Abstract
Background
We investigated the association between younger-onset type 2 diabetes, duration of diabetes, and cancer risk based on data from the Da Qing Diabetes Prevention Outcome Study (DQDPOS).
Methods
The analysis recruited 620 younger-onset (age≤50 years) and 649 older-onset (age>50 years) patients with type 2 diabetes, and 310 younger non-diabetes controls (age≤50 years). Multiple regression analysis was used to test the influence of younger-onset diabetes and duration of diabetes on the long-term risk of cancer.
Results
The annual incidence of all cancer among the non-diabetes, younger-, and older-onset type 2 diabetes was significantly different (3.7, 5.5, and 4.0/1000 person-years, respectively). The standard Cox analysis revealed that the patients with younger-onset diabetes had a significantly higher risk of cancer than those with older-onset diabetes (hazard ratio [HR]:1.81; 95% confidence interval [CI]:1.20–2.73) and younger non-diabetic controls (HR:2.43; 95% CI:1.34–4.41) after adjustment for diabetes duration and other confounders. Stepwise general linear regression model analysis revealed that a longer diabetes-free time was associated with longer lifetime cancer-free years (partial R2 = 0.36, p < 0.001), in addition to the non-modifiable predictor duration of diabetes.
Conclusions
Younger-onset type 2 diabetes was significantly associated with an increased risk of cancer beyond the influence of diabetes duration.
Similar content being viewed by others
Introduction
Malignant tumors are major causes of death, and their association with type 2 diabetes has attracted much attention. Several studies have reported that high blood sugar levels in patients with type 2 diabetes are associated with increased risks of cancer [1,2,3,4]. In recent years, the link between type 2 diabetes and cancer risk in the young population has become a popular topic in scientific literature. One study reported that adolescents and young adults with primary invasive cancer are at an increased risk of developing type 2 diabetes [5]. Bertrand et al. recently reported that data from five cohorts (including 257,290 women aged <55 years) did not support the hypothesis that gestational diabetes mellitus is a risk factor for breast cancer in young women [5]. However, reports on the relationship between a younger age at diabetes onset and subsequent cancer risk in long-term follow-up studies are limited.
One recent Chinese study has reported on the relationship between the onset age of type 2 diabetes and subsequent 8-year risk of pancreatic cancer; however, it did not consider the influence of diabetes duration [6]. The question of whether the onset age of type 2 diabetes affect cancer risk beyond the duration of diabetes remains unanswered [7]. This question is particularly important in the Chinese population because of the modern unhealthy lifestyle of eating more and exercising less in the younger and middle-aged population had greatly contributed to the apparent expansion of younger onset type 2 diabetes population in the past 20 years. Three national diabetes surveys in China (2002 to 2020), have consistently demonstrated a steady increase in the prevalence of type 2 diabetes in people aged 40–50 years [7,8,9]. The middle-aged trend of type 2 diabetes population may potentially increase the risk of cancer, and the burden related to cancer treatment in China if younger-onset type 2 diabetes truly increases cancer risk.
Previously, we found that, in participants with impaired glucose tolerance (IGT), progression to diabetes (considered a time-dependent variable) was associated with a 121% higher risk of cancer after adjusting for covariates [10]. The present study aimed to investigate the association between young age at type 2 diabetes onset, duration of diabetes, and cancer risk in the Chinese adults based on the over 30-year long-term follow-up data from the Da Qing Diabetes Prevention Outcome Study (DQDPOS).
Materials and methods
Study design and participants
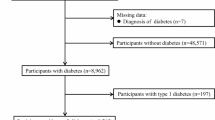
The DQDPOS is an observational prospective cohort study derived from the original Da Qing Diabetes Prevention Study (DQDPS) which enrolled 1725 adults including people with normal and impaired glucose tolerance (NGT and IGT), and new diagnosed type 2 diabetes (NDD) by oral glucose tolerance tests (OGTTs) in 1986. The DQDPOS, which has been ongoing for over 30 years, aims to investigate long-term outcomes such as death, diabetes-related macro- and microvascular complications, and cancer. The outcomes of death and diabetes-related macro- and microvascular complications have been previously reported [11, 12]. The present analysis reports the outcome of cancer in relation to the age at diabetes onset and the duration of diabetes. The original study design and population have been reported previously [13,14,15,16]. The present analysis recruited 1579 adults (accounting for 91.5% of the total participants of the DQDPOS), of whom 649 (41.1%) were diagnosed with type 2 diabetes after 50 years of age (Mean 51.1, standard deviation [SD] 8.7), 620 (39.3%) were diagnosed with type 2 diabetes before 50 years of age (Mean 41.4, SD 6.0), and 310 (19.6%) were diagnosed without diabetes before or equal to 50 years of age (Mean 40.0, SD 6.9) and maintaining non-diabetes over 30 follow-up study (Supplementary Fig. 1).
