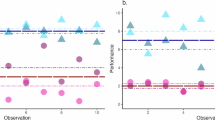
Abstract
Cognitive performance is classically measured through measures of central tendency. However, intraindividual cognitive variability (IIV) also holds important information about cognitive functioning. Smartphone-based ecological momentary cognitive testing (EMCT) can capture IIV across days. This study examined predictors of IIV, including demographics, affect, and mean performance, in EMCTs completed on the NeuroUX platform among two US-based samples: adults and older adults with high rates of mild cognitive impairment. The adult sample (n = 375) completed eight EMCTs assessing memory, processing speed, executive functioning, and working memory; each test was self-administered five times over ten days. The older adult sample (n = 94) completed three EMCTs assessing memory, processing speed/executive function, and working memory at three difficulty levels; each test at each difficulty level was self-administered five times over thirty days. Mean performance demonstrated the strongest association with IIV across groups. In the adult group, better mean performance was associated with less variability on tests of memory, executive functioning, and two out of three tests of processing speed. On tests of working memory, better mean performance was associated with greater variability, possibly due to the difficulty of these measures at higher performance levels. In the older adult sample, better mean performance was associated with two of three versions of the memory and working memory EMCTs and all difficulty levels of the processing speed/executive function task. Better average performance was associated with greater consistency across most measures. Broader and more diverse data collection methods like EMCT can provide valuable insights into cognitive functioning beyond traditional mean-based measures.
Lay summary
This study used smartphone-based mobile cognitive tests to explore how consistently people perform across days, which is called intraindividual variability. Participants included two groups: adults and older adults, many with mild cognitive impairment. Results showed that individuals who performed better on average, in both groups, tended to have more consistent scores across days. These findings suggest that mobile cognitive testing may be a tool to provide insight into cognitive functioning, such as cognitive variability.
Similar content being viewed by others
Introduction
Traditional paper and pencil neuropsychological testing has long been the gold standard for cognitive assessment. Neuropsychological tests were developed to detect cognitive impairment following illness or injury, rather than subtle changes in cognition such as in the preclinical phases of a disease. Additionally, standard neuropsychological testing occurs in a controlled environment with administration by trained technicians, which does not fully mimic the cognitive demands of everyday life. Thus, there is need for neuropsychologists to develop and validate cognitive assessments that better reflect real-world cognitive abilities and are sensitive to more subtle, within-person changes [1].
One way to assess everyday cognitive functioning is with mobile sampling methods adapted from ecological momentary assessment (EMA). Ecological momentary cognitive testing (EMCT) borrows from EMA methods to include brief objective cognitive assessments that occur in daily life [2]. EMCT allows multiple “snapshots” of cognitive performance that can be measured over the course of days to understand the dynamics of everyday cognition and how cognition fluctuates with other life factors (e.g., stress, activities) across time [3,4,5].
One of the strengths of EMCT is that both intraindividual mean and variability in cognitive performance can be indexed by multi-day, burst assessment data. Intraindividual variability (IIV) is defined as within-person fluctuations in behavioral performance. From a cognitive perspective, IIV can be measured either as variability across multiple test scores within the same evaluation (dispersion), or as variability in performance of the same task across multiple time points (inconsistency). Both fluctuations in accuracy and reaction time can be used for measuring IIV. Data from traditional neuropsychological evaluations show that IIV across different neuropsychological tests within a single testing session is increased in psychiatric (e.g., severe mental illness [6, 7]), neurological (e.g., traumatic brain injury [8], multiple sclerosis [9], neurodegenerative diseases [10,11,12]), and medical conditions (e.g., HIV [13]; breast cancer survivors [14]). For example, in the context of Alzheimer’s disease (AD), greater within session but across test IIV predicts future decline and may be a more sensitive marker of decline than mean performance [11]. IIV is also sensitive to the mild changes that occur in cognitive aging, a phenomenon that has been established for many years [15, 16]. There are several ways to measure IIV such as between test variation as described above, and inter-trial reaction time variability on measures of processing speed. IIV measured by inter-trial reaction time variability shows a U-shaped association with age such that variability is highest in childhood, reaches a nadir in early adulthood, and increases again in mid to late life [15, 17, 18]. Additionally, IIV is elevated in persons with high risk of AD [10, 11], suggesting that IIV may be an important and distinct marker of cognition.
Traditional neuropsychological measures of IIV have weaknesses. Testing sessions typically last hours and are not always accessible, which makes measuring IIV across tests (dispersion) time intensive. Moreover, traditional neuropsychological testing often compares variability across different measures within a testing session that often lack a shared normative sample, potentially reducing precison. For example, many of the most most recent and popular tests in neuropsychological measures [19] (e.g., Wechsler Adult Intelligence Scale-IV/5, California Verbal Learning Test II/III, Halstead Reitan Battery) are not cross-normed with one another. Lastly, IIV from traditional neuropsychological measures derived across tests given within a few hours on the same day lacks the potential for examining day-to-day variations. EMCT-derived IIV can be calculated across multiple days and times, making it potentially more sensitive to conditions with known temporal variations, such as circadian disruption in AD [20, 21] and diurnal variation in depression [22, 23]. Despite its appeal, research on EMCT-derived IIV is limited, with only a handful of published studies to date, primarily in the context of cognitive aging and AD [24,25,26]. Preliminary research demonstrates that EMCT-derived IIV has potential as a marker of cognitive vulnerability as, for example, it can distinguish mild cognitive impairment (MCI) from normal cognitive functioning [25].
