Abstract
In advanced non-small cell lung cancer (NSCLC), programmed death-ligand 1 (PD-L1) expression is a well-established but suboptimal biomarker for predicting response to immune checkpoint inhibitors (ICIs). Tumor-infiltrating lymphocytes (TILs), particularly CD8+ subsets, have demonstrated potential as complementary biomarker. Despite existing data on each biomarker individually, the combined effect is not fully understood. A systematic search of Ovid/Medline, Embase, and Web of Science identified studies on CD8+ TILs and PD-L1 in NSCLC patients treated with ICIs. The primary outcomes were progression-free survival (PFS) and overall survival (OS). Secondary endpoints included objective response rate (ORR) and durable clinical benefit (DCB). Study quality was assessed using the Newcastle-Ottawa Scale. Thirteen studies (2490 patients) were included. PD-L1 expression was associated with longer PFS in 6 of 8 studies (HR: 0.67, 95% CI: 0.49–0.90) but did not significantly correlate with OS. TILs alone showed no significant predictive value for PFS or OS. However, combining both biomarkers provided the strongest predictive value for PFS (HR: 0.39, 95% CI: 0.27–0.57) and OS (HR: 0.42, 95% CI: 0.31–0.56). Combining PD-L1 and TILs may more effectively predict PFS and OS than either biomarker alone, though their clinical application remains complex.
Similar content being viewed by others
Introduction
Lung cancer remains the leading cause of cancer-related mortality worldwide, with non-small cell lung cancer (NSCLC) accounting for ~85% of cases [1]. In patients with unresectable stage III/IV NSCLC, immune checkpoint inhibitors (ICIs) are a key treatment option, used as monotherapy or in combination with chemotherapy [2].
While ICIs have improved survival, therapeutic responses are heterogeneous. Programmed Cell Death Ligand 1 (PD-L1) expression is currently the standard of care biomarker for predicting tumor responses to ICIs, yet its limitations are evident: up to 40% of patients with high PD-L1 expression may not respond, while around 10% of PD-L1-negative patients may still experience benefit. This variability has prompted exploration of additional biomarkers, such as tumor-infiltrating lymphocytes (TILs), particularly CD8 + T cells, which reflect the immunogenicity of the tumor microenvironment (TME) and have shown promise in predicting immunotherapy efficacy [3, 4].
Depsite their potential, TILs have not been widely adopted in routine clinical practice. As nearly all registrational studies have used tumor PD-L1 expression to define study populations, regulatory authorities such as the FDA and the EMA have approved ICI regimen based on PD-L1 expression levels, limiting the integration of TILs into guidelines [5, 6].
Combining PD-L1 expression and TILs density as biomarkers may improve predictive accuracy and support better treatment decisions by capturing complementary pathways of immune evasion. Therefore, the primary aim of this study was to systematically review the predictive and prognostic role of combining PD-L1 and TILs in advanced-stage NSCLC, to better understand their joint potential in identifying patients who are most likely to benefit from immunotherapy.
Methods
This review was conducted and reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA)-statement (www.prisma-statement.org) and is registered in the International Registry of Prospective Systematic Reviews (PROSPERO, CRD42023434509) [7, 8].
Search strategy
Three bibliographic databases (Ovid/Medline, Embase, and Web of Science Core Collection) were searched for relevant literature. The search strategy, developed with a medical information specialist, included free-text terms like “tumor infiltrating lymphocyte” and “non-small cell lung carcinoma,” without restrictions on study design, date, or language. Additional sources, including Google Scholar, conference proceedings, and references from relevant studies, were also reviewed. Duplicates were removed using automated and manual methods in Endnote. An overview of the search strings can be found in the Supplementary.
Study selection
Two reviewers (YD and SH) independently screened titles and abstracts for eligibility using Rayyan [9]. Full-text articles were reviewed if necessary, and disagreements were resolved by consensus. Studies were included if these: (1) involved NSCLC patients, (2) assessed CD8+ TILs and PD-L1 expression in clinical trials or cohort studies evaluating ICI or combination therapy, (3) reported outcomes such as overall survival (OS), progression-free survival (PFS), durable clinical benefit (DCB), objective response rate (ORR), and (4) included 20 or more patients. Exclusion criteria were: (1) patients with recurrent tumors at sites other than the lung, as well as those with lung tumors other than NSCLC, (2) non-original studies, (3) inability to extract relevant data, and (4) studies that could not be evaluated for their quality.
