Abstract
Inefficient drug delivery in tumors, especially in desmoplastic cancers, arises from blood vessel collapse due to tumor stiffening and mechanical compression. Vessel collapse also leads to hypoxia, immune evasion, and metastasis, reducing treatment efficacy. Mechanotherapeutics and ultrasound sonopermeation, which address tumor stiffness and enhance vessel permeability, respectively, show promise in restoring tumor microenvironment abnormalities and improving drug delivery. This perspective highlights their independent and combined potential to optimize cancer therapy.
Similar content being viewed by others
Introduction
Cancer is responsible for approximately one in every six deaths globally, highlighting its significant impact on public health. Cancer development is a lengthy process, characterized by the gradual accumulation of various symptoms and a significant failure in the mechanisms that typically inhibit abnormal cell growth1. For many years, patients diagnosed with cancer had limited treatment options, which primarily include surgery, radiation therapy, and chemotherapy. These treatments could be used individually or in combination, but they often come with considerable side effects and varying degrees of effectiveness, leaving many patients seeking alternative approaches or additional support in their battle against this complex disease. While traditional treatments can effectively reduce tumor size or achieve temporary remission, they frequently fall short of providing a long-term cure, particularly in aggressive or advanced cancers. The failure of standard therapies to cure different types of cancer is rooted in a variety of complex biological and physiological challenges. In fibrotic tumors, this is attributed in large part to insufficient and heterogeneous drug delivery to the tumor site owing to physical abnormalities in the tumor microenvironment (TME) that induce hypo-perfusion, which in turn creates harsh hypoxic conditions2,3,4,5.
Hypo-perfusion hinders the delivery of medicines and induces immunosuppression
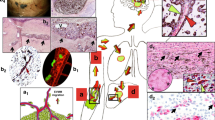
Although standard-of-care treatments can be highly potent and capable of eradicating cancer cells in controlled in vitro settings, their therapeutic outcomes are largely compromised in real-world applications due to inefficient drug delivery. Inadequate delivery prevents therapeutics from reaching the tumor mass in sufficient amounts thus, diminishing their effectiveness and limiting their ability to significantly improve treatment outcomes6. In cancers with a highly fibrotic nature, complex interactions between cancer cells, stromal cells, and the dense extracellular matrix (ECM)-characterized by the excessive deposition of components like collagen and hyaluronan- lead to significant tumor stiffening. This stiffening is accompanied by the buildup of mechanical forces within the tumor, which are exerted on intratumoral blood vessels. As a result, tumor blood vessels are compressed (Fig. 1), disrupting normal blood flow and contributing to the challenging treatment environment typical of these tumors7,8,9,10,11,12,13,14,15,16. In cancer types, such as pancreatic and breast tumors and sarcomas, it has been observed that a significant portion of intratumoral blood vessels—up to 95%—may become compressed, with as much as 80% collapsed entirely8,9,17,18,19,20. This widespread vessel collapse leads to severely reduced blood flow within the tumor, a condition known as hypo-perfusion. Hypo-perfusion, in turn, poses a major barrier to effective drug delivery, preventing therapeutic agents from reaching the tumor in adequate amounts. Additionally, this lack of proper blood flow creates a hypoxic environment within the tumor, further hindering the effectiveness of various cancer therapies by inducing the expression of genes associated with drug resistance. The lack of oxygen reduces the effectiveness of radiation therapy, which relies on oxygen to generate reactive oxygen species that damage DNA in cancer cells21,22. Furthermore, hypoxia enables cancer cells to evade the immune system and enhances their invasive and metastatic potential through processes such as epithelial–mesenchymal transition (EMT) and upregulation of genes involved in extracellular matrix modulation in a hypoxia-inducible factor 1-alpha (HIF1α)- dependent manner18,23,24,25,26,27. Hypo-perfusion can also limit immune cell infiltration into the tumor, while hypoxia creates an immunosuppressive TME, shifting tumor-associated macrophages (TAMs) from the immunosupportive M1 type to the immunosuppressive M2 type, and diminishing the cytotoxic capacity of effector immune cells13,28,29,30,31,32. Moreover, hypoxia fosters pro-tumor immune responses by increasing PD-L1 expression in myeloid-derived suppressor cells (MDSCs), TAMs, dendritic cells (DCs), and cancer cells33,34. It also attracts immunosuppressive T-regulator cells (Tregs) by triggering the expression of the chemokine CCL2835 and along with acidity36, affects differentiation and function of T-lymphocytes and myeloid cells37 and induces TAMs to promote angiogenesis38. In response to hypoxia, tumors often upregulate HIFs, which promote the expression of pro-angiogenic factors like vascular endothelial growth factor (VEGF) which leads to the formation of new blood vessels to supply the tumor with necessary nutrients and oxygen39. However, these newly formed vessels are frequently abnormal -leaky and poorly organized- resulting in uneven oxygen distribution and perpetuating hypoxic regions within the tumor40,41. This abnormal vasculature not only supports tumor growth but also contributes to increased tumor malignancy39. Indeed, higher perfusion levels, which indicate normoxia, serve as a predictive biomarker for therapy efficacy13,20,42,43,44.