Building on this emerging evidence and addressing current gaps in the literature, the present study aimed to further characterize the correlates of EMCT-derived IIV and evaluate it across diverse adult populations. Therefore, this study examined how demographic factors, affect, and mean performance correlates with EMCT-derived IIV collected via the NeuroUX platform over several days. These correlates were first examined in a large adult sample aged 20–79 years, participating remotely. To examine if these correlates extend beyond a general adult sample, we replicated these analyses in a multi-site study of older adults with high MCI rates, aiming to examine a broader range of cognitive performance. A secondary goal in this replication sample was to assess the extent to which EMCT IIV was associated with traditional neuropsychological testing performance.
Patients and methods
The two samples included in this study were drawn from independent research protocols with distinct aims and study designs. We leveraged available data from each to examine the replication and generalizability of findings across samples. While there is some protocol overlap, protocol differences reflect study-specific decisions tailored to the target populations and research questions. Data preprocessing procedures (e.g., exclusion criteria for EMCT data, as detailed below) were as similiar as possible to support comparability.
Adult sample participants and procedures
The methods of this study have been previously published in Paolillo et al. [27]. Three hundred and ninety-four participants were recruited through Prolific, an online research platform. Participants provided informed consent for this study through this platform. Verified and eligible participants enrolled in the study. Inclusion criteria included being between the ages of 20 and 79 years old, being born and residing in the United States, and speaking English as their first language. There were no other specific exclusion criteria. Participants’ data were fully anonymized during the data collection process; researchers cannot access identifiable information through the Prolific platform. This study received exempt status approval from the UC San Diego Institutional Review Board (IRB). No participants indicated that they had received a diagnosis of MCI or dementia. Demographic characteristics from this sample can be found in Table 1.
Over 10 days, participants completed eight NeuroUX EMCTs five times (i.e., four different EMCTs once per day) and a brief EMA survey on their personal smartphones. These procedures took between 8–10 min each day and could be completed between 7 am and 8 pm. Alternate forms were used for the EMCTs in each session [27].
Adult sample EMCT and EMA measures
The eight EMCTs included in the general adult sample are described in detail in Paolillo et al. [27] and included Memory List (recognition memory), Memory Matrix (visual working memory), Matching Pair (speed of information processing), Quick Tap 1 (speed of information processing), Quick Tap 2 (response inhibition), Odd One Out (visual working memory), CopyKat (visual working memory), and Hand Swype (cognitive flexibility). Raw scores for Quick Tap 1 and 2, Odd One Out, and Hand Swype represent the median reaction time of the trial whereas all other raw scores generally represent total correct. Raw scores from the EMCT measures were transformed to T-score distributions (not demographically corrected) using the means and standard deviations published for this sample in Paolillo et al. [27], with greater T-scores indicating better performance for all measures. Transforming raw scores into a T-distribution allows for standardization across tasks and places scores on a common metric to facilitate comparison across EMCTs.
EMA
Participants responded to EMA questions related to affect, substance use, and contextual factors (e.g., location). To assess negative affect, participants rated their current level of sadness/depression from 1 = not at all to 7 = extremely at each session, with averages calculated to explore the association between mean negative affect and IIV. Participants also reported alcohol or cannabis use earlier in the day.
Exclusions of EMCT trials
After reviewing the data, one participant performed at the floor on all EMCT tasks and was excluded from analysis. Prior to calculating the mean or IIV, outliers on specific trials that were thought to be reflective of invalid performance were removed as in Paolillo et al. [27]. If participants reported any alcohol or cannabis use in the past two hours or four or more drinks at any time that day, then EMCT trials were considered invalid for that day. After calculating IIV, if IIV was greater >3 interquartile ranges above the sample’s median, these were considered IIV outliers and thought to reflect variability due to non-cognitive factors (e.g., variable effort). Eighteen participants did not have four valid administrations of any test and were therefore excluded. In total, 375 participants were included in this analysis.
Older adult sample participants and procedures
To examine if correlates found in the general adult sample replicate and extend to a more clinically-based population, we also examined an older adult sample with and without cognitive impairment. The methods of this study focused on older adults who were either cognitively normal or had MCI (any subtype) have been published previously [28]. Data from 94 participants were collected from The University of Texas at Dallas, UC San Diego, and the University of Miami Miller School of Medicine. Inclusion criteria included being aged 50 and over, English-proficiency, and a familiar other who could serve as a study informant. Exclusion criteria included self-reported history of medical or neurological disorders that may affect brain function, dementia (i.e., clinical diagnosis or falling below the established cutoff scores on the MoCA [29]), self-reported head injury with loss of consciousness ≥15 min, vision or hearing impairment that would interfere with their ability to complete the study, intellectual disability (i.e., WRAT-4 Reading subtest standard score < 70), current substance use disorder, and history of a psychotic or bipolar disorder. Participant characteristics can be found in Table 1.
Procedures were approved by the IRBs of each respective university, and all participants provided written informed consent. Participants completed a baseline assessment to gather sociodemographic information and neuropsychological testing. As the study intended, about half of participants met criteria for any subtype of MCI using Jak/Bondi critera [30]. To mirror the general adult study, we chose to examine cognition longitudinally rather than by MCI status in order to examine if findings replicate in a broader range of cognitive abilities. Due to the evolving nature of COVID-19 restrictions, the baseline assessment was administered in person (32% of participants) and remotely (68% of participants). Additional details on COVID-19 adaptations can be found in Moore et al. [28].