Quality assessment
The full text of the selected articles was retrieved for further review. Two reviewers (YD and SH) independently evaluated the methodological quality of the full text papers using the Newcastle-Ottawa Scale (NOS) recommended by the Cochrane Collaboration [10]. Research quality was evaluated independently by two researchers (YD, SH), and disagreements were settled through discussions with the third researcher (IB).
Endpoints
The primary endpoints were PFS and OS, with the hypothesis that higher levels of PD-L1 or TILs may predict better responses to ICIs, resulting in enhanced PFS and OS. Secondary endpoints included objective response rate (ORR) and durable clinical response (DCB). Outcomes were reported as hazard ratios (HR) or odds ratios (OR) and 95% confidence intervals (CI), with forest plots used to illustrate the separate and combined effects of PD-L1 and TILs on survival. Data extracted from each study included study design, tumor details (type of tumor, stage), and biomarker information (PD-L1 expression levels, CD8 + TIL concentrations, and assessment methods). Treatment regimens, including ICIs or combination therapies were collected. Forest plots were generated using R (version 4.3.2; R Core Team, 2023) and displayed the biomarker cut-off values from each study, along with their heterogeneity and overall effect [11].
Results
Search and selection
The literature search generated a total of 2384 references: 609 in Ovid/Medline, 894 in Embase, and 881 in Clarivate Analytics/ Web of Science Core Collection. After removing duplicates, 1264 references remained. The flow chart of the search and selection process is presented in Fig. 1. After screening titles and abstracts, 1128 studies were excluded. The full text of the remaining 136 articles were reviewed, and a total of 11 articles (1770 patients) were included. On the 24th of September 2024, the search was reconducted, ultimately resulting in a total of 13 articles (2420 patients). Summary of each individual study can be found in the supplementary data.
The flowchart of the search and selection process is presented based on the PRISMA-guidelines [18]. Comments: (*) identified through databases Ovid/Medline, Embase and Web of Science Core Collection.
Study characteristics and key outcomes are shown in Table 1. For each included study quality assurance was performed according to NOS, as shown in Supplementary Table 1. None of the studies fell in the category of high risk of bias, 6 received a low risk of bias score, and 7 studies (54%) were of moderate risk of bias.
Biomarker accuracy
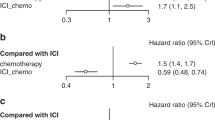
Figure 2 presents an overview of the HR for PFS and OS for PD-L1 and TILs CD8+ lymphocytes, both individually and combined.
A Forest plots for PFS based on PD-L1 expression (left) and CD8+ TILs density (right). B Forest plots for OS based on PD-L1 expression and CD8+ TILs density. C Forest plots for the combination of PD-L1 and CD8+ TILs for PFS and OS. For all panels, the x-axis represents the log-transformed hazard ratio (HR), and the y-axis lists the individual studies. Horizontal lines represent the 95% confidence interval (CI). Study-specific biomarker cut-offs are shown. PD-L1 programmed death-ligand 1, TILs tumor-infiltrating lymphocytes, HR hazard ratio, CI confidence interval.
For PFS, 6 of 8 evaluable studies showed that PD-L1 expression effectively discriminated for longer PFS. Interestingly, all these studies used a PD-L1 cutoff of 50%. The pooled HR for PD-L1-positive patients treated with ICIs was 0.67 (95% CI: 0.49–0.90). In terms of TILs, 5 out of 8 studies demonstrated that TILs could also differentiate for longer PFS. However, overall, there was no significant PFS benefit for patients with TILs expression treated with ICIs compared to those without TILs expression (HR: 0.81, 95% CI: 0.54–1.20). In contrast, when PD-L1 and TILs were combined, all 7 of 7 studies were able to predict longer PFS, with a p-value of <0.01.
