Abstract
Melatonin production is the gold-standard marker of the physiological “biological night”. Assessing it in athletes outside the laboratory enables individualized circadian interventions. This study quantified morning misalignment in elite athletes and examined its association with subjective well-being. Twenty-two elite female athletes (mean age = 26.8 ± 4.3 years) from the same national squad were monitored during a one-week training camp with standardized meal, training and sleep opportunities. Athletes wore actimetry devices to measure sleep and light exposure and completed daily sleep diaries with well-being ratings. On one day, salivary melatonin was self-sampled hourly from four hours before habitual sleep onset to one hour after, and again at wake time and one hour after waking. A longer phase angle between melatonin onset and sleep onset was associated with longer total sleep duration, whereas being an evening chronotype and sharing a room with a dissimilar chronotype were both independently associated with shorter total sleep duration. Elevated melatonin one hour after waking, indicative of morning circadian misalignment, was associated with lower subjective well-being. These findings demonstrate that real-life circadian timing influences both sleep and well-being in elite athletes and support the value of individualized circadian-informed strategies to optimize health and recovery.
Similar content being viewed by others
Introduction
Endogenous (circadian) time is set by the rhythmic activity of the hypothalamic suprachiasmatic nuclei (SCN), which is entrained to the 24-h day primarily by light, thereby aligning internal physiology with the external light-dark cycle1,2. In turn, the SCN rhythm drives a rhythm in pineal melatonin levels, with the onset and offset of high nocturnal melatonin levels defining the organism’s “biological night” 3, and low diurnal levels defining the “biological day”. There is, however, substantial inter-individual variation in both the timing (phase) and duration of biological night and day4.
Circadian timing is shaped by a complex interplay of environmental and behavioural time cues (Zeitgebers) such as light, meals, exercise, and social schedules5 modulating tissue-specific oscillators, where light is the dominant entraining signal for the SCN6,7,8. However, sensitivity to light is not uniform across individuals9,10,11,12. Individual differences in photic responsiveness and SCN function give rise to marked variability in circadian phase and in the duration of the biological night relative to the actual day and night. For instance, Phillips et al. showed that melatonin onset can shift by 12 to 137 min among individuals exposed to the same light levels11. Such differences may, in part, explain variability in sleep-wake patterns, often described in terms of chronotype13, and highlight why individuals exposed to the same real-world schedules can experience different degrees of circadian alignment. In applied settings such as elite sport, where athletes are exposed to shared schedules and environments, these individual differences may contribute to varying levels of circadian misalignment.
Subjectively, chronotype can also be represented as the individual’s daily preference for morningness or eveningness or even by assessing one’s activity and sleep timing throughout the week, using the midpoint of sleep as a reference point that can also reflect the individual’s phase of entrainment13,14. Objectively, chronotype can be assessed using physiological markers such as dim-light melatonin onset (DLMO), which marks the start of the biological night4,15,16,17. Although these measures are correlated, they do not always align perfectly. The interval between DLMO and sleep onset typically averages two hours4,15 but shows wide interindividual variability particularly in late chronotypes18, complicating the estimation of biological night and the assessment of circadian alignment in real-life contexts.
These individual differences in circadian timing are particularly relevant in athletes, specifically those performing team sports such as football19, who must synchronize training, recovery, and rest to shared schedules. These collective timetables may not match all individuals’ internal rhythms, leading to circadian misalignment, defined as a mismatch between internal biological timing and externally imposed behavioural or environmental schedules20,21,22. In practical terms, circadian misalignment occurs when individuals engage in daily activities, such as training or competition, at times that fall within their biological night23. In athletes, misalignment, especially wakefulness during the biological night, may impair recovery, general well-being, and ultimately compromise performance.
We previously showed that elite male football players sharing bedroom slept less than those in individual rooms24, but it remained unclear to what extent such differences reflect underlying chronotype variability or circadian phase misalignment. Moreover, our earlier findings focused solely on behavioural sleep duration and did not consider individual differences in biological timing. In the present study, we aimed to determine whether sleep duration and its relationship with room-sharing arrangements are influenced by athletes’ chronotype preferences and internal circadian phase, as indexed by melatonin onset. Understanding this interaction between social context and biological timing could reveal how external and internal factors jointly shape sleep in elite athletes. Elite training camps provide a relatively controlled environment for this purpose: athletes have shared schedules for training, meals, and sleep, which reduce external confounds, while still allowing individual variability in sleep timing to emerge.
Therefore, this study sought to investigate the expression and consequences of circadian misalignment in elite athletes within a real-world yet standardized environment. Specifically, we aimed to determine individual melatonin onset times and assess how they correspond to self-reported daily preferences, and to characterize the biological night and identify instances of circadian misalignment (wakefulness during elevated melatonin levels). We further examine whether such misalignment, beyond subjective chronotype, was associated with reduced morning well-being, and to explore how total sleep time relates to chronotype, circadian alignment, and room-sharing arrangements.