Eligible participants then participated in EMCTs and an EMA survey via the NeuroUX platform for 30 consecutive days. Participants received training on the EMCT protocol and completed a practice EMA survey and mobile cognitive testing session. Participants had the option to use their own personal smartphone or could borrow a study-owned Android smartphone (n = 6). Over the next 30 days, participants were sent text message notifications to take EMA surveys three times per day. Every other day, the surveys were paired with EMCTs (i.e., three different EMCTs every other day).
Older adult EMCT, EMA, and traditional neuropsychological measures
Details on these tests and the feasibility and validity of these tests in this sample are described in detail in Moore et al. [28]. The three different EMCTs in this study were Memory List (recognition memory; easy condition: 6-words; medium condition: 12-words and identical to the version in the normative sample; hard version: 18-words), Memory Matrix (visual working memory; easy version: 6 tiles; medium version: 12 tiles; hard version: 18 tiles), and Color Trick based on the classic Stroop paradigm (easy version: “Meaning-to-Meaning” to match the same words regardless of ink color testing speed of information processing; medium version: “Meaning-to-Color” to match the ink color to the word measuring inhibition; hard version: “Yes-No Mechanic” determining if the ink color matches a word measuring inhibition). For Memory List, scores were indicative of percent correct (i.e., 0–100%), and for Memory Matrix and Color Trick, scores ranged from 0–27, with greater scores indicating better performance. The EMCTs were counterbalanced by test type and difficulty level, resulting in a total of 5 easy, medium, and hard conditions of each of the three mobile cognitive tests. T-scores are not yet available for most of these EMCTs, so raw scores were utilized for these analyses.
EMA
EMA items to assess average negative affect and alcohol and cannabis use were the same as in the adult sample described above.
Exclusions of EMCT trials
Exclusions due to alcohol or cannabis use followed the same methodology as described for the adult sample. Following the same methodology of the adult sample, if an IIV calculation was >3 interquartile ranges above the median, then this IIV estimate was thought to reflect invalid performance and was removed from analysis. All 94 participants included in the study had at least one estimate of IIV.
Traditional neuropsychological measures (lab or remote administered at baseline)
Participants in the older adult sample completed a neuropsychological exam at baseline. For the purposes of this analysis, we chose to examine a subset of tests measuring general cognition (Montreal Cognitive Assessment-BLIND version 7.1 (MoCA-BLIND); general cognition and screener for the presence of dementia), premorbid IQ (WRAT-4 Word-Reading subtest), and four additional measures most analogous to the EMCT measures as in the initial Moore 2022 validation paper [28]: Hopkins Verbal Learning Test – Revised (HVLT-R) immediate recall, delayed recall, and recognition (verbal learning and memory); Brief Visuospatial Memory Test – Revised (BVMT-R) immediate recall, delayed recall, and recognition (visual learning and memory); WAIS-IV Letter-Number Span Test (working memory); and all subtests from the D-KEFS-Color Word Interference Test (processing speed, inhibition, and inhibition/switching). Raw scores were used to examine the correlations between IIV and traditional neuropsychological tests except for the WRAT-4, in which we used the standard score, which adjusts for age.
IIV
For both samples, IIV was calculated three ways: root mean square of successive differences (rMSSD) across five valid administrations, which accounts for gradual shifts in mean (e.g., due to practice effects); standard deviation (SD) from five valid administrations; and SD from the first four valid administrations. These were compared to determine which IIV estimate should be used, given that there is no “gold standard” approach to calculating IIV on EMCT. Coefficient of variation (CoV) was not considered. The CoV calculation includes the overall mean in the denominator. This study specifically examines associations with overall performance and variables (e.g., age, education) often associated with average performance. The CoV calculation has also been shown to amplify the IIV at low levels of mean [31].
Statistical analyses
Multivariable linear regressions were conducted to examine associations between IIV (the outcome) and demographics including age, sex, race/ethnicity (dichotomized to non-Hispanic white and all others; restricted numbers of minoritized individuals did not allow for additional subgroup analyses), and years of education (continuous), as well as average negative affect rating reported on EMA, and mean performance on the EMCT. Type of phone was included as a covariate given known differences in smartphone performance regarding EMCTs [27, 32, 33]. Smartphone type was categorized into three groups: iOS models, “Older Android” (≤Android 12), and “Newer Android” (i.e., ≥Android 13). This grouping of different phones was based on differences published in Paolillo et al. [27]. These analyses were done twice: once with the adult sample and then replicated in the older adult sample. Assumptions of linear regressions were checked. For some IIV outcomes, residuals were skewed. Therefore, for these analyses, a log-transformed outcome was utilized. Log-transformed IIV was utilized for QuickTap 1, Odd One Out, Hand Swype, and Memory List for the adult sample and 6-Word Version of Memory List, 12-Tile version of Memory Matrix, and all Color Trick tests for the older adult group. Model statistics from these models therefore represent the statistics from log-transformed outcomes; however, for continuity across the figures we display non-log transformed values. Quadratic associations were examined for continuous predictors and retained in the models if significant at p < 0.05. For linear regression analyses, we corrected for multiple comparisons using the Benjamini Hochberg False Discovery Rate [34]. Lastly, in the older adult sample, we used Spearman correlations to examine if IIV was associated with traditional neuropsychological test performance.
