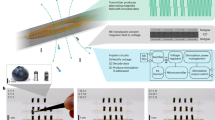
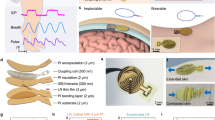
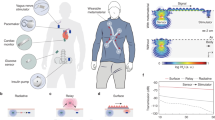
Abstract
Chronic cardiovascular, neurological and metabolic diseases affect billions of people worldwide, yet conventional tethered or battery-powered implants are constrained by bulk, infection risk and finite lifespans that necessitate surgical replacement. Here we examine recent advances in wireless and battery-free implantable sensors, spanning clinical motivations, device architectures, wireless communication strategies, energy solutions and representative perspectives. We discuss how tailored encapsulation and fixation, application-specific sampling strategies, and multimodal transduction pathways enable stable biointerfaces and high-fidelity physiological monitoring. We show that wireless telemetry has evolved from passive backscatter to hybrid active–passive schemes, enabling higher bandwidth, deeper implantation and improved signal robustness. We further summarize wireless power transfer and in-body energy-harvesting approaches that support sustainable device operation. We conclude by identifying key technical and translational challenges and outlining emerging directions towards clinically translatable, autonomous, wireless and battery-free bioelectronic systems.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
World Report on Ageing and Health (World Health Organization, 2015).
Mensah, G. A., Fuster, V. & Roth, G. A. A heart-healthy and stroke-free world: using data to inform global action. J. Am. Coll. Cardiol. 82, 25 (2023).
Sun, H. et al. IDF diabetes atlas: global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res. Clin. Pract. 183, 109119 (2022).
Moorman, J. R. The principles of whole-hospital predictive analytics monitoring for clinical medicine originated in the neonatal ICU. npj Digit. Med. 5, 41 (2022).
Ferrero, J. J. et al. Closed-loop electrical stimulation prevents focal epilepsy progression and long-term memory impairment. Nat. Neurosci. 28, 1753–1762 (2025).
Bocci, T. et al. Eight-hours conventional versus adaptive deep brain stimulation of the subthalamic nucleus in Parkinson’s disease. npj Parkinsons Dis 7, 88 (2021).
Kight, A. et al. VITALS: an implantable sensor network for postoperative cardiac monitoring in heart failure patients. npj Biomed. Innov. 2, 15 (2025).
Madhvapathy, S. R. et al. Implantable bioelectronics and wearable sensors for kidney health and disease. Nat. Rev. Nephrol. 21, 1–21 (2025).
Won, S. M., Cai, L., Gutruf, P. & Rogers, J. A. Wireless and battery-free technologies for neuroengineering. Nat. Biomed. Eng. 7, 405–423 (2023).
Lim, Y.-S. et al. Precise control of tibial nerve stimulation for bladder regulation via evoked compound action potential feedback mechanisms. Nat. Commun. 16, 4115 (2025).
Nair, V. et al. Miniature battery-free bioelectronics. Science 382, eabn4732 (2023).
Kim, H., Rigo, B., Wong, G., Lee, Y. J. & Yeo, W. H. Advances in wireless, batteryless, implantable electronics for real-time, continuous physiological monitoring. Nanomicro Lett. 16, 52 (2024).
Gutruf, P. et al. Wireless, battery-free, fully implantable multimodal and multisite pacemakers for applications in small animal models. Nat. Commun. 10, 5742 (2019).
Ates, H. C. et al. End-to-end design of wearable sensors. Nat. Rev. Mater. 7, 887–907 (2022).
Choi, Y. S. et al. Fully implantable and bioresorbable cardiac pacemakers without leads or batteries. Nat. Biotechnol. 39, 1228–1238 (2021).
Ouyang, W. et al. A wireless and battery-less implant for multimodal closed-loop neuromodulation in small animals. Nat. Biomed. Eng. 7, 1252–1269 (2023).
Wan, J. et al. Millimeter-scale magnetic implants paired with a fully integrated wearable device for wireless biophysical and biochemical sensing. Sci. Adv. 10, eadm9314 (2024).