The abnormal growth of cancer cells in normal host tissue, combined with interactions among stromal cells and the fibrotic ECM, creates mechanical forces that compress tumor blood vessels and stiffen the ECM, reducing tumor perfusion. This impaired blood supply limits immune cell infiltration, while hypoperfusion makes the TME immunosuppressive and supports pro-tumor immune responses. The use of mechanotherapeutics can normalize tissue stiffness, enhance perfusion, and boost immunostimulation, improving cytotoxic therapy effectiveness. However, mechanotherapeutics cannot decompress the majority of the vessels but only a percent. Ultrasound sonopermeation with microbubbles can enhance cell membrane permeability and decrease solid forces, increasing tumor perfusion and the uptake of therapies. However, sonopermeation cannot be effective in tumors with compressed vessels. Combining mechanotherapeutics with sonopermeation overcomes their limitations, effectively addressing tumor microenvironment abnormalities, enhancing drug delivery, and improving therapeutic outcomes. This approach increases tumor perfusion and oxygenation, activating effector immune cells while reducing immune regulator cells, resulting in greater drug delivery efficiency and more effective cancer cell elimination. Created with BioRender.com.
Strategies to modulate the tumor physical microenvironment
Mechanotherapeutics can improve perfusion, but not optimally
As we mentioned previously, vessel compression results from tumor stiffening and the buildup of mechanical forces within solid tumor components, which are modulated by cancer cells, cancer-associated fibroblasts (CAFs), collagen, and hyaluronan8,27,45. A therapeutic strategy to decompress vessels and improve perfusion is the use of mechanotherapeutics to alleviate stiffness and mechanical forces (Fig. 1)46. Mechanotherapeutics are often approved drugs (e.g., antihypertensive, anti-fibrotic, antihistamine) that are repurposed to modulate the TME by targeting ECM components or reprogramming CAFs, such as Transforming growth factor beta (TGFβ) inhibitors18,20,47 (Table 1). Modulation of the TME by selective targeting any or all of these components can re-open compressed vessels and improve perfusion and thus, delivery of drugs and oxygenation18,48,49. While short-term improvements in perfusion and drug delivery might increase treatment efficacy, the longer-term effects of modulation can create a more favorable environment for cancer cells to spread and metastasize50,51. However, treatment with mechanotherapeutics is short-lived as two weeks post-treatment the tumor elasticity returns to the untreated tumors level52. Additionally, treatment with mechanotherapeutics did not favor metastasis in preclinical tumor models20,53,54.
One mechanotherapeutic agent is the anti-hypertensive and angiotensin receptor blocker, losartan, which was successfully repurposed to improve the delivery of therapeutics in vivo by inducing blood vessel decompression and improving perfusion in various types of breast and pancreatic tumors in mice47,55,56. Importantly, losartan was the first mechanotherapeutic to successfully progress to clinical trials. It was found that the addition of losartan to the treatment regimen of FOLFIRINOX and radiation made 60% of previously unresectable, locally advanced pancreatic ductal adenocarcinoma tumors resectable57. Currently, losartan is under investigation in combination with chemoradiation and the immune checkpoint inhibitor (ICI) nivolumab for the treatment of pancreatic cancer (Clinical-Trials.gov identifier: NCT03563248). Losartan is a remarkable step forward and has established the term “mechanotherapeutics” as a new therapeutic strategy owing to first successes in clinical trials46,57. For this reason, it is considered a “gold standard” for mechanotherapeutics. However, in a clinical trial, the losartan dose could not be raised to decompress more vessels because of losartan’s strong anti-hypertensive effects, and normotensive and hypotensive patients could not tolerate losartan and were excluded from the study.
Another such agent is ketotifen, an anti-histamine drug that inhibits mast cell activation, suppresses CAF proliferation and ECM components, whereas none of the doses of ketotifen induced any toxicity in preclinical model58. Notably, a phase II clinical trial is underway to evaluate ketotifen’s potential in enhancing chemotherapy for sarcoma patients (EudraCT Number: 2022-002311-39). In this trial, the maximum tolerated dose (MTD) was administered to the patients, and many of them received ketotifen for a period longer than 100 days without any safety issues to have been reported. Additionally, in our previous studies, we successfully repurposed tranilast, an anti-fibrotic and antihistamine drug approved in Japan and South Korea, and pirfenidone, a globally approved anti-fibrotic drug for the treatment of idiopathic pulmonary fibrosis20,54. These therapeutic agents have demonstrated the ability to decrease the elastic modulus of tumors without affecting the elasticity of host organs and reduce both solid and fluid pressures within tumors, leading to improved tumor perfusion and a marked increase in the effectiveness of various cancer treatments. Specifically, they significantly enhanced the efficacy of chemotherapy, nanotherapy, and immunotherapy, not only in primary tumors but also in metastatic sites. The mechanism behind these improvements involved the suppression of TGFβ signaling, which plays a key role in tumor progression, as well as the downregulation of ECM components, including collagen and hyaluronan. By targeting these factors, the agents helped to remodel the TME, making it more receptive to therapeutic interventions and allowing for better drug delivery and immune response activation. Additional mechanotherapeutics include the antihyperglycemic agent metformin59, the corticosteroid dexamethasone47, and the endothelin receptor antagonist bosentan60. Notably, bosentan is being tested in a Phase I clinical trial in patients with unresectable pancreatic cancer (Clinical-Trials.gov identifier: NCT04158635). Another agent is paricalcitol—an agonist targeting the vitamin D receptor on CAFs61.
These successes demonstrate the clinical potential of mechanotherapeutics, but several challenges remain as mechanotherapeutics cannot decompress the majority of the vessels but only a percentage of them18,20,47,52.