Results
Associations between different methodologies to calculate intraindividual variability (IIV)
To inform our decision as to which IIV estimate to use for analyses, Spearman correlations were conducted to assess the association between IIV calculated from the SD from the first four valid EMCT administrations and IIV calculated from SD and rMSSD with all five EMCT administrations (i.e., since each trial was administered five times, this analysis required no missing data). There was a very strong correlation between SD calculated from the first four valid administrations compared to SD calculated from five assessments and rMSSD calculated from five assessments for most measures in the adult sample (see Table 2). More specifically, all were correlated at ρs > 0.76 except for Quick Tap 2 (ρ = 0.63, 0.68). Similarly, in the older adult group, all intraindividual SD calculated from four administrations were very strongly associated with SD and rMSSD calculated from five administrations (ρs > 0.81). These strong associations led to our decision to use the the four-administration statistic as our main outcome. This approach allowed for a larger sample size since some participants missed or had invalid data for all five administrations. Additionally, SD is more widely recognized and used in neuropsychology as a measure of IIV in traditional testing [13, 14], whereas rMSSD has been primarily utilized in psychophysiology for heart rate variability [35].
Adult sample associations with IIV
Older age was associated with lower IIV for Memory List (β = −0.121, p = 0.030) and Memory Matrix (β = −0.183 p = 0.011), but only the Memory Matrix association remained significant after adjusting for multiple comparisons. Additionally, for Matching Pair, greater age was also significantly associated with lower IIV (β = −0.272, p < 0.001; significant after correcting for multiple comparisons), but there was also a significant quadratic effect of age (β = 0.119, p = 0.041) indicating the negative association weakened as age increased, but this was no longer significant after correcting for multiple comparisons. More education was significantly associated with greater IIV on Odd One Out median reaction time (β = −0.124, p = 0.013), and remained significant after adjusting for multiple comparisons. There was a significant quadratic relationship between higher education levels and increased IIV for CopyKat. Specifically, the association between education and IIV was not linear (β = 0.016, p = 0.741), but instead followed a quadratic pattern, where the effect of education on IIV increased as education levels rose (quadratic: β = 0.129, p = 0.013; remains significant with multiple comparison adjustment).
Of all predictors examined, the greatest predictor of IIV for most tests was mean performance (i.e., demographically uncorrected average T-score). For Memory List, Quick Tap 1, Quick Tap 2, Hand Swype, and Odd One Out, worse mean performance was significantly associated with greater IIV. Additionally, Memory List, Quick Tap 1, Hand Swype, and Odd One Out also demonstrated a significant quadratic effect indicating that the slope (i.e., association) between IIV and mean performance is more steep (i.e., negative) at higher levels of mean performance. All remained significant after correcting for multiple comparisons except for the quadratic effect for Hand Swype. For CopyKat and Memory Matrix, better average performance was associated with greater IIV after correcting for multiple comparisons indicating a linear positive association. The only test in which IIV was not significantly associated with mean performance was Matching Pair. See Fig. 1 for the associations between mean performance and IIV and Supplementary Table 1 for comprehensive test statistics.
Regression lines are adjusted for age (centered), sex (reference: female), race/ethnicity (reference: non-Hispanic white), education (centered), average negative affect (centered), and smartphone type (reference: iOS). T-scores are not demographically corrected. *significant after adjusting for multiple comparisons. Log-transformed IIV was utilized for Quick Tap 1, Odd One Out, Hand Swype, and Memory List model statistics.
Older adult sample associations with IIV
Given the limited variability in scores for Memory Matrix 6-tile (i.e., most performed at the ceiling), there was very limited variability in IIV; thus Memory Matrix 6-tile was not examined in these analyses, but scores are presented in Fig. 2. Older age was associated with less IIV for Memory List 6-Word version (β = −0.222, p = 0.038) and Color Trick Meaning to Color (β = −0.227, p = 0.029), but they were no longer significant after correcting for multiple comparisons. Greater mean negative affect was associated with greater IIV for Memory List 12-Word version (β = 0.353, p = 0.001) and remained significant after correcting for multiple comparisons. Additionally, more education was associated with less variability on Memory Matrix 18-tiles version (β = −0.263, p = 0.024) but not after correcting for multiple comparisons.
Regression lines are adjusted for age (centered), sex (reference: female), race/ethnicity (reference: non-Hispanic white), education (centered), average negative affect (centered), and smartphone type (reference: iOS). *significant after adjusting for multiple comparisons. Log-transformed IIV was utilized for 6-Word Version of Memory List, 12-Tile version of Memory Matrix, and all Color Trick model statistics.
The strongest association with IIV for the majority of tests was mean performance (i.e., average raw score). For the 6-Word version of the Memory List Test, the 12-Tile version of the Memory Matrix, and all Color Trick Tests, poorer average performance was significantly associated with greater variability. Additionally, these tests also demonstrated a significant quadratic association. The 18-Tile Memory Matrix test also demonstrated a significant quadratic effect, but the linear association was not significant indicating an inverted U-shape pattern where individuals with both low and high average performance showed lower variability, and those in the midrange demonstrated the greatest variability. All of these findings remained significant after multiple comparison correction. The 12-Word and 18-Word version of the Memory List Test and Quick Tap 2 demonstrated a significant linear association between worse average performance and greater IIV; the quadratic effects were not significant and removed from the model. However, after correcting for multiple comparisons, the 18-Word version of the Memory List Test was no longer significant. See Fig. 2 for the associations between mean performance and IIV and and Supplementary Table 2 for comprehensive test statistics.