Of the patients with type 2 diabetes, half had prevalent diabetes, and the other half had incident diabetes. The remaining 146 people (in the original NGT and IGT groups) who remained non-diabetic during the entire follow-up period and with a baseline age more than 50 years were excluded because they were not suitable for inclusion as non-diabetic controls (Supplementary Fig. 1).
This study was approved by the ethics committee of Fuwai Hospital (approve number: 2020-1390). All participants, or representatives of deceased participants, provided written informed consent.
Identifying patients with type 2 diabetes
A diabetes survey was conducted from 1985 to 1986 in Da Qing City, Heilongjiang Province, Northeast China. A total of 110,660 adults were screened for type 2 diabetes and prediabetes. A total of 630 and 576 participants were identified as having NDD and IGT, respectively, by the standard 75-g OGTT, according to the World Health Organization diabetes diagnosis (1985) criteria [17]. Furthermore, 519 individuals with NGT and age- and sex-matched IGT were used as controls. Finally, 1,269 patients with type 2 diabetes (re-classified according to the American Diabetes Association [1999] diabetes criteria [18]) were included in the present analysis, including 659 patients with prevalent type 2 diabetes and 610 with incident type 2 diabetes. Of these, 78% (993/1269) were diagnosed by OGTT during systemic screening and interview, and 22% (281/1269) by physicians in clinical practice based on: elevated fasting or postprandial glucose values in medical records; diabetes-related symptoms (such as thirst and polyuria); and/or taking glucose-lowering medications, including oral hypoglycemic agents and insulin [19]. Once type 2 diabetes was diagnosed, patients were advised to regularly take medications to control hyperglycemia, concomitant hypertension, and dyslipidemia according to Chinese guidelines.
Identifying patients with incident cancer
Cancer outcome data were collected from all original study participants. For those who were still alive, data on cancer diagnosis were collected after the completion of a 6-year intervention trial and during follow-up interviews. For deceased participants, we asked proxy informants about the date of cancer diagnosis and the hospital where the cancer was diagnosed. For all participants, medical records including the pathological diagnosis of cancer, together with information from the informant interviews, were reviewed and adjudicated independently by two doctors to establish the underlying cause of cancer. Disagreements were resolved by a third senior physician. Cancer- and site-specific cancer diagnoses were based on International Classification of Diseases codes 10th (ICD10) Revision, Clinical Modification. Among the 258 cases of cancer, 194 were confirmed by medical records, 23 by cancer hospitals in Beijing, Shanghai, and other big cities in China, and 32 by a tertiary A hospital in Daqing City, where only nine cases were self-reported physician diagnosed (Supplementary Table S1). Of the 1579 participants, 25 (1.6%) lost follow-up.
Statistical analysis
Participants with type 2 diabetes were stratified into two subgroups according to the age at onset of diabetes: the younger diabetes group with a diabetes onset age of 50 years or less (n = 620) and an older diabetes group with a diabetes onset age of more than 50 years (n = 649). The 310 people aged 50 years or less at baseline and remaining non-diabetic over the whole follow-up period were taken as the younger non-diabetes control group.