Kwon, K. et al. A battery-less wireless implant for the continuous monitoring of vascular pressure, flow rate and temperature. Nat. Biomed. Eng. 7, 1215–1228 (2023).
Zhu, J. et al. End-of-life or second-life options for retired electric vehicle batteries. Cell Rep. Phys. Sci. 2, 100665 (2021).
Wu, T., Redouté, J.-M. & Yuce, M. R. A wireless implantable sensor design with subcutaneous energy harvesting for long-term IoT healthcare applications. IEEE Access 6, 35801–35808 (2018).
Cai, L. et al. Osseosurface electronics—thin, wireless, battery-free and multimodal musculoskeletal biointerfaces. Nat. Commun. 12, 6707 (2021).
Yoon, H.-J. & Kim, S.-W. Nanogenerators to power implantable medical systems. Joule 4, 1398–1407 (2020).
Feiner, R. & Dvir, T. Tissue–electronics interfaces: from implantable devices to engineered tissues. Nat. Rev. Mater. 3, 1–16 (2017).
Agarwal, K., Jegadeesan, R., Guo, Y.-X. & Thakor, N. V. Wireless power transfer strategies for implantable bioelectronics. IEEE Rev. Biomed. Eng. 10, 136–161 (2017).
Li, J., Dong, Y., Park, J. H. & Yoo, J. Body-coupled power transmission and energy harvesting. Nat. Electron. 4, 530–538 (2021).
Steins, H. et al. A flexible protruding microelectrode array for neural interfacing in bioelectronic medicine. Microsyst. Nanoeng. 8, 131 (2022).
Kim, D.-H. et al. Epidermal electronics. Science 333, 838–843 (2011).
Carnicer-Lombarte, A. et al. Ultraconformable cuff implants for long-term bidirectional interfacing of peripheral nerves at sub-nerve resolutions. Nat. Commun. 15, 7523 (2024).
Yao, M. et al. Microgel reinforced zwitterionic hydrogel coating for blood-contacting biomedical devices. Nat. Commun. 13, 5339 (2022).
Sun, H. et al. Liquid-based encapsulation for implantable bioelectronics across broad pH environments. Nat. Commun. 16, 1019 (2025).
Mariello, M. et al. Hermetic, hybrid multilayer, sub-5µm-thick encapsulations prepared with vapor-phase infiltration of metal oxides in conformal polymers for flexible bioelectronics. Adv. Funct. Mater. 34, 2403973 (2024).
Chung, Y. et al. Gigantic triboelectric power generation overcoming acoustic energy barrier using metal–liquid coupling. Joule 8, 2681–2695 (2024).
Zhang, Y. et al. Advances in bioresorbable materials and electronics. Chem. Rev. 123, 11722–11773 (2023).
Zhang, H. et al. Intrinsically low dielectric constant and low dielectric loss polyimides enabled by rigid-soft structure. Chin. J. Polym. Sci. 43, 1222–1230 (2025).
Maaß, M., Griessner, A., Steixner, V. & Zierhofer, C. Reduction of eddy current losses in inductive transmission systems with ferrite sheets. Biomed. Eng. Online 16, 3 (2017).
Jeon, S. et al. Ultrasound-driven highly stable implantable triboelectric nanogenerator with human-tissue acoustic impedance-matched polyether ether ketone. Adv. Mater. Technol. 9, 2400317 (2024).
Meng, X. et al. Self-contracting, battery-free triboelectric wound healing strip with strong wet adhesion. Nat. Commun. 16, 7220 (2025).
Mu, Q. et al. Force-triggered rapid microstructure growth on hydrogel surface for on-demand functions. Nat. Commun. 13, 6213 (2022).
Sun, Z. et al. A bioabsorbable mechanoelectric fiber as electrical stimulation suture. Nat. Commun. 15, 8462 (2024).
Yi, J. et al. Water-responsive supercontractile polymer films for bioelectronic interfaces. Nature 624, 295–302 (2023).
Wang, X. et al. Lateral and angular misalignments of coil in wireless power transfer system. Sens. Actuator A 341, 113577 (2022).
