Abstract
Mitochondrial diseases represent one of the most prevalent and debilitating categories of hereditary disorders, characterized by significant genetic, biological, and clinical heterogeneity, which has driven the development of the field of engineered mitochondria. With the growing recognition of the pathogenic role of damaged mitochondria in aging, oxidative disorders, inflammatory diseases, and cancer, the application of engineered mitochondria has expanded to those non-hereditary contexts (sometimes referred to as mitochondria-related diseases). Due to their unique non-eukaryotic origins and endosymbiotic relationship, mitochondria are considered highly suitable for gene editing and intercellular transplantation, and remarkable progress has been achieved in two promising therapeutic strategies—mitochondrial gene editing and artificial mitochondrial transfer (collectively referred to as engineered mitochondria in this review) over the past two decades. Here, we provide a comprehensive review of the mechanisms and recent advancements in the development of engineered mitochondria for therapeutic applications, alongside a concise summary of potential clinical implications and supporting evidence from preclinical and clinical studies. Additionally, an emerging and potentially feasible approach involves ex vivo mitochondrial editing, followed by selection and transplantation, which holds the potential to overcome limitations such as reduced in vivo operability and the introduction of allogeneic mitochondrial heterogeneity, thereby broadening the applicability of engineered mitochondria.
Similar content being viewed by others
Introduction
Mitochondria are derived from an endosymbiotic progenitor, and a significant portion of the genetic material possessed by the endosymbiont was either lost or integrated into the host genome throughout evolution.1,2 This integration ultimately resulted in the emergence of mitochondria as distinct cellular organelles. Due to their endosymbiotic origin, mitochondria possess two distinct membranes: the outer and inner membranes, which enclose the intermembrane gap and the innermost matrix, respectively. The inner membrane creates significant indentations, known as cristae, where most of the composites of the respiratory chain are primarily located. Numerous investigations have shed light on the pivotal role that mitochondria play in energy conversion, biosynthesis, and signal transduction. The view of mitochondria has evolved from adenosine triphosphate (ATP)-synthesizing machines serving in oxidative phosphorylation3,4 to multifunctional organelles.5 Mitochondria are involved in several pathways and processes, including metabolism (ATP,3,4 amino acids,6,7 lipids,8,9,10,11,12,13 ascorbate,14 carbonate,15 reactive oxygen species (ROS),16,17 sulfide,18,19,20 the iron–sulfur cluster,21,22,23 tricarboxylic acid and its derivatives,24,25,26 and one-carbon metabolism27,28,29,30), signal transduction31 (sensing,32,33,34 integrating,35 and signaling36,37), substance transport (calcium,38,39,40 sodium,38,41 mitochondrial protein,42 mitochondrial-derived vesicles,43,44 mitochondrial permeability45,46), redox homeostasis,47,48 inflammations,49 heat production,50,51,52 and cell death regulation.53 In addition, mitochondria dynamically recalibrate their biology according to organismal demands and stress, highlighting their multifaceted and complicated traits. Mitochondrial dysfunction and related molecular pathways were reviewed by Zong et al.54
Mitochondria are organelles with uniparental inheritance55 and possess their own genome. Mutations in mitochondria can lead to the occurrence of severe diseases,56,57 and mitochondrial diseases are a prevailing and devastating category of hereditary genetic disorders that exhibit clinical variability, diagnostic intricacies, and a lack of disease-modifying therapeutic interventions.58 Approximately 1000 children born in the United States and 200 children born in the United Kingdom each year are affected by mitochondrial disease.59 Recent technological advancements have encouraged the treatment of these disorders through two unique strategies for germline therapy60,61,62: in cases where oocytes or zygotes are susceptible to mitochondrial defects, potential solutions include remedial genome editing or substitution with unaffected mitochondria. In this review, genetically edited mitochondria and isolated mitochondria with or without modifications before transplantation are collectively referred to as “engineered mitochondria”. With the identification of the pathogenic effects of damaged mitochondria in increasingly common diseases, such as aging,63 oxidative disorders,64 inflammatory diseases,65 and cancer,66 the application potential of engineered mitochondria has expanded significantly (Fig. 1).
A schematic view of engineered mitochondria in disease therapy. A schematic view of engineered mitochondria in disease therapy. Mitochondrial gene editing precisely addresses abnormalities in mitochondria by clearing or repairing the mutant genome, while artificial mitochondrial transfer restores the activity level of mitochondria by adding functional mitochondria. (Generated by the authors with Adobe Illustrator)
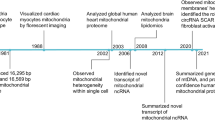
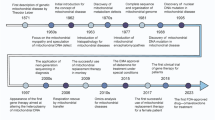
In the past decades, encouraging results have been achieved with mitochondrial gene editing and artificial mitochondrial transfer, two distinctive approaches that have developed in parallel. (Fig. 2) The timeline illustrates milestones in the important study of mitochondrial gene editing. In 1988, the identification of the first pathogenic mitochondrial DNA (mtDNA) mutation laid the foundation for understanding mitochondrial diseases.56 By 2001, researchers achieved mtDNA editing in vitro using mitochondrial restriction endonucleases (mitoREs).67 In 2008, mitochondrial zinc-finger nucleases (mitoZFNs) were used to achieve heteroplasmy shifting, which is a key step in altering the ratio of mutant to wild-type mtDNA.68 By 2013, this approach evolved into mitochondrial transcriptional activator-like effector nucleases (mitoTALENs), realizing the heteroplasmy shifting in vitro.69 In 2021, the development of mitochondrial-targeted meganucleases (mitoARCUS) enabled heteroplasmy shifting in vivo, a major step towards therapeutic applications.70 In 2022, researchers designed a new type of gene editing tools in vivo using a double-stranded DNA cytosine base editor (DdCBE) to enable novel mtDNA point mutations, and a de novo point mutation from A to G using TALE-linked deaminases (TALEDs).71 Both technologies further expand the toolkit for precision editing of the mtDNA. These milestones reflect the rapid progress in mitochondrial research and pave the way for potential therapeutic interventions in mitochondrial diseases. The journey of artificial mitochondrial transfer began in 1982 with Clark and Shay72 discovering that the transplantation of mitochondria containing antibiotic resistance genes into susceptible cells facilitated their survival in selective media, thereby establishing a novel avenue for research in this field. In 1997, the first human pregnancy after ooplasmic transfer (OT) marked a significant milestone,73 demonstrating the potential for mitochondrial manipulation in reproductive medicine. The detection of organelles moving between mammalian cells via tunneling nanotubes in 200474 provided insights into the mechanism of intercellular mitochondrial dynamics. By 2006, the discovery of normal mitochondria transferred from mesenchymal stem cells to mammalian cells75 led to numerous subsequent investigations of cellular repair and regeneration via mitochondrial transplantation. In 2014, the identification of extracellular vesicles containing mitochondria and free mitochondria from platelets76 suggested a new route of administration. The approval of mitochondrial donation techniques in the United Kingdom in 2015, with the first individual born using this method in Mexico,77 represented a major regulatory and clinical breakthrough. In 2021, the first reported mitochondrial transfer via the migrasome,78 introduced a novel mechanism for mitochondrial exchange. In 2024, research demonstrated that exogenous mitochondria can promote the function of endogenous mitochondria, providing new perspective on the therapeutic effect of transferred mitochondria.79,80
The development of mitochondrial engineering. This milestone timeline provides an overview of the major advancements associated with mitochondrial engineering over the past four decades. Mitochondrial gene editing has progressed from the initial identification of pathogenic mtDNA to the modification of mutants using various nuclease and base editing tools (purple background). Artificial mitochondrial transfer has evolved from the initial achievement of artificial mitochondrial transfer to the initiation of large-scale clinical trials (blue background). DdCBE DddA-derived cytosine base editor, mitoARCUS mitochondrial-targeted meganucleases, mitoREs mitochondrial-targeted restriction endonucleases, mitoTALEN mitochondrial-targeted transcription activator-like effector nucleases, mitoZFNs mitochondrial-targeted zinc finger nucleases, mtDNA mitochondrial DNA, TALEDs TALE-linked deaminases (Generated by the authors with Adobe Illustrator)
Engineered mitochondria have become a potential solution in regenerative medicine and for treating multiple diseases. In this paper, we comprehensively review the mechanisms and advances in engineered mitochondria for disease therapy and summarize the potential clinical applications and evidence from clinical trials.
Mechanisms and progress of engineered mitochondria
Mitochondrial gene editing
Mitochondrial transcription and translation
The human mitochondrial genome is a circular, double-stranded deoxyribonucleic acid (DNA) molecule encoding 2 ribosomal ribonucleic acids (RNAs), 22 transfer RNAs, and 13 proteins involved in mitochondrial oxidative phosphorylation (OXPHOS).81,82 The entire mitochondrial genome is transcribed from both strands, resulting in the production of extensive polycistronic transcripts. The classification of these strands is determined by their buoyancy in the density gradients of cesium chloride, resulting in the designation of heavy chains (H) or light chains (L).83,84,85 Initial transcription of mtDNA is driven by mitochondrial DNA-directed RNA polymerase (POLRMT), mitochondrial transcription factor A (TFAM) and mitochondrial transcription factor B2 (TFB2M), and during the elongation stage, transcription elongation factor (TEFM) promotes the processivity of POLRMT, and ultimately mtDNA undergoes bending and base-flipping promoted by mitochondrial termination factor 1 to induce transcription termination. Long polycistronic transcripts require multiple processing steps to form functional RNA species.86,87 Most mt-ribosomal ribonucleic acids (mt-rRNAs) and mt-messenger ribonucleic acids (mt-mRNAs) are separated by mt-tRNAs in precursor polycistronic transcripts, and these RNAs undergo different further modifications after being cleaved.88,89,90,91 In the translation apparatus, mRNAs, transfer ribonucleic acids (tRNAs), and the assembled mitoribosome converge, where translational factors govern the advancement of translation.92,93 Initial translation of mRNA matured by mtDNA post-transcriptional processing is driven by the mitochondrial initiation factors mtIF2 and mtIF3, and in the translation elongation phase, codon-anticodon site base pairing is carried out by the mitochondrial elongation factors EFTu (TUFM), EFTs (TSFM), and EFGM (GFM1) in a complex formed with the mitochondrial tRNAs, and ultimately, the stop codon at the A-site triggers the termination of translation and the release of peptides. Currently, the main strategies for mitochondrial gene editing are mtDNA or mitochondrial RNA (mtRNA) engineering (Fig. 3).
Schematic representation of mitochondrial gene editing strategies. mtDNA replication and transcription initiation: TFAM drives mtDNA replication and transcription, while POLRMT and TFB2M help form the initiation complex at the promoter region. Transcription elongation: After the dissociation of TFAM and TFB2M, POLRMT binds to TEFM for transcription elongation, producing longer transcripts. mtDNA mutant correction: mtDNA mutations are targeted and corrected by nuclease and base editors or suppressed during replication by blockers. Transcription maturation: Transcripts undergo maturation via hydrolytic cleavage by RNase P and ELAC2. a mtDNA programmable nucleases: A depiction of programmable nucleases, such as CRISPR-Cas9, designed to target specific sequences in the mitochondrial DNA (mtDNA), enabling precise editing or repair of genetic mutations within the mitochondria. b mtDNA base editors: Schematic showing the use of base editors to induce point mutations in mtDNA without causing double-strand breaks, offering a safer alternative for correcting specific genetic mutations. c mtDNA anti-replication: Illustration of strategies to inhibit mtDNA replication, including the use of specific blockers that prevent the replication of mutant mtDNA, thus controlling the spread of mutations. d mtRNA regulation: Depiction of methods for regulating mitochondrial RNA (mtRNA) expression, including the use of antisense oligonucleotides (ASOs), RNA interference (RNAi), or RNA-targeting nucleases to modulate mitochondrial gene expression. e mtRNA substitution: Illustration of the substitution strategy for defective mitochondrial RNAs (mtRNA), such as mt-mRNA, mt-tRNA, and mt-rRNA, to restore normal mitochondrial function by replacing mutated or defective RNA species with functional ones. ASOs antisense oligonucleotides, ELAC2 elaC ribonuclease Z2, HSD17B10 hydroxysteroid (17-beta) dehydrogenase 10, mtDNA mitochondrial DNA, mtRNA mitochondrial RNA, PIN PilT N-terminus, POLRMT mitochondrial DNA-directed RNA polymerase, PRORP proteinaceous RNase P, PUF Pumilio and FBF, RISC RNA-induced silencing complex, RNase P endonuclease P, RNAi RNA interference, shRNA short hairpin RNA, siRNA small interfering RNA, TEFM transcription elongation factor, TFAM mitochondrial transcription factor A, TFB2M mitochondrial transcription factor B2, TRMT10C tRNA methyltransferase 10C (Generated by the authors with Adobe Illustrator)
mtDNA engineering
The correlation between the quality of mtDNA and the function of mitochondria is considerable, and mutations in mtDNA could result in hereditary mitochondrial diseases.94,95 Unfortunately, the mutation susceptibility of mtDNA outweighs that of nuclear DNA for reasons including the proximity of mtDNA to the OXPHOS system,96,97,98 the limited number of DNA repair mechanisms inside mitochondria, the absence of canonical protection proteins, and the high replication and mistake rate.99,100,101 Pathogenic mutations in mtDNA lead to one of two states: heteroplasmy or homoplasmy. Heteroplasmic mutation is characterized by the coexistence of pathogenic mtDNA mutations with unaffected mtDNA molecules. Homoplasmic mutation refers to the condition in which cells only possess mutant mtDNA (devoid of wild-type mtDNA). In recent years, mitochondrial gene editing has substantially advanced the use of anti-replicative agents, programmable nucleases, and base editors.102,103,104 For heteroplasmic mutations, strategies include shifting the heteroplasmy ratio by selectively degrading mutant mtDNA or enhancing the replication of wild-type mtDNA.67 Restriction enzymes or programmable nucleases could shift the ratio of heteroplasmic mutations, while these strategies are unable to repair homoplasmic mutations.105 For homoplasmic mutations, techniques such as allotropic expression are being explored.96 The base editors have the potential to correct both heteroplasmic and homoplasmic mtDNA mutation71 (Fig. 4).
