking bioengineering for global health. Nat. Rev. Bioeng. 2, 444–446 (2024).
Dzau, V. et al. The 100 days mission: how a new medical-countermeasures network can deliver equity and innovation. Lancet 402, 1507–1510 (2023).
Zhang, G., Tang, T., Chen, Y., Huang, X. & Liang, T. mRNA vaccines in disease prevention and treatment. Signal Transduct. Target. Ther. 8, 365 (2023).
May, M. After COVID-19 successes, researchers push to develop mRNA vaccines for other diseases. Nat. Med. 27, 930–932 (2021).
Paczkowska, A. et al. The application of mRNA technology for vaccine production—current state of knowledge. Vaccines 13, 389 (2025).
Kaylor, A. Moderna’s COVID-19 vaccine mNEXSPIKE gains FDA approval. TechTarget https://www.techtarget.com/pharmalifesciences/news/366625275/Modernas-COVID-19-vaccine-mNEXSPIKE-gains-FDA-approval (2 June 2025).
Saraf, A. et al. An Omicron-specific, self-amplifying mRNA booster vaccine for COVID-19: a phase 2/3 randomized trial. Nat. Med. 30, 1363–1372 (2024).
Dolgin, E. Self-copying RNA vaccine wins approval: what’s next? Nature 624, 14 (2023).
Silva-Pilipich, N., Beloki, U., Salaberry, L. & Smerdou, C. Self-amplifying RNA: a second revolution of mRNA vaccines against COVID-19. Vaccines 12, 318 (2024).
Wilson, E. et al. Efficacy and safety of an mRNA-based RSV PreF vaccine in older adults. N. Engl. J. Med. 389, 2233–2244 (2023).
Chuang, Y.-M. et al. A mosquito AgTRIO mRNA vaccine contributes to immunity against malaria. npj Vaccines 8, 88 (2023).
Matarazzo, L. & Bettencourt, P. J. G. mRNA vaccines: a new opportunity for malaria, tuberculosis and HIV. Front. Immunol. 14, 1172691 (2023).
Vidal, S. J. et al. Mining the CD4 antigen repertoire for next-generation tuberculosis vaccines. Cell https://doi.org/10.1016/j.cell.2025.08.027 (2025). This article reports a systematic CD4+ T cell antigen screen that identifies a trivalent mRNA–LNP tuberculosis vaccine candidate with translational potential.
Rudman Spergel, A. K. et al. mRNA-based seasonal influenza and SARS-CoV-2 multicomponent vaccine in healthy adults: a phase 1/2 trial. Nat. Med. 31, 1484–1493 (2025).
Zubair, A., Ahmad, H., Arif, M. M. & Ali, M. mRNA vaccines against HIV: hopes and challenges. HIV Med. 26, 824–838 (2025).
Lusso, P. in Trends in mRNA Vaccine Research (eds Szabo, G. T. & Pardi, N.) 259–283 (Wiley, 2025).
Cevaal, P. M. et al. Efficient mRNA delivery to resting T cells to reverse HIV latency. Nat. Commun. 16, 4979 (2025).
Morris, L. mRNA vaccines offer hope for HIV. Nat. Med. 27, 2082–2084 (2021).
Willis, J. R. et al. Vaccination with mRNA-encoded nanoparticles drives early maturation of HIV bnAb precursors in humans. Science 389, eadr8382 (2025).
Parks, K. R. et al. Vaccination with mRNA-encoded membrane-anchored HIV envelope trimers elicited tier 2 neutralizing antibodies in a phase 1 clinical trial. Sci. Transl. Med. 17, eady6831 (2025).
Shim, I., Rogowski, L. & Venketaraman, V. Progress and recent developments in HIV vaccine research. Vaccines 13, 690 (2025).
Fan, T. et al. Therapeutic cancer vaccines: advancements, challenges and prospects. Signal Transduct. Target. Ther. 8, 450 (2023).
Lorentzen, C. L., Haanen, J. B., Met, Ö. & Svane, I. M. Clinical advances and ongoing trials of mRNA vaccines for cancer treatment. Lancet Oncol. 23, e450–e458 (2022).
Goyal, F. et al. Advancing cancer immunotherapy: the potential of mRNA vaccines as a promising therapeutic approach. Adv. Ther. 7, 2300255 (2024).
Türeci, Ö. et al. Targeting the heterogeneity of cancer with individualized neoepitope vaccines. Clin. Cancer Res. 22, 1885–1896 (2016).
Weber, J. S. et al. Individualised neoantigen therapy mRNA-4157 (V940) plus pembrolizumab versus pembrolizumab monotherapy in resected melanoma (KEYNOTE-942): a randomised, phase 2b study. Lancet 403, 632–644 (2024). This article reports results from a randomized phase IIb trial showing that an individualized neoantigen mRNA vaccine (mRNA-4157/V940) combined with pembrolizumab improves recurrence-free survival compared with pembrolizumab alone in patients with resected high-risk melanoma.
Rojas, L. A. et al. Personalized RNA neoantigen vaccines stimulate T cells in pancreatic cancer. Nature 618, 144–150 (2023).
Damle, S. R., Pillarisetty, V. G., Safyan, R. A. & Chiorean, E. G. A new dawn in cancer immunotherapy: the promise of mutant KRAS-specific vaccines. Transl. Gastroenterol. Hepatol. 10, 20 (2025).
Fu, Q. et al. mRNA vaccines in the context of cancer treatment: from concept to application. J. Transl. Med. 23, 12 (2025).
Fu, R. et al. Combination therapy with oncolytic virus and T cells or mRNA vaccine amplifies antitumor effects. Signal Transduct. Target. Ther. 9, 118 (2024).
Shariati, A. et al. mRNA cancer vaccines from bench to bedside: a new era in cancer immunotherapy. Biomark. Res. 12, 157 (2024).