Sonopermeation for instant mechano-modulation of the TME
Ultrasound is a widely utilized imaging modality in medical diagnostics, known for its safety and ability to provide real-time imaging. Over the past several decades, its use has expanded beyond diagnostics, gaining considerable attention in the field of therapeutic applications. Researchers have increasingly explored its potential in treating various medical conditions, recognizing the versatility and non-invasive nature of ultrasound technology in both imaging and therapy62,63,64,65. Because ultrasound can be focused, it is used to deliver energy to small volumes deep inside the body without affecting intermediate tissues. The use of ultrasound in the presence of exogenous gas bubbles (i.e., microbubbles) can lead to the development of local forces strong enough to cause membrane permeabilization of cells (Fig. 1). Specifically, sonopermeation works both through the formation of transient pores in endothelial cell membranes (traditional sonoporation), as well as via the opening of intercellular (tight) junctions66,67, stimulated endocytosis, transcytosis and/or exocytosis68,69 and causing macroscopic changes in perfusion70 and/or changes in the perivascular and extracellular space of tumors71. As pressure waves move through tissues, microbubbles expand during the low-pressure (rarefaction) phases and contract during the high-pressure (compression) phases, producing volume oscillations that align with the applied ultrasound frequency. This oscillation creates a circulating fluid flow called microstreaming. When microbubbles are close to the endothelium, they can deform the cell membrane and may induce pore formation. At higher acoustic pressures, the oscillations become larger, leading to bubble collapse. The oscillation and collapse of microbubbles can also generate free radicals, which may increase cytotoxicity and lead to potential cell death72. Sonopermeation effects last between 4 and 24 h, with no significant effect beyond 24 h73,74.
The promise of sonopermeation to improve perfusion and enhance the delivery of chemotherapeutics or nano-scale drugs to cancer has been extensively investigated in several preclinical studies. It has been demonstrated that combining sonopermeation with gemcitabine in a pancreatic adenocarcinoma mouse model resulted in more effective cancer treatment than gemcitabine alone, significantly inhibiting primary tumor growth, delaying metastasis formation, and extending the survival of the mice75. Recently, a study on pancreatic tumors found that combining sonopermeation with FOLFIRINOX (fluorouracil, irinotecan, oxaliplatin, and calcium folinate) significantly enhanced platinum uptake compared to untreated tumors or single-agent therapy76. Furthermore, when exposed to ultrasound and microbubbles, increased tumor uptake of nanoparticle liposomal doxorubicin (Doxil) has been demonstrated in murine subcutaneous colorectal77 and prostate adenocarcinoma78 models and enhanced encapsulation of cabazitaxel in prostate cancer79. In a recent study80, employment of sonopermeation in neuroblastoma xenografts increased Doxil uptake by increasing vascular lumen, disrupting tight junctions, inducing greater apoptosis, and resulting in increased survival. Assessment for side effects and toxicity in other organs, such as lung, liver, and kidney proved that treatment was safe. There was no evidence for injury in these organs, the red blood cell deposition within the lung capillaries was normal, as well as, the vasculature in the liver tissues. Also, there was no change in circulating tumor cells.
While sonopermeation primarily enhances drug delivery and therapeutic efficacy by facilitating the penetration of therapeutic agents into cells, it can also indirectly affect the TME through its influence on tumor cells and surrounding tissues. Besides forming transient pores, sonopermeation alters tumor perfusion and the TME. It has been reported that sonopermeation reduces intratumoral solid stress, improving perfusion in murine models of prostate adenocarcinoma and osteosarcoma81. Specifically, sonopermeation significantly enhanced perfusion, as shown by contrast-enhanced ultrasound (CEUS) imaging, and reduced tumor microvascular density, indicating vascular normalization of the TME in colon cancer82. These findings suggest that combining ultrasound with microbubbles can normalize TME and restore tumor perfusion. The enhanced perfusion may alter immune-suppressive factors, potentially increasing immune cell infiltration into hypo-vascular tumors. Effective immunotherapy relies heavily on sufficient CD8+ T-cell infiltration, and sonopermeation not only can improve immunotherapy delivery but also it can significantly increase the percentage of infiltrating cytotoxic CD8+ T-cells in murine models of colon cancer83 and melanoma84,85.
Sonopermeation efficacy has been studied in clinical trials, showing that ultrasound and microbubbles enhance the effects of conventional chemotherapy in patients with pancreatic cancer, liver metastases from colon cancer, and hepatic metastases from digestive system tumors86,87,88,89. A combination of gemcitabine and sonopermeation in ten inoperable pancreatic cancer patients has shown that can extend survival without causing additional toxicities86. Notably, the use of sonopermeation also allowed patients to undergo more treatment cycles, which corresponded to a longer period of well-being. However, recent findings indicated that sonopermeation did not enhance chemotherapy efficacy in patients with liver metastases88,89, but still, several studies are currently underway involving patients with breast cancer, glioblastoma, pancreatic cancer, liver metastases from breast and colorectal cancer, and brain metastases from melanoma90,91 (NCT03322813, NCT03477019, NCT04146441, NCT04021420, NCT03458975, and NCT03385200). These studies aim to further investigate the efficacy, safety, and clinical feasibility of this therapeutic strategy.
Despite these promising advancements, hypo-perfusion caused by vessel compression significantly undermines the effectiveness of sonopermeation, as microbubbles cannot be delivered effectively and uniformly in hypo-perfused tumors. This results in localized effects rather than a comprehensive therapeutic impact, ultimately compromising the overall efficacy of this treatment strategy75,92.
Mechanotherapeutics and sonopermeation uniquely complement each other and optimize treatment efficacy in sarcomas
From the above analysis, it is reasonable to argue that mechanotherapeutics, by opening up some tumor vessels and improving tumor perfusion can significantly increase the distribution of microbubbles within the tumor, thereby enhancing the efficacy of sonopermeation. Furthermore, it has been recently reported that sonopermeation can reduce intratumoral solid stress and thus, improve perfusion81. Consequently, sonopermeation could support the use of mechanotherapeutics to further alleviate mechanical forces in tumors and potentially open up more tumor vessels, allowing more microbubbles and anti-cancer drugs to enter the tumor and thus, creating a positive feedback loop. Therefore, it makes good sense to argue that the mechano-modulation of the TME by combining mechanotherapeutics and sonopermeation will not only have additive effects but, due to the synergistic mechanisms and complementarity of the two methods, may yield multiplicative improvements in perfusion and therapeutic outcomes (Fig. 1).