We hypothesized that athletes with an evening daily preference would show a later melatonin onset compared with morning types, reflecting a delayed biological night; that despite standardized schedules, a considerable proportion of athletes would wake during their biological night, indicating circadian misalignment; and that misaligned athletes would report lower morning well-being than aligned athletes.
Results
Study population and setting
This study included 22 elite female football players (mean age = 26.8 ± 4.3 years, Tier 4: Elite/International Level25) from the Portuguese national team during a residential training camp held from 8 to 15 November 2022, in Oeiras, Portugal (latitude ~39°N and longitude ~ -9°W). The photoperiod was 10.22 h (sunrise: 7:13, sunset: 17:26). Although conducted in a real-life setting, the training camp provided a relatively controlled environment: athletes followed standardized daily schedules for training sessions, meals (timing and composition), similar sleep-wake opportunities and were exposed to comparable levels of natural daylight and indoor artificial lighting.
Evening melatonin onset and morning melatonin offset were determined using a standardised salivary melatonin threshold of 3 pg/mL16 to define the biological night. Throughout the study, athletes wore actimetry devices for objective sleep and light exposure data and completed daily sleep diaries reporting bedtimes, wake times, and subjective ratings of morning well-being.
Chronotype daily preference and melatonin onset
Athletes were categorised into four chronotype groups based on their self-reported preference in the item 9 of the Athletes Sleep Screening Questionnaire (ASSQ)26 “Do you consider yourself to be a morning type person or an evening type person?”. Two considered themselves as “definitely morning type”, 9 as “more morning than evening type”, 6 as “more evening than morning type”, and 5 as “definitely evening type”.
A one-way ANOVA revealed no significant effect of chronotype on melatonin onset time. Although groups with stronger evening preference tended to show later onset, this trend was not consistent across categories. The earliest mean onset (20:07 ± 79 min) occurred in the “definitely morning type” group, whereas the latest mean onset (22:26 ± 31 min) was observed in the “more evening than morning type” group. Given the lack of significant differences in other parameters between the groups (e.g., melatonin levels, age) and to enhance the statistical power, we merged the categories into two broader preference groups: morning-type (n = 10) and evening-type (n = 7). In this comparison, evening-types had a significantly later melatonin onset (22:25 ± 19 min) than morning-types (21:08 ± 17 min; t₁₅ = 2.486, p = 0.025, two-tailed, mean difference = 1:17 ± 31 min, 95% CI: 0.18–2.38). The effect size was large (Hedges’ g = 1.16, 95% CI: 0.14–2.15) (see Supplementary Material Fig. S2). Variances were not significantly different between groups (F(9,6) = 2.031, p = 0.400).
Biological night and circadian misalignment
Using established melatonin cutoffs (3 pg/mL)16, biological night onset was determined for 19 athletes (14% lacked sufficient saliva for analyses). Owing to multiple missing immediate wake-up samples (due to insufficient saliva), we elected a priori to use the 1-h post-wake saliva sample for all participants. This decision is supported by data from participants with complete sampling, which show higher melatonin at wake followed by a decline to low daytime concentrations by 1 h post-wake (see Supplementary Material Fig. S3). Eleven athletes (58%) exhibited melatonin concentrations >3 pg/mL one hour after waking, indicating they awoke during their biological night, experiencing circadian misalignment. Melatonin concentrations 1 h after waking differed significantly between biological night and biological day groups (median: 7.41 vs. 2.33 pg/mL; Mann–Whitney U = 0, p < 0.001). The corresponding ranges were 4.64-21.88 pg/mL and 1.06-2.68 pg/mL, respectively, with a Hodges–Lehmann median difference of 5.54 pg/mL. This misalignment was not exclusive to evening preference-chronotypes: 3 morning-type athletes also awoke during elevated melatonin levels (Fig. 1), reinforcing the importance of objective measures over self-reported preference. One outlier showed an unusually early melatonin onset (19:47) but a delayed sleep onset (02:28). This individual self-identified as an evening type but was excluded from all analyses due to extreme circadian misalignment.
Frequency of athletes (n = 19) waking in the biological day or in the biological night according to each chronotype preference.
Phase relationship between melatonin onset and sleep timing
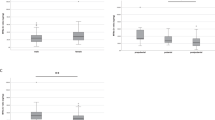
Morning- and evening-chronotype preference athletes showed a similar difference between melatonin onset and the sleep onset (Fig. 2A).
A Phase difference between melatonin onset time and sleep onset: morning (white circles) vs. evening (dark circles); B Phase difference between melatonin onset time and wake-up time: morning (white circles) vs. evening (dark circles); Each circle represents an individual, and the line in the box indicates the mean value and whiskers represent the standard deviation. The dashed line in (A) represents sleep onset time, while in (B) represents wake-up time. Statistical analyses were conducted using unpaired t-test. ns not significant; **p < 0.01.