Rohner, E., Yang, R., Foo, K. S., Goedel, A. & Chien, K. R. Unlocking the promise of mRNA therapeutics. Nat. Biotechnol. 40, 1586–1600 (2022).
Koeberl, D. et al. Interim analyses of a first-in-human phase 1/2 mRNA trial for propionic acidaemia. Nature 628, 872–877 (2024).
Attarwala, H., Lumley, M., Liang, M., Ivaturi, V. & Senn, J. Translational pharmacokinetic/pharmacodynamic model for mRNA-3927, an investigational therapeutic for the treatment of propionic acidemia. Nucleic Acid Ther. 33, 141–147 (2023).
Baek, R. et al. Characterizing the mechanism of action for mRNA therapeutics for the treatment of propionic acidemia, methylmalonic acidemia, and phenylketonuria. Nat. Commun. 15, 3804 (2024).
Geller, D. E., Crowley, C., Froehlich, J., Schwabe, C. & O’Carroll, M. WS10.03 Inhaled LUNAR®-CFTR mRNA (ARCT-032) is safe and well-tolerated: a phase 1 study. J. Cyst. Fibros. 23, S19 (2024).
Rowe, S. M. et al. Inhaled mRNA therapy for treatment of cystic fibrosis: interim results of a randomized, double-blind, placebo-controlled phase 1/2 clinical study. J. Cyst. Fibros. 22, 656–664 (2023).
Żak, M. M. & Zangi, L. Clinical development of therapeutic mRNA applications. Mol. Ther. 33, 2583–2609 (2025).
Zouali, M. Inverse vaccination for autoimmune diseases: insights into the role of B lymphocytes. Cells 14, 1085 (2025).
Zala, A. & Thomas, R. Antigen-specific immunotherapy to restore antigen-specific tolerance in type 1 diabetes and Graves’ disease. Clin. Exp. Immunol. 211, 164–175 (2023).
Chen, J. et al. LNP-mRNA vaccine prevents type 1 diabetes in non-obese diabetes mice. J. Control. Release 375, 513–523 (2024).
Krienke, C. et al. A noninflammatory mRNA vaccine for treatment of experimental autoimmune encephalomyelitis. Science 371, 145–153 (2021).
Liu, Y. et al. Generation of tolerogenic antigen-presenting cells in vivo via the delivery of mRNA encoding PDL1 within lipid nanoparticles. Nat. Biomed. Eng. https://doi.org/10.1038/s41551-025-01373-0 (2025). This article reports the in vivo generation of tolerogenic antigen-presenting cells through LNP delivery of mRNA encoding PD-L1, suppressing T cell activation and preventing disease progression in mouse models of autoimmune disease.
Liric Rajlic, I. et al. Translational kinetic-pharmacodynamics of mRNA-6231, an investigational mRNA therapeutic encoding mutein interleukin-2. CPT Pharmacomet. Syst. Pharmacol. 13, 1067–1078 (2024).
Shi, Y., Shi, M., Wang, Y. & You, J. Progress and prospects of mRNA-based drugs in pre-clinical and clinical applications. Signal Transduct. Target. Ther. 9, 322 (2024).
Hewitt, S. L. et al. Durable anticancer immunity from intratumoral administration of IL-23, IL-36γ, and OX40L mRNAs. Sci. Transl. Med. 11, eaat9143 (2019).
Hamouda, A. E. I. et al. Intratumoral delivery of lipid nanoparticle-formulated mRNA encoding IL-21, IL-7, and 4-1BBL induces systemic anti-tumor immunity. Nat. Commun. 15, 10635 (2024).
Li, Z. et al. Local administration of mRNA encoding cytokine cocktail confers potent anti-tumor immunity. Front. Immunol. 15, 1455019 (2024).
Gote, V. et al. A comprehensive review of mRNA vaccines. Int. J. Mol. Sci. 24, 2700 (2023).
Yeh, T.-F., Lin, C. & Sung, H.-C. A review of technological developments in lipid nanoparticle application for mRNA vaccination. Hum. Vaccines Immunother. 19, 2256040 (2023).
Lu, Y. et al. Advancing mRNA vaccines: a comprehensive review of design, delivery, and efficacy in infectious diseases. Int. J. Biol. Macromol. 319, 145501 (2025).
Kim, Y.-A. et al. Computational design of mRNA vaccines. Vaccine 42, 1831–1840 (2024).
Dolgin, E. ‘Remarkable’ AI tool designs mRNA vaccines that are more potent and stable. Nature https://doi.org/10.1038/d41586-023-01487-y (2023).
Castillo-Hair, S. M. & Seelig, G. Machine learning for designing next-generation mRNA therapeutics. Acc. Chem. Res. 55, 24–34 (2022).
Castillo-Hair, S. et al. Optimizing 5′UTRs for mRNA-delivered gene editing using deep learning. Nat. Commun. 15, 5284 (2024).
Kosuri, S. & Church, G. M. Large-scale de novo DNA synthesis: technologies and applications. Nat. Methods 11, 499–507 (2014).
Bang, D. & Church, G. M. Gene synthesis by circular assembly amplification. Nat. Methods 5, 37–39 (2008).
Karikó, K., Muramatsu, H., Ludwig, J. & Weissman, D. Generating the optimal mRNA for therapy: HPLC purification eliminates immune activation and improves translation of nucleoside-modified, protein-encoding mRNA. Nucleic Acids Res. 39, e142 (2011).
Jackson, N. A. C., Kester, K. E., Casimiro, D., Gurunathan, S. & DeRosa, F. The promise of mRNA vaccines: a biotech and industrial perspective. npj Vaccines 5, 11 (2020).
Verbeke, R., Lentacker, I., De Smedt, S. C. & Dewitte, H. The dawn of mRNA vaccines: the COVID-19 case. J. Control. Release 333, 511–520 (2021).