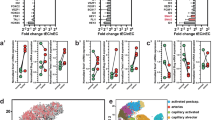
To test this hypothesis, we conducted in vivo studies on sarcoma models93 (Fig. 2). Our results showed that combining the mechanotherapeutic ketotifen with sonopermeation demonstrated optimal improvements in several key physiological parameters. Specifically, there was a statistically significant reduction in the tumor’s elastic properties, as measured by Shear Wave Elastography (SWE), indicating a decrease in tissue stiffness, presumably due to a reduction in hyaluronan levels. This dual approach not only improved tumor perfusion but also facilitated more efficient drug delivery resulting in enhanced anti-tumor effectiveness of nano-immunotherapy. Consequently, blood vessel functionality was enhanced, leading to improved infiltration of immune cells -including CD8+ T-cells- and a decrease of immunosuppressive Tregs, thereby strengthening the immune response against the tumor. To summarize, our study presented evidence that mechano-modulation of the TME through the combined use of mechanotherapeutics and sonopermeation can yield multiple, synergistic effects93. The combination of these strategies appears to amplify treatment efficacy beyond the benefits observed when each therapeutic method is applied separately.
a Experimental treatment protocol in MCA205 fibrosarcoma tumors. Created with BioRender.com. Combined treatment decreases b tumor growth, c tissue elasticity, d hyaluronan binding protein (bHABP1, green), and e boosts immune cell infiltration. Data adapted with permission from ref.93.
Conclusions
Tumor stiffening and elevation of mechanical forces within its solid components compress tumor blood vessels, reducing blood flow and hindering uniform and efficient drug delivery, ultimately weakening treatment effectiveness8,27,45. As a result, standard therapies struggle to treat highly desmoplastic tumors effectively2,3,4,5. One therapeutic approach to alleviate blood vessel compression and enhance perfusion involves the use of mechanotherapeutics, which work to reduce stiffness and relieve mechanical forces within tumors46. Ultrasound-mediated drug delivery with microbubbles is another strategy that could noninvasively enhance perfusion and thus, the transport of therapeutic agents to targeted tumors via sonopermeation. Although both strategies have shown promising results, each has encountered certain limitations.
The innovative concept of combining mechanotherapeutics with sonopermeation into a unified approach offers a potential solution to these challenges. This combined strategy could establish a new foundation for treating highly desmoplastic cancer types, ultimately enhancing therapeutic outcomes and overcoming barriers that individual methods alone cannot fully address. In our previous study93, we have shown for the first time, that mechano-modulation of the TME by combining mechanotherapeutics and sonopermeation can have multiplicative synergistic effects on improving perfusion and therapeutic outcomes. Importantly, mechanotherapeutics and sonopermeation synergistically modulated TME in sarcomas without causing any toxicity effects to the mice as the mouse weight, which was monitored throughout the treatment period for each group remained almost the same with no significant differences. While more research is needed, the current evidence suggests that mechanotherapeutics and sonopermeation have the potential to significantly improve drug delivery, enhance therapeutic response, and extend patient survival, offering a promising alternative for challenging cancers with poor prognosis.
Translating mechanotherapeutics and sonopermeation from preclinical models to humans involves challenges related to biological differences, safety, and feasibility. Variations in tissue structure and vascular properties between mice and humans can impact treatment efficiency and drug distribution. Scaling ultrasound parameters like frequency, intensity, and duration is crucial to maximize efficacy while avoiding off-target effects. Immune response differences and inadequate replication of the human tumor microenvironment in animal models further complicate translation. Regulatory hurdles and the need for standardized protocols add to the complexity. Glioblastomas may benefit most from this approach due to improved blood-brain barrier disruption, enhancing drug delivery. Other solid tumors, like pancreatic cancer, triple-negative breast cancer, and desmoplastic sarcomas, could also benefit by increasing drug penetration and immune cell infiltration. Effective translation to humans will require tailored ultrasound protocols and thorough safety evaluations of drugs to ensure a successful application.
Data availability
No datasets were generated or analyzed during the current study.
References
Ganesh, K. & Massague, J. Targeting metastatic cancer. Nat. Med. 27, 34–44 (2021).
Gill, J. & Gorlick, R. Advancing therapy for osteosarcoma. Nat. Rev. Clin. Oncol. 18, 609–624 (2021).
Murakami, T. et al. Role of the tumor microenvironment in pancreatic cancer. Ann. Gastroenterol. Surg. 3, 130–137 (2019).
Augsburger, D. et al. Current diagnostics and treatment of fibrosarcoma -perspectives for future therapeutic targets and strategies. Oncotarget 8, 104638–104653 (2017).
Torphy, R. J. et al. Stromal content is correlated with tissue site, contrast retention, and survival in pancreatic adenocarcinoma. JCO Precisi. Oncol. https://doi.org/10.1200/PO.17.00121 (2018).
Chauhan, V. P., Stylianopoulos, T., Boucher, Y. & Jain, R. K. Delivery of molecular and nanomedicine to tumors: Transport barriers and strategies. Annu. Rev. Chem. Biomol. Eng. 2, 281–298 (2011).
Jain, R. K., Martin, J. D. & Stylianopoulos, T. The role of mechanical forces in tumor growth and therapy. Annu. Rev. Biomed. Eng. 16, 321–346 (2014).
Stylianopoulos, T. et al. Causes, consequences, and remedies for growth-induced solid stress in murine and human tumors. Proc. Natl Acad. Sci. USA 109, 15101–15108 (2012).
