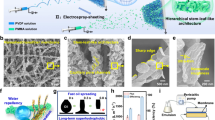
Abstract
Nanocarriers are indispensable in biomedicine, enabling breakthroughs in drug delivery, vaccines and diagnostics. Despite notable successes facilitated by microfluidic technologies, sustainable, low-cost and high-throughput production of monodisperse nanocarriers remains a longstanding challenge. Here we report surfactant-flux-induced interfacial instability, a self-driven nanoemulsification mechanism triggered by non-equilibrium surfactant partitioning at a water–oil interface. This strategy produces 5 l of highly uniform nanoemulsion (0.2 l of disperse phase) in 1 min, producing droplets with diameters as small as 34 nm and polydispersity indices below 0.10. This self-driven nanoemulsification is robust across a broad range of pH values (~3–11) and temperatures (~4–85 °C), demonstrating compatibility with over ten oil–water–surfactant systems and enabling the synthesis of diverse nanocarriers including nanodroplets, micelles, vesicles, polymeric nanoparticles and metal–organic framework nanocrystals. We further demonstrate that surfactant flux underpins the observed interfacial instability, offering a mechanism for nanoemulsification under far-from-equilibrium conditions. This work addresses a bottleneck in nanomanufacturing and provides insights into dynamic surfactant behavior at immiscible interfaces.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data Availability
Data supporting the findings of this study are available within the Article and its Supplementary Information. Source data are provided with this paper. Source data are also available via figshare at https://doi.org/10.6084/m9.figshare.31154185 (ref. 65).
References
Sheth, T., Seshadri, S., Prileszky, T. & Helgeson, M. E. Multiple nanoemulsions. Nat. Rev. Mater. 5, 214–228 (2020).
Carney, R. P. et al. Harnessing extracellular vesicle heterogeneity for diagnostic and therapeutic applications. Nat. Nanotechnol. 20, 14–25 (2025).
Grimaldi, N. et al. Lipid-based nanovesicles for nanomedicine. Chem. Soc. Rev. 45, 6520–6545 (2016).
Ma, X. B. et al. Bioengineered nanogels for cancer immunotherapy. Chem. Soc. Rev. 51, 5136–5174 (2022).
Beach, M. A. et al. Polymeric nanoparticles for drug delivery. Chem. Rev. 124, 5505–5616 (2024).
Allen, T. M. & Cullis, P. R. Liposomal drug delivery systems: from concept to clinical applications. Adv. Drug Deliv. Rev. 65, 36–48 (2013).
Cullis, P. R. & Felgner, P. L. The 60-year evolution of lipid nanoparticles for nucleic acid delivery. Nat. Rev. Drug Discov. 23, 709–722 (2024).
Li, X., Qin, Z., Wang, S., Zhang, L. & Jiang, X. Microfluidics-assembled nanovesicles for nucleic acid delivery. Acc. Chem. Res. 58, 570–582 (2025).
Moammeri, A. et al. Current advances in niosomes applications for drug delivery and cancer treatment. Mater. Today Bio 23, 100837 (2023).
Hou, X., Zaks, T., Langer, R. & Dong, Y. Lipid nanoparticles for mRNA delivery. Nat. Rev. Mater. 6, 1078–1094 (2021).
Liu, C., Feng, Q. & Sun, J. Lipid nanovesicles by microfluidics: manipulation, synthesis, and drug delivery. Adv. Mater. 31, 1804788 (2019).
Gu, F. et al. Precise engineering of targeted nanoparticles by using self-assembled biointegrated block copolymers. Proc. Natl Acad. Sci. USA 105, 2586–2591 (2008).
Cabral, H., Miyata, K., Osada, K. & Kataoka, K. Block copolymer micelles in nanomedicine applications. Chem. Rev. 118, 6844–6892 (2018).
Heuer-Jungemann, A. et al. The role of ligands in the chemical synthesis and applications of inorganic nanoparticles. Chem. Rev. 119, 4819–4880 (2019).
Desgouilles, S. et al. The design of nanoparticles obtained by solvent evaporation: a comprehensive study. Langmuir 19, 9504–9510 (2003).