When examining the phase relationship between melatonin onset and wake-up time, we found differences among the chronotype-preference groups. Evening types exhibited a shorter interval (-9.12 h ± 1.30 h) compared with morning types (-11.00 h ± 1.20 h; unpaired t-test, p = 0.008, η² = 0.387, Hedges’g = 1.44, 95% CI: 0.37-2.47; Fig. 2B). Average melatonin production profiles by chronotype group (A) and by biological day versus biological night (B) are shown in Fig. S3.
Total Sleep Time, chronotype and room-sharing
We aimed to examine the influence of chronotype daily preference, melatonin onset timing and room-sharing on athletes’ total sleep time (TST). Across the entire training camp, actigraphy-derived TST averaged 7.05 ± 1.28 h. When comparing chronotype groups, there were no significant differences in TST. However, athletes sharing rooms with mixed chronotypes had significantly longer TST compared with those sharing rooms with teammates of the same chronotype (mixed chronotypes: 7.37 h vs. same chronotype: 5.93 h; p = 0.012). No significant differences were found in melatonin onset timing (mixed chronotypes: 21:29 ± 1:17 vs. same chronotype: 22:12 ± 0:44; p = 0.318) or in the phase angle between melatonin onset and sleep onset times (mixed chronotypes: -2.44 ± 1.61 h vs. same chronotype: 3.00 h ± 1.07 h; p = 0.533) between room-sharing groups.
A multiple linear regression model including age, phase angle (melatonin onset-sleep onset), chronotype daily preference and room-sharing significantly predicted total sleep time (TST) (R² = 0.69, F(4,12) = 6.68, p = 0.004). The phase angle between melatonin onset and sleep onset was a significant positive predictor of TST (β = 0.40, 95% CI [0.09, 0.72], p = 0.015), indicating that athletes with a larger melatonin-to-sleep interval slept longer. Room-sharing was also a significant predictor (β = -1.38, 95% CI [-2.58, -0.18], p = 0.027), with athletes sharing rooms only with teammates of the same chronotype sleeping approximately 1.38 hours less than those in mixed-chronotype rooms. In addition, chronotype preference showed a significant effect (β = -1.06, 95% CI [-2.08, -0.05], p = 0.043), with evening-type athletes sleeping about 1.06 h less than morning types. Age was not a significant predictor of TST (β = -0.03, p = 0.822). Multicollinearity diagnostics were conducted prior to model estimation and revealed high collinearity between melatonin onset time and phase angle (melatonin onset-sleep onset). To avoid multicollinearity, melatonin onset time was excluded from the regression model before estimating coefficients. In the final model, no concerning multicollinearity was observed among the remaining predictors (all VIFs < 1.6).
Predictors of Melatonin Onset Time and Post-Wake Melatonin Levels
Light exposure is a key regulator of circadian rhythms and melatonin production. To identify potential predictors of melatonin onset timing and levels one hour after waking-up, we performed two multiple regression models using actigraphy-derived light exposure data, chronotype preference, age, and room-sharing as predictors. Average light exposure from two hours before the first saliva collection until sleep onset time (median [Q1, Q3]: 21.50 lux [13.39, 28.46]) used in model (1) and for model (2) from wake-up time until last collection (one hour after waking up) (median [Q1, Q3]: 41.05 lux [5.38, 71.68]). Model (1) for melatonin onset time the overall model was significant (R² = 0.64, F(4,11) = 4.79, p = 0.018). Among the predictors, chronotype preference significantly predicted melatonin onset time (β = 1.36, 95% CI [0.28, 2.44], p = 0.018), with evening types showing a later melatonin onset. Neither age (p = 0.419), pre-sleep light exposure (p = 0.064), nor room-sharing with mixed chronotypes (p = 0.763) were significant predictors.
Model (2), for melatonin concentration one hour after awakening, the model showed a trend toward significance (R² = 0.46, F(4,12) = 2.56, p = 0.093). None of the predictors reached the conventional significance threshold, although chronotype preference again showed a near-significant effect (β = 5.13, 95% CI [–0.35, 10.61], p = 0.064), with evening types presenting higher melatonin levels. Age (p = 0.930), post-wake light exposure (p = 0.164), and room-sharing type (p = 0.705) were not significant predictors.
Well-being and circadian alignment
To evaluate the impact of circadian misalignment on daytime functioning, we examined associations between melatonin-based indicators of misalignment and subjective well-being. Misaligned athletes reported lower morning well-being score (Mann-Whitney U = 17.5, p = 0.024, two-tailed; median [IQR]: 7.0, n = 11 vs. 8.5, n = 8; Hodges-Lehmann median difference = 2.0). In contrast, well-being did not differ significantly between morning and evening chronotypes (Mann–Whitney U = 44.5, p = 0.471; median: 8.0 vs. 7.5) (Fig. 3). This supports growing evidence that objective measures of circadian misalignment, rather than chronotype preference, are the primary determinant of well-being.