Regarding OS, neither PD-L1 nor TILs alone had a statistically significant impact on survival (PD-L1: HR: 0.81, 95% CI: 0.64–1.01; TILs: HR: 0.77, 95% CI: 0.54–1.09). However, the combination of PD-L1 and TILs was associated with a significant improvement in OS, with a pooled HR of 0.42 (95% CI: 0.31–0.56) and the test for overall effect showing significance at p < 0.05.
Discussion
General results
Here we present the results of a systematic review exploring the relationship between two key pathological biomarkers, i.e., PD-L1 and TILs, in predicting the efficacy of immunotherapy for NSCLC. Despite the important role of these biomarkers as predictors of immunotherapy response, the systematic search identified only 13 studies involving 2420 patients.
Preliminary analysis suggests that dual assessment of PD-L1 and TILs may improve the prediction of ICI efficacy and survival outcomes. While PD-L1 expression was more effective in identifying patients with favorable PFS, it demonstrated limited utility for distinguishing those with a favorable OS. TILs alone did not significantly differentiate for either PFS or OS. However, combining both biomarkers provided the strongest predictive value for PFS (HR: 0.39, 95% CI: 0.27–0.57) and OS (HR: 0.42, 95% CI: 0.31–0.56).
The biological rationale for this combined approach in improving predictive accuracy is well-supported. PD-L1 expression on both tumor and immune-suppressive cells can inhibit the function of effector T-cells, not only within the tumor area but also in the surrounding tumor stroma. This concept is central to the cancer immunity cycle model proposed by Chen and Mellman in 2013 and updated in 2023. The model emphasizes the critical role of T-cell activation and their migration through the stroma towards tumoral areas, illustrating how PD-L1 expression on both immune cells in the stroma and tumor cells in the tumorous area can disrupt this critical immune response cycle (see Fig. 3) [12, 13].
[13] Reviewed studies indicate a higher concentration of TILs in the stromal compartment compared to the tumoral compartment. PD-L1 expression on both cancer cells and immune cells in the tumoral compartment, as well as on immune cells in the stromal compartment, may lead to the inhibition of TILs and immunotherapy resistance. TIL tumor infiltrating lymphocyte.
Heterogeneity in measuring and defining PD-L1 and TILs
The main challenge in comparing studies in this field stems from the lack of standardized assessment methods for PD-L1 and TILs, along with significant variability in cutoff values across studies. An added complexity of PD-L1 expression is that its presence is most commonly on tumor cells, but can also appear on immune cell subsets. This distinction is important, as some studies focus solely on PD-L1 expression on tumor cells, while others include both tumor and immune cells. Hu-Lieskovan et al. demonstrated that PD-L1 on immune cells did not substantially improve the predictive accuracy of PD-L1 for ICI efficacy [14]. However, this study did not specifically explore patients with PD-L1-negative tumor cells, a particularly interesting subgroup. In a previous study by our group, patients who were PD-L1-negative on tumor cells but positive on immune cells responded to ICIs similarly to those with PD-L1-positive tumor cells [15].
Similar to PD-L1, the lack of a standardized assessment of TILs, complicates cross-study comparisons and makes correlating TILs with PD-L1 challenging. TILs include a variety of T and B cells, with CD8 + T cells being particularly important due to their cytotoxic potential. Many studies focus solely on CD8 + T cells as a defining subset of TILs, especially in pragmatic single-stain assessments [16]. Further variability arises in studies differentiating between TILs within tumor areas versus those in stromal areas. Larger studies by Hashemi et al., Rakaee et al., and Rodas et al., have shown that TILs concentrations are generally higher in stromal regions than in tumor regions [17,18,19]. These studies further suggest that stromal TILs concentration is a more reliable predictor of ICI efficacy than tumoral TIL concentration.
Building on the predictive significance of PD-L1 and TILs, Teng et al. and others proposed a four-category classification system for the tumor microenvironment, based on high/low levels of PD-L1 expression and TILs density [20,21,22]. Several studies in this review reference this classification, with the PD-L1-positive/TIL-high subgroup emerging as the most predictive for ICI efficacy.