Structure and function of typical mtDNA editing tools. The general architecture of editing tools with programmable nucleases, including mitoREs, mitoARCUS, mtZFN, and mtTALEN (purple background). mitoREs were the first tools used for mtDNA editing, consisting of MTS and endonucleases. mitoARCUS utilizes a significantly modified and simplified I-CreI homing endonuclease. For mitoZFNs and mitoTALENs, the ZFN or TALE domains are utilized to guide the Fok1 restriction endonuclease to target particular gene sequences in mtDNA. mitoCRISPR comprises an sgRNA and a Cas9 endonuclease. The schematic structure of editing tools with base editors, including DdCBEs, TALEDs, and ZFDs (blue background). DdCBEs contain programmable TALE and UGI, enabling the first C–G to T–A conversion. TALEDs utilize adenine deaminase TadA8e and DddA, promoting the conversion of A–G bases in mtDNA. ZFDs consisting of the zinc finger DNA-binding protein DddA and UGI, catalyze C-to-T conversion. AD adenosine deaminase; DdCBEs DddA-derived cytosine base editors; mitoARCUS mitochondrial-targeted meganuclease; mitoCRISPR mitochondrial-targeted clustered regularly interspaced short palindromic repeats; mitoREs mitochondrial-targeted restriction endonucleases; mitoTALENs mitochondrial-targeted transcription activator-like effector nucleases; mitoZFNs mitochondrial-targeted zinc finger nucleases; mtDNA mitochondrial DNA; MTS mitochondrial targeting sequence; PAM protospacer-adjacent motif; sgRNA guide RNA; TALEDs TALE-linked deaminases; UGI uracil glycosylase inhibitor; ZFDs zinc finger deaminases; ZFP zinc finger proteins (Generated by the authors with Adobe Illustrator)
mtDNA engineering with anti-replicative agents
The targeted disruption of pathogenic mtDNA replication can be achieved through specific small molecules capable of annealing with mutant sites. The first reported approach for shifting mtDNA heteroplasmy involves disrupting mtDNA replication by using peptide nucleic acid oligomers (PNAs).106 These small molecules with nucleobases linked to an achiral peptide backbone exhibit higher affinity than equivalent oligodeoxynucleotides when binding to single-stranded complementary DNA.107 However, delivering PNAs across the inner mitochondrial membrane is challenging.107 Another approach was developed using anti-replicative oligoribonucleotides that can be as efficient as synthetic PNAs.107 Because 5S rRNA is partially imported into human mitochondria,108 recombinant 5S rRNA molecules, which use 5S rRNA as a vector to deliver anti-replicative oligoribonucleotides, were specifically developed to regulate mtDNA heteroplasmy.109 However, these anti-replication agents were developed in vitro replication under physiological conditions, no ideal experimental results have been observed on either cell culture or isolated mitochondria. Mitochondria are highly dynamic, undergoing fusion and fission, and there is currently no consensus on the molecular mechanisms of nucleic acid trafficking within mitochondria or its potential functions. This gap in understanding complicates the stability of changes introduced by anti-replicative strategies.
mtDNA engineering with programmable nucleases
The presence of heteroplasmic characteristics in most mitochondrial disorders, the swift breakdown of mtDNA molecules that contain double-strand breaks (DSBs), and the precise regulation of mtDNA copy number give rise to a distinctive therapeutic concept known as “heteroplasmy shifting.” The use of programmable nucleases is the most widespread and efficient method.110,111,112,113,114 This approach relies on the specific cleavage of endonucleases at mutation sites in mtDNA, with the aim of degrading the entire pathogenic mtDNA.115 The absence of a nuclease recognition site in wild-type mtDNA results in its preservation, enabling the progressive replacement of mutant copies and subsequent restoration of mitochondrial function by wild-type mtDNA.69,116,117 mitoREs, mitoTALENs, mitoZFNs, and mitoARCUS are comprehensively described in the subsequent sections.
mitoCRISPR. The clustered regularly interspaced short palindromic repeats (CRISPR)/CRISPR-associated protein 9 (Cas9) system for nuclear gene editing was initially introduced in 2013 and can achieve sequence-specific DNA recognition and cleavage.118,119 The bacterial immune system incorporates CRISPR/Cas9 as a mechanism to safeguard cells against exogenous DNA.119,120,121,122,123,124 The system comprises a single guide RNA (sgRNA) that selectively identifies the gene sequence and a Cas9 endonuclease that produces DSBs at the adjacent motif.125,126,127,128,129,130 CRISPR/Cas9 has been successfully employed to specifically target the Cox1 and Cox3 loci within mtDNA, disrupting the mitochondrial membrane and subsequently inhibiting cell growth.131,132 Researchers have made progress in the targeted cleavage of mtDNA in HEK293T cells and zebrafish.133,134
However, this method is unsuitable for modifying the mammalian mitochondrial genome. Introducing sgRNA into human mitochondria remains challenging, limiting its widespread acceptance.135,136 The current lack of resolution in addressing this limitation highlights the inefficiency of this approach. Several researchers have made noteworthy efforts in the delivery of sgRNA; nevertheless, none of the studies conducted thus far have resulted in a shift in heteroplasmy, and this approach has consequently failed to achieve widespread acceptance within the research community.133,134,137 Until recently, the application of CRISPR-based systems for mtDNA manipulation has not been consistent.135,138,139,140
mitoREs. mitoREs were an early tool used to create site-specific DNA DSBs to cleave gene sequences. Pathogenic mtDNA variants can lead to the development of distinct restriction sites, and these tools can efficiently eliminate pathogenic mtDNA and facilitate heteroplasmy shifting. Certain nucleases, such as mitoPstI and mitoApaLI, can transport mitochondrial target signals (MTSs) via the mitochondrial transport machinery.67,141 Once they enter the mitochondria, mitoREs specifically target restriction sites in the mutant, causing targeted breaks that lead to mtDNA elimination and heteroplasmy shifting. These tools have shown promising results in in vitro models of primary mitochondrial diseases and in vivo models, such as mice with asymptomatic mtDNA heteroplasmy.141,142 In addition, human cybrid cell lines have been used to assess the effectiveness of mitoREs. The application of mitoREs to these cells decreased the extent of the mutation and improved physiological processes, such as oxidative phosphorylation activity and ATP production.143,144,145 Although these tools are effective in gene editing for mtDNA, these approaches have strict sequence limitations, making them the least versatile option in the mitochondrial gene editing toolbox.
mitoZFNs and mitoTALENs. mitoZFNs and mitoTALENs are modified nucleases that specifically target mitochondria. ZFN or transcription activator-like effector (TALE) domains are utilized to guide the Fok1 restriction endonuclease to target particular gene sequences in mtDNA. This approach overcomes the constraint of mitoREs, which are limited by the number of unique recognition sites for restriction endonucleases in the mtDNA variant.69,115,146,147 ZFN dimers cleave mtDNA at precise locations. These ZFNs comprise a sequence-independent endonuclease and a DNA-specific recognition domain.148 The homodimerization of FokI domains in both ZFN and TALEN monomers necessitates a high degree of closeness, leading to DSBs.149 mitoZFNs and mitoTALENs were subsequently created using MTS technology.69,150,151 Nevertheless, the practical application of mitoTALENs is restricted due to their substantial dimensions and heterodimeric composition, necessitating the packaging of each monomeric constituent into an independent viral vector.152
mitoZFNs have been evaluated in vivo using m.5024C > T tRNA-Ala mice. mitoZFNs were packaged with the cardiotropic Adeno-associated viruses (AAV) and administered through intravenous injection to male mice aged 2–8 months.153 The mice were euthanized 65 days after injection and exhibited heteroplasmy in the heart. The levels of tRNA-Ala, lactate, and pyruvate were restored, re-establishing the normal activity of cardiac mitochondria.68,154 In addition, mitoTALENs have been successfully used to detect mitochondrial alterations in mouse embryos.155 In the heteroplasmic NZB/BALB mouse model, the number of NZB mitochondrial genomes was selectively reduced in M-II oocytes utilizing mitoTALENs, preventing NZB mitochondrial genome transmission to the next generation. Following the successful introduction of the pathogenic human m.14459G > A and m.T9176T > C mutations, mouse oocytes were targeted using a relevant set of mitoTALENs in similar attempts. After mRNA microinjection and mitoTALEN expression, total mtDNA levels were not recovered because oocytes and preimplantation embryos cannot replicate mtDNA.156,157 Therefore, given the elevated mutant load of oocytes, the possibility of producing nonviable embryos following a mitonuclease treatment must be considered.
mitoARCUS. mitoARCUS was created to overcome the obstacles presented by the previously mentioned tools.70 ARCUS gene editing technology, created by Precision Biosciences, utilizes a significantly modified and simplified I-CreI homing endonuclease. mitoARCUS can be adjusted to target and cleave nearly any base sequence using computational evolution methods.158 The gene editing technique was evaluated in the MT-TA m.5024C > T animal model, known for reduced tRNA-Ala levels and mitochondrial cardiomyopathy.70 MitoARCUS packed with AAV9 was administered via retro-orbital injection to mice aged 2.5 or 6 weeks, and the mice were euthanized 6–24 weeks after the injection.159 The heteroplasmy levels were evaluated in various organs, including the heart, kidney, liver, and spleen. The most significant change occurred in the liver, possibly because of the pronounced hepatic tropism exhibited by AAV9. Moreover, the levels of tRNA-Ala in the same organ were effectively restored.
mtDNA engineering with base editors
Mito-nucleases can eliminate mutant mtDNA but cannot repair defective genomes. Therefore, these techniques are ineffective in rescuing pathogenic, homoplasmic mutations.160 This situation requires a distinct method to alter mitochondrial DNA.
Mok et al. discovered an interbacterial toxin, DddA, that catalyzes the deamination of cytidines within dsDNA; engineered split-DddA halves were developed that remained inactive until they interacted on target DNA.62 Several mitochondrial base editors have been developed based on this tool, including DdCBEs, TALEDs, and zinc-finger deaminases (ZFDs).
DdCBEs. Mok et al. utilized DddA, a double-stranded DNA deaminase of Burkholderia cepacia origin, and fused and assembled it with a programmable TALE and uracil glycosylase inhibitor (UGI) to generate cytosine base editors of DddA origin, enabling the first specific and efficient C–G to T–A conversion in mtDNA.62 Since DddA is a bacterial enzyme that is toxic to mammalian cells, researchers split DddA into two halves and fused each to a mitochondrion-targeted TALE array, thus ensuring that the two halves regained catalytic activity only when bound together at the targeted gene regions. The dU formed after cytosine deamination is excised by endogenous uracil glycosylase (UNG). Two TALEs are fused to UGI to prevent dU from being excised, and after DNA replication or repair, the final base-pair transition from C–G to T–A is achieved. In addition, DdCBEs have been utilized to construct disease models in human cells with mtDNA mutations, resulting in changes in respiration rates and oxidative phosphorylation. DdCBEs can decrease the proportion of mutant mtDNA without affecting the total number of copies.62 Editing efficiencies vary from 5% to 50%, which has implications for researching mitochondrial diseases and treating them instead of deleting mtDNA copies resulting from specific nuclease activity.62,161
DdCBEs have been effectively introduced into postnatal mice.162 Perdo et al. used AAV9.45 to deliver DdCBEs into the heart tissue of 8-week-old and 1-day-old mice by inducing two specific edits at the MT-Nd3 site: m.9576G > A and m.9577G > A.163 DdCBE-specific edits were detected in both adult and newborn mice after injection. However, significant mitochondrial off-target editing was observed at any time after treatment.164,165 DdCBEs have been used in rats to generate mutations equivalent to the human pathogenic m.8363G > A and m.14710G > A mutations to investigate whether DdCBEs can edit mtDNA in vivo and construct animal models that mimic human mtDNA mutant diseases.163,166,167,168,169 Lee et al. microinjected DdCBE-encoding mRNA into mouse embryos to introduce the m.12336C > T nonsense mutation into the mitochondrial Nd5 gene, mimicking the Leber hereditary optic neuropathy (LHON), and Leigh syndrome in human diseases.170 Shen et al. microinjection of DdCBE-encoding mRNA into zebrafish to generate the m.4247G > A, m.14076G > A, and m.8892G > A, stimulating m.3733G > A, m.13513G > A, and m.8363G > A mutations in human mtDNA to creat various disease models.166 These efforts have provided important animal models for studying clinical gene therapy options and the pathogenic mechanisms of mitochondrial diseases.
TALEDs. The first mitochondrial base editor to achieve A-to-G base editing of human mtDNA used transcriptional activator-like effector-linked deaminases called TALEDs, which were developed after the discovery of DdCBEs.71 Each of the three primary parts of TALEDs has a distinct purpose. TALE, a DNA binding protein that targets particular DNA sequences, is the first component. TALE can attach to particular mitochondrial DNA sequences via mitochondrial localization signals once they enter the mitochondria. The adenine deaminase TadA8e, which promotes the conversion of A–G bases in mtDNA, is involved in the second pathway. DddA, the third component, facilitates the easier editing of mtDNA by TadA8e.171 This base editor targets any A within the spacer region, regardless of the context or strand, with a preference for those located in the central part rather than at the edges. This innovative tool has been evaluated in various human cell lines as a proof-of-concept, successfully editing 17 different target regions within the mitochondrial genome. Although most of the changes did not exhibit any mitochondrial characteristics, the editing of the gene MT-RNR2 resulted in a mutation that conferred chloramphenicol resistance.71 Moreover, when this medication was introduced, the modified cell line ultimately became homoplasmic for the mutation.
ZFDs. ZFDs, consisting of the zinc finger DNA-binding protein DddA and UGI, catalyze C-to-T conversion. ZFDs combine MTS and nuclear output signal sequences, forming a functional ZFD system, achieving base editing efficiencies of 60% in nuclear DNA and 30% in mtDNA.172 Zinc finger arrays (2 × 0.3–0.6 kb) in ZFDs are compact and, therefore, smaller in size than the TALE arrays (2 × 1.7–2 kb) in DdCBE. Additionally, compared to TALEs, zinc fingers lack bulky structural domains at both the C-terminal and N-terminal ends, making them somewhat easier to use: a split DddA can be fused to either end of the zinc finger arrays. Direct delivery of the purified ZFD protein into human cells via electroporation resulted in up to 27% targeted C-to-T conversion. These properties make ZFDs an ideal nuclear and organelle DNA base editing platform.173
Collectively, mitoCRISPR offers simplicity of design, high flexibility, and efficiency in creating targeted DSBs, making it a powerful tool for various genetic modifications. However, it has the highest risk of off-target effects and faces significant delivery challenges due to the lack of effective delivery systems. mitoREs are highly specific for target sequences, which reduces off-target effects, but they have a limited targeting scope and can only recognize a narrow range of sequences. mitoTALENs and mitoZFNs combine high specificity with a broader targeting scope compared to mitoREs, but their construction is complex and difficult in packaging. mitoARCUS provides high specificity and minimal off-target effects, but similar to mitoTALENs and mitoZFNs, it involves challenging engineering processes. DdCBEs enable precise base editing without introducing DSBs, while they have a limited targeting range and can be difficult to deliver effectively. TALED offers precise editing with minimal off-target effects, but its application is limited by the complexity of TALEN design and delivery issues. ZFDs are highly specific and versatile, but their engineering is complex, and they have a higher potential for off-target effects compared to some other tools. Each tool’s relevance to mitochondrial diseases depends on balancing these strengths and weaknesses, with delivery and specificity being critical factors for therapeutic.