A Well-being scores by chronotype preference: morning (white circles) vs. evening (dark circles); B Well-being scores by wake-up timing relative to biological day or night: biological day (white triangles) vs. biological night (dark triangles). Waking up during the biological night was classified for individuals with melatonin > 3 pg/ml one hour after waking up. Each dot represents an individual, and the line in the box median and the whiskers represent the first (Q1) and third (Q3) quartiles. Statistical analyses were conducted using the Mann-Whitney test. ns not significant; a.u., arbitrary units; *p < 0.05.
Discussion
In this real-world but standardized environment, more than half of the athletes woke during their biological night, indicating substantial morning circadian misalignment despite having identical schedules and controlled conditions. Although melatonin onset did not differ significantly when daily preference was analysed across four chronotype categories, collapsing the categories into morning versus evening types revealed a significant difference, likely reflecting increased statistical power and reduced within-group variability. Melatonin onset was strongly associated with chronotype preference, yet subjective chronotype alone did not predict alignment. Importantly, misaligned athletes reported poorer morning well-being, suggesting functional consequences of biological-behavioural mismatch. In addition, room-sharing arrangements were found to influence total sleep time: athletes sharing rooms with mixed chronotypes slept significantly longer than those with teammates of the same chronotype. This highlights that social and environmental context may further modulate sleep together with endogenous circadian phase. We have previously reported in male athletes that room sharing was associated with shorter sleep duration in comparison with individual rooms24. Moreover, poor sleep has been shown to reduce performance and increases the risk of injuries in football players27. In the present study, room-sharing configuration and chronotype preference independently contributed to total sleep time, with evening-type athletes sleeping less overall and athletes in mixed-chronotype rooms obtaining longer sleep duration. While these findings suggest that mixed chronotype room-sharing may be more favourable for sleep than same-chronotype arrangements, the present analyses do not allow determination of whether these effects differ by chronotype or specific room-pairing combinations. This finding is consistent with evidence from the UK Biobank showing that sleep duration and timing are interrelated between cohabiting partner’s28. In our study, total sleep duration was predicted by a combination of social and endogenous factors, underscoring the importance of considering both when assigning room arrangements for athletes. Not notably, rooms shared by two evening chronotypes had the lowest average total sleep time. The phase angle between melatonin onset and sleep onset were on average -2.56 h, ranging from -5.08 h until -0.08 h, in line with what reported in the literature4. With evening chronotypes these phase angles can even be larger going from 5.5 h before sleep onset, until 1.2 h after sleep onset time6,18. We identified one athlete with an extreme phase angle of 6.68 h between melatonin onset and sleep onset. This value was considered an outlier and was excluded from the group-level analyses due to the degree of circadian misalignment. However, it is important to acknowledge that for individualized circadian interventions such extreme values are highly relevant, as any intervention would need to be tailored to the individual’s specific physiological timing.
The fact that 58% of athletes woke during elevated melatonin levels demonstrates that considerable circadian variability persists despite the highly structured daily routines and the outdoor nature of the sport, which involves substantial daytime light exposure. According to circadian entrainment principles6,29, such environmental conditions would be expected to favour a predominantly morning-type chronotype profile. Previous work by Cox et al. showed that 74.4% of individuals wake with melatonin levels above 3 pg/mL indicated that they are still in their biological night30. In our dataset, we relied on melatonin levels one hour after waking to assess circadian misalignment due to a high number of insufficient wake-time saliva samples. Consequently, the actual percentage of athletes waking while still biologically misaligned was likely underestimated. This is especially plausible given that participants in both studies had the same average age (~26 years) and similar average melatonin onset times (21:40 vs. 21:14 h), with our sample showing the later value. Furthermore, despite being located within the Western European time zone, contrary to the Central European time zone like Spain, Portugal shows a well-documented tendency toward later sleep-wake behaviour compared with other countries14,31, which may further contribute to this pattern of circadian lateness.
We acknowledge that the use of a fixed 3 pg/mL threshold to define biological night represents one of several possible methodological approaches, and that alternative criteria, such as individualized thresholds or a more conservative cut-off (e.g., 4 pg/mL), could also be applied. However, in the present dataset, group classification was robust to such variation. Athletes categorized as waking during their biological night exhibited melatonin concentrations ranging from 4.64–21.88 pg/mL, whereas those classified as waking during the biological day ranged from 1.06-2.68 pg/mL. Consequently, participant classification—and the resulting interpretations—would have remained unchanged regardless of whether a 3 pg/mL or a 4 pg/mL threshold had been used. Chronotype remained a consistent predictor of melatonin onset, with evening types showing later melatonin onset time. Light exposure, both pre-sleep and post-wake, did not reach significance in our regression models, suggesting that in this cohort and setting, individual biological timing differences outweighed immediate environmental effects. Room-sharing with mixed chronotypes did not significantly influence melatonin timing or morning melatonin levels, indicating that the effects of social environment may primarily affect sleep duration rather than circadian phase. Similarly, melatonin concentration one hour after awakening showed a near-significant trend with chronotype. Given the lack of statistical significance, these results should be interpreted cautiously and explored in future studies.