Older adult sample associations with traditional neuropsychological assessment
Spearman correlations between each EMCT-derived IIV measure and the traditional neuropsychological assessment measures can be found in Fig. 3. Overall, there were more significant correlations with traditional neuropsychological measures for the 6-Word Memory List, 12-Tile Memory Matrix, and all measures of Color Trick. All significant correlations indicated a medium association and indicated that greater variability on these measures was associated with worse performance on traditional neuropsychological measures.
MoCA Montreal Cognitive Assessment, WRAT Wide Range Achievement Test, HVLT-R Hopkins Verbal Learning Test-Revised, BVMT-R Brief Visuospatial Memory Test-Revised, WAIS-IV Wechsler Adult Intelligence Scale-IV, CWIT Delis-Kaplan Executive Functioning System (D-KEFS) Color Word Interference Test. *p < 0.05; **p < 0.01.
Discussion
This study examined predictors of within-person variability on several repeated smartphone-based EMCTs in two US-based samples: a general adult sample and an older adult sample. In the general adult sample, participants who performed better on average exhibited greater consistency in their performance across most EMCT measures. Many tests demonstrated a quadratic association in which at lower levels of average performance IIV was greater overall, whereas at higher levels of average performance there was a more linear negative association. The opposite pattern of correlations was observed in working memory tests, possibly due to the difficulty of these tasks at higher levels of performance (i.e., no ceiling effects). These patterns largely replicated in the older adult sample with a broader range of cognitive performance, further supporting the robustness of these observed associations. Again, in this sample we observed that better average performance consistently associated with less variability with a similar quadratic association observed with many tests as in the adult sample. Additionally, some tests (e.g., Memory Matrix 18-Tile) showed lower variability in the lowest and highest performers with those in the middle demonstrating the greatest variability. Taken together, these findings suggest both linear and curvilinear associations between IIV and average performance. Individuals with lower levels of performance tended to either perform consistently poorly (e.g., as demonstrated by the Memory Matrix 18-tile) or show greater variability across time, likely depending on the difficulty of the task. However, generally, those with those with higher levels of cognitive performance did consistently well across most tests. Overall, our findings in two different samples indicate that associations with IIV vary by ability and task difficulty, which aligns with other research [36, 37]. However, this pattern has not been well explored due to the reliance on traditional cognitive measures that often exhibit ceiling effects.
These findings may suggest that the reliability of neuropsychological measures across time may vary as a function of average cognitive performance. In other words, this could imply that single test administrations may be less reliable at lower performance levels and assuming equal reliability across performance levels is potentially problematic. Many neuropsycholgoical tests, such as the Wechsler Adult Intelligence Scale-IV [38], the most widely used assessment for adults [19], do not examine differences in reliability across time at different levels of performance. Thus, further investigation into reliability across time at different performance levels may be a critical next step for improving the psychometric properties of single-administration neuropsychological assessments.
As noted in the introduction, increased variability is commonly observed across a range of populations with or at risk for cognitive impairment, thus we believe this finding reflects meaningful variability that has been established with other methods. This interpretation is further supported by the consistency of findings across both independent samples, including the older adult group, which was well-characterized using traditional neuropsychological assessments and demonstrated multiple associations between EMCT-derived IIV and conventional measures. Thus, we interpret our results as evidence that temporal variability captured by EMCT, particularly with further refinement, may offer clinically useful insights and ultimately supports the continued development of these tools for aging and cognitively at-risk populations.
Regarding the older adult sample, the finding that worse mean performance correlated with greater variability aligns with existing literature, which indicates that greater IIV is associated with an increased risk of developing age-related neurodegenerative disorders like AD [11, 39]. A systematic review of 22 studies found that greater IIV was a risk factor of cognitive impairment and mortality and that variability was predictive over and above traditional measures of central tendency [40]. Further, IIV is a marker of neurodegenerative diseases such as Parkinson’s disease [10] and Alzheimer’s disease and related dementias [41]. The current study contributes to the limited literature examining IIV across time in older adults using EMCT. For example Cerino et al. [25], found that EMCT-derived IIV in processing speed and short-term memory tasks was greater in persons with MCI, even when accounting for mean performance. Similarly, Aschenbrenner et al. 2024 found that IIV in older adults was not consistently associated with demographic factors like age or education [26]. While the literature on the relationship between age and IIV is mixed [42,43,44], our results suggest that increased age may not be associated with increased IIV, but rather that IIV may be more related to cognitive decline and underlying pathology. This possibility aligns with evidence that greater IIV reflects integrity of frontal gray matter density and function, as well as white matter volume [18]. Functional connectivity of the default mode and limbic networks are associated with IIV [45], indicating both structural and functional underpinnings of cognitive fluctuations.
Additionally, we found that greater IIV on EMCTs was associated with worse performance on several in-person neuropsychological assessments. This finding may further demonstrates that lower cognitive performance (as demonstrated by traditional neuropsychological assessments) is associated with greater within-person variability, independent of contextual and environmental factors, as this finding was observed in both standardized and unstandardized environments. Notably, variability on the shortest and easiest memory list measure displayed some of the most robust associations with traditional neuropsychological performance, suggesting that variability on easier tests may be sensitive to overall cognitive functioning. However, further refinement is needed to identify which measures hold the greatest clinical significance across different clinical populations.