Targeted editing of mtDNA is now possible through the specific removal of mtDNA using nucleases. The use of artificial nucleases allows for more versatile design and application. mtDNA base editors may be used in heteroplasmy and homoplasmy situations and can create new mtDNA mutations at particular locations. Thus, these editors are a valuable tool for developing mtDNA mutation models, filling the gap left by existing models. Table 1 outlines the features of the currently available primary mitochondrial DNA editing tools.67,70,71,115,134,150,165,172
mtRNA engineering
The frequency of RNA synthesis is comparable to that of RNA encoded from a single strand of mtDNA.83,174,175,176,177 Nonetheless, gene expression is strictly regulated by posttranscriptional processing and translation.178 With the advances in mitochondrial RNA engineering,179,180,181,182,183 strategies at various genetic levels, such as mtRNA processing, stability, or transcript translation, have become promising for treating mitochondrial diseases178,184 (Fig. 3).
mtRNA engineering with nucleic acid tools
Various nucleic acid tools specifically target RNA molecules to regulate gene expression by influencing transcript processing, stability, or translation.185,186,187,188 Nucleic acid tools provide exceptional specificity and the potential for quick and efficient manufacturing.189,190 mRNA-based medicines and alternative wild-type mtRNAs can serve as effective treatments to address faulty mtRNAs and proteins.107,191,192
Antisense oligonucleotides (ASOs). ASOs have been successfully transported into mitochondria for several decades and have been demonstrated to function in mitochondria.193,194 In particular, new transport tools based on ASOs improve the safety and efficiency of mitochondrial gene silencing.195 Cruz-Zaragoza et al. developed a new ASO system, morpholino oligonucleotides (MOs), to selectively and efficiently inhibit mtRNA translation.196 Researchers have further utilized the mitochondrial protein Jac1 as a vector to facilitate the import of MOs into mitochondria given the protein’s small size, simple folding, solubility, and the presence of only one cysteine.197
mitoRNAi. RNA interference (RNAi) can silence genes through the RNA-induced silencing complex using various molecules, such as small interfering RNAs (siRNAs), microRNAs (miRNAs), short hairpin RNAs (shRNAs), long non-coding RNAs (lncRNAs), and circular RNAs (circRNAs). These molecules target specific mRNAs to reduce abnormal protein levels or regulate noncoding RNAs (ncRNAs) associated with various diseases.198,199,200,201
Although the intrinsic mitochondrial RNAi mechanism remains unknown, targeting mitochondrial RNA is therapeutically promising, given the existing understanding of mitochondrial biology and pathologies.190,202,203,204 Multiple therapeutic RNAi agents have been shown to suppress the translation of their target mitochondrial mRNAs.
miRNAs are present and active in mitochondria.205,206 In 2004, Zhang et al. discovered that miRNAs and their effector protein Ago2 are present in mitochondria and increase mitochondrial gene expression during muscle development, indicating the ability of miRNAs to penetrate mitochondria.198 In addition, siRNAs designed using Ago2-binding peaks might selectively target matching mtDNA-encoded transcripts in an Ago2-dependent manner. Mitochondrial function is greatly impacted by knocking down mitochondria-encoding subunits with mitoRNAi. These RNAi agents offer significant potential for modulating mitochondrial gene expression and improving mitochondrial function. (Table 2) Their relevance to mitochondrial status lies in their ability to specifically target and regulate mitochondrial mRNAs, offering therapeutic benefits in various mitochondrial diseases. The observed effects on mitochondrial function, such as improved respiration, reduced oxidative stress, and enhanced mitochondrial biogenesis, highlight their potential as promising therapeutic tools. This approach might offer a possible remedy for disorders resulting from mitochondrial DNA mutations.207
RNA supplementation. Another therapeutic nucleic acid tool is replacing or compensating for defective RNAs in the mitochondrial matrix using RNA supplements, including functional tRNAs, rRNAs, and mRNAs. Researchers have effectively restored mitochondrial respiration rates in patient-derived fibroblasts after targeting and delivering pre-tRNAPhe and 12S rRNA to mitochondria with the m.G625A mutation in the tRNAPhe gene and the m.A1555G mutation in the 12S rRNA gene.208,209 Additionally, the delivery of coding RNAs translated into one or more proteins to the mitochondrial matrix is an effective therapeutic strategy for treating mitochondrial diseases. For example, the delivery of polyribonucleic acid with eight in vitro-assembled RNA import complex (RIC) subunits to the mitochondria of Kearns–Sayre syndrome mice effectively restored mitochondrial respiratory efficiency and quality.210 The introduction of mRNA containing the human COX2 gene into the mitochondria of mouse embryonic fibroblasts resulted in the successful production of functional COX2 protein in the matrix.192 Yamada et al. reported that introducing mRNA for the ND3 gene into the mitochondria of fibroblasts from Leigh syndrome patients with the m.T10158C mutation in the ND3 gene also improved the mitochondrial respiration rate.211,212
mtRNA engineering with protein-based tools
In addition to nucleic acid tools, many synthetic protein tools have been designed to act on mitochondrial transcriptional processes, including coordinated RNA processing, polyadenylation, maturation, and RNA attenuation, by selectively binding to specific RNA targets within the mitochondria. Artificial site-specific RNA endonucleases (ASREs) contain a customizable, sequence-specific RNA-binding structural domain with an RNA nuclease structural domain.213 The structural domain of the Pumilio and FBF (PUF) targets an 8-nucleotide RNA sequence, and the PilT N-terminus (PIN) structural domain of SMG6 nonspecifically cleaves RNAs. PUF–PIN-based ASREs specifically repress the transcription of mitochondrial target genes.214 ASREs bind to the N-terminal targeting peptide of ornithine transcarbamylase to form mitoASREs, facilitating protein-targeted import into diseased mitochondria. Choudhury et al. designed mitoASREs with dehydrogenase (ubiquinone) Fe-S protein 4 (NADH) dehydrogenase subunit 5 (MT-ND5) as a target gene, and the transcript levels of MT-ND5 decreased by 20–30% after these mitoASREs were transfected into human cells.213
Mitochondrial gene delivery
Mitochondria have their own genetic system but rely heavily on importing several macromolecules encoded by the nucleus to maintain gene expression.215,216 Certain noncoding genes from the nuclear genome are partly transferred to the mitochondria in all eukaryotes. This phenomenon raises questions about the functional importance of these genes and the exact processes of transportation.217,218 Understanding gene import phenomena relies heavily on the effectiveness of the analytic tools used to identify and describe imported genes.219 Recent methodological advances using specifically designed in situ and interactomic techniques show potential for addressing many of these problems. Mitochondrial gene import requires three main components: import determinants within the gene, a mechanism to redirect RNA to the mitochondria, and a pathway to transport nucleic acid across the mitochondrial membrane.220,221,222,223,224,225
Introducing nucleic acid therapeutic tools into mitochondria by breaking through complex barriers, such as bilayer membrane structures, is necessary to realize gene therapy for mitochondrial diseases.226,227,228 Research on mitochondrial gene delivery has a four–decade–long history. Therapeutic nucleic acid medications are susceptible to degradation by nucleases in the blood and are quickly eliminated by the reticuloendothelial system and kidneys,229,230 impeding their efficient accumulation in mitochondria.231,232,233 Therefore, appropriate carriers are required to safeguard therapeutic nucleic acids and transport them to mitochondrial targets.234,235,236,237,238 Progress in drug delivery technology has allowed the containment of different cargos, including tiny pharmaceuticals, nucleic acids, and proteins, and the precise targeting of certain tissues and cell types to improve delivery effectiveness.239,240,241,242 In recent years, mitochondrial gene delivery has been developed to control the intracellular transportation of delivery vehicles to target specific organelles.243,244,245,246 Targeting the appropriate organelle can improve treatment effectiveness and reduce negative side effects.247,248 The essential activities of mitochondria rely on crucial proteins expressed by both nuclear and mitochondrial genes. Thus, the advancement of gene delivery methods has been a focal point of research, leading to the creation of several tools.236,249,250,251,252
Physical approaches
Physical gene delivery is the direct transfer of therapeutic nucleic acids into the cytoplasm or nucleus through membrane penetration without the use of chemicals or other carrier molecules, such as viruses. Microinjection directly delivers exogenous genes into cells through fine glass microtubes. In addition, hydrodynamic delivery increases hydrodynamic pressure by rapidly pushing a large volume of solution into the bloodstream, temporarily enhancing the permeability of the cell and allowing therapeutic nucleic acid tools to enter the cytoplasm, which they would not normally be able to do when unable to cross the cell membrane. Yasuzaki et al. showed that plasmid DNA could be injected intravenously into rat liver mitochondria by hydrodynamic injection.253,254 In contrast to chemical and biological processes, physical approaches do not cause toxicity when linked to carrier molecules. However, physical agents are equally dispersed in the cytoplasm and reach the mitochondrial matrix randomly. With the evolution of technology, the ability to deliver genes that are truly targeted to mitochondria via physical approaches will likely emerge.
Chemical approaches
Most methods developed for mitochondrial gene delivery are chemical. Physical methods enhance the absorption of nucleic acid drugs by cells through physical forces, thereby improving gene delivery efficiency, whereas chemical methods utilize the hydrophobic and negatively charged nature of mitochondrial membranes to deliver nucleic acid drugs through special chemical interactions with mitochondrial membranes. Nanotechnology has become a significant focus of chemical approach research with the advancement of the nanoscience era.255,256,257 Lipid-based, polymer-based, mitochondrial-targeted peptides, and inorganic-based nanoparticles have facilitated the creation of innovative and unique breakthroughs in mitochondrial gene delivery, which have been adapted to reduce immunogenicity, enhance safety profiles, and the flexibility to load gene materials.258,259
Lipophilic cations. The use of lipophilic cations as ligand molecules, such as triphenylphosphine cation (TPP+), which has high lipophilicity and a large ionic radius, effectively reduces the activation energy required for transmembrane transport and allows the effective targeting of genes to mitochondria. Compared with hydrophilic cations (e.g., Na+), lipophilic cations (e.g., TPP+) can accelerate the transport of bioactive molecules across the mitochondrial membrane by as much as 107–108 times. Faria et al. coupled TPP with Polyethylene Glycol (PEG)–Polyethylenimine (PEI) to form PEG–PEI–TPP. After complexing pDNA for targeted mitochondrial delivery, PEG–PEI–TPP significantly increased the efficiency of ND4 gene and protein expression.260
Dequinium chloride (DQA) is a bicationic compound comprising two symmetric molecules, and the vesicle-like aggregates formed by DQA with diameters ranging from 70 to 700 nm are known as DQAsomes, which are recognized as effective carriers that target mitochondria.261,262,263 DQAsomes containing pDNA expressing green fluorescent protein (GFP) stimulate mitochondrial gene expression in cultivated cells.264 The transfection efficacy of DQAsomes in the mitochondria was only 5%. Choi and colleagues combined DQA with additional lipids, 1,2-Dioleoyl-3-trimethylammonium-propane (DOTAP) and dioleoylphosphatidylethanolamine (DOPE), to enhance the functionalization of DQAsomes.265 Compared with the cellular uptake and endosomal escape capabilities of DQAsomes alone, those of DQA, in combination with DOTAP/DOPE, were enhanced, increasing mitochondrial gene expression.
Previous research has revealed that a carrier based on octaarginine (R8)-modified liposomes, referred to as MITO-Porters, has strong mitochondrial fusion activity.266,267,268 Similar to conventional cationic liposomes, these particles can be internalized by macropinocytosis rather than clathrin-mediated endocytosis.269
Dendrimers. Dendrimers are nanoscale polymers with a dendritic backbone and a spherical shape with many active functional groups attached to the ends of the dendritic branches. Drugs can be either encapsulated in the internal cavities of the dendrimer backbone or chemically coupled to the functional groups on the surface of the dendrimer.270,271,272,273 Dendrimers possess a dynamic structure and synthesis process that allows them to be easily tailored for specific applications. Dendrimers have a core-shell structure that enables the creation of branch points during synthesis. These branch points can either be encapsulated in the internal cavities of the dendrimer backbone or chemically coupled to the functional groups on the dendrimer surface.274,275 Wang et al. investigated the transport of mitochondrial genes utilizing dendrimers as carriers. They evaluated the generation 5 polyamidoamine (G5-PAMAM) dendrimer with an ethylenediamine core and amine terminations, to which TPP was added, in HeLa and COS-7 cells.275 The transfection efficacy of synthesized G5-TPP dendrimers is much higher than non-modified G5 dendrimers, which might contribute to efficient endosomal escape and mitochondrial targeting ability. HeLa cells (52%) showed a higher transfection efficiency compared to COS-7 cells (40%). This variation might be attributed to differences in endocytic pathways, mitochondrial dynamics, cellular metabolism, and mitochondrial activity between the cancerous (HeLa) and non-cancerous (COS-7) cell lines. Biswas et al. examined a comparable system using a TPP ligand and a G5-PAMAM dendrimer. TPP-modified dendrimers exhibited lower cytotoxicity than TPP-free dendrimers in normal mouse fibroblasts (NIH-3T3). Moreover, the TPP systems exhibited a high degree of selectivity for mitochondria and a strong capacity for cellular internalization.275,276
Inorganic nanoparticles. Inorganic nanoparticles can facilitate the transmission of large plasmid DNA and small DNA molecules. Rhodamine is a fluorescent chemical that attracts mitochondria, enabling the monitoring of mitochondrial entry into cells and the assessment of mitochondrial membrane potential.277 Santos et al. investigated this characteristic of rhodamine for targeting mitochondria. Researchers have created rhodamine nanoparticles using plasmid DNA by utilizing a coprecipitation approach with CaCl₂ and Na2CO3.278 The above inorganic compounds effectively encapsulated several plasmids and promoted cellular endocytosis; their targeting to mitochondria was verified by confocal microscopy experiments.278 Costa’s team created CaCO₃–pDNA–Rho123 nanoparticles utilizing the coprecipitation technique.279 These vectors formed nanoparticles of an appropriate size and surface charge after encapsulating plasmids with the GFP gene. In fluorescence confocal microscopy images, the nanoparticles showed targeted delivery into the mitochondria of fibroblasts and HeLa cells.279 Costa et al. also constructed a compound [16]phenN2 fluorescently labeled calcium carbonate delivery system in which p53 and ND1–GFP plasmids were encapsulated to achieve therapeutic effects of targeted intervention in mitochondria.280 Confocal fluorescence images showed that [16]phenN2 fluorescence intensity was significantly greater in mitochondria than in the cytoplasm or lysosomes.280
Biological approaches
Mitochondria-targeting signal peptides. Given that the precursor proteins of most mitochondrial proteins are synthesized in cytoplasmic ribosomes, these precursor proteins require specific MTSs that can be recognized by mitochondrial surface receptors and transported into the mitochondrion via membrane passage orientations.281 The translocase of the outer membrane (TOM) complex is the primary entry point for the recognition of MTS sequences to induce the entry of therapeutic tools into the mitochondria, and the entry channel subunit is an intact membrane β-barrel protein, Tom40. Upon crossing the TOM complex, the protein substrate containing the presequence is delivered to the inner membrane via the translocase of the inner membrane (TIM) complex, and the TOM and TIM form a supercomplex during translocation to achieve effective input of therapeutic tools across the mitochondrial double membrane.281,282,283,284,285,286 Khan et al. designed a protocol delivery system comprising three domains: (i) protein transduction domains that promote cellular uptake, (ii) mitochondrion-targeted MTS domains, and (iii) TFAM that recombines with wild-type mitochondrial DNA.286,287 Targeted transport of protein transduction domains (PTD)–MTS–TFAM to mitochondria can effectively reduce the number of mutated mitochondrial DNA copies, improve the cellular mitochondrial respiration rate, and increase the DNA copy number.288 Injecting PTD–MTS–TFAM compounded with wild-type mtDNA into mice via the tail vein can effectively increase the respiratory rate of the mouse brain and skeletal muscle mitochondria and simultaneously increase the levels of mtDNA and TFAM, demonstrating a synergistic therapeutic effect.289,290,291,292 In addition, PTD–MTS–TFAM was also used to construct LHON model by expressing a pathogenic mtDNA carrying the G11778A mutation associated with LHON disease.293
Framework of the nucleic acid-based delivery system. Highly editable DNA has enhanced structural flexibility, allowing lengthy DNA strands to fold into various two- or three-dimensional shapes.294,295,296 The earliest DNA nanostructures were fixed four-arm branches; branch structures and three-dimensional structures, such as DNA tetrahedra, DNA prisms, DNA polyhedra, and DNA nanolanterns, were then developed to meet the need for more complex and diverse DNA nanostructures.297,298,299 More editable and replaceable positions are available given the precise spatial design, flexible programmability, and sequence specificity of DNA nanostructures.300,301 Modified DNA framework architectures can transport therapeutic medicines into the mitochondria.302,303,304 Chan et al.299 attached MTS to DNA oligomers using a series of chemical modifications, and the modified DNA oligomers were then combined with Cy5-labeled DNA nanocages (DNA-NCs) to form the final MTS DNA-NCs, with additional thymidine units added to prevent steric clashes. The framework structure can be targeted and delivered to mitochondria by connecting the MTS to the branching structure of DNA nanostructures, indicating the huge potential of those nanostructures in targeting organelles.