Schoenmaker, L. et al. mRNA-lipid nanoparticle COVID-19 vaccines: structure and stability. Int. J. Pharm. 601, 120586 (2021).
Verbeke, R., Lentacker, I., De Smedt, S. C. & Dewitte, H. Three decades of messenger RNA vaccine development. Nano Today 28, 100766 (2019).
Roces, C. B. et al. Manufacturing considerations for the development of lipid nanoparticles using microfluidics. Pharmaceutics 12, 1095 (2020).
Cullis, P. R. & Hope, M. J. Lipid nanoparticle systems for enabling gene therapies. Mol. Ther. 25, 1467–1475 (2017).
Crommelin, D. J. A., Anchordoquy, T. J., Volkin, D. B., Jiskoot, W. & Mastrobattista, E. Addressing the cold reality of mRNA vaccine stability. J. Pharm. Sci. 110, 997–1001 (2021).
Wouters, O. J. et al. Challenges in ensuring global access to COVID-19 vaccines: production, affordability, allocation, and deployment. Lancet 397, 1023–1034 (2021).
Kis, Z., Kontoravdi, C., Dey, A. K., Shattock, R. & Shah, N. Rapid development and deployment of high-volume vaccines for pandemic response. J. Adv. Manuf. Process. 2, e10060 (2020).
vander Straeten, A. et al. A microneedle vaccine printer for thermostable COVID-19 mRNA vaccines. Nat. Biotechnol. 42, 510–517 (2024). This article reports a standalone microneedle vaccine printer that enables decentralized manufacture of thermostable mRNA–LNP COVID-19 vaccines without cold-chain requirements.
Han, J. et al. On-patient medical record and mRNA therapeutics using intradermal microneedles. Nat. Mater. 24, 794–803 (2025). This article reports a quantum dot-encoded on-patient medical record platform using microneedles delivering mRNA–LNP vaccine, enabling reliable tracking of vaccine administration in low-resource settings.
Skerritt, J. H., Tucek-Szabo, C., Sutton, B. & Nolan, T. The platform technology approach to mRNA product development and regulation. Vaccines 12, 528 (2024).
Knezevic, I., Liu, M. A., Peden, K., Zhou, T. & Kang, H.-N. Development of mRNA vaccines: scientific and regulatory issues. Vaccines 9, 81 (2021).
Skerritt, J. H. Considerations for mRNA product development, regulation and deployment across the lifecycle. Vaccines 13, 473 (2025).
Daniel, S., Kis, Z., Kontoravdi, C. & Shah, N. Quality by design for enabling RNA platform production processes. Trends Biotechnol. 40, 1213–1228 (2022).
Gunter, H. M. et al. mRNA vaccine quality analysis using RNA sequencing. Nat. Commun. 14, 5663 (2023).
Jones, C. H. et al. Breaking the mold with RNA—a “RNAissance” of life science. npj Genom. Med. 9, 2 (2024).
Patel, P. et al. Regulatory agilities impacting review timelines for Pfizer/BioNTech’s BNT162b2 mRNA COVID-19 vaccine: a retrospective study. Front. Med. 10, 1275817 (2023).
Bouderhem, R. Challenges faced by states and the WHO in efficiently regulating the use of mRNA vaccines. Med. Sci. Forum 26, 1 (2024).
Cavaleri, M. et al. A roadmap for fostering timely regulatory and ethics approvals of international clinical trials in support of global health research systems. Lancet Glob. Health 13, e769–e777 (2025).
Chisholm, O. & Critchley, H. Future directions in regulatory affairs. Front. Med. 9, 1082384 (2022).
Slaoui, M. & Hepburn, M. Developing safe and effective covid vaccines — operation warp speed’s strategy and approach. N. Engl. J. Med. 383, 1701–1703 (2020).
Rathore, A. S. & Winkle, H. Quality by design for biopharmaceuticals. Nat. Biotechnol. 27, 26–34 (2009).
Bouderhem, R. Access to COVID-19 vaccines: a new global approach. Vaccines 10, 1795 (2022).
Zosso-Pavic, M., Li, Q., Atiek, E., Wolfer, A. & Rohr, U.-P. Effect of Project Orbis participation by the Swiss regulator on submission gaps, review times, and drug approval decisions between 2020 and 2022: a comparative analysis. Lancet Oncol. 25, 770–778 (2024).
Algorri, M. et al. Meeting report: advancing accelerated regulatory review with Real-Time Oncology Review (RTOR), Project Orbis, and the Product Quality Assessment Aid. AAPS Open 8, 19 (2022).
de Claro, R. A. et al. Project Orbis: global collaborative review program. Clin. Cancer Res. 26, 6412–6416 (2020).
Paremoer, L. & Pollock, A. “A passion to change the landscape and drive a renaissance”: the mRNA Hub at Afrigen as decolonial aspiration. Front. Public Health 10, 1065993 (2022).
Herder, M. & Benavides, X. ‘Our project, your problem?’ A case study of the WHO’s mRNA technology transfer programme in South Africa. PLoS Glob. Public Health 4, e0003173 (2024).
Akanmori, B. D. et al. The role of the African Vaccine Regulatory Forum (AVAREF) in the accelerated clinical evaluation of Ebola vaccine candidates during the large West Africa epidemic. J. Immunol. Sci. 2, 75–79 (2018).
Abdulwahab, A. A. et al. The African medicines agency and medicines regulation: progress, challenges, and recommendations. Health Care Sci. 3, 350–359 (2024).
Wairagkar, N. et al. The African medicines agency — a potential gamechanger that requires strategic focus. PLOS Glob. Public Health 5, e0004276 (2025).
Wairagkar, N. et al. Regulatory reforms will boost African vaccine production and access. Nat. Med. 30, 3409–3412 (2024).