Overall, we believe our findings suggest that EMCT-derived IIV could serve as a marker of cognitive vulnerability. Future work should explore its potential to identify people at very early or preclinical stages of disease. A few studies have examined the relationship between cognitive dispersion across traditional neuropsychological measures and AD pathology or MRI markers with mixed results [46,47,48,49]. For instance, one EMCT study showed that genetic AD risk (i.e., presence of an APOE e4 allele) was associated with greater variability on EMCT [26], and a follow-up study by the same group found that within-trial variability was associated with genetic AD risk. Another promising direction is that EMCT derived IIV may also correlate and/or predict everyday functioning and quality of life, which neuropsychological test scores do not consistently predict [50]. Cognitive dispersion on traditional measures has been associated with future functional decline [51] and worse current everyday functioning [52]. Greater IIV may reflect less consistent cognitive abilities in real-world settings and thus correlates with worse real-world functioning (e.g., occupational, social).
This study has several limitations. First, the absence of traditional neuropsychological testing within the general adult sample restricts the scope of our findings. Therefore, we cannot rule out the presence of objective cognitive impairment in this group. Additionally, the generalizability of our results may be limited, particularly within the older adult group, which was predominantly composed of non-Hispanic white individuals with higher levels of education. Our analyses were also restricted to monolingual English speakers within the U.S. Future research should adopt more inclusive sampling strategies and investigate IIV across different languages and cultural backgrounds. Another limitation is the relatively low number of EMCT administrations (i.e., four per task) used to estimate IIV. While prior IIV work has used a higher number of administrations [24], we prioritized feasibility to align with the demands of larger-scale studies and potential clinical applications, where repeated assessments over extended periods may not be feasible. Although our findings suggest that, across two different samples, meaningful variability patterns can be detected even with a limited number of trials, future research should examine the minimum number of administrations needed to yield stable and generalizable IIV estimates across populations and cognitive domains. Additonally, while we covaried for smartphone type, this may not fully account for known software and hardward differences.
In conclusion, this research highlights the potential usefulness of EMCT for assessing within-person variability, offering a valuable tool for further investigating several populations prone to cognitive fluctuations and groups with subtle impairments. A promising area for future exploration is to explore whether EMCT-derived IIV can predict long-term outcomes. If successful, EMCT could be used clinically to monitor individuals who appear cognitively normal according to traditional neuropsychological testing but may still be at risk for decline, facilitating early detection and intervention.
Data availability
The adult sample data has been submitted to the ICPSR repository and is available for download and at https://www.icpsr.umich.edu/web/pages/ICPSR/index.html with appropriate IRB approval. The older adult sample data has been submitted to the National Data Archive under the collection title: Introspective Accuracy, Bias, and Everyday Functioning in Severe Mental Illness.
Change history
03 December 2025
We are updating the html to include a link to the podcast which provides a short overview of that article.
References
Fanning J, Brinkley TE, Campbell LM, Colon-Semenza C, Czaja SJ, Moore RC, et al. Research centers collaborative network workshop on digital health approaches to research in aging. Innov Aging. 2024;8:igae012.
Moore RC, Swendsen J, Depp CA. Applications for self-administered mobile cognitive assessments in clinical research: a systematic review. Int J Methods Psychiatr Res. 2017;26:e1562.
Campbell LM, Paolillo EW, Heaton A, Tang B, Depp CA, Granholm E, et al. Daily activities related to mobile cognitive performance in middle-aged and older adults: an ecological momentary cognitive assessment study. JMIR Mhealth Uhealth. 2020;8:e19579.
Wilks H, Aschenbrenner AJ, Gordon BA, Balota DA, Fagan AM, Musiek E, et al. Sharper in the morning: cognitive time of day effects revealed with high-frequency smartphone testing. J Clin Exp Neuropsychol. 2021;43:825–37.
Hyun J, Sliwinski MJ, Smyth JM. Waking up on the wrong side of the bed: The effects of stress anticipation on working memory in daily life. J Gerontol B Psychol Sci Soc Sci. 2019;74:38–46.
Haatveit B, Westlye LT, Vaskinn A, Flaaten CB, Mohn C, Bjella T, et al. Intra-and inter-individual cognitive variability in schizophrenia and bipolar spectrum disorder: an investigation across multiple cognitive domains. Schizophrenia. 2023;9:89.
Sánchez-Torres AM, García de Jalón E, Gil-Berrozpe GJ, Peralta V, Cuesta MJ, PEPsNA G. Cognitive intraindividual variability, cognitive impairment and psychosocial functioning in first-episode psychosis patients. Psychiatry Res. 2023;328:115473.
Merritt VC, Clark AL, Crocker LD, Sorg SF, Werhane ML, Bondi MW, et al. Repetitive mild traumatic brain injury in military veterans is associated with increased neuropsychological intra-individual variability. Neuropsychologia. 2018;119:340–8.
Wojtowicz MA, Ishigami Y, Mazerolle EL, Fisk JD. Stability of intraindividual variability as a marker of neurologic dysfunction in relapsing remitting multiple sclerosis. J Clin Exp Neuropsychol. 2014;36:455–63.
Burton CL, Strauss E, Hultsch DF, Moll A, Hunter MA. Intraindividual variability as a marker of neurological dysfunction: a comparison of Alzheimer’s disease and Parkinson’s disease. J Clin Exp Neuropsychol. 2006;28:67–83.
Costa AS, Dogan I, Schulz JB, Reetz K. Going beyond the mean: Intraindividual variability of cognitive performance in prodromal and early neurodegenerative disorders. Clin Neuropsychol. 2019;33:369–89.