Liu, Y. et al. Formulation of nanoparticles using mixing-induced nanoprecipitation for drug delivery. Ind. Eng. Chem. Res. 59, 4134–4149 (2020).
Hornig, S., Heinze, T., Becer, C. R. & Schubert, U. S. Synthetic polymeric nanoparticles by nanoprecipitation. J. Mater. Chem. 19, 3838–3840 (2009).
Silva, A. C. et al. Preparation, characterization and biocompatibility studies on risperidone-loaded solid lipid nanoparticles (SLN): high pressure homogenization versus ultrasound. Colloids Surf. B Biointerfaces 86, 158–165 (2011).
Zhang, C.-L. & Yu, S.-H. Nanoparticles meet electrospinning: recent advances and future prospects. Chem. Soc. Rev. 43, 4423–4448 (2014).
Amstad, E. et al. Production of amorphous nanoparticles by supersonic spray-drying with a microfluidic nebulator. Science 349, 956–960 (2015).
Solans, C., Morales, D. & Homs, M. Spontaneous emulsification. Curr. Opin. Colloid Interface Sci. 22, 88–93 (2016).
López-Montilla, J. C., Herrera-Morales, P. E., Pandey, S. & Shah, D. O. Spontaneous emulsification:mechanisms, physicochemical aspects, modeling, and applications. J. Dispers. Sci. Technol. 23, 219–268 (2002).
Cholakova, D., Vinarov, Z., Tcholakova, S. & Denkov, N. D. Self-emulsification in chemical and pharmaceutical technologies. Curr. Opin. Colloid Interface Sci. 59, 101576 (2022).
Naseema, A., Kovooru, L., Behera, A. K., Kumar, K. P. P. & Srivastava, P. A critical review of synthesis procedures, applications and future potential of nanoemulsions. Adv. Colloid Interface Sci. 287, 102318 (2021).
Ren, G. H. et al. Nanoemulsion formation by the phase inversion temperature method using polyoxypropylene surfactants. J. Colloid Interface Sci. 540, 177–184 (2019).
Tcholakova, S. et al. Efficient self-emulsification via cooling-heating cycles. Nat. Commun. 8, 15012 (2017).
Saberi, A. H., Fang, Y. & McClements, D. J. Thermal reversibility of vitamin E-enriched emulsion-based delivery systems produced using spontaneous emulsification. Food Chem. 185, 254–260 (2015).
Wang, L. et al. New low-energy method for nanoemulsion formation: pH regulation based on fatty acid/amine complexes. Langmuir 36, 10082–10090 (2020).
Chen, L. H., Cheng, L. C. & Doyle, P. S. Nanoemulsion-loaded capsules for controlled delivery of lipophilic active ingredients. Adv. Sci. 7, 2001677 (2020).
Perazzo, A., Preziosi, V. & Guido, S. Phase inversion emulsification: current understanding and applications. Adv. Colloid Interface Sci. 222, 581–599 (2015).
Chen, C. et al. Controllable fabrication of highly uniform sub-10 nm nanoparticles from spontaneous confined nanoemulsification. Small 19, 2300801 (2023).
Browne, K. P., Walker, D. A., Bishop, K. J. M. & Grzybowski, B. A. Self-division of macroscopic droplets: partitioning of nanosized cargo into nanoscale micelles. Angew. Chem. Int. Ed. 49, 6756–6759 (2010).
Ravera, F., Dziza, K., Santini, E., Cristofolini, L. & Liggieri, L. Emulsification and emulsion stability: the role of the interfacial properties. Adv. Colloid Interface Sci. 288, 102344 (2021).
Singh, Y. et al. Nanoemulsion: concepts, development and applications in drug delivery. J. Control. Release 252, 28–49 (2017).
Komaiko, J. S. & McClements, D. J. Formation of food-grade nanoemulsions using low-energy preparation methods: a review of available methods. Compr. Rev. Food Sci. Food Saf. 15, 331–352 (2016).
Feng, J. et al. Nanoemulsions obtained via bubble-bursting at a compound interface. Nat. Phys. 10, 606–612 (2014).
Feng, J. et al. A scalable platform for functional nanomaterials via bubble-bursting. Adv. Mater. 28, 4047–4052 (2016).
