Abstract
Pregnant women are often excluded from randomized clinical trials due to safety concerns, yet the increasing prevalence of pre-existing conditions and pregnancy complications necessitates medication use. Observational cohort data can provide valuable insights to support clinical decision-making. We developed a web-based tool that presents population-level data on medication use and preterm birth risk. By integrating real-world evidence, this tool helps clinicians assess medication-related outcomes and improve maternal and neonatal health.
Similar content being viewed by others
Introduction
Pregnant women are typically excluded from randomized clinical trials due to concerns about fetal safety. However, with increasing maternal age and rising obesity rates, a growing proportion of pregnant individuals require medications for pre-existing conditions and pregnancy-related complications1,2,3,4. Despite the widespread use of prescription medications in pregnancy, clinical guidance remains limited, making it difficult for healthcare providers to assess potential risks and benefits.
Observational cohort data offer an alternative approach to randomized trials, providing large-scale, real-world evidence to support clinical decision-making. Preterm birth, defined as delivery before 37 weeks of gestation, affects approximately 10%5 of pregnancies worldwide and is a leading cause of neonatal morbidity and mortality5,6,7. It is associated with significant long-term health and economic consequences, including increased disability-adjusted life-years (DALYs)8, higher risks of neurodevelopmental impairments9, respiratory complications10, and future cardiovascular diseases11. These impacts highlight the critical need for predictive tools to guide medication decisions during pregnancy.
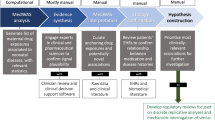
To address this knowledge gap, we developed an interactive web-based tool that provides clinicians with population-level data on prescription drug use during pregnancy and its association with preterm birth.
This tool was built using data from the Alberta Pregnancy Cohort, a longitudinal dataset containing information on 238,676 mothers and 328,834 singleton live births in Alberta, Canada, between 2009 and 2018. The dataset includes pharmaceutical claims, gestational age, birth outcomes, and maternal demographic data. By integrating these data into an interactive dashboard, we provide clinicians with an accessible platform to assess medication-related risks in real time.
Users can start by selecting a medication of interest from a drop-down menu (Supplementary Fig. 1), which generates graphical summaries of four key types of findings:
-
1.
Preterm birth incidence by medication use: This shows four findings: the incidence of term versus preterm births among women who did (versus did not) have a prescription for the medication during pregnancy (Supplementary Fig. 2).
-
2.
Maternal age and medication use: The incidence of term versus preterm births among women who did (versus did not have) a prescription for the medication, stratified by maternal age categories (Supplementary Fig. 3).
-
3.
Timing of first prescription: The incidence of term versus preterm births according to the trimester (one, two, or three) in which women first received a prescription for the medication (Supplementary Fig. 4).
-
4.
Primary indications for medication use: The 10 most frequent medical reasons for which women who received the medication visited their physician during pregnancy (Supplementary Fig. 5).
This tool provides an evidence-based approach to prescribing decisions by summarizing trends from a large observational dataset. However, the tool’s reliance on observational data may lead to selection bias; therefore, future enhancements will incorporate adjusted statistical models to control for potential confounding factors (e.g., maternal age, comorbidities, and socioeconomic factors). We also acknowledge the possibility of confounding by indication, where the underlying condition for which a medication is prescribed may itself influence the risk of preterm birth. While not addressed in the current version, stratifying outcomes by primary clinical indications is an important direction for future work to support more clinically meaningful interpretation. Expanding the dataset to include real-time updates from other provinces will further enhance its utility12.
Methods
Study design
This study used a retrospective cohort design, analyzing all singleton births in Alberta between 2009 and 2018. Inclusion criteria required maternal residency in Alberta for at least one year before and throughout pregnancy. Records with missing gestational age or birth weight were excluded.
Medication classification
Medication exposure was identified through pharmaceutical claims data and categorized using the first three levels of the Anatomical Therapeutic Chemical (ATC) classification system. To ensure compliance with privacy regulations, we restricted the display of results to ATC codes with at least 20 exposed pregnant individuals, in line with widely accepted disclosure control practices13.
Dashboard development
The web-based dashboard was developed using Streamlit and Python, enabling interactive visualization of preterm birth incidence, maternal age distributions, timing of prescription initiation, and common indications for medication use.
Data availability
The dataset used in this study is derived from the Alberta Pregnancy Cohort. Access to data is subject to provincial health data privacy regulations. The dashboard website can be found at https://alberta-pregnancy-preterm-info-621757242756.us-central1.run.app.