Duchek JM, Balota DA, Tse C-S, Holtzman DM, Fagan AM, Goate AM. The utility of intraindividual variability in selective attention tasks as an early marker for Alzheimer’s disease. Neuropsychology. 2009;23:746–58.
Vance DE, Del Bene VA, Frank JS, Billings R, Triebel K, Buchholz A, et al. Cognitive intra-individual variability in HIV: An integrative review. Neuropsychol Rev. 2022;32:855–76.
Vance DE, Collette C, Frank JS, Billings R, Deaver J, Del Bene VA, et al. Cognitive intra-individual variability in breast cancer survivors: a systematic review. Appl Neuropsychol Adult. 2023. 1–15. https://doi.org/10.1080/23279095.2023.2270097.
Salthouse TA. Implications of within-person variability in cognitive and neuropsychological functioning for the interpretation of change. Neuropsychology. 2007;21:401–11.
Bielak AA, Hultsch DF, Strauss E, MacDonald SW, Hunter MA. Intraindividual variability is related to cognitive change in older adults: evidence for within-person coupling. Psychol Aging. 2010;25:575–86.
Williams BR, Hultsch DF, Strauss EH, Hunter MA, Tannock R. Inconsistency in reaction time across the life span. Neuropsychol. 2005;19:88–96.
MacDonald SW, Nyberg L, Bäckman L. Intra-individual variability in behavior: Links to brain structure, neurotransmission and neuronal activity. Trends Neurosci. 2006;29:474–80.
Rabin LA, Barr WB, Burton LA. Assessment practices of clinical neuropsychologists in the United States and Canada: A survey of INS, NAN, and APA division 40 members. Arch Clin Neuropsychol. 2005;20:33–65.
Ahmad F, Sachdeva P, Sarkar J, Izhaar R. Circadian dysfunction and Alzheimer’s disease–An updated review. Aging Med. 2023;6:71–81.
Rigat L, Ouk K, Kramer A, Priller J. Dysfunction of circadian and sleep rhythms in the early stages of Alzheimer’s disease. Acta Physiologica. 2023;238:e13970.
de Leeuw M, Verhoeve SI, van der Wee NJ, van Hemert AM, Vreugdenhil E, Coomans CP. The role of the circadian system in the etiology of depression. Neurosci Biobehav Rev. 2023;153:105383.
Chen Z, Zhao S, Tian S, Yan R, Wang H, Wang X, et al. Diurnal mood variation symptoms in major depressive disorder associated with evening chronotype: evidence from a neuroimaging study. J Affect Dis. 2022;298:151–9.
Welhaf MS, Wilks H, Aschenbrenner AJ, Balota DA, Schindler SE, Benzinger T, et al. Naturalistic assessment of reaction time variability in older adults at risk for Alzheimer’s disease. J Int Neuropsychol Soc. 2024;30:428–38.
Cerino ES, Katz MJ, Wang C, Qin J, Gao Q, Hyun J, et al. Variability in working memory performance on mobile devices is sensitive to mild cognitive impairment: results from the Einstein Aging Study: neuropsychology/early detection of cognitive decline with neuropsychological tests. Front Dig Health. 2021;3:758031.
Aschenbrenner AJ, Hassenstab J, Morris JC, Cruchaga C, Jackson JJ. Relationships between hourly cognitive variability and risk of Alzheimer’s disease revealed with mixed-effects location scale models. Neuropsychology. 2023;38:69–80.
Paolillo EW, Bomyea J, Depp CA, Henneghan AM, Raj A, Moore RC. Characterizing performance on a suite of english-language neuroUX mobile cognitive tests in a US adult sample: ecological momentary cognitive testing study. J Med Internet Res. 2024;26:e51978.
Moore RC, Ackerman RA, Russell MT, Campbell LM, Depp CA, Harvey PD, et al. Feasibility and validity of ecological momentary cognitive testing among older adults with mild cognitive impairment. Front Digit Health. 2022;4:946685.
Rossetti HC, Lacritz LH, Cullum CM, Weiner MF. Normative data for the montreal cognitive assessment (MoCA) in a population-based sample. Neurology. 2011;77:1272–5.
Jak AJ, Bondi MW, Delano-Wood L, Wierenga C, Corey-Bloom J, Salmon DP, et al. Quantification of five neuropsychological approaches to defining mild cognitive impairment. Am J Geriatr Psychiatry. 2009;17:368–75.
Sørensen JB. The use and misuse of the coefficient of variation in organizational demography research. Sociol Methods Res. 2002;30:475–91.
Passell E, Strong RW, Rutter LA, Kim H, Scheuer L, Martini P, et al. Cognitive test scores vary with choice of personal digital device. Behav Res Methods. 2021;53:2544–57.
Nicosia J, Wang B, Aschenbrenner AJ, Sliwinski MJ, Yabiku ST, Roque NA, et al. To BYOD or not: Are device latencies important for bring-your-own-device (BYOD) smartphone cognitive testing? Behav Res Methods. 2023;55:2800–12.
Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B. 1995;57:289–300.
Pham T, Lau ZJ, Chen SA, Makowski D. Heart rate variability in psychology: a review of HRV indices and an analysis tutorial. Sensors. 2021;21:3998.
Goldhammer F, Naumann J, Greiff S. More is not always better: the relation between item response and item response time in Raven’s matrices. J Intell. 2015;3:21–40.