Baseline characteristics are presented as means ± SD for normally distributed continuous variables and median (IQR) for non-normally distributed continuous variables, and as numbers and percentages for categorical variables. We used 1986 as the baseline year for this analysis because it was the year of enrollment of the DQDPS when incident type 2 diabetes cases were identified. The correlation between diabetes-free time (equal to diabetes onset age) and age at cancer diagnosis was analyzed using a multivariate general linear regression model (GLM) after adjusting for confounders such as age, sex, body mass index (BMI), smoking status, blood pressure, plasma glucose, and diabetes duration. Diabetes duration was defined (in years) as the difference between the date of diabetes diagnosis and the date of death, last follow-up, cancer diagnosis, or end of follow-up (December 2020), whichever occurred first. Cancer-free time was calculated from the birth year to the date of cancer diagnosis, death, last follow-up, or the end of follow-up (December 2020), whichever occurred first. Multivariate fractional polynomial (MFP) analysis [20] was used to test for potential nonlinear relationships between age of onset of type 2 diabetes or diabetes duration and cancer risk. Cox proportional hazards regression analysis was used to estimate hazard ratios (HRs) and 95% confidence intervals (CIs) for cancers related to diabetes onset age after adjusting for confounders. Cox regression analysis competing for non-cancer death was used to control for the influence of premature non-cancerous deaths on cancer development to further confirm whether the age of diabetes onset was associated with an increased cause-specific hazard rate for cancer [21]. The different influences of important factors, such as diabetes duration, premature death, and smoking status, on the association between the age of onset of diabetes and cancer risk were tested. No evidence of violation of the proportional hazard assumption was found (tested using the likelihood ratio test to compare models with and without product terms between exposure and log-transformed follow-up times). Medications for treating hyperglycemia, hypertension, and dyslipidemia were also included as adjusted variables. The correlation between diabetes- and cancer-free time was analyzed using a GLM combined with stepwise analysis to evaluate the importance of diabetes onset age and diabetes duration on the long-term or lifetime risk of cancer [22]. All analyses were performed using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA) and Stata SE software (version 16.0; Stata Corp.). All statistical tests were two-sided, with a p-value of less than 0.05 indicating statistical significance.
Results
Participant characteristics
In participants with type 2 diabetes, the average age at diabetes diagnosis (diabetes-onset age) was 52.4 ± 11.5 years and diabetes duration were 18.3 ± 10.7 years. The average age for cancer-free years was 70.3 ± 9.9 years in all participants. Across the two groups with a diabetes-onset age of 50 years or less and more than 50 years, the diabetes-onset age was 42.4 ± 5.4 and 61.0 ± 8.4 years, and diabetes duration was 27 (18–23) and 13 (6–20) 8 years, respectively. Compared with the younger group, the older group had a higher prevalence of hypertension, males, and smokers (p < 0.05) and lower plasma glucose levels (p < 0.01); however, both had a similar BMI (p > 0.05). There were 93 (13.6%) and 113 (9.3%) incident cancer cases in the younger- and older-onset groups, respectively.
All variables at baseline were significantly lower in the younger non-diabetic control group than in the younger diabetic group, except for the frequency of males and smokers.
During the follow-up period, 69.1%, 42.5%, and 35.8% of participants in the younger- onset diabetes, older-onset diabetes, and younger non-diabetic control groups received regular medications for hypertension and hyperlipidemia or hyperglycemia, respectively. During the follow-up period, 258 cases of cancer occurred, of which 52, 93, and 113 were in the non-diabetes, younger-, and older-onset diabetes groups, respectively (Table 1).
Association between diabetes-onset age and subsequent long-term cancer risk and the influence of diabetes duration and related factors
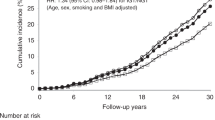
The 1000 person-years incidence of cancer among the non-diabetes, younger-, and older-onset diabetes groups was 3.7 (95% CI: 2.7–5.1), 5.5 (95% CI: 4.3–6.9) and 4.0 (95% CI: 3.1–5.2), respectively (Table 1). The annual incidence of cancer in the younger- and older-onset diabetes subgroups stratified by diabetes duration were showed in Supplementary Table S2 and Fig. S2. Patients with a diabetes duration of less than or equal to 20 years had a cancer incidence rate per 1000 person years of 9.11 (95% CI: 5.32–15.02) and 3.72 (95% CI: 2.88–5.30) in the younger- and older-onset diabetes groups after the adjustment of age, sex, smoking and non-cancer death (p < 0.0001), respectively, whereas in those with a diabetes duration of more than 20 years, this incidence was 5.67 (95% CI: 4.38–7.24) and 3.66 (95% CI: 2.15–6.05), respectively, (p = 0.043). The results clearly showed a greater increased long-term risk of cancer in patients with younger-onset diabetes than that in older-onset regardless of diabetes duration. In addition, compared with individuals with diabetes for more than 20 years, diabetes duration had a greater influence on the association between age of diabetes onset and cancer risk among individuals with diabetes for less than 20 years.
The results of fractional polynomial regression analysis showed that the age of onset of diabetes (as a continuous variable) was linearly negatively associated with subsequent cancer risk over 30 years (p < 0.001, Fig. 1a) and the duration of diabetes was negatively associated with the risk of cancer for those who had diabetes for more than ten years. The cancer risk was slightly increased before ten years of diabetes duration but declined thereafter (Fig. 1b).
a Diabetes onset age and (b) Diabetes duration.