Stylianopoulos, T. et al. Coevolution of solid stress and interstitial fluid pressure in tumors during progression: Implications for vascular collapse. Cancer Res. 73, 3833–3841 (2013).
Nia, H. T. et al. Solid stress and elastic energy as measures of tumour mechanopathology. Nat. Biomed. Eng. https://doi.org/10.1038/s41551-016-0004 (2016).
Papageorgis, P. et al. Tranilast-induced stress alleviation in solid tumors improves the efficacy of chemo- and nanotherapeutics in a size-independent manner. Sci. Rep. 7, 46140 (2017).
Polydorou, C., Mpekris, F., Papageorgis, P., Voutouri, C. & Stylianopoulos, T. Pirfenidone normalizes the tumor microenvironment to improve chemotherapy. Oncotarget 8, 24506–24517 (2017).
Panagi, M. et al. TGF-beta inhibition combined with cytotoxic nanomedicine normalizes triple negative breast cancer microenvironment towards anti-tumor immunity. Theranostics 10, 1910–1922 (2020).
Voutouri, C., Mpekris, F., Papageorgis, P., Odysseos, A. D. & Stylianopoulos, T. Role of constitutive behavior and tumor-host mechanical interactions in the state of stress and growth of solid tumors. PloS ONE 9, e104717 (2014).
Mpekris, F., Angeli, S., Pirentis, A. P. & Stylianopoulos, T. Stress-mediated progression of solid tumors: effect of mechanical stress on tissue oxygenation, cancer cell proliferation, and drug delivery. Biomech. Model. Mechanobiol. 14, 1391–1402 (2015).
Stylianopoulos, T., Munn, L. L. & Jain, R. K. Reengineering the physical microenvironment of tumors to improve drug delivery and efficacy: from mathematical modeling to bench to bedside. Trends Cancer 4, 292–319 (2018).
Chauhan, V. P. et al. Compression of pancreatic tumor blood vessels by hyaluronan is caused by solid stress and not interstitial fluid pressure. Cancer Cell 26, 14–15 (2014).
Mpekris, F. et al. Sonic-hedgehog pathway inhibition normalizes desmoplastic tumor microenvironment to improve chemo- and nanotherapy. J. Controlled Release261, 105–112 (2017).
Stylianopoulos, T. The solid mechanics of cancer and strategies for improved therapy. J. Biomech. Eng. https://doi.org/10.1115/1.4034991 (2017).
Mpekris, F. et al. Normalizing the Microenvironment Overcomes Vessel Compression and Resistance to Nano-immunotherapy in Breast Cancer Lung Metastasis. Adv. Sci. 8, 2001917 (2021).
Renaudin, X. Reactive oxygen species and DNA damage response in cancer. Int. Rev. Cell Mol. Biol. 364, 139–161 (2021).
Telarovic, I., Wenger, R. H. & Pruschy, M. Interfering with Tumor Hypoxia for Radiotherapy Optimization. J. Exp. Clin. Cancer Res.40, 197 (2021).
Helmlinger, G., Netti, P. A., Lichtenbeld, H. C., Melder, R. J. & Jain, R. K. Solid stress inhibits the growth of multicellular tumor spheroids. Nat. Biotechnol. 15, 778–783 (1997).
Cheng, G., Tse, J., Jain, R. K. & Munn, L. L. Micro-environmental mechanical stress controls tumor spheroid size and morphology by suppressing proliferation and inducing apoptosis in cancer cells. PloS ONE 4, e4632 (2009).
Samuel, M. S. et al. Actomyosin-mediated cellular tension drives increased tissue stiffness and beta-catenin activation to induce epidermal hyperplasia and tumor growth. Cancer Cell 19, 776–791 (2011).
Hidalgo, M. & Von Hoff, D. D. Translational therapeutic opportunities in ductal adenocarcinoma of the pancreas. Clin. Cancer Res.18, 4249–4256 (2012).
Padera, T. P. et al. Pathology: cancer cells compress intratumour vessels. Nature 427, 695 (2004).
Joyce, J. A. & Fearon, D. T. T cell exclusion, immune privilege, and the tumor microenvironment. Science 348, 74–80 (2015).
Mariathasan, S. et al. TGFbeta attenuates tumour response to PD-L1 blockade by contributing to exclusion of T cells. Nature 554, 544–548 (2018).
Wilson, W. R. & Hay, M. P. Targeting hypoxia in cancer therapy. Nat. Rev. Cancer 11, 393–410 (2011).
Barsoum, I. B., Smallwood, C. A., Siemens, D. R. & Graham, C. H. A mechanism of hypoxia-mediated escape from adaptive immunity in cancer cells. Cancer Res. 74, 665–674 (2014).
Majidpoor, J. & Mortezaee, K. Angiogenesis as a hallmark of solid tumors - clinical perspectives. Cell. Oncol. 44, 715–737 (2021).
Noman, M. Z. et al. PD-L1 is a novel direct target of HIF-1alpha, and its blockade under hypoxia enhanced MDSC-mediated T cell activation. J. Exp. Med. 211, 781–790 (2014).
Noman, M. Z. et al. Hypoxia: a key player in antitumor immune response. A Review in the Theme: Cellular Responses to Hypoxia. Am. J. Physiol. Cell. Physiol. 309, C569 (2015).
Facciabene, A. et al. Tumour hypoxia promotes tolerance and angiogenesis via CCL28 and T(reg) cells. Nature 475, 226–230 (2011).
Calcinotto, A. et al. Modulation of microenvironment acidity reverses anergy in human and murine tumor-infiltrating T lymphocytes. Cancer Res. 72, 2746–2756 (2012).