The association between morning misalignment and lower well-being supports the idea that circadian phase—rather than chronotype label—better captures physiological readiness. Circadian misalignment is well established as a risk factor for both physical and psychiatric disorders32. More specifically, morning circadian misalignment has been shown to reduce insulin sensitivity in healthy individuals subjected to restricted sleep33. In our study, athletes experiencing morning misalignment reported lower well-being, reinforcing the importance of aligning daily routines with endogenous biological rhythms to support optimal health and functioning. This has clear implications for elite athletes, where even subtle reductions in daily well-being can impair performance, recovery, and decision-making, highlighting the importance of minimizing circadian misalignment in high-performance environments to protect both physical and mental readiness34. Wherever possible, training and competition schedules should therefore aim to consider individual circadian profiles to promote better alignment between biological rhythms and daily demands. We recognize, however, that full circadian alignment may not always be feasible in elite-sport contexts due to fixed team schedules, logistical constraints, and the need for standardized routines. In situations where schedule individualization cannot be implemented, alternative strategies such as optimized light exposure, strategic timing of training sessions, individualized sleep education, and tailored recovery planning should be considered to mitigate the adverse effects of unavoidable circadian misalignment.
This study has some limitations, the most significant being the sample composition, which included only female participants. This limits the generalization of the findings to male athletes. Moreover, although menstrual cycle phase can influence sleep and circadian regulation, this information was not collected and may have contributed to interindividual variability. Future studies should incorporate both male and female participants and consider menstrual cycle phase to determine whether the results observed here can be replicated in a broader population. Additionally, the sample size is relatively small. However, the study was conducted in real-life setting within a training camp, where environmental conditions such as accommodation, training schedules and meals were standardized. This provided a relatively controlled environment, reducing external variability. We assessed melatonin as our primary circadian marker, however, incorporating additional physiological measures (e.g., core body temperature or cortisol rhythms) would provide a more comprehensive view of circadian alignment. A relevant limitation is that morning circadian misalignment was estimated primarily from a single post-wake melatonin sample. Although the fixed-threshold method has been recommended for field studies with restricted sampling opportunities16, it provides less precision than a full melatonin onset assessment and may underestimate the degree of misalignment in some individuals. The gold standard for assessing melatonin rhythms is measurement under controlled dim-light conditions16,35. However, to characterize the output of the SCN in relation to real-world sleep-wake behaviour and to quantify circadian misalignment, it is also important to obtain measurements that capture environmental influences—an approach central to the present study. For this reason, throughout the manuscript we refer to “melatonin onset” rather than “dim-light melatonin onset,” as participants were not studied in a strictly controlled dim-light environment. The final morning sample was collected one hour after awakening; however, several participants still had melatonin levels above the threshold at that time. Because no additional samples were obtained, we could not determine their exact melatonin-off values. Furthermore, several wake samples were missing due to insufficient saliva volume. We recommend that future studies account for these issues by collecting additional morning samples to allow for accurate determination of melatonin-off and, consequently, the duration of melatonin production (biological night). The current study additionally reveals that room-sharing among mismatched chronotypes may influence total sleep time, emphasizing the need to consider social environment in future investigations. Future studies should recruit larger, mixed-sex cohorts, control for hormonal phase, and incorporate multi-parameter circadian measures to fully capture the interaction between biological, behavioural, and social factors.
Even within a structured environment, many elite athletes woke during their biological night, demonstrating that standardized schedules do not ensure circadian alignment. Objective biological measures, rather than subjective chronotype, most accurately captured misalignment and its association with reduced morning well-being. Moreover, room-sharing arrangements may play a role in modulating sleep duration, highlighting the relevance of social and environmental factors. Integrating circadian assessment and consideration of social sleep environments into athlete monitoring could enhance recovery and health through personalized timing strategies.