In Cox regression model, the β estimate of the association between diabetes onset age and the risk of cancer was −0.0345 after adjustment for age at baseline and sex. After adding the adjustment of smoking status, BMI, systolic blood pressure (SBP), and 2-h plasma glucose after 75 g of glucose load (PG2h), the β-estimate was −0.0226. However, further adjusting for non-cancer death, the β-estimate moved to −0.0351. Finaly, put diabetes duration into the previous adjustment, the β-estimate drastically increased to −0.3567 (p = 0.029, Supplementary Table S3).
Comparison of long-term cancer risk among the younger-onset diabetes, older-onset diabetes, and younger non-diabetes control groups
The cancer risk among the younger-onset diabetes, older-onset diabetes, and younger non-diabetes control groups were compared. In the standard Cox model, compared with the age of onset of diabetes more than 50 years, participants with a diabetes onset age of 50 years or less had an increased risk of cancer (HR: 1.81; 95%CI: 1.20–2.73) after the full adjustment for baseline age, sex, BMI, SBP, PG2h, smoking status, follow-up period medications (for treatment of hyperglycemia, hypertension, and hyperlipidemia), and diabetes duration (Table 2 and Fig. 2). The risk of cancer in patients with younger-onset diabetes was also higher than that in those who were younger and non-diabetic (HR: 2.43; 95%CI: 1.34–4.41) after the same adjustment. Moreover, baseline age (HR: 1.06; 95% CI: 1.04–1.08), PG2h (mmol/l, HR: 1.09; 95% CI: 1.05–1.12), and smoking status (HR: 1.33; 95% CI 1.004–1.76) were also significantly associated with cancer risk. The Cox analysis competing for the influence of non-cancer death showed a similar result: the younger-onset diabetes group had a significant higher risk of cancer than the older-onset (HR: 1.82; 95% CI: 1.20–2.74) and non-diabetes groups (HR: 2.43; 95% CI: 1.34–4.41) after the same adjustment (Table 2). Regarding the influence of medications on cancer risk, the use of antihypertensives and lipid-lowering drugs did not increase the risk of cancer (HR: 0.53; 95% CI: 0.39–0.73 and HR: 0.41; 95% CI: 0.29–0.58, p < 0.0001, respectively). Stepwise Cox model analysis in participants with type 2 diabetes alone showed that diabetes onset age first entered the model with R2 = 0.28 (p < 0.001) over a 30-year follow-up period, and with R2 = 0.36 (p < 0.001) in those who with type 2 diabetes died or lived to 80 years and over (Table 3).
DM diabetes mellitus, BMI body mass index, SBP systolic blood pressure.
Correlation between diabetes- and cancer-free time
The increase in diabetes-free time (equivalent to age of onset of diabetes in the analysis) from 42.4 ± 5.4 years to 61.1 ± 8.4 years in the groups with diabetes-onset ages of 50 years or less and more than 50 years (Table 1) was accompanied by a 7.8-year increase in cancer-free time across the two groups (from 66.4 ± 9.8 to 74.2 ± 8.1 years). The general linear model assessment revealed that the diabetes-free time was significantly positively correlated with the cancer-free time (β-estimate=0.99, p < 0.0001) after adjusting for age, sex, smoking status, BMI, hypertension, PG2h, medications, and diabetes duration (Supplementary Table S4). The stepwise regression analysis revealed that in the entire diabetes group, diabetes-free time entered the model (partial R2 = 0.28) in the first step, followed by diabetes duration and smoking status (a well-known risk factor for cancer), with model R2 = 0.96 (Table 3).
A separate stepwise analysis among patients with diabetes who died or lived to 80 years and over during the study period revealed that the cancer-free lifespan was significantly associated with diabetes-free time and diabetes duration (p < 0.0001). The diabetes-free time first entered the model with a partial R2 = 0.36 (p < 0.0001), followed by the duration of diabetes (R2 = 0.60, p < 0.0001). Age at baseline was entered into the model in the third step with a very small partial R2 = 0.002 (p = 0.039). Interestingly, other factors such as sex, smoking status, SBP, PG2h, and medications for the treatment of hyperglycemia, hypertension, and dyslipidemia were not included in the model (p = 0.10, Table 3).