Palazon, A., Aragones, J., Morales-Kastresana, A., de Landazuri, M. O. & Melero, I. Molecular pathways: hypoxia response in immune cells fighting or promoting cancer. Clin. Cancer Res.18, 1207–1213 (2012).
Laoui, D. et al. Tumor hypoxia does not drive differentiation of tumor-associated macrophages but rather fine-tunes the M2-like macrophage population. Cancer Res. 74, 24–30 (2014).
Jain, R. K. Antiangiogenesis strategies revisited: from starving tumors to alleviating hypoxia. Cancer cell 26, 605–622 (2014).
Jing, X. et al. Role of hypoxia in cancer therapy by regulating the tumor microenvironment. Mol. Cancer 18, 157 (2019).
Jain, R. K. Transport of molecules across tumor vasculature. Cancer Metastasis Rev. 6, 559–593 (1987).
Zheng, X. et al. Increased vessel perfusion predicts the efficacy of immune checkpoint blockade. J. Clin. Investig. 128, 2104–2115 (2018).
Chen, I. X. et al. Blocking CXCR4 alleviates desmoplasia, increases T-lymphocyte infiltration, and improves immunotherapy in metastatic breast cancer. Proc. Natl Acad. Sci. USA 116, 4558–4566 (2019).
Chauhan, V. P. et al. Reprogramming the microenvironment with tumor-selective angiotensin blockers enhances cancer immunotherapy. Proc. Natl Acad. Sci. USA 116, 10674–10680 (2019).
Griffon-Etienne, G., Boucher, Y., Brekken, C., Suit, H. D. & Jain, R. K. Taxane-induced apoptosis decompresses blood vessels and lowers interstitial fluid pressure in solid tumors: clinical implications. Cancer Res. 59, 3776–3782 (1999).
Sheridan, C. Pancreatic cancer provides testbed for first mechanotherapeutics. Nat. Biotechnol. 37, 829–831 (2019).
Chauhan, V. P. et al. Angiotensin inhibition enhances drug delivery and potentiates chemotherapy by decompressing tumor blood vessels. Nature Commun. https://doi.org/10.1038/ncomms.3516 (2013).
Mpekris, F. et al. Combining microenvironment normalization strategies to improve cancer immunotherapy. Proc. Natl Acad. Sci. USA 117, 3728–3737 (2020).
Mpekris, F., Voutouri, C., Papageorgis, P. & Stylianopoulos, T. Stress alleviation strategy in cancer treatment: Insights from a mathematical model. Z. Angew. Math. Mech. 98, 2295–2306 (2018).
Wei, X. et al. Mechanisms of vasculogenic mimicry in hypoxic tumor microenvironments. Mol. Cancer 20, 7 (2021).
Li, H. et al. Light-driven self-recruitment of biomimetic semiconducting polymer nanoparticles for precise tumor vascular disruption. Adv. Mater. 35, e2210920 (2023).
Panagi, M. et al. Stabilizing tumor-resident mast cells restores T-cell infiltration and sensitizes sarcomas to PD-L1 inhibition. Clin. Cancer Res. 30, 2582–2597 (2024).
Panagi, M. et al. Polymeric micelles effectively reprogram the tumor microenvironment to potentiate nano-immunotherapy in mouse breast cancer models. Nat. Commun. 13, 7165 (2022).
Mpekris, F. et al. Pirfenidone-loaded polymeric micelles as an effective mechanotherapeutic to potentiate immunotherapy in mouse tumor models. ACS Nano 17, 24654–24667 (2023).
Diop-Frimpong, B., Chauhan, V. P., Krane, S., Boucher, Y. & Jain, R. K. Losartan inhibits collagen I synthesis and improves the distribution and efficacy of nanotherapeutics in tumors. Proc. Natl Acad. Sci. USA 108, 2909–2914 (2011).
Liu, J. et al. TGF-beta blockade improves the distribution and efficacy of therapeutics in breast carcinoma by normalizing the tumor stroma. Proc. Natl Acad. Sci. USA 109, 16618–16623 (2012).
Murphy, J. E. et al. Total neoadjuvant therapy with FOLFIRINOX in combination with losartan followed by chemoradiotherapy for locally advanced pancreatic cancer: a phase 2 clinical trial. JAMA Oncol. 5, 1020–1027 (2019).
Panagi, M. et al. Stabilizing Tumor-Resident Mast Cells Restores T-Cell Infiltration and Sensitizes Sarcomas to PD-L1 Inhibition. Clin Cancer Res. 30, 2582–2597 (2024).
Incio, J. et al. Metformin reduces desmoplasia in pancreatic cancer by reprogramming stellate cells and tumor-associated macrophages. PloS ONE 10, e0141392 (2015).
Voutouri, C. et al. Endothelin inhibition potentiates cancer immunotherapy revealing mechanical biomarkers predictive of response. Adv. Therap. https://doi.org/10.1002/adtp.202000289 (2021).
Sherman, M. H. et al. Vitamin D receptor-mediated stromal reprogramming suppresses pancreatitis and enhances pancreatic cancer therapy. Cell 159, 80–93 (2014).
Czarnota, G. J. et al. Ultrasound imaging of apoptosis: high-resolution non-invasive monitoring of programmed cell death in vitro, in situ and in vivo. Br. J. Cancer 81, 520–527 (1999).
Arvanitis, C. D., Ferraro, G. B. & Jain, R. K. The blood-brain barrier and blood-tumour barrier in brain tumours and metastases. Nat. Rev. Cancer 20, 26–41 (2020).
Sennoga, C. A. et al. Microbubble-mediated ultrasound drug-delivery and therapeutic monitoring. Expert Opin. Drug Deliv. 14, 1031–1043 (2017).