Guha, I. F., Anand, S. & Varanasi, K. K. Creating nanoscale emulsions using condensation. Nat. Commun. 8, 1371 (2017).
Anand, S., Galavan, V. & Mulik, M. U. Continuous synthesis of nanoscale emulsions by vapor condensation (EVC). Adv. Sci. 11, 2307443 (2024).
Zhu, P. & Wang, L. Thermo-induced reversible nanoemulsification. Matter 8, 102103 (2025).
Colon-Quintana, G. S., Clarke, T. B. & Dick, J. E. Interfacial solute flux promotes emulsification at the water|oil interface. Nat. Commun. 14, 705 (2023).
Iglicki, D., Goubault, C., Mahamoud, M. N., Chevance, S. & Gauffre, F. Shedding light on the formation and stability of mesostructures in ternary ‘Ouzo’ mixtures. J. Colloid Interface Sci. 633, 72–81 (2023).
Balaj, R. V., Xue, W., Bayati, P., Mallory, S. & Zarzar, L. D. Dynamic partitioning of surfactants into nonequilibrium emulsion droplets. J. Am. Chem. Soc. 146, 26340–26350 (2024).
Su, Y.-Y. et al. Wetting-induced interfacial instability: a mechanism for droplet emission at air-liquid interfaces. Sci. Adv. 11, eads1065 (2025).
Vitale, S. A. & Katz, J. L. Liquid droplet dispersions formed by homogeneous liquid-liquid nucleation: ‘the Ouzo effect’. Langmuir 19, 4105–4110 (2003).
Schmidt, S. et al. Microfluidic spontaneous emulsification for generation of O/W nanoemulsions—opportunity for in-space manufacturing. Adv. Healthcare Mater. 12, 2203363 (2023).
Toprakcioglu, Z., Challa, P. K., Morse, D. B. & Knowles, T. Attoliter protein nanogels from droplet nanofluidics for intracellular delivery. Sci. Adv. 6, eaay7952 (2020).
Zahariev, T. K., Tadjer, A. V. & Ivanova, A. N. Transfer of non-ionic surfactants across the water-oil interface: a molecular dynamics study. Colloids Surf. A 506, 20–31 (2016).
Tian, J.-Q. et al. Interfacial energy-mediated bulk transport across artificial cell membranes. Nat. Chem. Eng. 1, 450–461 (2024).
Yang, Z., Wei, J., Sobolev, Y. I. & Grzybowski, B. A. Systems of mechanized and reactive droplets powered by multi-responsive surfactants. Nature 553, 313–318 (2018).
Lach, S., Yoon, S. M. & Grzybowski, B. A. Tactic, reactive, and functional droplets outside of equilibrium. Chem. Soc. Rev. 45, 4766–4796 (2016).
Zarzar, L. D. et al. Dynamically reconfigurable complex emulsions via tunable interfacial tensions. Nature 518, 520–524 (2015).
Wang, W. et al. Hole-shell microparticles from controllably evolved double emulsions. Angew. Chem. Int. Ed. 52, 8084–8087 (2013).
Ravera, F., Ferrari, M., Liggieri, L., Miller, R. & Passerone, A. Measurement of the partition coefficient of surfactants in water/oil systems. Langmuir 13, 4817–4820 (1997).
Catanoiu, G., Carey, E., Patil, S. R., Engelskirchen, S. & Stubenrauch, C. Partition coefficients of nonionic surfactants in water/n-alkane systems. J. Colloid Interface Sci. 355, 150–156 (2011).
Fong, C., Le, T. & Drummond, C. J. Lyotropic liquid crystal engineering-ordered nanostructured small molecule amphiphile self-assembly materials by design. Chem. Soc. Rev. 41, 1297–1322 (2012).
Massarweh, O. & Abushaikha, A. The use of surfactants in enhanced oil recovery: a review of recent advances. Energy Rep. 6, 303–318 (2020).
Miura, S. et al. pH-induced motion control of self-propelled oil droplets using a hydrolyzable gemini cationic surfactant. Langmuir 30, 7977–7985 (2014).