Discussion
This analysis, based on data from the DQDOS, which screened 110,660 adults (approximately 40% of the residents in Da Qing city), enrolled the NDD, IGT and matched NGT groups in 1986 and followed up to 2020, revealed a significant association between the age of onset of type 2 diabetes and long-term cancer risk. The age of onset of type 2 diabetes was significantly and negatively associated with the long-term cancer risk and adults with a diabetes-onset age of 50 years or less have a significantly higher risk of cancer than those with diabetes-onset age more than 50 years over 30-year follow-up after adjusted for potential confounders including diabetes duration. Furthermore, the diabetes onset age (diabetes-free time) was found to be positively significantly associated with the cancer-free lifetime.
A retrospective matched cohort study in Chinese population had reported that individuals who develop type 2 diabetes at a young age are at a higher relative risk of any cancer than their age and sex matched non-diabetic counterparts. Interestingly, the biggest HR was found between diabetes and non-diabetes controls in the age below 50 years subgroup, then the HRs gradually declined with the increase of age of the diabetes and control pares [23]. In consistent, our study also revealed that the cancer risk in the younger onset type 2 diabetes compared with the age-sex matched NGT controls but also demonstrated the younger onset diabetes had a significant higher risk of all cancer compared with older onset diabetes after adjustment of confounders including diabetes duration. These findings indicate type 2 diabetes developed before the age of 50 years are potentially at a higher risk of cancer not only than the normal glucose controls but also significantly higher than those developed diabetes later, which highlight that preventing type 2 diabetes before the age of 50 years is a major challenge for cancer prevention, and prevent development of type 2 diabetes forever or delaying the onset of type 2 diabetes in the younger population may favor long-term cancer prevention. A nationwide cohort study in Sweden (1964–2015) also found that a diabetes diagnosis at less than 50 years of age was associated with a 1.9-fold increased risk of colorectal cancer (CRC) compared to that at more than 50 years [24]. The study further demonstrated that patients with diabetes had a similar lifetime risk of CRC before the age of 50 years to those with only a family history of CRC. This emphasizes the importance of early-onset diabetes on the risk of developing cancer prematurely. These findings indicate that in both Chinese and Western populations, those with younger-onset type 2 diabetes have a higher risk of cancer than those with older-onset.
To date, there are insufficient data to determine whether the role of blood sugar in triggering cancer is stronger in young people than in older people. Our findings suggest that hyperglycemia-related factors may contribute to an increased cancer risk in young people with type 2 diabetes. First, their lifetime diabetes-free years were shorter; hence, they were exposed to hyperinsulinemia and hyperglycemia earlier and for longer periods than those in the older-onset diabetes group. Second, poor β-cell function, a key defect in diabetes, appears to develop earlier and plays an important role in cancer development [25]. Together, these factors demonstrate that except for earlier hyperinsulinemia in the prediabetes stage, long-term hyperglycemia in the younger-onset diabetes population is highly likely to promote mitosis. Diabetes-level glucose can promote the degradation of the cancer inhibitory protein p53 through a set of complex and sophisticated processes, which in turn, enhances the uptake of glucose and glycolysis by cancer cells and promotes cancer progression [26]. Sattar et al. discussed the potential mechanisms for the adverse effects of year loss of type 2 diabetes diagnosis at younger ages and addressed the role of concomitant smoking and higher BMI, in addition to higher glycemic levels, in their study population [27]. In our study, except for higher glycemic levels over time, the younger group was not more obese and had fewer smokers than the older group. These findings support the potentially important influence of better glucose control after the diagnosis of diabetes on subsequent long-term cancer risk [10].
Another notable finding of this study was the strong impact of diabetes duration on the association between the age at diabetes onset and long-term risk of cancer. The multivariate analyses revealed that the diabetes-onset age related cancer risk was substantially higher after adjusting for the duration of diabetes. This indicates that diabetes duration drastically enhanced the influence of type 2 diabetes onset age on cancer risk. Meanwhile, in patients with a diabetes duration of 20 years or less, we found that the incidence of cancer in the younger-onset and older-onset diabetes groups were 9.1 and 3.7 per 1000 person years, respectively, while in those with a diabetes duration of more than 20 years, the incidence was 5.7 and 3.7 per 1000 person years, respectively. This shows that the difference in cancer incidence between patients with younger- and older-onset diabetes varied with the duration of diabetes and that the influence of type 2 diabetes onset age on cancer risk was much stronger in patients with diabetes duration less than 20 years. This suggests that there may be a population difference in the association between age at type 2 diabetes onset and long-term cancer risk; details of this difference may need to be determined in future large cohort studies.