Code availability
The dashboard code is available upon reasonable request.
References
Mitchell, A. A. et al. Medication use during pregnancy, with particular focus on prescription drugs: 1976–2008. Am. J. Obstet. Gynecol. 205, 51.e1–8 (2011).
Thunbo, M. Ø, Vendelbo, J. H., Witte, D. R., Larsen, A. & Pedersen, L. H. Use of medication in pregnancy on the rise: study on 1.4 million Danish pregnancies from 1998 to 2018. Acta Obstet. Gynecol. Scand. 103, 1210–1223 (2024).
Alyami, A. A., Alem, M. M., Dorgham, S. R. & Alshamandy, S. A. Trends of over-the-counter and prescribed medication use during pregnancy: a cross-sectional study. J. Multidiscip. Healthc. 16, 3847–3856 (2023).
Mansour, O. et al. Prescription medication use during pregnancy in the United States from 2011 to 2020: trends and safety evidence. Am. J. Obstet. Gynecol. 231, 250.e1–250.e16 (2024).
Ohuma, E. O. et al. National, regional, and global estimates of preterm birth in 2020, with trends from 2010: a systematic analysis. Lancet 402, 1261–1271 (2023).
Cao, G., Liu, J. & Liu, M. Global, regional, and national incidence and mortality of neonatal preterm birth, 1990–2019. JAMA Pediatr. 176, 787–796 (2022).
Liang, X., Lyu, Y., Li, J., Li, Y. & Chi, C. Global, regional, and national burden of preterm birth, 1990–2021: a systematic analysis from the global burden of disease study 2021. EClinicalMedicine 76, 102840 (2024).
GBD 2021 Diseases and Injuries Collaborators. Global incidence, prevalence, years lived with disability (YLDs), disability-adjusted life-years (DALYs), and healthy life expectancy (HALE) for 371 diseases and injuries in 204 countries and territories and 811 subnational locations, 1990–2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet 403, 2133–2161 (2024).
Inder, T. E., Volpe, J. J. & Anderson, P. J. Defining the neurologic consequences of preterm birth. N. Engl. J. Med. 389, 441–453 (2023).
Course, C. W., Kotecha, E. A., Course, K. & Kotecha, S. The respiratory consequences of preterm birth: from infancy to adulthood. Br. J. Hosp. Med. 85, 1–11 (2024).
Clayton, G. L. et al. Preterm birth and cardiometabolic health trajectories from birth to adulthood: the Avon Longitudinal Study of Parents and Children. J. Am. Heart Assoc. 14, e030823 (2025).
Bérard, A. et al. The Canadian Mother–Child Cohort Active Surveillance Initiative (CAMCCO): comparisons between Quebec, Manitoba, Saskatchewan, and Alberta. PLoS ONE 17, e0274355 (2022).
Treasury Board of Canada SecretariatPrivacy Implementation Notice 2020-03: Protecting Privacy When Releasing Information About a Small Number of Individuals. Treasury Board of Canada Secretariat (2020).
Acknowledgements
This study was supported by the Alberta Machine Intelligence Institute (AMII), Natural Sciences and Engineering Research Council of Canada (NSERC), and the Alberta Innovates Graduate Student Scholarship for A.K.P. R.G. was supported by AMII, the Canadian Institute for Advanced Research (CIFAR), and NSERC. P.K. is the Tier 1 Canada Research Chair in Women and Children’s Cardiometabolic Health; she holds the Canadian Institute of Health Research (CIHR) Chair in Sex and Gender Science and the Heart & Stroke Foundation Chair in Cardiovascular Research. The funders had no role in study design, data collection, data analysis, data interpretation, or writing of the manuscript.
Author information
Authors and Affiliations
Contributions
Study design was contributed by A.K.P. and S.V.K. A.K.P. performed the data analysis, drafted the manuscript, and developed the dashboard interface. S.V.K., R.G., and P.K. reviewed and edited the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the University of Alberta Research Ethics Board (Pro00114981). Patients’ written informed consent was waived by the University of Alberta Research Ethics Board as the study used data from anonymized electronic health records.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Paul, A.K., Kalmady, S.V., Greiner, R. et al. Developing point-of-care tools to inform decisions regarding prescription medication use in pregnancy. npj Womens Health 3, 43 (2025). https://doi.org/10.1038/s44294-025-00093-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44294-025-00093-9