Virus-based mitochondrial targeting. Adenoviruses have been utilized as effective tools for the transportation of mitochondria-targeting nucleases. The mito-ApaLI enzyme has been effectively administered to the hearts of NZB/BALB mice using recombinant adenovirus type 5 to target and remove mitochondrial DNA.142 However, the limited duration of expression and robust immunological reactions restrict the use of adenoviruses.142
AAV is another commonly utilized viral tool for mitochondrial gene therapy because of its extended expression and established clinical safety. AAV vectors have a maximum packaging capacity of under 5 kilobases. This limitation restricts the transgene size and its effective distribution.305,306 Although AAV vectors have limited DNA packaging capacities, they are preferred for in vivo research involving mitochondrion-targeted nucleases. Research has demonstrated that individuals exposed to AAV develop an immunological reaction, which hinders the administration of effective genetic therapy.306 Recombinant adeno-associated virus (rAAV) is nonpathogenic and has minimal immunogenicity. Various tissues and cells can be transduced by different AAV serotypes by modifying the tissue specificity of rAAV.307 AAV serotypes, such as AAV6 and AAV9, exhibit specificity for skeletal and cardiac muscle. AAVs containing DNA survive as episomes for an extended period in transduced cells, making AAV vectors a potential therapeutic option for homoplasmic or heteroplasmic mutation. These vectors can deliver therapeutic genes to patients’ nerves and muscles.308,309 Creating secure and efficient AAV delivery techniques to introduce mtDNA-editing tools into specific cell types is a significant yet worthwhile undertaking.
Recent clinical trials have utilized the nuclear delivery of recombinant AAV to re-express the mtDNA-encoded ND4 gene, which is mutated in LHON. The efficacy of this impact was demonstrated in a phase 3 clinical study in which visual function was enhanced in individuals diagnosed with LHON.310,311,312,313 rAAV2, rAAVrh10, and rAAV9 have been utilized in therapeutic settings to transport modified genes to the central nervous system and might be beneficial in treating Alzheimer’s disease (AD) and spinal muscular atrophy.314 Various methods, including in silico analysis, rational design, and directed evolution, have been employed to modify AAV capsid proteins, enhancing tissue selectivity and reducing off-target transduction.315 Tissue specificity can be attained by utilizing tissue-specific promoters, such as the alpha-1 antitrypsin promoter for liver transduction, PGDF and NSE for neurons, and desmin for skeletal muscle.316,317,318,319
Additionally, when utilizing AAV viral delivery for mtDNA modification tools, it is important to address the immunogenicity of the AAV capsid, the neutralizing antibodies, and the immunological response to AAV-encoded transgenes. Therefore, creating new AAV serotypes with fewer immune reactions will enable the use of AAV to deliver mtDNA editing tools in clinical therapy. The bacteria-derived toxins FokI, TALE, and DddA may influence T-cell responses.320 An altered AAV vector has been created by combining an outer capsid protein with an MTS to deliver DNA to mitochondria in living organisms. It is uncertain how MTS–AAV reaches the mitochondria and releases encapsulated DNA inside the organelle despite the documented effective delivery, recombination, and expression of DNA in the mitochondrial matrix.321 Second, there is inefficient recombination in mammalian mitochondrial DNA. Therefore, unlike in yeast or green algae, DNA transferred to the mitochondria cannot be readily incorporated into the host genome. Moreover, due to the polycopy nature of mtDNA, it is challenging to isolate specific mutant mtDNA from other mtDNA combinations to reach the necessary quantities for experimental or therapeutic purposes. Hence, a technique that can decrease the concentration of mitochondrial nuclease-encoding AAV is essential for therapeutic use. Possible solutions include enhancing AAV capsid specificity, implementing temporary immunosuppressive procedures, delivering injections locally to certain organs, and incorporating mitoZFNs or mitoTALEN monomers into a single AAV capsid. The development of AAV technology that can safely deliver mitoZFNs or mitoTALENs while being compatible with the current recombinant AAV manufacturing capacity and clinical standards is a significant challenge.252,322 In addition, the significant expense associated with clinical studies and therapies poses a major barrier to implementing mitochondrial gene therapy.323
The interplay among physical, chemical, and biological approaches lies in their complementary strengths and limitations. Physical methods provide direct delivery but with low specificity; chemical methods enhance targeting but may involve complex synthesis; biological methods offer high specificity but can be limited by immune responses. Combining elements from each—such as using chemical carriers with biological targeting signals—could lead to more effective mitochondrial gene therapies by maximizing delivery efficiency while minimizing adverse effects.
While mitochondrial gene editing focuses on directly modifying genetic defects, an alternative approach, artificial mitochondrial transfer, offers a different strategy to address mitochondrial dysfunction. This method involves the replacement of damaged mitochondria with healthy ones from a donor, providing a holistic solution by entirely substituting the malfunctioning organelles. Both strategies aim to rectify mitochondrial deficiencies but differ fundamentally in their execution and potential applications.
Artificial mitochondrial transfer
Mitochondrial transfer observed in health and disease
The initial observation of organelle migration across mammalian cells through tunneling nanotubes (TNTs) was made by Rustom et al. in 2004,74 and the initial demonstration of functional mitochondrial transfer from mesenchymal stem cells (MSCs) to cells with dysfunctional mitochondria was conducted by Spees et al. in 2006.75 Subsequently, mitochondrial transfer was observed between different cells, tissue types, and even species. Extracellular vesicles (EVs)-containing mitochondria and free mitochondria were later observed during the transfer process.76 The transferred mitochondria may have significant and enduring consequences due to the potential for cross-species fusion between foreign mitochondria and host cell mitochondria.324 In vitro, mitochondria function for a minimum of 45 cell passages, equivalent to 135 days.325
Mitochondrial transfer contributes to the structural and functional connectivity of tissue homeostasis and is related to recipient-cell mitochondrial supplementation, donor-cell mitochondrial quality control, and cell-to-cell signal transduction. The detailed molecular mechanism of mitochondrial transfer has been reviewed in previous reviews.326,327
Transfer between differentiated cells
Mitochondrial transfer is mainly observed in vitro in cultures of the same cell type (for example, mitochondria between normal rat kidney cells by TNT328) or in cocultures of different types of cells (for example, mitochondria exchange between cardiomyocytes and cardiofibroblasts,329 between rat cardiomyocytes and human endothelial progenitor cells,330 and between mouse osteocytes and endothelial cells (ECs) of transcortical vessels331).
In recent years, evidence of mitochondrial transfer in the neural systems has been reported, and a primary network has been constructed that might provide some guidance for studies in other systems. In the nervous system, mitochondrial transfer is multidirectional. Neurons can be the donor, transferring mitochondria to neurons and glial cells: functional mitochondria can be transferred from normal neuroblastoma cells to mitochondria-defective neuroblastoma cells through tunnel nanotubes,332 and neurons at the optic disc release a significant quantity of mitochondria, which are then broken down by the lysosomes of nearby glial cells.333 It was assumed that the transcellular degradation of mitochondria occurs in other regions of the central nervous system due to structurally comparable accumulations of deteriorating mitochondria along neurites in the superficial regions of the cortex. Conversely, neurons can be recipients, internalizing mitochondria from neurons (stated above), astrocytes, and macrophages. Astrocyte-derived mitochondria normalize the neuronal calcium dynamics impaired by cisplatin,334 protecting neurons against apoptosis in ischemia.335 Macrophages play an active role in the remote modulation of inflammatory pain elimination by delivering mitochondria to sensory neurons.336 Additionally, mitochondria secreted by astrocytes can enter microglia, improving recovery from intracerebral hemorrhage.337 Mitochondrial transfer can occur in multiple stages in the nervous system: dysfunctional mitochondria spread from microglia to astrocytes to neurons.338
Transfer related to stem cells
Stem cells possess substantial differentiation potential and high levels of mitochondrial activity. As an optimal mitochondrial resource, stem cells provide functional mitochondria and facilitate the homeostasis of target cells. In an in vitro ischemia-reperfusion model, the restoration of injured endothelial cells was achieved through the transfer of mitochondria via TNTs between MSCs and human umbilical vein endothelial cells (HUVECs).339 Bronchial epithelial cells obtain functional mitochondria when cocultured with mesenchymal stromal cells, regardless of whether the bronchial epithelial cells are derived from the lung parenchyma or bronchoalveolar lavage fluid.340 Interestingly, in addition to restoring physiological functions, transferred mitochondria appear to have the potential to reprogram differentiated cells. The reprogramming of cardiomyocytes to a more immature state was achieved through the transfer of mitochondria from human multipotent adipose-derived stem cells to cardiomyocytes.341 Mitochondrial transfer from stem cells appears to provide somatic cells with differentiation abilities to a certain extent. Conversely, somatic cell-derived mitochondria provide differentiation cues to stem cells. The regulation of MSC differentiation in coculture necessitates the transfer of mitochondria from vascular smooth muscle cells (VSMCs) to MSCs.342 Similarly, the bidirectional mitochondrial transfer between human mesenchymal multipotent stromal cells (MMSCs) and rat RTCs induces the differentiation of MMSCs into kidney tubular cells.343 More stem cell artificial mitochondrial transfer therapies are discussed in detail in the preclinical research section.
Transfer related to cancers
Mitochondria play a vital role in most metabolic pathways, as they contribute significantly to the synthesis of energy and biomass while also serving as metabolic sensors. From this perspective, mitochondria finely regulate signaling pathways linked to cancer cell metabolism.344
From host cells to cancer cells. The equilibrium between glycolysis and OXPHOS in cancer cells can be modified to align with biomass generation and energy requirements. Regardless of whether cancer cells use OXPHOS or glycolysis as their main method of energy generation, pathways with excessive rates contribute to detrimental effects on mitochondria. Increasing evidence reveals that mitochondrial transfer from host cells helps cancer cells overcome mitochondrial impairment.
Wharton’s jelly-derived MSCs transfer their mitochondria to mtDNA-depleted osteosarcoma cells (143B) and restore the original defective OXPHOS.325 Similarly, human MSCs deliver mitochondria to 143B cells lacking mitochondrial function to rescue proliferation.345 Host mtDNA can also be transferred to tumor cells with impaired respiratory function, restoring respiration and promoting tumor initiation.346
Transfer between cancer cells. Cellular interactions in the tumor microenvironment have been extensively studied in tumor development.347 Mitochondrial transfer between cancer cells has been found to contribute to the tumor communication network. Mitochondria are transmitted in a bidirectional manner between cancer cells, such as primary cells from human laryngeal squamous cell carcinoma,348 mesothelioma cells (MSTO-211H),349 and GBM stem-like cells.350 In addition to the simple exchange of mitochondria between cancer cells of the same type, alterations in the characteristics of cancer cells after receiving mitochondria from the microenvironment have been observed. Cancer-associated fibroblasts facilitate the aggressiveness of prostate cancer (PC3, DU145) through mitochondrial transfer.351 Mitochondrial transport from aggressive bladder cancer cells (T24) to nonaggressive bladder cancer cells (RT4) has been found to increase bladder cancer cell invasiveness.352 Coinjection of human cancer-associated fibroblasts (CAFs) and human prostate cancer (PCa) cells into severe combined immunodeficient mice showed that mitochondria from CAFs enhance the motility and lactate metabolism of cancer cells.351
Mitochondrial transfer and chemoresistance. Regardless of chemotherapy or radiotherapy, anticancer medicines result in mitochondrial impairment via oxidative or related stress, and chemoresistance mediated by mitochondrial transfer is a major challenge: both the transfer of mitochondria into cancer cells (normal mitochondrial replenishment) and the transfer of mitochondria from cancer cells (abnormal mitochondrial clearance) can result in chemoresistance. Acute myeloid leukemia (AML)-derived cells acquire normal mitochondria from cocultured murine or human bone marrow stem cells (BMSCs), leading to increased survival rates after chemotherapy.353 MSCs also transport mitochondria to rescue acute lymphoblastic leukemia (ALL) B-cell precursors from ROS-inducing chemotherapy.354 Tumor-activated stromal cells increase GBM proliferation and resistance to standard treatments via mitochondrial transfer.355 In the tri-culture system (MCF7 cells, MSCs, and E4+ECs), mitochondrial transfer exhibited comparable patterns between cancer cells and MSCs or ECs. However, the exchange of mitochondria between ECs and cancer cells is more pronounced, increasing cancer cell chemoresistance.356 Pheochromocytoma (PC) 12 cells can survive after ultraviolet light (UV) treatment by obtaining functional mitochondria from untreated PC12 cells.357 Mitochondrial transfer from Jurkat cells to MSCs may also result in chemoresistance since damaged mitochondria are removed from cancer cells.358
However, the detailed mechanisms of chemoresistance via mitochondrial transfer remain unclear. Conflicting reports exist regarding the effect of mitochondrial transfer on cancer cells. The proliferation of cancer cells (MCF-7/ADR) was found to be inhibited by normal mitochondria derived from epithelial MCF-12A cells, whereas susceptibility to chemotherapeutic drugs was enhanced.359 The authors stated that “ROS will be elevated” and that “depriving cancer cells of glycolytic intermediates”360 could explain their results. However, these findings might be oversimplified since the effects of new mitochondria are multifaceted, especially in different cancer types. For example, mitochondrial transfer has been especially studied in AML since ATP generation in AML depends on oxidative phosphorylation, contrary to the common Warburg effect in solid tumors.361 In vivo, normal mitochondria are transferred from human BMSCs to leukemic blasts, causing multiple myeloma cells to rely on oxidative phosphorylation.362,363 Future investigations might provide insight into the unique role of energy metabolism in AML and novel therapeutic strategies.
Technologies for artificial mitochondrial transfer
Mitochondrial imaging technologies
Fluorescent dyes are widely employed as a prevalent technique for quantifying mitochondrial transport in vitro.364 The use of fluorescent dyes such as mitoTracker provides a visible and quantitative method for studying mitochondrial transport in vitro. Fluorescent dyes enable the continuous tracking of mitochondrial transport in real-time via flow cytometry or confocal microscopy.365,366 mtDNA sequencing is an alternative technique employed to quantify mitochondrial transport in vitro via the identification of donor mtDNA in recipient cells.367 The mtDNA sequencing technique can even identify specific mitochondria with edited genes. Furthermore, Zhang et al. utilized single-cell sequencing techniques to investigate the dynamics of mitochondrial transport between cancer cells and T cells via a novel deconvolution process.368 Applying this method, they accurately predicted the recipient cells and their relative mitochondrial compositions and identified a reproducible mitochondrial transfer phenotype.
Fluorescent mitochondrial labeling and mtDNA sequencing369 can also be performed in vivo. Special mtDNA sequencing focused on mtDNA haplotypes has also been frequently used in vivo. Mitochondrial DNA haplotypes are specific sequences of mitochondrial DNA that can aggregate with the mtDNA of other mitochondria, providing insights into evolutionary origins. MtDNA haplotypes are highly valuable for investigating mitochondrial trafficking from mothers to fetuses during pregnancy.370
Advanced imaging techniques have facilitated the observation of mitochondrial transport within living organisms. One such technique involves using two-photon microscopy to monitor the movement of mitochondria in vivo.371 This approach enables the real-time imaging of mitochondrial dynamics and intercellular travel, providing essential insights into the fundamental mechanics of mitochondrial trafficking in vivo.