Patients with actionable genomic alterations (AGAs)
The TME in non-oncogenic driven lung cancers differs substantially from that of tumors with actionable genomic alerations (AGAs). Clinical evidence demonstrates that patients with AGAs typically respond poorly to ICIs, even if the tumors exhibits some PD-L1 expression [23]. Only a minority of studies explicitly documented their inclusion of AGA-positive patients, and those that did reported widely varying proportions (e.g., 26% in Hu-Lieskovan et al. versus 13% in Shirasawa et al.) [14, 24]. This variability raises important questions about the potential confounding effect on study outcomes. However, this reduced response does not seem to diminish the overall predictive value of PD-L1 and TILs. For example, Shimoda’s study, which specifically examined EGFR-positive patients, found that a combination of high PD-L1 expression and a high concentration of tumoral CD8+ TILs was a strong predictor of PFS and ORR in ICI treatments [25].
Different stages and treatment regimens
This review primarily analyzed studies involving patients with advanced stage NSCLC, most of whom received ICI monotherapy as first- or second-line treatment. However, some studies also included patients treated with concurrent chemotherapy with ICIs, introducing potential confounding effect on the predictive value of both biomarkers. Sun et al. demonstrated that in patients receiving chemoimmunotherapy, both PD-L1 expression and TIL levels (measured via mRNA quantification) remained predictive of treatment response [26]. This correlation was also observed in locally advanced stage disease, where patients receiving durvalumab consolidation therapy showed improved outcomes when pre-chemoradiotherapy biopsies indicated high TIL density or elevated PD-L1 expression [27].
Patient characteristics
The predictive value of PD-L1 and TILs must be interpreted within the broader context of patient-specific factors, including overall health status and immune system status. Hashemi et al.‘s comprehensive multivariate analysis evaluated these biomarkers alongside established clinical parameters (age, sex, performance status) and laboratory values (LDH, albumin). Stromal TIL density emerged as the strongest independent predictor for both PFS and OS, outperforming conventional markers. For PFS, albumin levels provided secondary predictive value, while for OS, both albumin and LDH offered additional prognostic information. These findings highlight that while multiple clinical factors influence outcomes, stromal TIL assessment provides superior predictive capability [18].
Limitations and future perspectives
Several limitations should be acknowledged. First, the small number of included studies with the inherent risk of publication bias limits the overall impact of the findings. Additionally, the lack of uniform definitions for TILs and PD-L1, combined with considerable variations in assessment methods, complicate interpretation and potentially introduce bias. Immunohistochemistry (IHC) was the most commonly used technique; here, variations in antibody clones, platforms, cutoffs, and interobserver variability challenge comparability. Due to the variability in study designs and biomarker evaluation protocols, quantitative synthesis was not feasible. Results are displayed visually through forest plots to illustrate between-study differences.
Future research should focus on standardization of assessment methods. IHC remains a practical approach due to its accessibility and feasibility in routine clinical settings, yet achieving greater interobserver consistency remains challenging. Automated methods, particularly machine learning algorithms, represent a promising avenue for reducing observer-related variability. Rakaee et al.‘s findings suggest that these algorithms can produce results that align closely with traditional assessments while enhancing consistency [19]. Nonetheless, it’s important to recognize that these algorithms are still trained on pathologist-assessed data, introducing its own set of limitations. As such, careful validation and optimization of machine learning models on large, diverse datasets will be necessary to maximize their utility in routine biomarker evaluation.
Furthermore, advanced techniques like imaging mass cytometry (IMC) offer unique advantages in understanding the spatial organization of the TME, as demonstrated by Lopez Rodas et al [17]. By allowing multi-staining of a single formalin-fixed paraffin-embedded slide with a range of stains, varying from 7 to 40, IMC can differentiate complex cellular communities and map complex interactions between cells within spatial distinct tumor areas. Although currently limited by high costs and labor-demands, IMC has a significant potential to become a common exploration tool as data processing capabilities improve and platform costs decrease in the future.