Rix, A. et al. Advanced ultrasound technologies for diagnosis and therapy. J. Nucl. Med.59, 740–746 (2018).
Sheikov, N., McDannold, N., Sharma, S. & Hynynen, K. Effect of focused ultrasound applied with an ultrasound contrast agent on the tight junctional integrity of the brain microvascular endothelium. Ultrasound Med. Biol. 34, 1093–1104 (2008).
Lentacker, I., De Cock, I., Deckers, R., De Smedt, S. C. & Moonen, C. T. Understanding ultrasound induced sonoporation: definitions and underlying mechanisms. Adv. Drug Deliv. Rev. 72, 49–64 (2014).
Meijering, B. D. et al. Ultrasound and microbubble-targeted delivery of macromolecules is regulated by induction of endocytosis and pore formation. Circ. Res. 104, 679–687 (2009).
Yuana, Y. et al. Microbubbles-Assisted Ultrasound Triggers the Release of Extracellular Vesicles. Int. J. Mol. Sci. https://doi.org/10.3390/ijms18081610 (2017).
Rix, A. et al. Influence of repetitive contrast agent injections on functional and molecular ultrasound measurements. Ultrasound Med. Biol. 40, 2468–2475 (2014).
Hersh, D. S. et al. Pulsed ultrasound expands the extracellular and perivascular spaces of the brain. Brain Res. 1646, 543–550 (2016).
Jain, A., Tiwari, A., Verma, A. & Jain, S. K. Ultrasound-based triggered drug delivery to tumors. Drug Deliv. Transl. Res. 8, 150–164 (2018).
Zhong, W., Sit, W. H., Wan, J. M. & Yu, A. C. Sonoporation induces apoptosis and cell cycle arrest in human promyelocytic leukemia cells. Ultrasound Med. Biol. 37, 2149–2159 (2011).
Yu, Z. W., Liang, Y. & Liang, W. Q. Low-frequency sonophoresis enhances rivastigmine permeation in vitro and in vivo. Die Pharm. 70, 379–380 (2015).
Kotopoulis, S. et al. Sonoporation-enhanced chemotherapy significantly reduces primary tumour burden in an orthotopic pancreatic cancer xenograft. Mol. Imaging Biol. 16, 53–62 (2014).
Haram, M. et al. Ultrasound and microbubbles increase the uptake of platinum in murine orthotopic pancreatic tumors. Ultrasound Med. Biol. 49, 1275–1287 (2023).
Lin, C. Y., Li, J. R., Tseng, H. C., Wu, M. F. & Lin, W. L. Enhancement of focused ultrasound with microbubbles on the treatments of anticancer nanodrug in mouse tumors. Nanomedicine 8, 900–907 (2012).
Olsman, M. et al. Ultrasound-mediated delivery enhances therapeutic efficacy of MMP sensitive liposomes. J. Controlled Release325, 121–134 (2020).
Snipstad, S. et al. Sonopermeation enhances uptake and therapeutic effect of free and encapsulated cabazitaxel. Ultrasound Med. Biol. 47, 1319–1333 (2021).
Sundland, R. M. et al. Sonopermeation with size-sorted microbubbles synergistically increases survival and enhances tumor apoptosis with L-DOX by increasing vascular permeability and perfusion in neuroblastoma xenografts. Ultrasound Med. Biol. 51, 348–357 (2025).
Soulheim, E. et al. Sonopermeation with nanoparticle-stabilized microbubbles reduces solid stress and improves nanomedicine delivery to tumors. Adv. Ther. 4, 2100147 (2021).
Li, N. et al. Tumor perfusion enhancement by ultrasound stimulated microbubbles potentiates PD-L1 blockade of MC38 colon cancer in mice. Cancer Lett. 498, 121–129 (2021).
Bulner, S., Prodeus, A., Gariepy, J., Hynynen, K. & Goertz, D. E. Enhancing checkpoint inhibitor therapy with ultrasound stimulated microbubbles. Ultrasound Med. Biol. 45, 500–512 (2019).
Liu, H. L. et al. Low-pressure pulsed focused ultrasound with microbubbles promotes an anticancer immunological response. J. Transl. Med. 10, 221 (2012).
Yang, C., Du, M., Yan, F. & Chen, Z. Focused ultrasound improves NK-92MI cells infiltration into tumors. Front. Pharmacol. 10, 326 (2019).
Dimcevski, G. et al. A human clinical trial using ultrasound and microbubbles to enhance gemcitabine treatment of inoperable pancreatic cancer. J. Controlled Release243, 172–181 (2016).
Kotopoulis, S., Dimcevski, G., Gilja, O. H., Hoem, D. & Postema, M. Treatment of human pancreatic cancer using combined ultrasound, microbubbles, and gemcitabine: a clinical case study. Med. Phys. 40, 072902 (2013).
Haram, M. et al. Treatment of liver metastases with focused ultrasound and microbubbles in patients with colorectal cancer receiving chemotherapy. Ultrasound Med. Biol. 49, 2081–2088 (2023).
Wang, Y. et al. Clinical study of ultrasound and microbubbles for enhancing chemotherapeutic sensitivity of malignant tumors in digestive system. Chin. J. Cancer Res.30, 553–563 (2018).
Idbaih, A. et al. Safety and feasibility of repeated and transient blood-brain barrier disruption by pulsed ultrasound in patients with recurrent glioblastoma. Clin. Cancer Res.25, 3793–3801 (2019).
Snipstad, S. et al. Ultrasound and microbubbles to beat barriers in tumors: Improving delivery of nanomedicine. Adv. Drug Deliv. Rev. 177, 113847 (2021).
Snipstad, S. et al. Sonopermeation to improve drug delivery to tumors: from fundamental understanding to clinical translation. Expert Opin. Drug Deliv. 15, 1249–1261 (2018).