Mitochondrial transfer technologies
Currently, the mitochondrion is acknowledged as an endosymbiotic entity whose noneukaryotic derivation might increase its capacity for intercellular transplantation and fusion with the recipient cells’ original mitochondrial network.
Coculture was the first and most widely used artificial mitochondrial transfer method72 and has been demonstrated to be effective directly and in transwell systems.372 The coculture technique has notable benefits regarding safety and simplicity; nonetheless, it is essential to acknowledge that heterogeneity and efficiency pose considerable limitations. Therefore, many new methods have been developed to achieve reliable and efficient artificial mitochondrial transfer. Notably, there are several difficulties associated with artificial mitochondrial transfer. First, how to precisely drive mitochondria to move toward target cells is an issue. Second, naked mitochondria might be toxic to cells when located outside the cell membrane. An illustration of this phenomenon is the activation of proinflammatory processes in leukocytes by oxidized cardiolipin present on the mitochondrial membrane.373 Third, the mechanisms regulating the fate of transplanted mitochondria have not been fully elucidated.
The exploration of artificial mitochondrial transfer can be classified into two categories. The first requires the use of additional physical force to facilitate the internalization of free mitochondria into recipient cells, including microinjection,374,375 magnetomitotransfer,376 mitoCeption,377,378 photothermal nanoblade,379 mitoPunch,380,381 optical tweezers,382, and droplet microfluidics.383 The second adopts biological modifications or vectors to facilitate delivery, including small molecule peptides,384 polymers,385 and lipids,386 to optimize mitochondrial internalization.
Extra physical force. The advantage of using additional physical force is that the manipulator can determine the appropriate timing of the transplant and the number of transplanted mitochondria. However, these approaches are device-dependent and challenging to use in vivo (Fig. 5).
Strategies for artificial mitochondrial transfer. Physical methods for facilitating the transplantation of free mitochondria into cells include coculture, microinjection, magnetomitotransfer, mito-ception, mitopunch, fluidFM, photothermal nanoblade, optical tweezers, droplet microfluidics, etc. These methods utilize natural intercellular mitochondrial transfer or apply additional driving forces using various devices. Biochemical methods involve utilizing mitochondrial surface modifications or vectors for delivering mitochondria, including exosomes, polymer coatings, targeting peptides, and so on. (Generated by the authors with Adobe Illustrator)
Microinjection of exogenous mitochondria was initially applied in oocytes harboring mutants related to mitochondrial disease.374,375 Microinjection is appropriate for manipulation and achieving exact efficacy for a single germ cell. Procedures with greater efficiency and larger scale are needed for diseases related to the degeneration of mitochondria in multiple cells. Magnetomitotransfer adds magnetic bead-labeled mitochondria to recipient cells under the magnetic field produced by a specialized device.376 mitoCeption377,378 involves using centripetal force (~1500 g) created within a centrifuge to localize mitochondria to target cells. The mitoPunch platform is a highly parallel, pressure-driven instrument designed for large-scale transplantation.380,381 The mitoPunch technique employs a plunger to induce the physical transformation of a flexible reservoir that houses isolated mitochondria. The plunger’s motion propels the mitochondria in the reservoir through a membrane with tiny holes, where adherent cells are cultivated.387 In contrast to mitoCeption, this rapid, cost-effective, and high-throughput method can be used in any laboratory setting. Additionally, this method requires a minimal quantity of donor mitochondria.380 Both mitoPunch and mitoCeption can generate cells that permanently retain exogenous mtDNA.380
FluidFM388,389 uses hollow cantilevers for artificial mitochondrial transfer with atomic force microscopy (AFM)-assisted control. Researchers have integrated AFM and nanofluidic techniques to achieve force and volume control under real-time inspection. The photothermal nanoblade379,390 has a modified microneedle using a laser-induced bubble to generate perforations in the cell membrane, facilitating the efficient transport of mitochondria into the cell via coordinated flow. Optical tweezers employ a concentrated light beam to generate trapping force magnitudes in the range of piconewtons.391 Shakoor et al. used an automated optical tweezer-based micromanipulation system to control transplantation.382 This device combines optical tweezers and automated systems to transport healthy mitochondria to recipient cells using endocytosis with high accuracy and efficiency. Droplet microfluidics selectively distributes a continuous stream of items to be processed into distinct droplets of micrometer dimensions.383 The enclosed microenvironment of the droplets decreases the distance that the isolated mitochondria have to travel and increases their chances of coming into contact with the cell, improving the effectiveness of artificial mitochondrial transfer. The quantitative control of mitochondria transplantation can be achieved by altering the proportion of the mitochondrial solution.383
Surface modification and delivery vectors. Although cell-free mitochondria are reportedly abundant, normal components of blood,76,392 surface modification, and delivery vectors have several benefits in the application of artificial mitochondrial transfer.
-
1.
Enhanced stability and internalization.
Although spontaneous mitochondrial transport has been widely detected in coculture systems, two unexpected outcomes of transplanted mitochondria in recipient cells have been documented: 1. The absence of modifications in mitochondria may result in their failure to escape endosomes, subsequently leading to their degradation. 2. During spontaneous transfer, mitochondria undergo degradation after their internalization.333,335 Therefore, one main goal of mitochondrial modification and delivery is to protect mitochondria (prolonging circulation durations by preventing protein adsorption and identification by macrophages, a phenomenon commonly referred to as “stealthiness”),393 enhance stability, improve internalization, and avoid direct exposure to the membrane.
Extracellular vesicles are potential candidates since mitochondria can be naturally secreted via EVs.394,395 With a mitochondria-specific dye or size filter, EVs containing mitochondria can be easily screened out. However, one main limitation of this strategy is that the mitochondria naturally secreted via EVs are often damaged,396,397 limiting their therapeutic effects.
The outer membranes of mitochondria are abundantly enriched with anionic lipids that are negatively charged under biological circumstances.398,399 Due to this characteristic, charge-based mitochondrial coatings have been researched and demonstrated to be reliable and effective. Cationic gelatin nanospheres have the potential to be readily immobilized on the mitochondrial surface through electrostatic interactions, enhancing the efficiency of mitochondrial internalization.400 Comparably, coating with a mixture of DOTAP and DOPE using a reverse emulsion technique386 or with the dextran‐TPP polymer385,401 also improved artificial mitochondrial transfer and efficacy. Furthermore, a layered architecture with flexible thickness could be created using the layer-by-layer (LbL) approach. LbL is an effective option for modifying mitochondria with minimal disruption of their natural bioactivity.402 Chen et al. identified chitosan and poly(acrylic acid) (PAA) as promising options for mitochondrial LbL modification owing to their exceptional compliance with mitochondrial membranes.403 The researchers detected the presence of a nanomaterial coating with a thickness of ~45 nm after six rounds of LbL assembly. This coating effectively preserved the integrity of the mitochondrial membrane, resulting in no noticeable distortion or collapse. Interestingly, charge-based coatings also overcome the barrier of electrostatic repulsion when the target cells, for example, cancer cells, are negatively charged.403
-
2.
Enhancing the ability to penetrate
Maeda et al. reported that cell penetration peptide (CPP) and glucan modification of mitochondria can significantly increase the efficiency of artificial mitochondrial transfer, reducing oxidative stress and apoptosis in cardiomyocytes.404 Similar improvements in penetration were widely reported when CPP-modified mitochondria were applied to treat mitochondrial disease,384 breast cancer cells (MCF-7 cells),405 and dopaminergic neurons.406
Engineered mitochondria incorporating a Janus surface structure were also developed, drawing inspiration from the natural bacterial mechanism. This system utilizes glucose oxidase to drive movement, enabling it to navigate toward tumors and autonomously infiltrate the depths of tumor tissues. Furthermore, the system exhibited a prolonged retention period.407
-
3.
Incorporate additional synergists
A two-in-one strategy involving mixing mitochondria in EVs and exogenous 27-kDa heat shock protein (HSP27) has been developed to preserve brain endothelial cell tight junction integrity.394 A synthetic biological system has been created to enhance photodynamic therapy by combining a photosensitizer with mitochondria.408 In addition to the direct combination of mitochondria and synergists, optogenetic tools have been applied to modify the mitochondrial membrane to realize light-activated engineering.409
Artificial mitochondria
In several recent studies, researchers have attempted to construct artificial mitochondria by transplanting existing ATP-producing structures. Escherichia coli cells were successfully transplanted into bacteriogenic protocells to allow mitochondria to produce ATP.410 Another study revealed that camouflaged nanophotosynthetic thylakoids could achieve quick penetration into chondrocytes by fusing with the cell membrane, preventing the destruction of the lysosomes.165 Moreover, exposure to light increases intracellular ATP.165 Although these novel strategies are promising, more researchers have adopted a from-bottom-to-up method. According to this theory, artificial mitochondria have three fundamental components: the membrane, the proton gradient generator, and the ATPase.411 The membranous compartment separates the reactive site from the outer environment. The generator produces a proton gradient through the membrane, which the ATPase subsequently utilizes to facilitate the synthesis of ATP.
The membranous compartment. Lipids were most frequently used in the initial attempts to form the membranous compartment, whereas novel strategies adopting synthetic polymers were also appealing because of their stability, flexibility, and durability.412 Specific synthetic polymers automatically form stable vesicles413 with dimensions similar to those of a typical lipid bilayer (5 nm).414 Moreover, combining synthetic polymers with lipids makes it possible to create hybrid lipid‒polymer membranes exhibiting tunable distribution. Hybrid membranes have demonstrated substantial potential in artificial mitochondria.415,416
Researchers have attempted to integrate multiple optimally reconstituted elements into one compartment using biological-mediated417 or charge-mediated418 methods. Biologically mediated methods reconstitute each protein separately in liposomes and then fuse the different membrane protein-containing vesicles to construct a vesicle containing multiple membrane proteins without altering the orientation of any component.419,420 Charge-mediated fusion, the fusion of liposomes containing cationic lipids with liposomes with a negative charge, was first described in 1988.421 Charge-mediated fusion has a significantly shorter duration (for several minutes) than biologically mediated fusion but preserves the orientation of the membrane protein.418
The proton gradient generator. The fundamental approach employed in artificial mitochondrial models to generate transmembrane proton gradients involves the utilization of oxidation or electron transport of specific substrates catalyzed by a sequence of enzymes.
Most ATP in living cells is generated through the collaborative activity of respiratory complexes within the mitochondria. However, integrating all the complexes into a single compartment in artificial structures is challenging because each protein needs an individual specialized mechanism for reconstitution.422 Hence, employing a solitary constituent derived from the electron transport chain is a pragmatic approach.423,424 Similarly, artificial bacterial respiratory system components are effective; for example, cytochrome bo3.418,425,426,427 In addition, glucose oxidase can generate protons via oxidative reactions.428,429,430
Self-adaptation or homeostasis maintenance, when the external environment is altered, is impossible for the above artificial mitochondria, and the efficiency of ATP generation may correspondingly decrease significantly. In particular, fluctuations in osmotic pressure can impact synthetic mitochondrion behavior and perhaps lead to the destruction of these synthetic organelles. Further investigations are needed to determine the ability of synthetic mitochondria to generate ATP sustainably across diverse situations. More information on the process of artificial mitochondria synthesis could be found in Biner et al.’s review.422
Application of engineered mitochondria in disease
The field of engineered mitochondria has seen significant strides through the development of both mitochondrial gene editing and artificial mitochondrial transfer. Mitochondrial gene editing enables precise alterations to mitochondrial DNA, offering a targeted approach to rectifying genetic defects at their source. Meanwhile, artificial mitochondrial transfer provides a method to replace dysfunctional mitochondria with healthy ones, thus restoring cellular function. These complementary approaches not only highlight the potential for innovative treatments but also underscore the versatility of engineered mitochondria-based therapies. By now, an increasing number of preclinical and clinical studies have yielded encouraging results (Fig. 6).
Preclinical research on the application of engineered mitochondria in diseases. Only limited in vivo studies have demonstrated the effectiveness of mitoTALEN, mtZFN, and mitoARCUS in clearing mutant mtDNA in specific tissues (in red). Concurrently, various mitochondrial diseases involving different mutant genes have been extensively investigated in vivo to determine the therapeutic effects of nuclear genome editing (in purple). With the rapid advancement in mitochondrial gene editing, the application may be soon extended, particularly to scenarios that have been researched for previous nuclear genome editing. The administration of artificial mitochondrial transfer in multiple animal models with diseases related to mitochondrial dysfunction has shown promising results, including in the cardiovascular, nervous, digestive, visual, urinary, and musculoskeletal systems (in blue) (Generated by the authors with Adobe Illustrator)
Application of gene-edited mitochondria in disease treatment
Gene editing strategies targeting mitochondrial diseases encompass two distinct approaches: the editing of nuclear genes encoding mitochondrial-related proteins and the direct editing of mitochondrial genes. Both approaches have shown promising progress in preclinical studies (Table 3).