Furthermore, the age at type 2 diabetes onset was positively and significantly correlated with cancer-free lifetime after controlling for traditional confounders and duration of diabetes. This suggests that patients with older-onset diabetes have a longer cancer-free lifetime than those with younger-onset diabetes. Moreover, the stepwise analysis revealed that diabetes-onset age entered the model before diabetes duration and other predictors of cancer. These findings suggesting a potential influence of delaying diabetes onset age on prolong cancer-free years. The findings in patients with type 2 diabetes who died during follow-up or lived up to 80 years of age suggest that diabetes-free time may be recognized as a potential contributor to a longer cancer-free life expectancy. The younger-onset diabetes group showed a consistent and significantly higher cancer risk than the older-onset diabetes group, even after full adjustment for covariates.
A few long follow-up studies have focused on the association between age at diabetes onset and cancer risk, and those accounting for the influence of diabetes duration are even rarer. Although it is believed that the duration of diabetes is closely associated with the age at diabetes onset clinically, finding evidence of a constant positive association between the duration of diabetes and the long-term risk of cancer was not easy. The Nurses’ Health Study (1978–2014) and the Health Professionals Follow-up Study (1988–2014) reported that diabetes duration was significantly related to cancer risks, while the cancer risk was reached its highest level 4–8 years after type 2 diabetes diagnosis and then gradually decreased afterward [28]. The 34-year follow-up of the DQDOS provides data to answer this question as it has detailed data on both broad-range diabetes onset age and decades-long diabetes duration. Figure 1b showed that the cancer risk slightly increased before ten years of diabetes duration but declined thereafter. The finding supports the results of the previous two reports. The plausible mechanism is that the diabetes duration in a real long-term study may be largely affected by premature cardiovascular death related to the early onset of type 2 diabetes. Those people with a higher rate of premature death would miss enough time to develop cancer and have a relatively short duration of diabetes, then those who survived had a longer diabetes duration and maybe not have a higher risk of cancer. Interestingly, in our study, the younger onset diabetes group still showed a higher long-tern risk of cancer compared with those patients with older onset diabetes even after accounting for the competing for non-cancer death and diabetes duration, and other traditional confounders. These results demonstrate the potential stable influence of a younger diabetes onset age on the long-term increased risk of cancer.
Strengths and limitations
This study has several strengths. First, this was a prospective cohort study with an extended follow-up period of more than 30 years (24.4 years on average), with a total 31,047 person-years contributed. Second, all participants had detailed data on their age at type 2 diabetes diagnosis and/or cancer, and 78% of the type 2 diabetes cases were diagnosed using the OGTT. Third, the participants had a long lifespan, approximately 70% (876/1274) of them had lived for at least 80 years (data not shown); thus, reasonable diabetes- and cancer-free time data were available. Finally, the association between age at diabetes onset and increased cancer risk after diabetes persisted after accounting for the effect of diabetes duration and the competing risk of non-cancer death. This demonstrates the independent impact of younger-onset diabetes on cancer risk beyond the longer duration of diabetes and premature non-cancer death in the younger population with type 2 diabetes.
However, this study also has some limitations. First, there were no records of a family history of cancer; hence, we could not discuss its influence on cancer risk. Second, due to the limited number of cancer cases, we could not investigate the relationship between site-specific cancer risk and diabetes onset. Finally, we had inadequate data regarding medication prescriptions, including dose changes and treatment duration. Therefore, we were unable to discuss the benefits of these medications in detail.
Conclusions
In conclusion, younger onset type 2 diabetes is significantly associated with an increased long-term risk of cancer in Chinese population despite the strong and complex influence of diabetes duration on cancer risk. This finding revealed that patients with younger-onset type 2 diabetes had a long-term higher risk of developing cancer than those with older-onset. Therefore, early detection and effective prevention of type 2 diabetes in young adults and timely cancer screening in patients with younger-onset diabetes may be warranted. Future studies with more detailed patient characteristics may be warranted to adjust for more confounding factors.
Data availability
Data cannot be shared publicly because of ethical reasons pertaining to patient confidentiality. De-identified participant data are available for researchers who meet the requirements for access to confidential patient data and after approval from the Study Steering Committee of the Da Qing Diabetes Study. Data requestors will be required to sign a data access agreement before data are released. Request for access to data can be made to the Study Steering Committee of the Da Qing Diabetes Study, Fuwai Hospital (dqds_china@fuwaihospital. org).