In addition to these morphological techniques, functional analyses such as RNA and DNA sequencing add a valuable layer of insight as seen in the studies by Fumet and Sun [26, 28]. RNA sequencing allows for a more objective assessment of PD-L1 expression and the specific concentration of TIL subsets; while DNA sequencing, such as whole exome sequencing, offers further immune context, such as capturing the diversity of T-cell receptors, though it is limited by sample selection and platform sensitivity.
Conclusion
Combining PD-L1 expression and TILs density demonstrates superior predictive accuracy for immunotherapy outcomes in NSCLC than either biomarker alone. However, the major challenge in comparing studies in this area is the lack of standardized assessments for PD-L1 and TILs, along with variability in cutoff values.
Addressing these limitations through standardized immunohistochemistry measurements, coupled with advanced technologies like machine learning, IMC, and DNA/RNA sequencing, could pave the way for more refining personalized therapeutic decision-making in NSCLC immunotherapy.
Key messages
What is already known on this topic:
-
PD-L1 expression is an established but imperfect biomarker for predicting immunotherapy response in NSCLC. Tumor-infiltrating lymphocytes (TILs), particularly CD8 + T cells, show promise but are not yet standard in clinical practice. The combined predictive value of PD-L1 and TILs remains underexplored.
What this study adds:
-
This systematic review demonstrates that combining PD-L1 and TILs offers superior predictive value for both progression-free survival (PFS) and overall survival (OS) in NSCLC patients treated with ICIs compared to using either biomarker alone.
How this study might affect research, practice, or policy:
-
These findings support the integration of combined PD-L1 and TILs assessments into clinical workflows for better patient stratification in NSCLC immunotherapy. Standardization of biomarker assessment and the development of advanced analytic tools are needed to enhance the reproducibility and clinical adoption of these methods.
Data availability
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study. All data from this systematic review is available in the studies that were included, as can be found in the manuscript. The search strings are provided in the supplementary files.
References
Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA Cancer J Clin. 2024;74:12–49. https://doi.org/10.3322/caac.21820
Postmus PE, Kerr KM, Oudkerk M, Senan S, Waller DA, Vansteenkiste J, et al. Early and locally advanced non-small-cell lung cancer (NSCLC): ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2017;28:iv1–iv21. https://doi.org/10.1093/annonc/mdx222
Zeng DQ, Yu YF, Ou QY, Li XY, Zhong RZ, Xie CM, et al. Prognostic and predictive value of tumor-infiltrating lymphocytes for clinical therapeutic research in patients with non-small cell lung cancer. Oncotarget. 2016;7:13765–81. https://doi.org/10.18632/oncotarget.7282
Tian C, Lu S, Fan Q, Zhang W, Jiao S, Zhao X, et al. Prognostic Significance of Tumor-infiltrating CD8+ or CD3+ T Lymphocytes and Interleukin-2 Expression in Radically Resected Non-small Cell Lung Cancer. Chin Med J (Engl). 2015;128:105–10. https://doi.org/10.4103/0366-6999.147828
Díaz-Carrasco MS, González-Haba E, García-Soler JI, Espuny-Miró A. Predictive biomarkers of response to immune checkpoint inhibitors. Farm Hosp. 2020;44:141–8. https://doi.org/10.7399/fh.11328
Houda I, Dickhoff C, Uyl-de Groot CA, Reguart N, Provencio M, Levy A, et al. New systemic treatment paradigms in resectable non-small cell lung cancer and variations in patient access across Europe. Lancet Reg Health Eur. 2024;38:100840 https://doi.org/10.1016/j.lanepe.2024.100840
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med. 2009;6:e1000100. https://doi.org/10.1371/journal.pmed.1000100
National Institute for Health and Care Excellence. PROSPERO: International prospective register of systematic reviews [Internet]. Available from: https://www.crd.york.ac.uk/prospero/. Accessed 21 May 2024.
Ouzzani M, Hammady H, Fedorowicz Z, Elmagarmid A. Rayyan-a web and mobile app for systematic reviews. Syst Rev. 2016;5:210. https://doi.org/10.1186/s13643-016-0384-4
Ottawa Hospital Research Institute. The Oxford Centre for Evidence-Based Medicine [Internet]. Available from: https://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. Accessed 21 May 2024.