Nuclear genome editing in mitochondrial diseases
Ethylmalonic acid encephalopathy (EE) is a type of mitochondrial encephalopathy resulting from toxic sulfide buildup due to mutations in the ETHE1 gene. The ETHE1 protein encoded by the ETHE1 gene is a mitochondrial sulfur dioxygenase located in the mitochondrial matrix that is involved in sulfur metabolism and converts sulfides into less toxic substances. ETHE1 protein deficiency leads to the accumulation of hydrogen sulfide (H2S) and ethylmalonic acid (EMA) in the brain, decreased enzyme activity, and mitochondrial energy metabolism disorders.431 H2S is toxic at relatively high concentrations and inhibits enzymes such as cytochrome c oxidase (COX)432 and short-chain acyl-CoA dehydrogenase,433 resulting in intellectual and motor lag, persistent diarrhea, skin ecchymosis and petechiae, orthostatic cyanosis of hands and feet, and brain gray matter damage.434 Common brain MRI abnormalities include symmetrical basal ganglia and brainstem necrotic lesions, similar to mitochondrial encephalopathy. Half of affected children die in the first 2 years due to metabolic crisis, and there is currently no specific treatment for this disease. Di et al. injected a ssAAV2/8 vector carrying human hETHE1 cDNA into Eth1 KO mice, significantly improving symptom phenotypes and survival during the early stages of EE disease. This impressive outcome was linked to the improvement of the disease’s primary metabolic and biochemical indicators, such as EMA and thiosulfate levels in plasma and COX activity in tissues.435 Leigh syndrome is a progressive encephalopathy caused by defects in the NDUFS4, NDUFS3, and SurF1 genes and is the most common childhood mitochondrial disease.436 Leigh syndrome can result in a wide range of neurodegenerative disease phenotypes, including ataxia, dystonia, vision loss, and hearing loss. Reynaud-Dulaurier et al. intravenously injected AAV-PHP into Ndufs4-KO adult mice and showed reduced gliosis, significant body weight and locomotor activity improvement, and a significantly prolonged survival time.437
Mitochondrial neurogastrointestinal encephalomopathy is a rare multisystem disorder caused by mutations in the TYMP gene encoding thymidine phosphorylase (TP) and is inherited as an autosomal recessive disorder. The TYMP gene encodes thymidine phosphorylase, an enzyme necessary for the breakdown and transformation of certain nucleosides in the body. Mutations in the TP gene result in very low thymidine phosphorylase activity in cells and tissues. A lack of TP can lead to abnormally high levels of nucleoside, deoxyuridine, and thymidine in the body. Cabrera and her colleagues applied liver-specific promoter sequences, including TBG, PGK, HLP, and AAT, to drive hTYMP expression in a deoxyribonucleoside stress model. Compared with the activity of the commonly used promoter PGK, the activity of the liver-specific promoter sequence AAV2/8 is high in TYMP transgenic mice. Even at the lowest intravenous injection dose of 5 × 1011, the liver TP activity of knockout mice aged 8–11 weeks was effectively restored, and dThd levels returned to near-normal levels.316 Marta et al. utilized lipid nanoparticles or polymeric nanoparticles to synergistically deliver CRISPR/Cas9 and TYMP cDNA to liver cells, achieving targeted integration of the TYMP gene into the liver cells of the Tymp/Upp1 mouse model. The experimental results showed that after treatment, Tymp/Upp1 promoted the synthesis and secretion of TYMP mRNA and active enzymes in the liver in Tymp/Upp1 mice, resulting in an effective decrease in plasma nucleoside levels that persisted for 1 year.438 Similarly, Battani et al. investigated mitochondrial hepatocerebral disease and delivered the MPV17 into an MPV17 knockout mouse model gene using AAV8; the supramolecular complex level of MPV17 was effectively restored, and the progression of liver failure was delayed.439
The adenine nucleotide transporter protein encoded by ANT1 is the most abundant protein in the mitochondrial inner membrane. Under normal circumstances, the biological function of ANT1 is to form nonspecific pores that cause mitochondrial inner membrane permeability, output ATP to exchange for cytoplasmic ADP, and act as an electric pump.440 ANT1 mutations cause mitochondrial myopathy (MM), characterized by ragged red muscular fibers and progressive external ophthalmoplegia due to extraocular muscle paralysis. Flierl et al. were the first to conduct preclinical gene therapy research on MM by constructing an AAV2 vector carrying ANT1 cDNA and injecting it intramuscularly into ANT1 knockout mice. The ANT1 knockout mice exhibited significant exercise intolerance and clinical manifestations of metabolic acidosis. After AAV2 vector injection of ANT1 cDNA, ANT1 was stably expressed in muscle precursor cells and differentiated muscle fibers long-term, effectively improving mitochondrial respiratory efficiency and energy metabolism and reversing MM-related histopathological and clinical symptom changes.441 Wang et al. used intravenous TAZ gene replacement by AAV in a cardiomyocyte-specific TAZ knockout mouse model of Barth syndrome; TAZ gene replacement effectively prevented the progression of cardiomyopathy and fibrosis in a dose-dependent manner.442 Friedreich’s ataxia (FRDA) is caused by mutations in the FXN gene that result in severely reduced levels of expression of its encoded protein frataxin, leading to a reduction in iron-sulfur clusters and ATP production, which in turn affects neurological, cardiac, and pancreatic functioning in patients, with approximately 60% of patients passing away from cardiovascular problems.443 Perdomini et al. injected AAVrh10-CAG-hFXN into Mck-CKO mice, which effectively improved frataxin protein expression, delayed the progression of myocardial fibrosis, and prolonged the survival time.444
Over the past 10 years, clinical trials on gene therapy for mitochondrial diseases have mostly concentrated on the most common mutation in LHON: the m.11778G > A variation in MT-ND4.445 GS010, a recombinant AAV vector serotype 2 containing cDNA for the wild-type ND4 protein, was unilaterally injected intravitreally in clinical studies (NCT02652780 and NCT02652767). This treatment led to visual improvement in persons with LHON.323,446,447 Studies conducted in retinal ganglion cells and patient fibroblasts showed that the normal ND4 generated by the therapeutic gene appropriately moved to mitochondria due to MTS.448,449 On the ETDRS visual acuity chart, the best corrected visual acuity (BCVA) of the treated eyes improved by an average of 15 letters, while that of the eyes treated with sham surgery improved by an average of 13 letters.323 The visual improvement in untreated eyes may be due to the diffusion of rAAV2/2-ND4 between the optic nerve and the chiasma.323,446,450 Phase III studies have demonstrated that intravitreal administration of rAAV2-ND4 leads to enhanced visual acuity, exhibits favorable safety and systemic tolerance, and significantly advances the regulatory approval procedure.
Mitochondrial genome editing in mitochondrial diseases
Programmable nuclease-based mitochondrial gene editing tools are gradually being applied in preclinical studies. Bacman et al. intramuscularly injected mitoTALENs into the m.50624C > T mouse model of mitochondrial cardiomyopathy at a 1.0–1.5 × 1012 vg/dose of AAV9 to specifically recognize the disease-causing variant; this approach induced the degradation of the variant in muscle and heart via mtDNA cleavage, effectively restored the mtDNA level to that of the WT, restored the normal expression of tRNAAla, and ameliorated the symptoms of mitochondrial cardiomyopathy in mice.115 In a similar m.50624C > T mouse model, Gammage et al. injected the mitoZFNs-containing AAV 9.45 vector intravenously at a dose of 5 × 1012 vg, and their experimental results showed that mitochondrial tRNAAla expression levels in the heart of the mice were also effectively restored and that the tissue levels of lactate, pyruvate, and aspartate were improved.146 Zekonyte et al. delivered mitoARCUS into m.50624C > T juvenile and adult mouse models using an AAV9 vector, which effectively reduced the amount of mutant mtDNA in organs such as the heart, skeletal muscles, kidney, and liver by inducing specific dsDNA breaks; additionally, the level of mt-tRNAAla expression was significantly increased.70
As recently emerging technologies, mitochondria base editors have not been directly used in disease therapy in vivo; however, some initial studies have demonstrated the substantial potential of these tools in this setting. DdCBEs successfully edit mtDNA in the cardiac tissue of mice after delivery by AAV in vivo.163 Similarly, zinc finger-DdCBEs delivered by AAV, which are capable of base editing of both mitochondrial and nuclear DNA in vivo, have also been developed.173 In addition, the off-target effect of zinc finger DdCBEs is reduced by impeding spontaneous split DddA reassembly.173
In addition, mtDNA base editing technology can induce the generation of specific sites, providing useful tools for constructing mtDNA mutation models for mitochondrial disease research. In zebrafish, the DdCBE editing efficiency of mtDNA can reach 88.32% via embryo microinjection in the F0 generation.166 Another editing tool, FusX TALE Base Editor (FusXTBE), with an efficient editing site design script based on Python, induced 90+% editing efficiency in zebrafish.451 In mammalian models, the editing efficiency is much lower. The editing efficiency of DdCBE in F0 rats was reported to be 36.33%,162 and the initial mtDNA editing efficiency of DdCBE via embryo microinjection in mice was reported to be 0.25–23%.170 Some strategies have been developed to enhance this efficiency. DdCBE combined with a nuclear export signal (DdCBE-NES) enhances mtDNA editing by 38.9%.452 Coinjecting mitoTALENs with DdCBEs or DdCBE-NES yielded a 1.7–3-fold increase in editing efficiency.452
Application of artificial mitochondrial transfer
Mitochondrial transplantation
Preclinic studies
Most preclinical studies involve directly injecting mitochondria into the target organ or tissue, including the muscles,453,454 cardium,455,456 dorsal root ganglion,457 spinal cord,372 cerebral cortex,458 vitreous cavity of the eyes,459 internal carotid artery,324 left coronary ostium,458 renal capsule,460 lateral ventricles,461,462 striatum,463 femoral artery,463 ex vivo lung perfusion (EVLP),464 medial forebrain bundle (MFB),406 and renal artery.465 A few systematic administrations, including intravenous injection, have also achieved satisfactory results.466,467,468,469,470 Shi et al. conducted an intravenous injection of isolated mitochondria in mice and observed the distribution of exogenous mitochondria in multiple organs.470 Nakamura et al. reported that intravenously administered mitochondria can traverse the blood-brain barrier and disperse throughout the brain.469
In addition to the above direct administration, mitochondria can also exert their effects by being loaded in BMSCs,365,471 human peripheral blood mononuclear cells,472 and EVs.395 The details of preclinical studies on mitochondrial transplantation are summarized in Table 4.
Musculoskeletal system. BMSCs containing extra mitochondria from donor rats increased OXPHOS activity, ATP production, and bone repair in cranial defect model rats.471 Mitochondria from hUCMSCs promoted the recovery of motor function in the gastrocnemius muscle of acute limb Ischemia-reperfusion injury (IRI) model mice by increasing adipocyte browning and ATP production and reducing apoptosis.454 Tail vein injection of mitochondria isolated from donor mouse livers accelerated muscle repaired and restored muscular function in a muscle injury mouse model,466 and similar results have also been reported in a muscular atrophy rat model.453 Notably, recent research revealed an attractive result: the new autologous-produced mitochondria stimulated by the degradation process of transplanted mitochondria in ECs, rather than the transplanted mitochondria, enhanced metabolic activity and vessel formation.79,80
Circulation system. Myocardial ischemia in mice,455,473 rabbits,456,474 and pigs458,475 is improved by local mitochondrial injection. The related mechanism may include reducing cell apoptosis, ROS, endothelial cell senescence, and fibrosis and promoting ATP production, endothelial cell proliferation, and angiogenesis. Although most transplanted mitochondria were isolated from stem cells, autologous pectoralis could also provide optimal mitochondria without affecting the immune response or mtDNA heterogeneity. Most preclinical studies have involved directly injecting mitochondria into the myocardium, while Blitzer458 injected mitochondria into the left coronary ostium. Considering that coronary angiography techniques are quite mature in the clinic, mitochondrial transplantation via coronary arteries might be practical and potentially useful.
Neural system. Cerebral injury: Similar to myocardial ischemia, mitochondrial transplantation has been applied in cerebral ischemia mainly to enhance cellular functioning by decreasing ROS levels and regulating cellular metabolism. Cerebral ischemia models in both rats324,461,462,463,476 and mice469 have been confirmed to improve cognitive function and motor function and reduce infarct size and neurobehavioral deficits. However, whether mitochondria can pass through the blood-brain barrier is controversial. Xie et al.324 reported that no mitochondria pass through the intact blood-brain barrier, while Shi et al.470 and Nakamura et al.469 observed mitochondria in the brain after intravenous injection. One potential explanation is that disruption of the blood-brain barrier may occur during stroke,477 allowing the penetration of mitochondria. From this perspective, Dave et al. suggested a synergistic approach including mitochondria and HSP27 to enhance the viability of brain endothelial cells and enhance the structural stability of tight junctions.394 In addition to cerebral ischemia, injection of mitochondria into the cerebral cortex of traumatic brain injury (TBI) model mice improved spatial memory and alleviated anxiety.478 Notably, the findings obtained in vitro demonstrate the facile internalization of foreign mitochondria by neurons, astrocytes, and microglia,478 suggesting that mitochondrial transplantation might exert effects via multiple pathways.
Spinal Cord Injury: Mitochondrial transplantation into spinal cord injury (SCI) rat372,457 and mouse467 models mitigates apoptosis and neuroinflammation, improves locomotor functional recovery, and promotes tissue regeneration. An engineered mitochondrial compound (mitochondria-Tpp-CAQK-FAM) has been designed.467 Tpp are used to bind mitochondria via charge attraction. The CAQK peptide can efficiently bind to proteoglycan molecules in the damaged region after SCI, and FAM fluorescent molecules can be integrated for real-time monitoring. Transplanted mitochondria isolated from mouse bone marrow-differentiated macrophages were absorbed by recipient macrophages in an SCI model and effectively modulated inflammation. The ability to regulate and target inflammation shows significant potential for SCI management.
Neural degeneration: In Parkinson’s disease (PD) mice470 and rats,406 mitochondrial transplantation decreased ROS levels, prevented cell apoptosis and necrosis, and improved locomotive activity. In AD model mice, mitochondrial transplantation significantly improved cognitive performance.468 In addition to the central nervous system, peripheral nerve degeneration can be alleviated by mitochondrial transplantation. Jiang459 injected mitochondria from human induced pluripotent stem cells (iPSC-MSCs) into the vitreous cavity of the eyes of a retinal ganglion cell (RGC) degeneration model and showed a significant increase in RGC survival and retinal function.
Respiratory system. Islam et al. used live optical tools and observed that intratracheally instilled mBMSCs transferred mitochondria to the alveolar epithelium, leading to a protective effect.365 Subsequently, mitochondrial transplantation via lipopolysaccharide (LPS) was explored in mouse acute lung injury models, and reduced inflammation and lung injury were observed.395,479 In addition, rotenone (Rot)-induced asthma in a mouse model was alleviated by mitochondrial transplantation, resulting in the rescue of injured epithelial cells and a reduction in epithelial-mediated amplification of the immune response.480 Using mitochondria via EVLP to preserve lungs for transplantation could also be beneficial. Cloer et al. reported that mitochondrial transplantation reduced resistance and inflammation in both porcine and human ex vivo lungs, potentially reducing graft rejection.464
Immune system. Although the details of signal transduction between transplanted mitochondria and immune cells remain unknown, the biological function of immunocompetent cells is directly linked to their energetic status, which is influenced by mitochondria.481 Transplantation of mitochondria from human umbilical cord-derived mesenchymal stem cells (UC-MSCs) to human peripheral blood mononuclear cells (PBMCs) induced Treg differentiation in vitro.472 Administering mitochondria-treated T cells in a graft-versus-host disease (GVHD) mouse model greatly enhanced survival and decreased the invasion of specific T cells, revealing that mitochondrial transplantation has substantial potential for T‐cell reprogramming. Macrophages are another type of target cell for mitochondrial transplantation. Morrison et al. transplanted human MSC-derived mitochondria into alveolar macrophages (AMs) in vitro, and mitochondria-containing AMs significantly reduced inflammation in acute lung injury model mice.395 Xu et al. transplanted mitochondria from mouse bone marrow-derived macrophages (BMDMs) into the SCI environment to modulate inflammation via absorption by recipient macrophages.467
Urinary system. Chronic kidney injury related to diabetes mellitus in mouse482 and rat460 models was alleviated by mitochondrial transplantation by switching Macrophages from M1 to M2, inhibiting the inflammatory response and improving the cellular morphology of renal proximal tubular epithelial cells and the architecture of the basement membrane. In addition, acute kidney injury in rat483 and porcine465 models was alleviated by transplanting autologous mitochondria via renal arterial injection.
Notably, the administered mitochondria in swine were promptly absorbed by the kidneys and were not detected in any other organs, highlighting the efficiency and safety of intrarenal arterial mitochondria injection in treating kidney impairment.
Cancer. As stated above, the effects of transferred mitochondria in cancers remain controversial. No therapeutic application of mitochondrial transplantation has been applied for cancer treatment in vivo. Only limited pilot studies have been conducted on the behavior of transplanted mitochondria related to cancer cells in vivo. The aggressiveness of prostate cancer cells (PC3 and DU145) in vivo is enhanced when they are coinjected with cancer-associated fibroblasts capable of providing mitochondria.351 Similarly, AML-derived cells acquire normal mitochondria from coinjected BMSCs, increasing survival times after chemotherapy.353 Coinjected MSCs also transport mitochondria to rescue ALL B-cell precursors from ROS-inducing chemotherapy.354 Given the in vivo findings, blocking mitochondrial transfer rather than transplanting mitochondria might be beneficial in cancer therapy.