References
Rey-Renones C, Baena-Diez JM, Aguilar-Palacio I, Miquel C, Grau M. Type 2 diabetes mellitus and cancer: epidemiology, physiopathology and prevention. Biomedicines. 2021;9:1429.
Renehan AG, Yeh HC, Johnson JA, Wild SH, Gale EA, Moller H, et al. Diabetes and cancer (2): evaluating the impact of diabetes on mortality in patients with cancer. Diabetologia. 2012;55:1619–32.
Johnson JA, Carstensen B, Witte D, Bowker SL, Lipscombe L, Renehan AG, et al. Diabetes and cancer (1): evaluating the temporal relationship between type 2 diabetes and cancer incidence. Diabetologia. 2012;55:1607–18.
Wu BU, Butler RK, Lustigova E, Lawrence JM, Chen W. Association of glycated hemoglobin levels with risk of pancreatic cancer. JAMA Netw Open. 2020;3:e204945.
Holowatyj AN, Viskochil R, Ose D, Tingey B, Haaland B, Wilson D, et al. Diabetes, body fatness, and insulin prescription among adolescents and young adults with cancer. J Adolesc Young Adult Oncol. 2021;10:217–25.
Li Y, Tian J, Hou T, Gu K, Yan Q, Sun S, et al. Association between age at diabetes diagnosis and subsequent incidence of cancer: a longitudinal population-based cohort. Diabetes Care. 2024;47:353–61.
Zhang Y, Song M, Cao Y, Eliassen AH, Wolpin BM, Stampfer MJ, et al. Incident early- and later-onset type 2 diabetes and risk of early- and later-onset cancer: prospective cohort study. Diabetes Care. 2023;46:120–9.
Wang L, Peng W, Zhao Z, Zhang M, Shi Z, Song Z, et al. Prevalence and treatment of diabetes in China, 2013-2018. JAMA. 2021;326:2498–506.
Li Y, Teng D, Shi X, Qin G, Qin Y, Quan H, et al. Prevalence of diabetes recorded in mainland China using 2018 diagnostic criteria from the American Diabetes Association: national cross sectional study. BMJ. 2020;369:m997.
He S, Qian X, Chen Y, Shen X, Zhang B, Chen X, et al. Risk of death and heart failure among patients with type 2 diabetes treated by metformin and nonmetformin monotherapy: a real-world study. J Diabetes Res. 2021;2021:1–10.
Tancredi M, Rosengren A, Svensson AM, Kosiborod M, Pivodic A, Gudbjornsdottir S, et al. Excess mortality among persons with type 2 diabetes. N Engl J Med. 2015;373:1720–32.
Duckworth W, Abraira C, Moritz T, Reda D, Emanuele N, Reaven PD, et al. Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med. 2009;360:129–39.
Pan XR, Li GW, Hu YH, Wang JX, Yang WY, An ZX, et al. Effects of diet and exercise in preventing NIDDM in people with impaired glucose tolerance. The Da Qing IGT and Diabetes Study. Diabetes Care. 1997;20:537–44.
Li G, Zhang P, Wang J, Gregg EW, Yang W, Gong Q, et al. The long-term effect of lifestyle interventions to prevent diabetes in the China Da Qing Diabetes Prevention Study: a 20-year follow-up study. Lancet. 2008;371:1783–9.
Li G, Zhang P, Wang J, An Y, Gong Q, Gregg EW, et al. Cardiovascular mortality, all-cause mortality, and diabetes incidence after lifestyle intervention for people with impaired glucose tolerance in the Da Qing Diabetes Prevention Study: a 23-year follow-up study. Lancet Diabetes Endocrinol. 2014;2:474–80.
Gong Q, Zhang P, Wang J, Ma J, An Y, Chen Y, et al. Morbidity and mortality after lifestyle intervention for people with impaired glucose tolerance: 30-year results of the Da Qing Diabetes Prevention Outcome Study. Lancet Diabetes Endocrinol. 2019;7:452–61.
Diabetes mellitus. Report of a WHO study group. World Health Organ Tech Rep Ser. 1985;727:1–113.
Mellitus TEC. Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care. 1997;20:1183–97.
Ali Khan U, Fallah M, Sundquist K, Sundquist J, Brenner H, Kharazmi E. Risk of colorectal cancer in patients with diabetes mellitus: A Swedish nationwide cohort study. PLoS Med. 2020;17:e1003431.