Posit team. RStudio: Integrated Development Environment for R. Posit Software, PBC, Boston, MA. (2025). http://www.posit.co/.
Chen DS, Mellman I. Oncology meets immunology: the cancer-immunity cycle. Immunity. 2013;39:1–10. https://doi.org/10.1016/j.immuni.2013.07.012
Mellman I, Chen DS, Powles T, Turley SJ. The cancer-immunity cycle: Indication, genotype, and immunotype. Immunity. 2023;56:2188–205. https://doi.org/10.1016/j.immuni.2023.09.011
Hu-Lieskovan S, Lisberg A, Zaretsky JM, Grogan TR, Rizvi H, Wells DK, et al. Tumor characteristics associated with benefit from pembrolizumab in advanced non-small cell lung cancer. Clin Cancer Res. 2019;25:5061–8. https://doi.org/10.1158/1078-0432.CCR-18-4275
Ulas EB, Hashemi SMS, Houda I, Kaynak A, Veltman JD, Fransen MF, et al. Predictive value of combined positive score and tumor proportion score for immunotherapy response in advanced NSCLC. JTO Clin Res Rep. 2023;4:100532. https://doi.org/10.1016/j.jtocrr.2023.100532
Steele KE, Tan TH, Korn R, Dacosta K, Brown C, Kuziora M, et al. Measuring multiple parameters of CD8+ tumor-infiltrating lymphocytes in human cancers by image analysis. J Immunother Cancer. 2018;6:20. https://doi.org/10.1186/s40425-018-0326-x
Lopez de Rodas M, Nagineni V, Ravi A, Datar IJ, Mino-Kenudson M, Corredor G, et al. Role of tumor infiltrating lymphocytes and spatial immune heterogeneity in sensitivity to PD-1 axis blockers in non-small cell lung cancer. J Immunother Cancer. 2022;10:e004440. https://doi.org/10.1136/jitc-2021-004440
Hashemi S, Fransen MF, Niemeijer A, Ben Taleb N, Houda I, Veltman J, et al. Surprising impact of stromal TIL’s on immunotherapy efficacy in a real-world lung cancer study. Lung Cancer. 2021;153:81–89. https://doi.org/10.1016/j.lungcan.2021.01.013
Rakaee M, Adib E, Ricciuti B, Sholl LM, Shi W, Alessi JV, et al. Association of machine learning-based assessment of tumor-infiltrating lymphocytes on standard histologic images with outcomes of immunotherapy in patients with NSCLC. JAMA Oncol. 2023;9:51–60. https://doi.org/10.1001/jamaoncol.2022.4933
Teng MW, Ngiow SF, Ribas A, Smyth MJ. Classifying cancers based on T-cell Infiltration and PD-L1. Cancer Res. 2015;75:2139–45. https://doi.org/10.1158/0008-5472.CAN-15-0255
Kim TK, Vandsemb EN, Herbst RS, Chen L. Adaptive immune resistance at the tumour site: mechanisms and therapeutic opportunities. Nat Rev Drug Discov. 2022;21:529–40. https://doi.org/10.1038/s41573-022-00493-5
Zhang Y, Chen L. Classification of advanced human cancers based on tumor immunity in the MicroEnvironment (TIME) for cancer immunotherapy. JAMA Oncol. 2016;2:1403–4. https://doi.org/10.1001/jamaoncol.2016.2450
Vokes NI, Pan K, Le X. Efficacy of immunotherapy in oncogene-driven non-small-cell lung cancer. Ther Adv Med Oncol. 2023;15:17588359231161409. https://doi.org/10.1177/17588359231161409
Shirasawa M, Yoshida T, Shimoda Y, Takayanagi D, Shiraishi K, Kubo T, et al. Differential immune-related microenvironment determines programmed cell death protein-1/programmed death-ligand 1 blockade efficacy in patients with advanced NSCLC. J Thorac Oncol. 2021;16:2078–90. https://doi.org/10.1016/j.jtho.2021.07.027
Shimoda Y, Shibaki R, Yoshida T, Murakami S, Shirasawa M, Torasawa M, et al. Concurrent high PD-L1 expression and CD8+ immune cell infiltration predict PD-1 blockade efficacy in advanced EGFR-mutant NSCLC patients. Clin Lung Cancer. 2022;23:477–86. https://doi.org/10.1016/j.cllc.2022.04.001