Clinical studies
Emani et al. injected mitochondria from autologous nonischemic skeletal muscle into 5 children requiring central extracorporeal membrane oxygenation (ECMO) assistance due to myocardial dysfunction following heart surgery.484 Four patients were successfully withdrawn from ECMO assistance. Guariento et al. conducted a comparative study (MT group, n = 10; control group, n = 14) with similar settings given these encouraging results.485 The rates of successful separation from ECMO (80% vs. 29%), median time to functional recovery after revascularization (2 days vs. 9 days), and incidence of cardiovascular events (20% vs. 79%) were significantly greater in the mitochondrial transduction group.
Jacoby et al. intravenously infused autogenous CD34+ cells augmented ex vivo (hematopoietic stem cells) that received mitochondria from maternal PBMCs into six patients with single large-scale mitochondrial DNA deletion syndrome (SLSMD).486 The technique was well tolerated, and all major adverse events during the study were related to the leukapheresis process or the underlying disease at the beginning of the study. After mitochondrial transplantation, all 6 patients exhibited an elevation in mtDNA content in their peripheral blood cells, ranging from 6 to 12 months, compared to the initial measurements. Two patients had a notable enhancement in their aerobic capabilities.
Considering the substantial potential of mitochondrial transplantation, several clinical trials, including those involving cerebral ischemia (NCT04998357), myocardial ischemia/reperfusion injury (NCT05669144), ECMO (NCT02851758), refractory polymyositis or dermatomyositis (NCT04976140), are ongoing. These trials might lead to a breakthrough in the application of mitochondrial transplantation (Table 5).
To date the primary artificial mitochondrial transfer techniques applied in clinical trials for mitochondrial diseases or mitochondrial-related disorders involve direct injection or ex vivo coculture. Most of these techniques remain in the laboratory or preclinical research stages. Nevertheless, their rapid progress is highly encouraging. For research purposes, these technologies provide precise, quantifiable, and highly reproducible methods for mitochondrial transplantation, enabling the exploration of post-transplant mitochondrial behavior and mechanisms. From a clinical application perspective, the advancement in miniaturization and non-invasiveness of transfer tools, along with improvements in the safety and targeting of surface modifications, offer promising prospects for future clinical applications. Additionally, the adoption of indirect transplantation strategies may facilitate the rapid clinical translation of many techniques. This approach involves extracting patient cells for ex vivo expansion and mitochondrial transfer before returning them to the patient. Such a method could significantly broaden the application of mitochondrial transfer techniques, achieving almost the same extensive applicability as cellular therapies.
Mitochondrial replacement therapy (MRT)
MRT was initially conceived to address infertility in older women and subsequently refined to prevent mitochondrial diseases, influencing a large population.487,488 At present, the available options for a couple to prevent the spread of mitochondrial problems are minimal. These include oocyte or embryo donation and the termination of a pregnancy upon prenatal diagnosis.
Mitochondria play a vital role in maintaining the quality of oocytes, promoting normal fertilization, sustaining subsequent embryo formation via energy generation, maintaining Ca2+ balance, and regulating oxidative stress.489 Mitochondrial manipulation has emerged as a new approach for enhancing the effectiveness of in vitro fertilization therapies in women with oocyte quality issues.490
MRTs involve two primary methodologies: heterologous and autologous. In the heterologous technique, mitochondria are obtained from an exogenous source. Heterologous procedures involve transferring the cytoplasm to the recipient oocyte or, vice versa, transferring the nucleus. Ooplasmic transfer (OT), also called partial cytoplasm transplantation, was the first technique developed to address defects in oocyte quality.491 OT involves transplanting cytoplasm components from a donor’s oocyte to the recipient oocyte, which contains functional mitochondria.492 The first pregnancy involving OT was announced in 1997.73 Conversely, total cytoplasm transplantation transports genetic material from a defective oocyte or zygote to a healthy cytoplasm, which can be achieved with various cytoplasm providers, including germinal vesicles (GVs),493 spindles,494,495 pronuclei,496 polar bodies,497,498 and blastomeres.499 However, the issue of mitochondrial heteroplasmy remains unresolved and leads to ethical conflicts, such as ‘three-parent’ babies.500
The autologous technique effectively solves the ethical and safety concerns related to heteroplasmy. Alternative sources of autologous mitochondria have been widely researched.500 Suggested autologous mitochondria sources include undeveloped oocytes,124 granulosa cells,501 germline stem cells (GSCs),502 and other stem cells.503
Due to the large difference between germ cell mitochondrial replacement and somatic cell mitochondrial transplantation, this review focused mainly on somatic cell mitochondrial transplantation. Detailed information on germ cell mitochondrial replacement has been reviewed.490,500,504,505
Conclusions and perspectives
Over the past decade, there have been significant advances in engineered mitochondria, including new mitochondrial gene editing tools and artificial mitochondrial transfer platforms. Comprehensive knowledge of the pathogenic genes, mitochondrial subcellular distribution, and the toxicity of therapeutic vectors is essential for the successful clinical implementation of these therapies. Appropriate cellular and animal models related to mitochondrial disease and precise biomarkers are necessary for designing effective trials.
Mitochondrial gene editing faces several significant challenges, with one of the most pressing issues being in vivo delivery and off-target effects. One promising direction that current research is actively exploring is the development of highly efficient targeted delivery systems for editing tools. Enhancing the specificity and effectiveness of these delivery systems could significantly improve the precision of mitochondrial gene editing in living organisms. An alternative direction that circumvents delivery issues is to manipulate reproductive cells or embryos to prevent the transmission of genetic disorders from the outset. In these scenarios, optimizing editing tools based on the precise identification of mutations using the latest detection technologies, such as modified mitochondrial genome sequencing,506,507 is crucial.
Regarding mitochondrial transplantation, a major limitation is its current applicability primarily to conditions characterized by “inadequate normal mitochondria,” rather than “excessive abnormal mitochondria.” Research into enhancing the clearance of abnormal mitochondria through mechanisms such as mitophagy and mitocytosis may address this gap. Additionally, bidirectional mitochondrial transfer329,508—a spontaneous phenomenon observed in several studies—presents another potential solution. Understanding and regulating the direction and process of mitochondrial transfer could be a crucial element in optimizing engineered mitochondria applications. Therefore, further research relying on advancements in tracing technologies is needed to understand mitochondrial transfer better. Long-term in vivo observation of transplanted mitochondria will provide valuable insights into their behavior and fate.
Future widespread clinical applications of mitochondrial gene editing are likely to be first realized in scenarios that are relatively easier to manipulate and observe. These include interventions in the reproductive cells or zygotes of parents with hereditary mitochondrial diseases or causative genes, and in mitochondrial diseases mainly affecting single or limited organs, such as LHON. In these cases, engineered mitochondria could be confined to a specific area, improving targeting and delivery effectiveness. As for mitochondrial transplantation, autologous resources might be first adopted for their reliable safety. Insufficient mitochondrial functions in the tissues that are supposed to have highly active mitochondria, such as myocardial insufficiency and neurological dysfunction, are optimal indications. Given the rapid and continuous technological advancements in engineered mitochondria through both gene editing and artificial mitochondrial transfer, their potential application is expected to continually expand across a wide range of diseases, from genetic mitochondrial diseases to non-genetic scenarios. Furthermore, a potentially more readily implementable approach is to perform ex vivo editing on mitochondria, followed by the selection and transfer of successfully edited mitochondria into the target organ. This method may simultaneously address specific challenges such as poor in vivo operability and the introduction of heterogeneity, thereby broadening the application boundaries.
References
Roger, A. J., Muñoz-Gómez, S. A. & Kamikawa, R. The origin and diversification of mitochondria. Curr. Biol. 27, R1177–r1192 (2017).
Zimorski, V., Ku, C., Martin, W. F. & Gould, S. B. Endosymbiotic theory for organelle origins. Curr. Opin. Microbiol. 22, 38–48 (2014).
Mitchell, P. Coupling of phosphorylation to electron and hydrogen transfer by a chemi-osmotic type of mechanism. Nature 191, 144–148 (1961).
Mitchell, P. & Moyle, J. Chemiosmotic hypothesis of oxidative phosphorylation. Nature 213, 137–139, (1967).
Monzel, A. S., Enríquez, J. A. & Picard, M. Multifaceted mitochondria: moving mitochondrial science beyond function and dysfunction. Nat. Metab. 5, 546–562 (2023).
Borst, P. The malate-aspartate shuttle (Borst cycle): how it started and developed into a major metabolic pathway. IUBMB Life 72, 2241–2259 (2020).
Benevenga, N. J. & Blemings, K. P. Unique aspects of lysine nutrition and metabolism. J. Nutr. 137, 1610s–1615s (2007).
Bassi, G., Sidhu, S. K. & Mishra, S. The expanding role of mitochondria, autophagy and lipophagy in steroidogenesis. Cells 10, 1851 (2021).
Miller, W. L. Steroid hormone synthesis in mitochondria. Mol. Cell Endocrinol. 379, 62–73 (2013).
Omura, T. & Gotoh, O. Evolutionary origin of mitochondrial cytochrome P450. J. Biochem. 161, 399–407 (2017).
Schlame, M. & Greenberg, M. L. Biosynthesis, remodeling and turnover of mitochondrial cardiolipin. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 1862, 3–7 (2017).
Eiyama, A. et al. Disturbed intramitochondrial phosphatidic acid transport impairs cellular stress signaling. J. Biol. Chem. 296, 100335 (2021).
Miliara, X. et al. Structural determinants of lipid specificity within Ups/PRELI lipid transfer proteins. Nat. Commun. 10, 1130 (2019).
Smirnoff, N. L-ascorbic acid biosynthesis. Vitam. Horm. 61, 241–266 (2001).
Aspatwar, A. et al. Mitochondrial carbonic anhydrase VA and VB: properties and roles in health and disease. J. Physiol. 601, 257–274 (2023).
Hernansanz-Agustín, P. & Enríquez, J. A. Generation of reactive oxygen species by mitochondria. Antioxidants 10, 415 (2021).
Murphy, M. P. How mitochondria produce reactive oxygen species. Biochem J. 417, 1–13 (2009).
Cecchini, G. Complexities of complex II: sulfide metabolism in vivo. J. Biol. Chem. 298, 101661 (2022).
Kumar, R. et al. A redox cycle with complex II prioritizes sulfide quinone oxidoreductase-dependent H(2)S oxidation. J. Biol. Chem. 298, 101435 (2022).
Landry, A. P. et al. A catalytic trisulfide in human sulfide quinone oxidoreductase catalyzes coenzyme a persulfide synthesis and inhibits butyrate oxidation. Cell Chem. Biol. 26, 1515–1525.e1514 (2019).
Braymer, J. J. & Lill, R. Iron-sulfur cluster biogenesis and trafficking in mitochondria. J. Biol. Chem. 292, 12754–12763 (2017).
Hinton, T. V., Batelu, S., Gleason, N. & Stemmler, T. L. Molecular characteristics of proteins within the mitochondrial Fe-S cluster assembly complex. Micron 153, 103181 (2022).
Read, A. D., Bentley, R. E., Archer, S. L. & Dunham-Snary, K. J. Mitochondrial iron-sulfur clusters: structure, function, and an emerging role in vascular biology. Redox Biol. 47, 102164 (2021).
O’Neill, L. A. J. & Artyomov, M. N. Itaconate: the poster child of metabolic reprogramming in macrophage function. Nat. Rev. Immunol. 19, 273–281 (2019).
Inigo, M., Deja, S. & Burgess, S. C. Ins and outs of the TCA cycle: the central role of anaplerosis. Annu. Rev. Nutr. 41, 19–47 (2021).
Williams, N. C. & O’Neill, L. A. J. A role for the Krebs cycle intermediate citrate in metabolic reprogramming in innate immunity and inflammation. Front. Immunol. 9, 141 (2018).
Bennett, C. F. et al. Peroxisomal-derived ether phospholipids link nucleotides to respirasome assembly. Nat. Chem. Biol. 17, 703–710 (2021).
Fang, J. et al. Dihydro-orotate dehydrogenase is physically associated with the respiratory complex and its loss leads to mitochondrial dysfunction. Biosci. Rep. 33, e00021 (2013).
Brosnan, M. E., MacMillan, L., Stevens, J. R. & Brosnan, J. T. Division of labour: how does folate metabolism partition between one-carbon metabolism and amino acid oxidation? Biochem. J. 472, 135–146, (2015).
Zheng, Y. & Cantley, L. C. Toward a better understanding of folate metabolism in health and disease. J. Exp. Med. 216, 253–266 (2019).
Picard, M. & Shirihai, O. S. Mitochondrial signal transduction. Cell Metab. 34, 1620–1653 (2022).
Sterling, K., Milch, P. O., Brenner, M. A. & Lazarus, J. H. Thyroid hormone action: the mitochondrial pathway. Science 197, 996–999, (1977).
Sterling, K. et al. Mitochondrial thyroid hormone receptor: localization and physiological significance. Science 201, 1126–1129 (1978).
Abadir, P. M. et al. Identification and characterization of a functional mitochondrial angiotensin system. Proc. Natl. Acad. Sci. USA 108, 14849–14854 (2011).
Picard, M. & Sandi, C. The social nature of mitochondria: Implications for human health. Neurosci. Biobehav. Rev. 120, 595–610 (2021).
Burtscher, J. et al. Mitochondrial stress and mitokines in aging. Aging Cell 22, e13770 (2023).
Kasahara, A. & Scorrano, L. Mitochondria: from cell death executioners to regulators of cell differentiation. Trends Cell Biol. 24, 761–770, (2014).
Palty, R. et al. NCLX is an essential component of mitochondrial Na+/Ca2+ exchange. Proc. Natl. Acad. Sci. USA 107, 436–441 (2010).
Baughman, J. M. et al. Integrative genomics identifies MCU as an essential component of the mitochondrial calcium uniporter. Nature 476, 341–345 (2011).
De Stefani, D. et al. A forty-kilodalton protein of the inner membrane is the mitochondrial calcium uniporter. Nature 476, 336–340 (2011).
Mitchell, P. & Moyle, J. Translocation of some anions cations and acids in rat liver mitochondria. Eur. J. Biochem. 9, 149–155, (1969).
Pfanner, N., Warscheid, B. & Wiedemann, N. Mitochondrial proteins: from biogenesis to functional networks. Nat. Rev. Mol. Cell Biol. 20, 267–284 (2019).
Neuspiel, M. et al. Cargo-selected transport from the mitochondria to peroxisomes is mediated by vesicular carriers. Curr. Biol. 18, 102–108 (2008).
König, T. & McBride, H. M. Mitochondrial-derived vesicles in metabolism, disease, and aging. Cell Metab. 36, 21–35 (2024).
Carrer, A. et al. Defining the molecular mechanisms of the mitochondrial permeability transition through genetic manipulation of F-ATP synthase. Nat. Commun. 12, 4835 (2021).
Bonora, M., Giorgi, C. & Pinton, P. Molecular mechanisms and consequences of mitochondrial permeability transition. Nat. Rev. Mol. Cell Biol. 23, 266–285 (2022).
Hernansanz-Agustín, P. & Enríquez, J. A. Functional segmentation of CoQ and cyt c pools by respiratory complex superassembly. Free Radic. Biol. Med. 167, 232–242 (2021).
Nesci, S., Trombetti, F. & Pagliarani, A. Nicotinamide nucleotide transhydrogenase as a sensor of mitochondrial biology. Trends Cell Biol. 30, 1–3 (2020).
Marchi, S. et al. Mitochondrial control of inflammation. Nat. Rev. Immunol. 23, 159–173 (2023).
Bertholet, A. M. et al. Mitochondrial uncouplers induce proton leak by activating AAC and UCP1. Nature 606, 180–187 (2022).