Lim, J., Zoss, P. A., Powley, T. L., Lee, H. & Ward, M. P. A flexible, thin-film microchannel electrode array device for selective subdiaphragmatic vagus nerve recording. Microsyst. Nanoeng. 10, 16 (2024).
Zhang, X. et al. Ingestible optoelectronic capsules enable bidirectional communication with engineered microbes for controllable therapeutic interventions. Nat. Microbiol. 10, 1841–1853 (2025).
Lu, B. et al. Pure PEDOT:PSS hydrogels. Nat. Commun. 10, 1043 (2019).
Choi, M. et al. Light-guiding hydrogels for cell-based sensing and optogenetic synthesis in vivo. Nat. Photon. 7, 987–994 (2013).
Shin, J. et al. Bioresorbable optical sensor systems for monitoring of intracranial pressure and temperature. Sci. Adv. 5, eaaw1899 (2019).
Chahari, M., Salman, E., Stanacevic, M., Willing, R. & Towfighian, S. Performance of piezoelectric and triboelectric transducers under gait loading for energy harvesting and load monitoring in total knee replacements. Nano Energy 141, 111117 (2025).
Wu, J., Liu, H., Chen, W., Ma, B. & Ju, H. Device integration of electrochemical biosensors. Nat. Rev. Bioeng. 1, 346–360 (2023).
Seo, D. et al. Wireless recording in the peripheral nervous system with ultrasonic neural dust. Neuron 91, 529–539 (2016).
Yang, Q. et al. Photocurable bioresorbable adhesives as functional interfaces between flexible bioelectronic devices and soft biological tissues. Nat. Mater. 20, 1559–1570 (2021).
Zebda, A. et al. Single glucose biofuel cells implanted in rats power electronic devices. Sci. Rep. 3, 1516 (2013).
Rao, Y. et al. Fabrication and characterization of a thermoelectric generator with high aspect ratio thermolegs for electrically active implants. Adv. Mater. Technol. 9, 2301157 (2024).
Yu, H. et al. Flexible temperature-pressure dual sensor based on 3D spiral thermoelectric Bi2Te3 films. Nat. Commun. 15, 2521 (2024).
Flavin, M. T. et al. Bioelectronics for targeted pain management. Nat. Rev. Electr. Eng. 2, 407–424 (2025).
Olenik, S., Lee, H. S. & Guder, F. The future of near-field communication-based wireless sensing. Nat. Rev. Mater. 6, 286–288 (2021).
Stuart, T., Cai, L., Burton, A. & Gutruf, P. Wireless and battery-free platforms for collection of biosignals. Biosens. Bioelectron. 178, 113007 (2021).
Soares, I. V. et al. Wireless powering efficiency of deep-body implantable devices. IEEE Trans. Microw. Theory Tech. 71, 2680–2692 (2023).
Jiang, L. & Wu, J. Emerging ultrasonic bioelectronics for personalized healthcare. Prog. Mater. Sci. 136, 101110 (2023).
Yan, F. & Liu, J. Analog front-end input-impedance boosting techniques for bio-potential monitoring—a review. IEEE Trans. Instrum. Meas. 73, 1–14 (2024).
Tang, H. et al. Injectable ultrasonic sensor for wireless monitoring of intracranial signals. Nature 616, 123–128 (2024).
Zhang, H. et al. Wireless, battery-free optoelectronic systems as subdermal implants for local tissue oximetry. Sci. Adv. 5, eaaw0873 (2019).
Fan, S. et al. An ultralow quiescent current power management system with maximum power point tracking (MPPT) for batteryless wireless sensor applications. IEEE Trans. Power Electron 33, 7326–7337 (2018).
Cho, M. et al. Fully bioresorbable hybrid opto-electronic neural implant system for simultaneous electrophysiological recording and optogenetic stimulation. Nat. Commun. 15, 2000 (2024).
Kim, H. W. et al. Transparent, metal-free PEDOT:PSS neural interfaces for simultaneous recording of low-noise electrophysiology and artifact-free two-photon imaging. Nat. Commun. 16, 4032 (2025).
Lu, D. et al. Bioresorbable, wireless, passive sensors as temporary implants for monitoring regional body temperature. Adv. Healthc. Mater. 9, 2000942 (2020).