Mpekris, F. et al. A synergistic approach for modulating the tumor microenvironment to enhance nano-immunotherapy in sarcomas. Neoplasia 51, 100990 (2024).
Izumi, K. et al. Tranilast inhibits hormone refractory prostate cancer cell proliferation and suppresses transforming growth factor beta1-associated osteoblastic changes. Prostate 69, 1222–1234 (2009).
Hashemzehi, M. et al. Inhibition of transforming growth factor-beta by Tranilast reduces tumor growth and ameliorates fibrosis in colorectal cancer. EXCLI J. 20, 601–613 (2021).
Fujiwara, A. et al. Effects of pirfenidone targeting the tumor microenvironment and tumor-stroma interaction as a novel treatment for non-small cell lung cancer. Sci. Rep. 10, 10900 (2020).
Yao, Y. et al. Dexamethasone inhibits pancreatic tumor growth in preclinical models: Involvement of activating glucocorticoid receptor. Toxicol. Appl. Pharmacol. 401, 115118 (2020).
Martin, J. D. et al. Dexamethasone increases cisplatin-loaded nanocarrier delivery and efficacy in metastatic breast cancer by normalizing the tumor microenvironment. ACS Nano 13, 6396–6408 (2019).
Yousef, M. & Tsiani, E. Metformin in lung cancer: review of in vitro and in vivo animal studies. Cancers https://doi.org/10.3390/cancers9050045 (2017).
Chen, K. et al. Metformin suppresses cancer initiation and progression in genetic mouse models of pancreatic cancer. Mol. Cancer 16, 131 (2017).
Cufi, S. et al. Metformin-induced preferential killing of breast cancer initiating CD44+CD24-/low cells is sufficient to overcome primary resistance to trastuzumab in HER2+ human breast cancer xenografts. Oncotarget 3, 395–398 (2012).
Sahra, I. et al. The antidiabetic drug metformin exerts an antitumoral effect in vitro and in vivo through a decrease of cyclin D1 level. Oncogene 27, 3576–3586 (2008).
Buzzai, M. et al. Systemic treatment with the antidiabetic drug metformin selectively impairs p53-deficient tumor cell growth. Cancer Res. 67, 6745–6752 (2007).
Tomic, T. et al. Metformin inhibits melanoma development through autophagy and apoptosis mechanisms. Cell Death Dis. 2, e199 (2011).
Gautam, S. K. et al. Endothelin-axis antagonism enhances tumor perfusion in pancreatic cancer. Cancer Lett. 544, 215801 (2022).
Schwartz, G. G. et al. 19-nor-1 alpha,25-dihydroxyvitamin D2 (paricalcitol) inhibits the proliferation of human pancreatic cancer cells in vitro and in vivo. Cancer Biol. Ther. 7, 430–436 (2008).
Xun, J. et al. JMJD3 suppresses stem cell-like characteristics in breast cancer cells by downregulation of Oct4 independently of its demethylase activity. Oncotarget 8, 21918–21929 (2017).
Mitjans, F. et al. In vivo therapy of malignant melanoma by means of antagonists of alphav integrins. Int. J. Cancer 87, 716–723 (2000).
Burke, P. A. et al. Cilengitide targeting of alpha(v)beta(3) integrin receptor synergizes with radioimmunotherapy to increase efficacy and apoptosis in breast cancer xenografts. Cancer Res. 62, 4263–4272 (2002).
Zhao, Y. et al. Losartan treatment enhances chemotherapy efficacy and reduces ascites in ovarian cancer models by normalizing the tumor stroma. Proc. Natl Acad. Sci. USA 116, 2210–2219 (2019).
Zhao, J. et al. Fructose-coated nanoparticles: a promising drug nanocarrier for triple-negative breast cancer therapy. Chem. Commun. 50, 15928–15931 (2014).
Cun, X. et al. A dual strategy to improve the penetration and treatment of breast cancer by combining shrinking nanoparticles with collagen depletion by losartan. Acta Biomater. 31, 186–196 (2016).
Zhao, Q. et al. Enhanced therapeutic efficacy of combining losartan and chemo-immunotherapy for triple negative breast cancer. Front. Immunol. 13, 938439 (2022).
Datta, M. et al. Losartan controls immune checkpoint blocker-induced edema and improves survival in glioblastoma mouse models. Proc. Natl Acad. Sci. USA 120, e2219199120 (2023).
Hashemzehi, M. et al. Angiotensin receptor blocker Losartan inhibits tumor growth of colorectal cancer. EXCLI J. 20, 506–521 (2021).
Acknowledgements
This project received funding from the European Research Council (ERC) under the European Union’s Horizon 2020 and Horizon Europe research and innovation program (grant agreement no. 101076425 to FM) and from the Republic of Cyprus through the Research and Innovation Foundation (CULTURE/AWARD-YR/0523 B/0006 to FM). Views and opinions expressed are, however, those of the authors only and do not necessarily reflect those of the European Union or the European Research Council Executive Agency. Neither the European Union nor the granting authority can be held responsible for them.
Author information
Authors and Affiliations
Contributions
All authors contributed to the conceptualization, methodology, validation, formal analysis, investigation, resources, data curation, writing—review editing, and visualization of the article. C.N. and F.M. were responsible for writing the original draft. T.S. and F.M. were responsible for supervision and project administration and funding acquisition.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Neophytou, C., Stylianopoulos, T. & Mpekris, F. The synergistic potential of mechanotherapy and sonopermeation to enhance cancer treatment effectiveness. npj Biol. Phys. Mech. 2, 13 (2025). https://doi.org/10.1038/s44341-025-00017-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44341-025-00017-3