Hood, S., Campbell, B. & Baker, K. Culturally Informed Community Engagement: Implications for Inclusive Science and Health Equity (RTI, 2023).
Lythgoe, M. P. & Middleton, P. Comparison of COVID-19 vaccine approvals at the US Food and Drug Administration, European Medicines Agency, and Health Canada. JAMA Netw. Open 4, e2114531 (2021).
Suphap, S., Luksameesate, P., Nerapusee, O. & Anantachoti, P. A scoping review of authorisation pathway for COVID-19 vaccines among selected countries. J. Pharm. Policy Pract. 18, 2520861 (2025).
Lurie, N., Saville, M., Hatchett, R. & Halton, J. Developing Covid-19 vaccines at pandemic speed. N. Engl. J. Med. 382, 1969–1973 (2020).
Cheuvart, B. et al. Harmonizing the collection of solicited adverse events in prophylactic vaccine clinical trials. Expert Rev. Vaccines 22, 849–859 (2023).
Moshkovits, I. & Shepshelovich, D. Emergency use authorizations of COVID-19-related medical products. JAMA Intern. Med. 182, 228–229 (2022).
Chen, D. & Kristensen, D. Opportunities and challenges of developing thermostable vaccines. Expert Rev. Vaccines 8, 547–557 (2009).
Karlsson, L. C. et al. Fearing the disease or the vaccine: the case of COVID-19. Pers. Individ. Differ. 172, 110590 (2021).
Karlsson, L. C. et al. The association between vaccination confidence, vaccination behavior, and willingness to recommend vaccines among Finnish healthcare workers. PLoS One 14, e0224330 (2019).
Giurgea, L. T., Han, A. & Memoli, M. J. Universal coronavirus vaccines: the time to start is now. npj Vaccines 5, 43 (2020).
McCall, B. COVID-19 and artificial intelligence: protecting health-care workers and curbing the spread. Lancet Digit. Health 2, e166–e167 (2020).
Mei, X. et al. Artificial intelligence–enabled rapid diagnosis of patients with COVID-19. Nat. Med. 26, 1224–1228 (2020).
Budd, J. et al. Digital technologies in the public-health response to COVID-19. Nat. Med. 26, 1183–1192 (2020).
Parvin, N., Joo, S. W. & Mandal, T. K. Enhancing vaccine efficacy and stability: a review of the utilization of nanoparticles in mRNA vaccines. Biomolecules 14, 1036 (2024).
Suzuki, Y. & Ishihara, H. Difference in the lipid nanoparticle technology employed in three approved siRNA (Patisiran) and mRNA (COVID-19 vaccine) drugs. Drug Metab. Pharmacokinet. 41, 100424 (2021).
Arte, K. S. et al. Recent advances in drying and development of solid formulations for stable mRNA and siRNA lipid nanoparticles. J. Pharm. Sci. 114, 805–815 (2025).
Ball, R. L., Bajaj, P. & Whitehead, K. A. Achieving long-term stability of lipid nanoparticles: examining the effect of pH, temperature, and lyophilization. Int. J. Nanomed. 12, 305–315 (2016).
Fan, Y. et al. Physicochemical and structural insights into lyophilized mRNA-LNP from lyoprotectant and buffer screenings. J. Control. Release 373, 727–737 (2024).
Kim, B. et al. Optimization of storage conditions for lipid nanoparticle-formulated self-replicating RNA vaccines. J. Control. Release 353, 241–253 (2023).
Lamoot, A. et al. Successful batch and continuous lyophilization of mRNA LNP formulations depend on cryoprotectants and ionizable lipids. Biomater. Sci. 11, 4327–4334 (2023).
Li, M. et al. Lyophilization process optimization and molecular dynamics simulation of mRNA-LNPs for SARS-CoV-2 vaccine. npj Vaccines 8, 153 (2023).
Ai, L. et al. Lyophilized mRNA-lipid nanoparticle vaccines with long-term stability and high antigenicity against SARS-CoV-2. Cell Discov. 9, 9 (2023).
Mata, E. et al. Preserved efficacy of lyophilized SARS-CoV-2 mRNA vaccine incorporating novel ionizable lipids after one year at 25 °C. npj Vaccines 10, 135 (2025). This article reports a lyophilized mRNA–LNP vaccine incorporating ionizable lipids that preserve immunogenicity and protective efficacy after up to 1 year of storage at 25 °C, addressing cold-chain limitations.
Hashiba, K. et al. Overcoming thermostability challenges in mRNA–lipid nanoparticle systems with piperidine-based ionizable lipids. Commun. Biol. 7, 556 (2024).
Ripoll, M. et al. An imidazole modified lipid confers enhanced mRNA-LNP stability and strong immunization properties in mice and non-human primates. Biomaterials 286, 121570 (2022). This article reports an imidazole-modified ionizable lipid that enhances mRNA–LNP stability while retaining strong immunogenicity in mice and non-human primates.
Wu, J. et al. Rapidly separable bubble microneedle-patch system present superior transdermal mRNA delivery efficiency. Int. J. Pharm. 674, 125427 (2025).
Doan, T. N. K., Davis, M. M. & Croyle, M. A. Identification of film-based formulations that move mRNA lipid nanoparticles out of the freezer. Mol. Ther. Nucleic Acids 35, 102179 (2024).
Friis, K. P. et al. Spray dried lipid nanoparticle formulations enable intratracheal delivery of mRNA. J. Control. Release 363, 389–401 (2023).
Sarode, A. et al. Inhalable dry powder product (DPP) of mRNA lipid nanoparticles (LNPs) for pulmonary delivery. Drug Deliv. Transl. Res. 14, 360–372 (2024).
Roozen, G. V. T. et al. Safety and immunogenicity of intradermal fractional dose administration of the mRNA-1273 vaccine: a proof-of-concept study. Ann. Intern. Med. 175, 1771–1774 (2022).