Eastoe, J. & Dalton, J. S. Dynamic surface tension and adsorption mechanisms of surfactants at the air-water interface. Adv. Colloid Interface Sci. 85, 103–144 (2000).
Tadros, T., Izquierdo, P., Esquena, J. & Solans, C. Formation and stability of nano-emulsions. Adv. Colloid Interface Sci. 108–109, 303–318 (2004).
Blau, B., Krzeczek, O., Heinrich, C. & Klein, M. Analysis of water-in-gasoline emulsions via experiments and direct numerical simulations. Exp. Comput. Multiph. Flow 7, 178–196 (2025).
Hashemnejad, S. M., Badruddoza, A. M., Zarket, B., Castaneda, C. R. & Doyle, P. S. Thermoresponsive nanoemulsion-based gel synthesized through a low-energy process. Nat. Commun. 10, 2749 (2019).
Shahidzadeh, N., Bonn, D. & Meunier, J. Dynamics of spontaneous emulsification for fabrication of oil in water emulsions. Langmuir 16, 9703–9708 (2000).
Davis, C. R., Martinez, C. J., Howarter, J. A. & Erk, K. A. Diffusion-controlled spontaneous emulsification of water-soluble oils via micelle swelling. Langmuir 36, 7517–7527 (2020).
Deng, N.-N. et al. Dataset. figshare https://doi.org/10.6084/m9.figshare.31154185 (2026).
Acknowledgements
We acknowledge financial support from the National Natural Science Foundation of China (22278281 (W.W.), 22494712 (L.-Y.C.) and 22278264 (N.-N.D.)), Sichuan Science and Technology Program (2025YFHZ0287 (W.W.) and 2025ZNSFSC0335 (N.-N.D.)), Shanghai Municipal Science and Technology Major Project (N.-N.D.) and Shanghai Pilot Program for Basic Research (N.-N.D.).
Author information
Authors and Affiliations
Contributions
Conceptualization, Q.C., N.-N.D. and W.W. Supervision, N.-N.D. and W.W. Methodology, Q.C., P.Z., H.-Y.Y., T.-Y.H., N.-N.D. and W.W. Investigation, Q.C. Visualization, Q.C., P.Z., H.-Y.Y., T.-Y.H., N.-N.D. and W.W. Resources, D.-W.P., X.-J.J., Z.L., R.X., N.-N.D., W.W. and L.-Y.C. Project administration, N.-N.D., W.W. and L.-Y.C. Funding acquisition, N.-N.D., W.W. and L.-Y.C. Writing—original draft, Q.C., N.-N.D. and W.W. Writing—review and editing, N.-N.D. and W.W.
Corresponding authors
Ethics declarations
Competing interests
Sichuan University has filed a Chinese patent application (CN202510294623.2, China, filed on 13 March 2025) based on this work.
Peer review
Peer review information
Nature Chemical Engineering thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Notes 1–11, Figs. 1–33, Tables 1–5 and refs. 1 and 2.
Supplementary Video 1 (download MP4 )
Interfacial instability of an LR300-dyed DCM droplet in water with 0.1 wt% of F127.
Supplementary Video 2 (download MP4 )
Interfacial instability of a Cur-dyed DCM droplet in water with 0.1 wt% of RhB-F127.
Supplementary Video 3 (download MP4 )
Self-nanoemulsification process of an LR300-dyed DCM droplet in water with 0.1 wt% of F127.
Supplementary Video 4 (download MP4 )
Fast, manually shaken production of nanoemulsions within 60 s via self-nanoemulsification.
Supplementary Video 5 (download MP4 )
Fast production of 5 l of nanoemulsions within 60 s via self-nanoemulsification.
Supplementary Video 6 (download MP4 )
Fast production of 1 l of PLGA-containing nanoemulsions within 60 s via self-nanoemulsification.
Supplementary Data 1 (download ZIP )
Statistical source data.
Source data
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cao, Q., Zhang, P., Yang, HY. et al. Non-equilibrium surfactant partitioning drives self-nanoemulsification for scalable nanocarrier production. Nat Chem Eng 3, 181–192 (2026). https://doi.org/10.1038/s44286-026-00367-2
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44286-026-00367-2