Sun D, Liu J, Zhou H, Shi M, Sun J, Zhao S, et al. Classification of tumor immune microenvironment according to programmed death-ligand 1 expression and immune infiltration predicts response to immunotherapy plus chemotherapy in advanced patients with NSCLC. J Thorac Oncol. 2023;18:869–81. https://doi.org/10.1016/j.jtho.2023.03.012
Haratani K, Nakamura A, Mamesaya N, Mitsuoka S, Yoneshima Y, Saito R, et al. Tumor microenvironment landscape of NSCLC reveals resistance mechanisms for programmed death-ligand 1 blockade after chemoradiotherapy: a multicenter prospective biomarker study (WJOG11518L:SUBMARINE). J Thorac Oncol. 2023;18:1334–50. https://doi.org/10.1016/j.jtho.2023.06.012
Fumet JD, Richard C, Ledys F, Klopfenstein Q, Joubert P, Routy B, et al. Prognostic and predictive role of CD8 and PD-L1 determination in lung tumor tissue of patients under anti-PD-1 therapy. Br J Cancer. 2018;119:950–60. https://doi.org/10.1038/s41416-018-0220-9
Niemeijer AN, Sahba S, Smit EF, Lissenberg-Witte BI, de Langen AJ, Thunnissen E. Association of tumour and stroma PD-1, PD-L1, CD3, CD4 and CD8 expression with DCB and OS to nivolumab treatment in NSCLC patients pre-treated with chemotherapy. Br J Cancer. 2020;123:392–402. https://doi.org/10.1038/s41416-020-0888-5
Shirasawa M, Yoshida T, Imabayashi T, Okuma K, Matsumoto Y, Masuda K, et al. Baseline PD-L1 expression and tumour-infiltrated lymphocyte status predict the efficacy of durvalumab consolidation therapy after chemoradiotherapy in unresectable locally advanced patients with non-small-cell lung cancer. Eur J Cancer. 2022;162:1–10. https://doi.org/10.1016/j.ejca.2021.11.013
Nojima Y, Shimizu K, Saisho S, Maeda AI, Kurosaki T, Kurose K, et al. Tumor PD-L1 and VEGF expression, and CD8 T cell infiltration predict clinical response to immune checkpoint inhibitors in non-small cell lung cancer. Anticancer Res. 2021;41:5469–75. https://doi.org/10.21873/anticanres.15359
Ghiringhelli F, Bibeau F, Greillier L, Fumet JD, Ilie A, Monville F, et al. Immunoscore immune checkpoint using spatial quantitative analysis of CD8 and PD-L1 markers is predictive of the efficacy of anti-PD1/PD-L1 immunotherapy in non-small cell lung cancer. EBioMedicine. 2023;92:104633. https://doi.org/10.1016/j.ebiom.2023.104633
Acknowledgements
The authors declare no acknowledgements.
Author information
Authors and Affiliations
Contributions
YD: Conceptualization, Methodology, Formal analysis, Investigation, Writing—Original Draft, Review & Editing, Visualization, Project administration. SMH: Supervision, Conceptualization, Methodology, Formal analysis, Investigation, Writing—Original Draft, Review & Editing, Visualization. EBU: Conceptualization, Methodology, Formal analysis, Investigation, Writing—Original Draft, Review & Editing, Visualization. KAZ: Software, Validation. BL-W; Validation, Visualization, TR: Writing—Reviewing and Editing, IB: Supervision, Writing—Original Draft, Review & Editing, Visualization, Project administration. IB: Supervision, Writing—Original Draft, Review & Editing, Visualization, Project administration.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Derraze, Y., Hashemi, S.M.S., Ulas, E.B. et al. Evaluating the significance of combining PD-L1 and TILs as biomarkers in non-small cell lung cancer patients treated with immunotherapy: a systematic review. BJC Rep 3, 65 (2025). https://doi.org/10.1038/s44276-025-00174-x
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44276-025-00174-x