Prins, M. L. M. et al. Immunogenicity and reactogenicity of intradermal mRNA-1273 SARS-CoV-2 vaccination: a non-inferiority, randomized-controlled trial. npj Vaccines 9, 1 (2024).
Roozen, G. V. T. et al. Intradermal delivery of the third dose of the mRNA-1273 SARS-CoV-2 vaccine: safety and immunogenicity of a fractional booster dose. Clin. Microbiol. Infect. 30, 930–936 (2024).
Darade, A. R. et al. Effect of mRNA delivery modality and formulation on cutaneous mRNA distribution and downstream eGFP expression. Pharmaceutics https://doi.org/10.3390/pharmaceutics14010151 (2022).
Hassan, J., Haigh, C., Ahmed, T., Uddin, M. J. & Das, D. B. Potential of microneedle systems for COVID-19 vaccination: current trends and challenges. Pharmaceutics 14, 1066 (2022).
Kirtane, A. R., Tang, C., Freitas, D., Bernstock, J. D. & Traverso, G. Challenges and opportunities in the development of mucosal mRNA vaccines. Curr. Opin. Immunol. 85, 102388 (2023).
Coffey, J. W., Gaiha, G. D. & Traverso, G. Oral biologic delivery: advances toward oral subunit, DNA, and mRNA vaccines and the potential for mass vaccination during pandemics. Annu. Rev. Pharmacol. Toxicol. 61, 517–540 (2021).
Eshaghi, B. et al. The role of engineered materials in mucosal vaccination strategies. Nat. Rev. Mater. 9, 29–45 (2024).
Liu, S. et al. Charge-assisted stabilization of lipid nanoparticles enables inhaled mRNA delivery for mucosal vaccination. Nat. Commun. 15, 9471 (2024).
Lokugamage, M. P. et al. Optimization of lipid nanoparticles for the delivery of nebulized therapeutic mRNA to the lungs. Nat. Biomed. Eng. 5, 1059–1068 (2021).
Zhang, H., Leal, J., Soto, M. R., Smyth, H. D. C. & Ghosh, D. Aerosolizable lipid nanoparticles for pulmonary delivery of mRNA through design of experiments. Pharmaceutics 12, 1042 (2020).
Keikha, R., Hashemi-Shahri, S. M. & Jebali, A. The evaluation of novel oral vaccines based on self-amplifying RNA lipid nanparticles (saRNA LNPs), saRNA transfected Lactobacillus plantarum LNPs, and saRNA transfected Lactobacillus plantarum to neutralize SARS-CoV-2 variants alpha and delta. Sci. Rep. 11, 21308 (2021).
Sung, J., Alghoul, Z., Long, D., Yang, C. & Merlin, D. Oral delivery of IL-22 mRNA-loaded lipid nanoparticles targeting the injured intestinal mucosa: a novel therapeutic solution to treat ulcerative colitis. Biomaterials 288, 121707 (2022).
Luo, P. K. et al. pH-responsive β-glucans-complexed mRNA in LNPs as an oral vaccine for enhancing cancer immunotherapy. Adv. Mater. 36, e2404830 (2024).
Esih, H. et al. Mucoadhesive film for oral delivery of vaccines for protection of the respiratory tract. J. Control. Release 371, 179–192 (2024).
Meany, E. L. et al. Generation of an inflammatory niche in a hydrogel depot through recruitment of key immune cells improves efficacy of mRNA vaccines. Sci. Adv. 11, eadr2631 (2025).
Neshat, S. Y. et al. Polymeric nanoparticle gel for intracellular mRNA delivery and immunological reprogramming of tumors. Biomaterials 300, 122185 (2023).
Zhu, Y. et al. An mRNA lipid nanoparticle-incorporated nanofiber-hydrogel composite for cancer immunotherapy. Nat. Commun. 16, 5707 (2025).
Sharifnia, Z. et al. In-vitro transcribed mRNA delivery using PLGA/PEI nanoparticles into human monocyte-derived dendritic cells. Iran. J. Pharm. Res. 18, 1659–1675 (2019).
Karlsson, J., Rhodes, K. R., Green, J. J. & Tzeng, S. Y. Poly(beta-amino ester)s as gene delivery vehicles: challenges and opportunities. Expert Opin. Drug Deliv. 17, 1395–1410 (2020).
Kim, H. et al. Gastrointestinal delivery of an mRNA vaccine using immunostimulatory polymeric nanoparticles. AAPS J. 25, 81 (2023).
Qin, M., Du, G. & Sun, X. Recent advances in the noninvasive delivery of mRNA. Acc. Chem. Res. 54, 4262–4271 (2021).
Bus, T., Traeger, A. & Schubert, U. S. The great escape: how cationic polyplexes overcome the endosomal barrier. J. Mater. Chem. B 6, 6904–6918 (2018).
Suberi, A. et al. Polymer nanoparticles deliver mRNA to the lung for mucosal vaccination. Sci. Transl. Med. 15, eabq0603 (2023).
Zhao, W. et al. Lipid polymer hybrid nanomaterials for mRNA delivery. Cell. Mol. Bioeng. 11, 397–406 (2018).
Yadava, S. K., Reddy, B. P. K., Prausnitz, M. R. & Cicerone, M. T. Hybrid lipid nanocapsules: a robust platform for mRNA delivery. ACS Appl. Mater. Interfaces 16, 15981–15992 (2024).
Meyer, R. A., Hussmann, G. P., Peterson, N. C., Santos, J. L. & Tuesca, A. D. A scalable and robust cationic lipid/polymer hybrid nanoparticle platform for mRNA delivery. Int. J. Pharm. 611, 121314 (2022).
Lim, C. C., Chia, L. Y. & Kumar, P. V. Dendrimer-based nanocomposites for the production of RNA delivery systems. OpenNano 13, 100173 (2023).