Methods
Participants
Before starting the training camp, athletes completed a screening questionnaire to assess eligibility, followed by an interview conducted by the clinical team and a certified sleep medicine researcher. The screening included the Athlete Sleep Screening Questionnaire (ASSQ)26. Participants with a sleep difficulty score greater than 12 were excluded. Chronotype preference was assessed using item 9 of the questionnaire, which asks: “Do you consider yourself to be a morning type person or an evening type person?”. Note that the ASSQ score reflects sleep difficulty (higher scores indicate greater sleep difficulties) and is not related to chronotype; chronotype was assessed using this single self-report item. The Munich Chronotype Questionnaire (MCTQ)14 was used to assess habitual sleep-wake behaviour, which was fundamental for establishing the saliva collections protocol, as recommended16. Since restless leg syndrome is relatively prevalent in the general population, especially among women, we also included a single-item screening tool to exclude participants with restless legs syndrome36. As a result of the screening, one athlete was excluded for meeting the criteria for insomnia, another was excluded due to recent time zone crossing (3 h), and a third outlier for melatonin onset time was excluded due to extreme circadian misalignment (melatonin onset time at 19:47 h, but delayed sleep onset (02:28 h).
During the one-week training camp, all participants underwent actigraphy (for objective sleep measures and light), complemented by a diary. During the training camp, athletes followed a standardized daily schedule designed and implemented by the team staff. Meals and activities occurred at fixed times for all participants as follows: breakfast until 09:15, team meeting at 10:00, morning training session at 10:30, lunch at 12:30, afternoon snack at 16:00, team meeting at 17:00, evening training session at 17:30, dinner at 20:00, and designated quiet time at 23:00. A detailed overview of room assignments by chronotype and athlete distribution is presented in Supplementary Table S1.
The study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of the Portugal Football School (CE PFS 6/2021). All participants provided written informed consent prior to participation.
Activity, Sleep, and Light Exposure Assessment
Data were continuously collected at 1-min intervals across multiple days using the CondorTM Actrust 2 actigraphy devices (Condor, Brazil). The devices were removed during collective training sessions due to legal and safety constraints. The devices recorded activity (PIM) and light total light (lux). The operational range for light measurement was 0.01—8388.61μW/cm2 with a precision of 10% at 10 μW/cm2. Sleep periods were calculated using an adaptation of the Cole-Kripke algorithm37 via ActStudio software. Missing data were excluded with the manufacturer’s software and verified through visual inspection. Calculated sleep and wake-up times were also cross-checked with the diary reports.
For light quantification, we use the average total light (in lux) retrieved from the actigraphy device from the days of saliva collections and the values used for each specific model were as follows: model (1) from two hours before the first saliva collection until sleep onset time (median [Q1, Q3]: 21.50 lux [13.39, 28.46]) and for model (2) from wake up time until last collection (one hour after waking up) (median [Q1, Q3]: 41.05 lux [5.38, 71.68]) (see Fig. S1 for detailed scheme). Sleep timings (sleep onset and wake time) used for light exposure calculations were derived from actigraphy data from saliva collections day and not the average days. Average daytime light exposure could not be calculated because the athletes were required to remove and/or cover the actimeter during the training sessions. The average sleep duration was retrieved from all days of actigraphy across the training camp.
Diary
Across all days, athletes were asked to report their bedtime, sleep time, time taken to fall asleep, wake-up time, and get-up time. Additionally, each morning, participants reported their sleep quality and well-being on a scale from 0 (poor) to 10 (excellent), the values reported in the manuscript report to the day of saliva collections (during the morning saliva collection). Mealtimes, meal content and training schedules were standardized for all participants.
Saliva collections
All athletes performed the saliva collections on the same day, at the beginning of the training camp, between the second and third day. Participants were instructed to dim the lights in their rooms, and light exposure was objectively measured using an actimetry device. During saliva collection days, participants were instructed to refrain from using toothpaste and from consuming coffee, tea, alcohol, energy drinks, chocolate, and bananas. Food intake was not permitted within 30 minutes prior to any saliva sample. Accordingly, after waking, participants were instructed to brush their teeth at least 30 minutes before the 1 h post-waking saliva sample and to postpone breakfast until after this sample was collected. Athletes were asked to refrain from exercise and to restrict their activity level during the sampling period. However, posture and light exposure after waking were not otherwise standardized, as participants were allowed to engage in their usual morning activities. Light exposure was subsequently included as a covariate in the statistical models. Saliva samples were collected at time points relative to each participant’s reported average habitual sleep time (via MCTQ): 4, 3, 2, and 1 h before sleep time; habitual sleep time; 1 h after sleep time; at wake-up time; and 1 h after waking up (Fig. S1). Participants were instructed to remain awake until the collection of the final saliva sample of the evening (1 h after their habitual sleep time). In total, eight saliva samples were obtained per athlete. Participants were requested to mark an event in the actimetry device for each saliva collection to increase saliva timing accuracy for determining melatonin onset timing. Saliva samples were collected with Salivette tubes (Sarstedt Ag & Co, Germany) and analysed for melatonin levels. Salivary melatonin was measured using the Melatonin Direct Saliva RIA (RE29301, IBL International). The analytical sensitivity (limit of detection) of the assay was 0.3 pg/mL and a measuring range of 0.3–300 pg/mL. Melatonin onset was estimated using a linear interpolated fixed 3 pg/ml threshold.