Bailey, W. H. et al. Synopsis of IEEE Std C95.1TM-2019 ‘IEEE standard for safety levels with respect to human exposure to electric, magnetic, and electromagnetic fields, 0 Hz to 300 GHz’. IEEE Access 7, 171346–171356 (2019).
Song, K. et al. Subdermal flexible solar cell arrays for powering medical electronic implants. Adv. Healthc. Mater. 5, 1572–1580 (2016).
Shadid, R. & Noghanian, S. A literature survey on wireless power transfer for biomedical devices. Int. J. Antennas Propag. 2018, 1–11 (2018).
Shankar, H. & Pagel, P. S. Potential adverse ultrasound-related biological effects: a critical review. Anesthesiology 115, 1109–1124 (2011).
Lazaro, A., Boada, M., Villarino, R. & Girbau, D. Study on the reading of energy-harvested implanted NFC tags using mobile phones. IEEE Access 8, 2200–2221 (2020).
Lee, D.-M. et al. Ultrasound-mediated triboelectric nanogenerator for powering on-demand transient electronics. Sci. Adv. 8, eabl8423 (2022).
Liu, Z. et al. A self-powered intracardiac pacemaker in swine model. Nat. Commun. 15, 507 (2024).
Fang, H. et al. Ultrathin, transferred layers of thermally grown silicon dioxide as biofluid barriers for biointegrated flexible electronic systems. Proc. Natl Acad. Sci. USA 113, 11682–11687 (2016).
Dong, G. et al. Oriented Bi2Te3-based films enabled high performance planar thermoelectric cooling device for hot spot elimination. Nat. Commun. 15, 9695 (2024).
Nithianandam, P. et al. Flexible, miniaturized sensing probes inspired by biofuel cells for monitoring synaptically released glutamate in the mouse brain. Angew. Chem. Int. Ed. 62, e202310245 (2023).
Karan, S. K. et al. Magnetic field and ultrasound induced simultaneous wireless energy harvesting. Energy Environ. Sci. 17, 2129–2144 (2024).
Kiani, M. & Ghovanloo, M. An RFID-based closed-loop wireless power transmission system for biomedical applications. IEEE Trans. Circuits Syst. II 57, 260–264 (2010).
Azimi, S. et al. Self-powered cardiac pacemaker by piezoelectric polymer nanogenerator implant. Nano Energy 83, 105781 (2021).
Beck, H. et al. 50th anniversary of the first successful permanent pacemaker implantation in the United States: historical review and future directions. Am. J. Cardiol. 106, 810–818 (2010).
Shichiri, M., Yamasaki, Y., Kawamori, R., Hakui, N. & Abe, H. Wearable artificial endocrine pancreas with needle-type glucose sensor. The Lancet 320, 1129–1131 (1982).
Bartels, J. et al. Neurotrophic electrode: method of assembly and implantation into human motor speech cortex. J. Neurosci. Methods 174, 168–176 (2008).
Bhatia, S. N. et al. Wireless, battery-free and fully implantable organ interfaces. Chem. Rev. 124, 2205–2280 (2024).
Herbert, R., Lim, H.-R., Rigo, B. & Yeo, W.-H. Fully implantable wireless batteryless vascular electronics with printed soft sensors for multiplex sensing of hemodynamics. Sci. Adv. 8, eabm1175 (2022).
Li, S. et al. Bioresorbable, wireless, passive sensors for continuous pH measurements and early detection of gastric leakage. Sci. Adv. 10, eadj0268 (2024).
Madhvapathy, S. R. et al. Miniaturized implantable temperature sensors for the long term monitoring of chronic intestinal inflammation. Nat. Biomed. Eng. 8, 1040–1052 (2024).
Lee, S. et al. Ultrasoft electronics to monitor dynamically pulsing cardiomyocytes. Nat. Nanotechnol. 14, 156–160 (2019).
Kang, S.-K. et al. Bioresorbable silicon electronic sensors for the brain. Nature 530, 71–76 (2016).
Ho, J. S. et al. Wireless power transfer to deep-tissue microimplants. Proc. Natl Acad. Sci. USA 111, 7974–7979 (2014).