Meshanni, J. A. et al. Targeted delivery of TGF-β mRNA to murine lung parenchyma using one-component ionizable amphiphilic Janus Dendrimers. Nat. Commun. 16, 1806 (2025).
Chahal, J. S. et al. Dendrimer-RNA nanoparticles generate protective immunity against lethal Ebola, H1N1 influenza, and Toxoplasma gondii challenges with a single dose. Proc. Natl Acad. Sci. USA 113, E4133–E4142 (2016).
Udhayakumar, V. K. et al. Arginine-rich peptide-based mRNA nanocomplexes efficiently instigate cytotoxic T cell immunity dependent on the amphipathic organization of the peptide. Adv. Healthc. Mater. https://doi.org/10.1002/adhm.201601412 (2017).
van den Brand, D. et al. Peptide-mediated delivery of therapeutic mRNA in ovarian cancer. Eur. J. Pharm. Biopharm. 141, 180–190 (2019).
Zhong, R. et al. Hydrogels for RNA delivery. Nat. Mater. 22, 818–831 (2023).
Ou, B. S. et al. Broad and durable humoral responses following single hydrogel immunization of SARS-CoV-2 subunit vaccine. Adv. Healthc. Mater. 12, e2301495 (2023).
Blakney, A. K., Ip, S. & Geall, A. J. An update on self-amplifying mRNA vaccine development. Vaccines 9, 97 (2021).
Liu, X. et al. Circular RNA: an emerging frontier in RNA therapeutic targets, RNA therapeutics, and mRNA vaccines. J. Control. Release 348, 84–94 (2022).
Voigt, E. A. et al. A self-amplifying RNA vaccine against COVID-19 with long-term room-temperature stability. npj Vaccines 7, 136 (2022).
Pollock, K. M. et al. Safety and immunogenicity of a self-amplifying RNA vaccine against COVID-19: COVAC1, a phase I, dose-ranging trial. eClinicalMedicine 44, 101262 (2022).
Hick, T. A. H. et al. Safety concern of recombination between self-amplifying mRNA vaccines and viruses is mitigated in vivo. Mol. Ther. 32, 2519–2534 (2024). This article reports that recombination between saRNA vaccines and viruses is not observed in vivo, supporting the safety of saRNA vaccines.
Hồ, N. T. et al. Safety, immunogenicity and efficacy of the self-amplifying mRNA ARCT-154 COVID-19 vaccine: pooled phase 1, 2, 3a and 3b randomized, controlled trials. Nat. Commun. 15, 4081 (2024).
Zuo, L. et al. Self-amplifying mRNA vaccines protect elderly BALB/c mice against a lethal respiratory syncytial virus infection. Mol. Ther. 33, 499–513 (2025).
Krähling, V. et al. Self-amplifying RNA vaccine protects mice against lethal Ebola virus infection. Mol. Ther. 31, 374–386 (2023).
Chang, C. et al. Self-amplifying mRNA bicistronic influenza vaccines raise cross-reactive immune responses in mice and prevent infection in ferrets. Mol. Ther. Methods Clin. Dev. 27, 195–205 (2022).
Crosby, E. J. et al. Long-term survival of patients with stage III colon cancer treated with VRP-CEA(6D), an alphavirus vector that increases the CD8+ effector memory T cell to Treg ratio. J. ImmunoTher. Cancer https://doi.org/10.1136/jitc-2020-001662 (2020).
Li, Y. et al. In vitro evolution of enhanced RNA replicons for immunotherapy. Sci. Rep. 9, 6932 (2019).
Zhang, Y. et al. Small circular RNAs as vaccines for cancer immunotherapy. Nat. Biomed. Eng. 9, 249–267 (2025).
Huang, K. et al. Circular mRNA vaccine against SARS-COV-2 variants enabled by degradable lipid nanoparticles. ACS Appl. Mater. Interfaces 17, 4699–4710 (2025).
Liu, X. et al. A single-dose circular RNA vaccine prevents Zika virus infection without enhancing dengue severity in mice. Nat. Commun. 15, 8932 (2024).
Fukuchi, K. et al. Internal cap-initiated translation for efficient protein production from circular mRNA. Nat. Biotechnol. https://doi.org/10.1038/s41587-025-02561-8 (2026).
Chen, H. et al. Chemical and topological design of multicapped mRNA and capped circular RNA to augment translation. Nat. Biotechnol. 43, 1128–1143 (2025).
Chen, R. et al. Engineering circular RNA for enhanced protein production. Nat. Biotechnol. 41, 262–272 (2023).
Qu, L. et al. Circular RNA vaccines against SARS-CoV-2 and emerging variants. Cell 185, 1728–1744.e16 (2022). This article reports circular RNA vaccines that exhibit enhanced stability and durable antigen expression, eliciting potent humoral and cellular immunity and broad protection against SARS-CoV-2 variants in mice and non-human primates.
Gong, Z. et al. Recent advances and perspectives on the development of circular RNA cancer vaccines. npj Vaccines 10, 41 (2025).
Li, H. et al. Circular RNA cancer vaccines drive immunity in hard-to-treat malignancies. Theranostics 12, 6422–6436 (2022).
Hanna, A. R., Issadore, D. A. & Mitchell, M. J. High-throughput platforms for machine learning-guided lipid nanoparticle design. Nat. Rev. Mater. https://doi.org/10.1038/s41578-025-00831-0 (2026).
Zhang, H. et al. Algorithm for optimized mRNA design improves stability and immunogenicity. Nature 621, 396–403 (2023).
Bravi, B. Development and use of machine learning algorithms in vaccine target selection. npj Vaccines 9, 15 (2024).
Ong, E., Wong, M. U., Huffman, A. & He, Y. COVID-19 coronavirus vaccine design using reverse vaccinology and machine learning. Front. Immunol. 11, 1581 (2020).