Data analysis
The normality of data distribution was assessed using the Shapiro-Wilk test, due to the small sample size38. Bivariate group comparisons were performed using either independent-samples t-test or Mann-Witney U test, depending on data distribution. For normally distributed data, results are reported as mean ± standard deviation, whereas for non-normally distributed data, results are reported as the median and interquartile range (Q1–Q3), along with the Hodges-Lehmann estimate of the median difference. Effect sizes are reported for all group comparisons. Standardized means differences were quantified using Hedges’ g, which corrects for sample size bias, with values of 0.2, 0.5, and 0.8 interpreted as small, medium, and large effects, respectively. To compare melatonin onset across the four chronotype groups, we performed a one-way ANOVA and report the test values along with η² for the variance explained (small effect: 0.01, medium effect: 0.06, large effect: 0.14)39.
To examine the effect of light exposure, mixed-chronotype rooms and chronotype daily preference (adjusted for age) on melatonin onset time (beginning of the biological night), melatonin levels one hour after wake-up and total sleep time, we used multivariable regression models. We performed Linear Regression Models with bootstrapping (1000 resamples), including age, light levels, mixed-chronotype rooms and chronotype daily preference as independent variables. All analyses were performed using SPSS version 29 and GraphPad Prism version 10, and graphs were created with GraphPad Prism version 10. The significance level for all analyses was set at p < 0.05.
Data availability
The datasets generated and/or analysed during the current study are not publicly available due to privacy and confidentiality constraints but are available from the corresponding author on reasonable request.
References
Aschoff, J. & Pohl, H. Phase relations between a circadian rhythm and its zeitgeber within the range of entrainment. Naturwissenschaften 65, 80–84 (1978).
Lewy, A. J., Wehr, T. A., Goodwin, F. K., Newsome, D. A. & Markey, S. P. Light suppresses melatonin secretion in humans. Science 210, 1267–1269 (1980).
Saper, C. B., Scammell, T. E. & Lu, J. Hypothalamic regulation of sleep and circadian rhythms. Nature 437, 1257–1263 (2005).
Kennaway, D. J. The Dim Light Melatonin Onset (DLMO) across ages, methodologies and sex and its relationship with morningness: eveningness. Sleep https://doi.org/10.1093/sleep/zsad033 (2023).
Aschoff, J. et al. Human circadian rhythms in continuous darkness: entrainment by social cues. Science (1979) 171, 213–215 (1971).
Duffy, J. F. & Wright, K. P. Entrainment of the human circadian system by light. J. Biol. Rhythms 20, 326–338 (2005).
Czeisler, C. A., Richardson, G. R., Zimmerman, R. C., Moore-Ede, M. C. & Weitzman, E. D. Entrainment of human circadian rhythms by light-dark cycles: a reassessment. Photochem Photobio. 43, 239–247 (1981).
Wright, K. P. et al. Entrainment of the human circadian clock to the natural light-dark cycle. Curr. Biol. 23, 1554–1558 (2013).
Abbott, S. M., Choi, J., Wilson, J. & Zee, P. C. Melanopsin-dependent phototransduction is impaired in delayed sleep–wake phase disorder and sighted non–24-hour sleep–wake rhythm disorder. Sleep https://doi.org/10.1093/sleep/zsaa184 (2020).
Stone, J. E. et al. The role of light sensitivity and intrinsic circadian period in predicting individual circadian timing. J. Biol. Rhythms 1–13. https://doi.org/10.1177/0748730420962598 (2020).
Phillips, A. J. K. et al. High sensitivity and interindividual variability in the response of the human circadian system to evening light. Proc. Natl. Acad. Sci. USA 116, 12019–12024 (2019).
Watson, L. et al. Increased sensitivity of the circadian system to light in delayed sleep-wake phase disorder. J. Physiol. 596, 6249–6261 (2018).
Roenneberg, T., Pilz, L. K., Zerbini, G. & Winnebeck, E. C. Chronotype and social jetlag: a (self-) critical review. Biology (Basel) 8 (2019).
Reis, C., Madeira, S. G., Lopes, L. V., Paiva, T. & Roenneberg, T. Validation of the portuguese variant of the munich chronotype questionnaire (MCTQPT). Front Physiol. 11 (2020).
Burgess, H. J. & Eastman, C. I. The dim light melatonin onset following fixed and free sleep schedules. J. Sleep. Res. 14, 229–237 (2005).
Murray, J. M. et al. A protocol to determine circadian phase by at-home salivary dim light melatonin onset assessment. J. Pineal Res. 76 (2024).
Keijzer, H. et al. Evaluation of salivary melatonin measurements for Dim Light Melatonin Onset calculations in patients with possible sleep-wake rhythm disorders. Clin. Chim. Acta 412, 1616–1620 (2011).