Piech, D. K. et al. A wireless millimetre-scale implantable neural stimulator with ultrasonically powered bidirectional communication. Nat. Biomed. Eng. 4, 207–222 (2020).
Kim, D.-H. et al. Materials for multifunctional balloon catheters with capabilities in cardiac electrophysiological mapping and ablation therapy. Nat. Mater. 10, 316–323 (2011).
Jun, J. J. et al. Fully integrated silicon probes for high-density recording of neural activity. Nature 551, 232–236 (2017).
Zhou, A. et al. A wireless and artefact-free 128-channel neuromodulation device for closed-loop stimulation and recording in non-human primates. Nat. Biomed. Eng. 3, 15–26 (2019).
Berényi, A., Belluscio, M., Mao, D. & Buzsáki, G. Closed-loop control of epilepsy by transcranial electrical stimulation. Science 337, 735–737 (2012).
Tinkhauser, G. et al. The modulatory effect of adaptive deep brain stimulation on beta bursts in Parkinson’s disease. Brain 140, 1053–1067 (2017).
Guo, L. et al. 68-channel neural signal processing system-on-chip with integrated feature extraction, compression, and hardware accelerators for neuroprosthetics in 22 nm FDSOI. Front. Neurosci. 18, 1432750 (2024).
Lee, J. et al. Neural recording and stimulation using wireless networks of microimplants. Nat. Electron. 4, 604–614 (2021).
Wang, M. L. & Arbabian, A. Exploiting spatial degrees of freedom for high data rate ultrasound communication with implantable devices. Appl. Phys. Lett. 111, 133503 (2017).
Luan, L. et al. Ultraflexible nanoelectronic probes form reliable, glial scar-free neural integration. Sci. Adv. 3, e1601966 (2017).
Wu, J. et al. Adhesive anti-fibrotic interfaces on diverse organs. Nature 630, 360–367 (2024).
Richter, A. et al. Improved silicon surface passivation by ALD Al2O3/SiO2 multilayers with in-situ plasma treatments. Adv. Mater. Interfaces 10, 2202469 (2023).
Driscoll, N. et al. MXene-infused bioelectronic interfaces for multiscale electrophysiology and stimulation. Sci. Transl. Med. 13, eabf8629 (2021).
Zhang, L. et al. Skin‑inspired, sensory robots for electronic implants. Nat. Commun. 15, 4777 (2024).
Kim, N. et al. Elastic conducting polymer composites in thermoelectric applications. Nat. Commun. 11, 1424 (2020).
Qin, S. et al. Triboelectric sensor with ultra-wide linear range based on water-containing elastomer and ion-rich interface. Nat. Commun. 15, 10640 (2024).
Li, Z. et al. Gelatin methacryloyl-based capacitive tactile sensor for medical wearables. Adv. Funct. Mater. 30, 2003601 (2020).
Song, J. et al. Hydrogel-based flexible materials for diabetes diagnosis, treatment, and management. npj Flex. Electron. 5, 26 (2021).
Xu, C. et al. A physicochemical-sensing electronic skin for stress response monitoring. Nat. Electron. 7, 168–179 (2024).
Zhou, Y. et al. Giant magnetoelastic effect in soft systems for bioelectronics. Nat. Mater. 20, 1670–1676 (2021).
Kutbee, A. T. et al. Flexible and biocompatible high-performance solid-state micro-battery for implantable orthodontic system. npj Flex. Electron. 1, 7 (2017).
Tsang, M. et al. Biodegradable magnesium/iron batteries with polycaprolactone encapsulation: a microfabricated power source for transient implantable devices. Microsyst. Nanoeng. 1, 15024 (2015).
Liu, X. et al. An ultrasound-driven implantable wireless energy harvesting system using a triboelectric transducer. Matter 5, 4315–4331 (2022).
Ryu, H. et al. Self-rechargeable cardiac pacemaker system with triboelectric nanogenerators. Nat. Commun. 12, 4374 (2021).
Shi, C. et al. Application of a sub–0.1-mm3 implantable mote for in vivo real-time wireless temperature sensing. Sci. Adv. 7, eabf6312 (2021).