Wayment-Steele, H. K. et al. Deep learning models for predicting RNA degradation via dual crowdsourcing. Nat. Mach. Intell. 4, 1174–1184 (2022).
Bae, S.-H. et al. Rational design of lipid nanoparticles for enhanced mRNA vaccine delivery via machine learning. Small 21, 2405618 (2025).
Chan, A. et al. Designing lipid nanoparticles using a transformer-based neural network. Nat. Nanotechnol. https://doi.org/10.1038/s41565-025-01975-4 (2025). This article reports a transformer-based deep learning framework that enables end-to-end prediction of LNP performance and stability across diverse, multi-component formulations, accelerating rational mRNA delivery design.
Wang, W. et al. Prediction of lipid nanoparticles for mRNA vaccines by the machine learning algorithm. Acta Pharm. Sin. B 12, 2950–2962 (2022).
Wang, W. et al. Artificial intelligence-driven rational design of ionizable lipids for mRNA delivery. Nat. Commun. 15, 10804 (2024).
Xu, Y. et al. AGILE platform: a deep learning powered approach to accelerate LNP development for mRNA delivery. Nat. Commun. 15, 6305 (2024).
Li, B. et al. Accelerating ionizable lipid discovery for mRNA delivery using machine learning and combinatorial chemistry. Nat. Mater. 23, 1002–1008 (2024). This article reports a machine learning-guided combinatorial chemistry approach that accelerates the discovery of ionizable lipids for mRNA delivery, identifying candidates that outperform benchmark lipids in vivo.
Bhujel, R., Enkmann, V., Burgstaller, H. & Maharjan, R. Artificial intelligence-driven strategies for targeted delivery and enhanced stability of RNA-based lipid nanoparticle cancer vaccines. Pharmaceutics 17, 992 (2025).
Kong, H. Advances in personalized cancer vaccine development: AI applications from neoantigen discovery to mRNA formulation. BioChem 5, 5 (2025).
Vitiello, A. & Zanetti, M. Neoantigen prediction and the need for validation. Nat. Biotechnol. 35, 815–817 (2017).
Maharjan, R., Kim, K. H., Lee, K., Han, H. K. & Jeong, S. H. Machine learning-driven optimization of mRNA-lipid nanoparticle vaccine quality with XGBoost/Bayesian method and ensemble model approaches. J. Pharm. Anal. 14, 100996 (2024).
Maharjan, R. et al. Comparative study of lipid nanoparticle-based mRNA vaccine bioprocess with machine learning and combinatorial artificial neural network-design of experiment approach. Int. J. Pharm. 640, 123012 (2023).
Boro, E. & Stoll, B. Barriers to COVID-19 health products in low-and middle-income countries during the COVID-19 pandemic: a rapid systematic review and evidence synthesis. Front. Public Health 10, 928065 (2022).
Santangelo, O. E., Provenzano, S., Di Martino, G. & Ferrara, P. COVID-19 vaccination and public health: addressing global, regional, and within-country inequalities. Vaccines 12, 885 (2024).
Dagovetz, M. et al. Global COVID-19 vaccination challenges: inequity of access and vaccine hesitancy. J. Med. Surg. Public. Health 6, 100197 (2025).
Batisani, K. The role of mRNA vaccines in infectious diseases: a new era of immunization. Trop. Dis. Travel Med. Vaccines 11, 12 (2025).
Uddin, M. N. & Roni, M. A. Challenges of storage and stability of mRNA-based COVID-19 vaccines. Vaccines https://doi.org/10.3390/vaccines9091033 (2021).
Scott, G. Y. et al. Enhancing vaccine quality and accessibility: strategies for efficient storage and distribution in resource-constrained Environment. Outbreak Manag. Response 1, 2443439 (2025).
Why a vaccine hub for low-income countries must succeed. Nature 607, 211–212 (2022).
Maxmen, A. The fight to manufacture COVID vaccines in lower-income countries. Nature 597, 455–457 (2021).
Prompetchara, E. et al. Immunogenicity and protective efficacy of SARS-CoV-2 mRNA vaccine encoding secreted non-stabilized spike in female mice. Nat. Commun. 14, 2309 (2023).
Puthanakit, T. et al. Phase II prefusion non-stabilised Covid-19 mRNA vaccine randomised study. Sci. Rep. 14, 2373 (2024).
Bisbas, G. mRNA technology transfer programme. Lancet Microbe 4, e578 (2023).
Kolawole, O., Ncube, C. & de Beer, J. Challenges implementing technology transfer as a viable pathway for equitable vaccine production and access: a case study of the mRNA vaccine hub in South Africa. Glob. Public Health 20, 2504698 (2025).
Maxmen, A. South African scientists copy Moderna’s COVID vaccine. Nature 602, 372–373 (2022).
Bero, L. et al. Ethical, legal, and social issues related to ingestible, implantable electronic devices and mRNA delivery devices. Device https://doi.org/10.1016/j.device.2025.100841 (2025).
Adekola, T. A. & Mercurio, B. mRNA technology transfer hub and intellectual property: towards a more equitable and sustainable model. World Trade Rev. 24, 302–320 (2025).
Lalani, H. S. et al. US public investment in development of mRNA covid-19 vaccines: retrospective cohort study. BMJ 380, e073747 (2023).
Lurie, N., Keusch, G. T. & Dzau, V. J. Urgent lessons from COVID 19: why the world needs a standing, coordinated system and sustainable financing for global research and development. Lancet 397, 1229–1236 (2021).
Solís Arce, J. S. et al. COVID-19 vaccine acceptance and hesitancy in low- and middle-income countries. Nat. Med. 27, 1385–1394 (2021).
Sommers, T. et al. Building trust and equity in vaccine communication through community engagement. Hum. Vaccines Immunother. 21, 2518636 (2025).