Dodonova YA, Dodonov YS. Faster on easy items, more accurate on difficult ones: cognitive ability and performance on a task of varying difficulty. Intelligence. 2013;41:1–10.
Wechsler D. Wechsler adult intelligence scale—fourth edition technical and interpretive manual. Pearson; San Antonio, TX: 2008.
Hilborn JV, Strauss E, Hultsch DF, Hunter MA. Intraindividual variability across cognitive domains: Investigation of dispersion levels and performance profiles in older adults. J Clin Exp Neuropsychol. 2009;31:412–24.
Haynes BI, Bauermeister S, Bunce D. A systematic review of longitudinal associations between reaction time intraindividual variability and age-related cognitive decline or impairment, dementia, and mortality. J Int Neuropsychol Soc. 2017;23:431–45.
Murtha S, Cismaru R, Waechter R, Chertkow H. Increased variability accompanies frontal lobe damage in dementia. J Int Neuropsychol Soc. 2002;8:360–72.
Mella N, Fagot D, de Ribaupierre A. Dispersion in cognitive functioning: age differences over the lifespan. J Clin Exp Neuropsychol. 2016;38:111–26.
Morse CK. Does variability increase with age? an archival study of cognitive measures. Psychol Aging. 1993;8:156–64.
Schmiedek F, Lövdén M, Lindenberger U. Keeping it steady: older adults perform more consistently on cognitive tasks than younger adults. Psychol Sci. 2013;24:1747–54.
Lin SS-H, McDonough IM. Intra-individual cognitive variability in neuropsychological assessment: A sign of neural network dysfunction. Neuropsychol Dev Cogn B Aging Neuropsychol Cogn. 2022;29:375–99.
Watermeyer T, Marroig A, Ritchie CW, Ritchie K, Blennow K, Muniz-Terrera G, et al. Cognitive dispersion is not associated with cerebrospinal fluid biomarkers of Alzheimer’s disease: results from the European prevention of Alzheimer’s dementia (EPAD) v500. 0 Cohort. J Alzheimer Dis. 2020;78:185–94.
Nicosia J, Aschenbrenner AJ, Balota DA, Sliwinski MJ, Tahan M, Adams S, et al. Unsupervised high-frequency smartphone-based cognitive assessments are reliable, valid, and feasible in older adults at risk for Alzheimer’s disease. J Int Neuropsychol Soc. 2023;29:459–71.
Holmqvist SL, Thomas KR, Edmonds EC, Calcetas A, Edwards L, Bangen KJ, et al. Cognitive dispersion is elevated in amyloid-positive older adults and associated with regional hypoperfusion. J Int Neuropsychol Soc. 2023;29:621–31.
Malek-Ahmadi M, Lu S, Chan Y, Perez SE, Chen K, Mufson EJ. Cognitive domain dispersion association with Alzheimer’s disease pathology. J Alzheimer Dis. 2017;58:575–83.
Chaytor N, Schmitter-Edgecombe M. The ecological validity of neuropsychological tests: a review of the literature on everyday cognitive skills. Neuropsychol Rev. 2003;13:181–97.
Bangen KJ, Weigand AJ, Thomas KR, Delano-Wood L, Clark LR, Eppig J, et al. Cognitive dispersion is a sensitive marker for early neurodegenerative changes and functional decline in nondemented older adults. Neuropsychology. 2019;33:599–608.
Fellows RP, Schmitter-Edgecombe M. Between-domain cognitive dispersion and functional abilities in older adults. J Clin Exp Neuropsychol. 2015;37:1013–23.
Funding
The adult sample study was supported by NeuroUX Inc. The older adult sample study was supported by R01-MH112620-03S1 from NIH/NIMH to AEP. Salary support was provided by NIH/NIMH (T32MH019934 and T32MH018399 to LMC), and NIH/NIA (K23AG076663; to AMW). Dr Twamley gratefully acknowledges the support of a VA Rehabilitation Research and Development Research Career Scientist Award. The content is solely the responsibility of the authors and does not necessarily represent the official position of the institution.
Author information
Authors and Affiliations
Contributions
AEP, RCM, RA, CAD, and PDH contributed to the conception, design, and obtaining funding for this study. LMC performed the statistical analysis. LMC, AMW, AH, and RCM wrote the manuscript. EWP, RA, JB, CAD, PDH, AEP, and EWT provided critical edits. All authors contributed to manuscript revisions and approved the submitted version.
Corresponding author
Ethics declarations
Competing interests
LMC serves as a consultant for NeuroUX Inc. EWP previously served as a data analytic consultant for NeuroUX Inc. RCM is a cofounder of KeyWise AI, Inc, and NeuroUX Inc. AMH previously served as a consultant for Prodeo, Inc. PDH has received consulting fees or travel reimbursements from Alkermes, BMS (Karuna Therapeutics), Boehringer Ingelheim, Merck Pharma, Minerva Neurosciences, DSP-Sunovion Pharma, and Wed MD; Psychiatrist.com. PDH is chief scientific officer of i-Function, Inc and Founder and Scientific Consultant to EMA Wellness, Inc. All other authors declare no commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Campbell, L.M., Weinstein, A.M., Henneghan, A. et al. Exploring the relationship between mean performance and within-person variability on smartphone-based cognitive testing in adults across the lifespan. NPP—Digit Psychiatry Neurosci 3, 15 (2025). https://doi.org/10.1038/s44277-025-00036-x
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44277-025-00036-x