Khalifa, A. et al. The microbead: a 0.009 mm³ implantable wireless neural stimulator. IEEE Trans. Biomed. Circuits Syst. 13, 971–985 (2019).
Shah, S. A. A. & Yoo, H. Radiative near-field wireless power transfer to scalp-implantable biotelemetric device. IEEE Trans. Microw. Theory Tech. 68, 2944–2953 (2020).
Agrawal, D. et al. Conformal phased surfaces for wireless powering of bioelectronic microdevices. Nat. Biomed. Eng. 1, 0043 (2017).
Kim, T.-I. et al. Injectable, cellular-scale optoelectronics with applications for wireless optogenetics. Science 340, 211–216 (2013).
Park, S. I. et al. Ultraminiaturized photovoltaic and radio frequency powered optoelectronic systems for wireless optogenetics. J. Neural Eng. 12, 056002 (2015).
Yener, U. C., Toymus, A. T., Esat, K., Alem, M. & Beker, L. Passive ultrasonic communication link for deep-tissue sensor implants. Device 3, 100755 (2025).
Ghanbari, M. M. et al. A sub-mm3 ultrasonic free-floating implant for multi-mote neural recording. IEEE J. Solid State Circuits 54, 3017–3030 (2019).
Standard for Real-time Display of Thermal and Mechanical Acoustic Output Indices on Diagnostic Ultrasound Equipment NEMA UD 3-2004 (NEMA, 2004).
Yu, M. et al. Self-closing stretchable cuff electrodes for peripheral nerve stimulation and electromyographic signal recording. ACS Appl. Mater. Interfaces 15, 7663–7672 (2023).
Ozeri, S., Shmilovitz, D., Singer, S. & Wang, C.-C. Ultrasonic transcutaneous energy transfer using a continuous wave 650 kHz Gaussian shaded transmitter. Ultrasonics 50, 666–674 (2010).
Guo, H. et al. Wireless implantable optical probe for continuous monitoring of oxygen saturation in flaps and organ grafts. Nat. Commun. 13, 3009 (2022).
Song, M. et al. A 1.66 Gb/s and 5.8 pJ/b transcutaneous IR-UWB telemetry system with hybrid ASK-PWM-UWB signaling for high-channel-count brain–machine interfaces. In 2022 IEEE International Solid-State Circuits Conference (ISSCC) 394–396 (IEEE, 2022).
Jung, D. et al. Sustainably powered, multifunctional flexible feedback implant by the bifacial design and si photovoltaics. Adv. Healthc. Mater. 10, 2001480 (2021).
Ahnood, A. et al. Diamond encapsulated photovoltaics for transdermal power delivery. Biosens. Bioelectron. 77, 589–597 (2016).
Song, K., Han, J. H., Yang, H. C., Nam, K. I. & Lee, J. Generation of electrical power under human skin by subdermal solar cell arrays for implantable bioelectronic devices. Biosens. Bioelectron. 92, 364–371 (2017).
Kim, J. et al. Active photonic wireless power transfer into live tissues. Proc. Natl Acad. Sci. USA 117, 16856–16863 (2020).
Acknowledgements
We acknowledge that this work was supported by the Basic Research Program (RS-2022-NR070716, RS-2025-18362970) and the Nano & Material Technology Development Program (RS-2025-25441823) through the National Research Foundation of Korea (NRF) funded by Ministry of Science and ICT.
Author information
Authors and Affiliations
Contributions
All authors contributed to discussions and editing of the paper. X.M., X.X., Y.H.K. and S.-W.K. conceived of and structured the article. Y.P. and Y.J.J. conducted literature collection and prepared the tables and references. X.M., X.X. and Y.H.K. drafted the paper. S.-W.K. revised and refined the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Sensors thanks Ada Shuk Yan Poon and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Table 1.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Meng, X., Xiao, X., Kwon, Y.H. et al. Wireless and battery-free implantable sensing technologies for translatable bioelectronics. Nat. Sens. 1, 290–304 (2026). https://doi.org/10.1038/s44460-026-00054-y
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44460-026-00054-y