Coronavirus misinformation needs researchers to respond. Nature 581, 355–356, (2020).
Subbaraman, N. This COVID-vaccine designer is tackling vaccine hesitancy — in churches and on Twitter. Nature 590, 377 (2021).
Ball, P. & Maxmen, A. The epic battle against coronavirus misinformation and conspiracy theories. Nature 581, 371–374 (2020).
Donovan, J. Social-media companies must flatten the curve of misinformation. Nature https://doi.org/10.1038/d41586-020-01107-z (2020).
Tregoning, J. S., Flight, K. E., Higham, S. L., Wang, Z. & Pierce, B. F. Progress of the COVID-19 vaccine effort: viruses, vaccines and variants versus efficacy, effectiveness and escape. Nat. Rev. Immunol. 21, 626–636 (2021).
Kozlov, M. COVID vaccines have higher approval in less-affluent countries. Nature https://doi.org/10.1038/d41586-021-01987-9 (2021).
Lu, X. et al. Engineered PLGA microparticles for long-term, pulsatile release of STING agonist for cancer immunotherapy. Sci. Transl. Med. 12, eaaz6606 (2020).
McHugh, K. J. et al. Fabrication of fillable microparticles and other complex 3D microstructures. Science 357, 1138–1142 (2017). This article reports the SEAL microfabrication platform, which enables high-resolution assembly of complex 3D polymer microstructures, highlighting injectable pulsatile systems for single-injection, self-boosting vaccines that could improve vaccine adherence in LMICs.
Kanelli, M. et al. A machine learning-optimized system for pulsatile, photo- and chemotherapeutic treatment using near-infrared responsive MoS2-based microparticles in a breast cancer model. ACS Nano 18, 30433–30447 (2024).
Wang, E. Y. et al. TIMED: temporal intervention with microparticle encapsulation and delivery — a programmed release system for post-myocardial infarction therapy. Cell Biomater. https://doi.org/10.1016/j.celbio.2025.100249 (2026).
Author information
Authors and Affiliations
Contributions
Conceptualization: B.E. and A.J. Writing — original draft, figures and tables: B.E. Writing — review & editing: B.E., R.L. and A.J. All authors reviewed and approved the manuscript prior to submission.
Corresponding authors
Ethics declarations
Competing interests
From fiscal year 2021 to the present, R.L. has received licensing fees (to patents in which he was an inventor on) from, invested in, consulted (or was on Scientific Advisory Boards or Boards of Directors) for, lectured (and received a fee), or conducted sponsored research at the Massachusetts Institute of Technology for which he was not paid, for the entities listed in the Supplementary Information. From fiscal year 2021 to the present, A.J. has received licensing fees (to patents on which she was an inventor) from, invested in, consulted (or was on Scientific Advisory Boards or Boards of Directors) for, lectured (and received a fee), or conducted sponsored research at the Massachusetts Institute of Technology for which she was not paid, for the following entities: The Estée Lauder Companies, Moderna Therapeutics, OmniPulse Biosciences, Particles for Humanity, SiO2 Materials Science, VitaKey.
Peer review
Peer review information
Nature Reviews Bioengineering thanks Nicaise Ndembi, Gregg Duncan, and the other, anonymous, reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
African Medicines Agency (AMA): https://www.nepad.org/microsite/african-medicines-agency-ama
African Vaccine Regulatory Forum (AVAREF): https://avaref.afro.who.int/
Afrigen: https://www.afrigen.co.za/
BioNet-Asia: https://bionet-asia.com/partnerships/
Chulalongkorn University: https://www.chulavrc.org/
Coalition for Epidemic Preparedness Innovations (CEPI): https://cepi.net/
COVID-19 Vaccines Global Access (COVAX): https://www.who.int/initiatives/act-accelerator/covax
Decentralized and Accelerated mRNA Vaccine Production System (DeCAFx): https://rnatherapeutics-vaccines-asia.com/seminar/building-a-decentralized-accelerated-mrna-vaccine-production-system-decafx-for-pandemic-preparedness/
First mRNA vaccine factory built from BioNTainer units: https://investors.biontech.de/news-releases/news-release-details/biontech-achieves-milestone-mrna-based-vaccine-manufacturing-0
Gavi, the Vaccine Alliance: https://www.gavi.org/
International Coalition of Medicines Regulatory Authorities (ICMRA): https://icmra.info/drupal/
International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH): https://www.ich.org/
Mutual Recognition Agreements (MRAs): https://www.fda.gov/international-programs/international-arrangements/mutual-recognition-agreements-mra
Ntensify mRNA platform: https://quantoom.com/ntensify-mrna-production-solution/ntensify-midi-mrna-production-system/
Planned expansions to Senegal and South Africa: https://investors.biontech.de/news-releases/news-release-details/update-first-biontainer-african-based-mrna-manufacturing
Project Orbis: https://www.fda.gov/about-fda/oncology-center-excellence/project-orbis
Supported by a US $40 million Gates Foundation: https://www.gatesfoundation.org/ideas/media-center/press-releases/2023/10/mrna-vaccine-manufacturing-africa
WHO mRNA Technology Transfer Hub: https://www.who.int/initiatives/mrna-technology-transfer-(mrna-tt)-programme
World Health Organization’s (WHO) Collaborative Registration Procedure (CRP): https://www.who.int/teams/regulation-prequalification/regulation-and-safety/facilitated-product-introduction/collaborative-registration-procedure/crp-for-medicines-and-vaccines-approved-by-the-stringent-regulatory-authorities
World Health Organization’s (WHO) Emergency Use Listing (EUL): https://www.who.int/teams/regulation-prequalification/eul
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Eshaghi, B., Langer, R. & Jaklenec, A. Expanding global access to mRNA vaccines. Nat Rev Bioeng (2026). https://doi.org/10.1038/s44222-026-00424-8
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44222-026-00424-8