Austin PC, Fang J, Lee DS. Using fractional polynomials and restricted cubic splines to model non-proportional hazards or time-varying covariate effects in the Cox regression model. Stat Med. 2022;41:612–24.
Lau B, Cole SR, Gange SJ. Competing risk regression models for epidemiologic data. Am J Epidemiol. 2009;170:244–56.
Lu X, Liu Z, Cui Q, Liu F, Li J, Niu X, et al. A polygenic risk score improves risk stratification of coronary artery disease: a large-scale prospective Chinese cohort study. Eur Heart J. 2022;43:1702–11.
Yang Z, Wu Y, Xu L, Zhu Z, Li T, Yu L, et al. Age at diagnosis modifies associations of type 2 diabetes with cancer incidence and mortality: a retrospective matched-cohort study. Diabetologia. 2023;66:1450–9.
Ali Khan U, Fallah M, Tian Y, Sundquist K, Sundquist J, Brenner H, et al. Personal history of diabetes as important as family history of colorectal cancer for risk of colorectal cancer: a nationwide cohort study. Am J Gastroenterol. 2020;115:1103–9.
Bao J, Liu D, Sun J, Su X, Cheng H, Qi L, et al. Pancreatic cancer-associated diabetes mellitus is characterized by reduced beta-cell secretory capacity, rather than insulin resistance. Diabetes Res Clin Pract. 2022;185:109223.
Su Y, Luo Y, Zhang P, Lin H, Pu W, Zhang H, et al. Glucose-induced CRL4COP1-p53 axis amplifies glycometabolism to drive tumorigenesis. Molecular Cell. 2023;83:2316–e2317.
Sattar N, Rawshani A, Franzen S, Rawshani A, Svensson AM, Rosengren A, et al. Age at diagnosis of type 2 diabetes mellitus and associations with cardiovascular and mortality risks. Circulation. 2019;139:2228–37.
Hu Y, Zhang X, Ma Y, Yuan C, Wang M, Wu K, et al. Incident type 2 diabetes duration and cancer risk: a prospective study in two US cohorts. J Natl Cancer Inst. 2021;113:381–9.
Acknowledgements
We thank all the study participants and their relatives. We thank Yanjun Liu (306 PLA Hospital, Beijing, China), and Yuqing Zhu (China-Japan Friendship Hospital, Beijing, China) for their help with data collection. Our special thanks go to the late Professor Xiaoren Pan (China-Japan Friendship Hospital, Beijing, China).
Funding
This work was supported from 1986 to 1992 by the World Bank, the Ministry of Public Health of the People’s Republic of China (to Guangwei Li), and Da Qing First Hospital (to Jinping Wang); from 2004 to 2009 by the US Centers for Disease Control and Prevention (CDC)-WHO Cooperative Agreement (to Guangwei Li, grant number: U58/CCU424123-01-02), China-Japan Friendship Hospital (to Guangwei Li), and Da Qing First Hospital (to Jinping Wang); from 2015 to 2018 by the US CDC–Chinese Center for Disease Control and Prevention Cooperative Agreement (to Guangwei Li, grant number: 5U19GH000636-05), National Center for Cardiovascular Diseases & Fuwai Hospital (to Guangwei Li), China-Japan Friendship Hospital (to Guangwei Li), and Da Qing First Hospital (to Jinping Wang); and in 2020 by Chinese Academy of Medical Sciences, Innovation Fund for Medical Sciences (to Yanyan Chen, grant number: 2020-I2M-2-006). The sponsors had no role in any aspect of the study design, the collection, analysis, and interpretation of data, or in the development of the manuscript.
Author information
Authors and Affiliations
Contributions
GL, SH, XQ, and JW conceived and designed the study. SH, JW, XS, BZ, XC, HL, and BC contributed to data acquisition. GL, XQ, YA, QG, YW, and GJ analyzed the data. SH and XQ contributed to the draft and revision of the paper. GL contributed to the study design, statistical analysis, and draft and revision of the paper. GL is the guarantor of this work and, as such, has full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
He, S., Qian, X., Wang, J. et al. Younger-onset type 2 diabetes associated with increased long-term cancer risk in Chinese adults: A 30-year follow-up of the Da Qing Diabetes Study. BJC Rep 3, 24 (2025). https://doi.org/10.1038/s44276-025-00142-5
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44276-025-00142-5