Reis, C., Pilz, L. K., Paiva, T., Hidalgo, M. P. & Wright, K. P. Sleep and circadian rhythms in delayed sleep–wake phase disorder: phenotypic differences between patients with and without comorbid depression. J. Sleep Res. https://doi.org/10.1111/jsr.14437 (2025).
Facer-Childs, E. & Brandstaetter, R. Circadian phenotype composition is a major predictor of diurnal physical performance in teams. Front Neurol. 6 (2015).
Klerman, E. B. et al. Keeping an eye on circadian time in clinical research and medicine. Clin. Transl. Med. 12, (2022).
Roenneberg, T. Having trouble typing? What on earth is chronotype?. J. Biol. Rhythms 30, 487–491 (2015).
Roenneberg, T., Wirz-Justice, A. & Merrow, M. Life between clocks: daily temporal patterns of human chronotypes. J. Biol. Rhythms 18, 80–90 (2003).
Hashemipour, S., Yazdi, Z. & Mahabad, N. Association of evening chronotype with poor control of type 2 diabetes: roles of sleep duration and insomnia Level. Int. J. Endocrinol. Metab. 18 (2020).
Costa, J. A. et al. Comparing Sleep in Shared and Individual Rooms During Training Camps in Elite Youth Soccer Players: A Short Report. J. Athl. Train. 58, 79–83 (2023).
McKay, A. K. A. et al. Defining Training and Performance Caliber: A Participant Classification Framework. Int J. Sports Physiol. Perform. 17, 317–331 (2022).
Bender, A. M., Lawson, D., Werthner, P. & Samuels, C. H. The clinical validation of the athlete sleep screening questionnaire: an instrument to identify athletes that need further sleep assessment. Sports Med Open 4, 23 (2018).
Clemente, F. M. et al. Relationships between sleep, athletic and match performance, training load, and injuries: a systematic review of soccer players. Healthcare 9, 808 (2021).
Richmond, R. C. et al. Correlations in sleeping patterns and circadian preference between spouses. Commun. Biol. 6, 1156 (2023).
Roenneberg, T., Daan, S. & Merrow, M. The Art of Entrainment. J. Biol. Rhythms 18, (2003).
Cox, R. C. et al. Distribution of dim light melatonin offset (DLMOff) and phase relationship to waketime in healthy adults and associations with chronotype. Sleep. Health 10, S76–S83 (2024).
Korman, M. et al. COVID-19-mandated social restrictions unveil the impact of social time pressure on sleep and body clock. Sci. Rep. 10, 22225 (2020).
Baron, K. G. & Reid, K. J. Circadian misalignment and health. Int. Rev. Psychiatry 26 (2014).
Eckel, R. H. et al. Morning circadian misalignment during short sleep duration impacts insulin sensitivity. Curr. Biol. 25 (2015).
Charest, J. & Grandner, M. A. Sleep and athletic performance. Sleep. Med. Clin. 15, 41–57 (2020).
Brown, T. M. et al. Recommendations for daytime, evening, and nighttime indoor light exposure to best support physiology, sleep, and wakefulness in healthy adults. PLoS Biol. 20, e3001571 (2022).
Möller, J. C. & Oertel, W. H. Single-question screen for restless legs syndrome. Nat. Clin. Pr. Neurol. 4, 132–133 (2008).
Cole, R. J., Kripke, D. F., Gruen, W., Mullaney, D. J. & Gillin, J. C. Automatic sleep/wake identification from wrist activity. Sleep 15, 461–469 (1992).
Ghasemi, A. & Zahediasl, S. Normality tests for statistical analysis: a guide for non-statisticians. Int J. Endocrinol. Metab. 10, 486–489 (2012).
Richardson, J. T. E. Eta squared and partial eta squared as measures of effect size in educational research. Educ. Res Rev. 6, 135–147 (2011).
Acknowledgements
The authors would like to acknowledge the Portuguese Football Federation for funding the study, and we thank Professor Kenneth P. Wright Jr. and Dr. William J. Schwartz for fruitful discussions and valuable comments into the manuscript.
Author information
Authors and Affiliations
Contributions
C.R.: Conceptualization, study design, data collection, statistical analyses, manuscript writing (original draft), manuscript review and editing. R.T.: Data collection, participant coordination, manuscript review and editing. V.C.: Data analyses, visualization, manuscript review and editing. J.A.C.: Data collection, project administration, manuscript review, and editing. J.B.: Conceptualization, funding acquisition, study supervision, manuscript review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Reis, C., Tomás, R., Cardoso, V. et al. Field assessment of melatonin timing reveals circadian misalignment beyond chronotype in elite female football players. npj Biol Timing Sleep 3, 13 (2026). https://doi.org/10.1038/s44323-026-00074-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44323-026-00074-4