Chouchani, E. T., Kazak, L. & Spiegelman, B. M. New advances in adaptive thermogenesis: UCP1 and beyond. Cell Metab. 29, 27–37 (2019).
Kazak, L. et al. A creatine-driven substrate cycle enhances energy expenditure and thermogenesis in beige fat. Cell 163, 643–655 (2015).
Bock, F. J. & Tait, S. W. G. Mitochondria as multifaceted regulators of cell death. Nat. Rev. Mol. Cell Biol. 21, 85–100 (2020).
Zong, Y. et al. Mitochondrial dysfunction: mechanisms and advances in therapy. Signal Transduct. Target. Ther. 9, 124 (2024).
Giles, R. E., Blanc, H., Cann, H. M. & Wallace, D. C. Maternal inheritance of human mitochondrial DNA. Proc. Natl. Acad. Sci. USA 77, 6715–6719 (1980).
Wallace, D. C. et al. Familial mitochondrial encephalomyopathy (MERRF): genetic, pathophysiological, and biochemical characterization of a mitochondrial DNA disease. Cell 55, 601–610 (1988).
Holt, I. J., Harding, A. E. & Morgan-Hughes, J. A. Deletions of muscle mitochondrial DNA in patients with mitochondrial myopathies. Nature 331, 717–719, (1988).
Rahman, J. & Rahman, S. Mitochondrial medicine in the omics era. Lancet 391, 2560–2574 (2018).
Gorman, G. S. et al. Mitochondrial donation—how many women could benefit? N. Engl. J. Med. 372, 885–887 (2015).
Chinnery, P. F. Mitochondrial replacement in the clinic. N. Engl. J. Med. 382, 1855–1857 (2020).
Adashi, E. Y. et al. Mitochondrial disease: replace or edit? Science 373, 1200–1201 (2021).
Mok, B. Y. et al. A bacterial cytidine deaminase toxin enables CRISPR-free mitochondrial base editing. Nature 583, 631–637 (2020).
Harrington, J. S. et al. Mitochondria in health, disease, and aging. Physiol. Rev. 103, 2349–2422 (2023).
Lin, M. T. & Beal, M. F. Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 443, 787–795, (2006).
Ho, G. T. & Theiss, A. L. Mitochondria and inflammatory bowel diseases: toward a stratified therapeutic intervention. Annu. Rev. Physiol. 84, 435–459 (2022).
Wallace, D. C. Mitochondria and cancer. Nat. Rev. Cancer 12, 685–698 (2012).
Srivastava, S. & Moraes, C. T. Manipulating mitochondrial DNA heteroplasmy by a mitochondrially targeted restriction endonuclease. Hum. Mol. Genet. 10, 3093–3099, (2001).
Minczuk, M. et al. Development of a single-chain, quasi-dimeric zinc-finger nuclease for the selective degradation of mutated human mitochondrial DNA. Nucleic Acids Res. 36, 3926–3938 (2008).
Bacman, S. R. et al. Specific elimination of mutant mitochondrial genomes in patient-derived cells by mitoTALENs. Nat. Med. 19, 1111–1113 (2013).
Zekonyte, U. et al. Mitochondrial targeted meganuclease as a platform to eliminate mutant mtDNA in vivo. Nat. Commun. 12, 3210 (2021).
Cho, S. I. et al. Targeted A-to-G base editing in human mitochondrial DNA with programmable deaminases. Cell 185, 1764–1776.e1712 (2022).
Clark, M. A. & Shay, J. W. Mitochondrial transformation of mammalian cells. Nature 295, 605–607, (1982).
Cohen, J. et al. Birth of infant after transfer of anucleate donor oocyte cytoplasm into recipient eggs. Lancet 350, 186–187 (1997).
Rustom, A. et al. Nanotubular highways for intercellular organelle transport. Science 303, 1007–1010 (2004).
Spees, J. L., Olson, S. D., Whitney, M. J. & Prockop, D. J. Mitochondrial transfer between cells can rescue aerobic respiration. Proc. Natl. Acad. Sci. USA 103, 1283–1288, (2006).
Boudreau, L. H. et al. Platelets release mitochondria serving as substrate for bactericidal group IIA-secreted phospholipase A2 to promote inflammation. Blood 124, 2173–2183 (2014).
Craven, L. et al. Research into policy: a brief history of mitochondrial donation. Stem Cells 34, 265–267 (2016).
Jiao, H. et al. Mitocytosis, a migrasome-mediated mitochondrial quality-control process. Cell 184, 2896–2910.e2813 (2021).
Lin, R. Z. et al. Mitochondrial transfer mediates endothelial cell engraftment through mitophagy. Nature 629, 660–668 (2024).
Evans, C. S. Cells destroy donated mitochondria to build blood vessels. Nature 629, 539–541 (2024).
Calvo, S. E., Clauser, K. R. & Mootha, V. K. MitoCarta2.0: an updated inventory of mammalian mitochondrial proteins. Nucleic Acids Res. 44, D1251–1257 (2016).
Smith, A. C. & Robinson, A. J. MitoMiner v3.1, an update on the mitochondrial proteomics database. Nucleic Acids Res. 44, D1258–1261 (2016).
Montoya, J. et al. Identification of initiation sites for heavy-strand and light-strand transcription in human mitochondrial DNA. Proc. Natl. Acad. Sci. USA 79, 7195–7199 (1982).
Montoya, J., Gaines, G. L. & Attardi, G. The pattern of transcription of the human mitochondrial rRNA genes reveals two overlapping transcription units. Cell 34, 151–159, (1983).
Chang, D. D. & Clayton, D. A. Precise identification of individual promoters for transcription of each strand of human mitochondrial DNA. Cell 36, 635–643, (1984).
Posse, V. & Gustafsson, C. M. Human mitochondrial transcription factor B2 is required for promoter melting during initiation of transcription. J. Biol. Chem. 292, 2637–2645 (2017).
Sologub, M. et al. TFB2 is a transient component of the catalytic site of the human mitochondrial RNA polymerase. Cell 139, 934–944 (2009).
Ramachandran, A. et al. Human mitochondrial transcription factors TFAM and TFB2M work synergistically in promoter melting during transcription initiation. Nucleic Acids Res. 45, 861–874 (2017).
Boehm, E. et al. FASTKD1 and FASTKD4 have opposite effects on expression of specific mitochondrial RNAs, depending upon their endonuclease-like RAP domain. Nucleic Acids Res. 45, 6135–6146 (2017).
Popow, J. et al. FASTKD2 is an RNA-binding protein required for mitochondrial RNA processing and translation. RNA 21, 1873–1884 (2015).
Antonicka, H. & Shoubridge, E. A. Mitochondrial RNA granules are centers for posttranscriptional RNA processing and ribosome biogenesis. Cell Rep. 10, 920–932 (2015).
Haque, M. E. & Spremulli, L. L. Roles of the N- and C-terminal domains of mammalian mitochondrial initiation factor 3 in protein biosynthesis. J. Mol. Biol. 384, 929–940 (2008).
Bhargava, K. & Spremulli, L. L. Role of the N- and C-terminal extensions on the activity of mammalian mitochondrial translational initiation factor 3. Nucleic Acids Res. 33, 7011–7018 (2005).
Ng, Y. S. et al. Mitochondrial disease in adults: recent advances and future promise. Lancet Neurol. 20, 573–584 (2021).
Gorman, G. S. et al. Mitochondrial diseases. Nat. Rev. Dis. Prim. 2, 1–22 (2016).
Silva-Pinheiro, P. & Minczuk, M. The potential of mitochondrial genome engineering. Nat. Rev. Genet 23, 199–214 (2022).
Ashar, F. N. et al. Association of mitochondrial DNA copy number with cardiovascular disease. JAMA Cardiol. 2, 1247–1255, (2017).
Herbers, E. et al. Tissue specific differences in mitochondrial DNA maintenance and expression. Mitochondrion 44, 85–92 (2019).
Howell, N., Kubacka, I. & Mackey, D. A. How rapidly does the human mitochondrial genome evolve? Am. J. Hum. Genet. 59, 501–509, (1996).
Jazin, E. et al. Mitochondrial mutation rate revisited: hot spots and polymorphism. Nat. Genet. 18, 109–110 (1998).
Raha, S. & Robinson, B. H. Mitochondria, oxygen free radicals, disease and ageing. Trends Biochem. Sci. 25, 502–508, (2000).
Kyriakouli, D. S., Boesch, P., Taylor, R. W. & Lightowlers, R. N. Progress and prospects: gene therapy for mitochondrial DNA disease. Gene Ther. 15, 1017–1023, (2008).
Kulkarni, C. A. et al. A novel triphenylphosphonium carrier to target mitochondria without uncoupling oxidative phosphorylation. J. Med. Chem. 64, 662–676 (2021).
Arafa, K. K., Hamzawy, M. A., Mousa, S. A. & El-Sherbiny, I. M. Mitochondria-targeted alginate/triphenylphosphonium-grafted-chitosan for treatment of hepatocellular carcinoma. RSC Adv. 12, 21690–21703 (2022).
Chen, X. et al. Correction of a homoplasmic mitochondrial tRNA mutation in patient-derived iPSCs via a mitochondrial base editor. Commun. Biol. 6, 1116 (2023).
Taylor, R. W., Chinnery, P. F., Turnbull, D. M. & Lightowlers, R. N. Selective inhibition of mutant human mitochondrial DNA replication in vitro by peptide nucleic acids. Nat. Genet. 15, 212–215, (1997).
Comte, C. et al. Mitochondrial targeting of recombinant RNAs modulates the level of a heteroplasmic mutation in human mitochondrial DNA associated with Kearns Sayre Syndrome. Nucleic Acids Res. 41, 418–433 (2013).
Entelis, N. S. et al. 5 S rRNA and tRNA import into human mitochondria. Comparison of in vitro requirements. J. Biol. Chem. 276, 45642–45653 (2001).
Loutre, R. et al. Anti-replicative recombinant 5S rRNA molecules can modulate the mtDNA heteroplasmy in a glucose-dependent manner. PLoS One 13, e0199258 (2018).
Nissanka, N. & Moraes, C. T. Mitochondrial DNA heteroplasmy in disease and targeted nuclease‐based therapeutic approaches. EMBO Rep. 21, e49612 (2020).
Zekonyte, U., Bacman, S. R. & Moraes, C. T. DNA‐editing enzymes as potential treatments for heteroplasmic mtDNA diseases. J. Intern. Med. 287, 685–697 (2020).
Rong, Z. et al. The mitochondrial response to DNA damage. Front. Cell Dev. Biol. 9, 669379 (2021).
Pereira, C. V. & Moraes, C. T. Current strategies towards therapeutic manipulation of mtDNA heteroplasmy. Front. Biosci. (Landmark Ed.) 22, 991 (2017).
Habbane, M. et al. Human mitochondrial DNA: Particularities and diseases. Biomedicines 9, 1364 (2021).
Bacman, S. R. et al. MitoTALEN reduces mutant mtDNA load and restores tRNA(Ala) levels in a mouse model of heteroplasmic mtDNA mutation. Nat. Med. 24, 1696–1700 (2018).
Castellani, C. A. et al. Thinking outside the nucleus: Mitochondrial DNA copy number in health and disease. Mitochondrion 53, 214–223 (2020).
Lee, W. et al. Mitochondrial DNA copy number is regulated by DNA methylation and demethylation of POLGA in stem and cancer cells and their differentiated progeny. Cell Death Dis. 6, e1664 (2015).
Cong, L. et al. Multiplex genome engineering using CRISPR/Cas systems. Science 339, 819–823 (2013).
Ran, F. A. et al. Genome engineering using the CRISPR-Cas9 system. Nat. Protoc. 8, 2281–2308 (2013).
Jiang, F. & Doudna, J. A. CRISPR–Cas9 structures and mechanisms. Annu. Rev. Biophys. 46, 505–529 (2017).
Hsu, P. D., Lander, E. S. & Zhang, F. Development and applications of CRISPR-Cas9 for genome engineering. Cell 157, 1262–1278 (2014).
Doudna, J. A. & Charpentier, E. The new frontier of genome engineering with CRISPR-Cas9. Science 346, 1258096 (2014).
Redman, M., King, A., Watson, C. & King, D. What is CRISPR/Cas9? Arch. Dis. Child Educ. Pract. Ed. 101, 213–215, (2016).
Zhai, J. et al. In vitro activation of follicles and fresh tissue auto-transplantation in primary ovarian insufficiency patients. J. Clin. Endocrinol. Metab. 101, 4405–4412 (2016).
Chylinski, K., Makarova, K. S., Charpentier, E. & Koonin, E. V. Classification and evolution of type II CRISPR-Cas systems. Nucleic Acids Res. 42, 6091–6105 (2014).
Zhang, F., Wen, Y. & Guo, X. CRISPR/Cas9 for genome editing: progress, implications and challenges. Hum. Mol. Genet. 23, R40–R46 (2014).
Mei, Y. et al. Recent progress in CRISPR/Cas9 technology. J. Genet. Genom. 43, 63–75 (2016).
Mahata, B. et al. Functional delivery of a cytosolic tRNA into mutant mitochondria of human cells. Science 314, 471–474 (2006).
Goswami, S. et al. A bifunctional tRNA import receptor from Leishmania mitochondria. Proc. Natl. Acad. Sci. USA 103, 8354–8359 (2006).
Mukherjee, S. et al. Necessary and sufficient factors for the import of transfer RNA into the kinetoplast mitochondrion. EMBO Rep. 8, 589–595 (2007).
Wang, B. et al. CRISPR/Cas9-mediated mutagenesis at microhomologous regions of human mitochondrial genome. Sci. China Life Sci. 64, 1463–1472 (2021).
Amai, T., Tsuji, T., Ueda, M. & Kuroda, K. Development of a mito-CRISPR system for generating mitochondrial DNA-deleted strain in Saccharomyces cerevisiae. Biosci. Biotechnol. Biochem. 85, 895–901 (2021).
Jo, A. et al. Efficient mitochondrial genome editing by CRISPR/Cas9. Biomed. Res. Int. 2015, 305716 (2015).
Bian, W. P. et al. Knock-in strategy for editing human and zebrafish mitochondrial DNA using Mito-CRISPR/Cas9 system. ACS Synth. Biol. 8, 621–632 (2019).
Gammage, P. A., Moraes, C. T. & Minczuk, M. Mitochondrial genome engineering: the revolution may not be CRISPR-Ized. Trends Genet. 34, 101–110 (2018).
Jeandard, D. et al. Import of non-coding RNAs into human mitochondria: a critical review and emerging approaches. Cells. 8, 286 (2019).
Antón, Z. et al. Mitochondrial import, health and mtDNA copy number variability seen when using type II and type V CRISPR effectors. J. Cell Sci. 133, jcs248468 (2020).
Patananan, A. N., Wu, T. H., Chiou, P. Y. & Teitell, M. A. Modifying the mitochondrial genome. Cell Metab. 23, 785–796, (2016).
Fox, T. D., Sanford, J. C. & McMullin, T. W. Plasmids can stably transform yeast mitochondria lacking endogenous mtDNA. Proc. Natl. Acad. Sci. USA 85, 7288–7292, (1988).
Moretton, A. et al. Selective mitochondrial DNA degradation following double-strand breaks. PLoS One 12, e0176795 (2017).
Bayona-Bafaluy, M. P. et al. Rapid directional shift of mitochondrial DNA heteroplasmy in animal tissues by a mitochondrially targeted restriction endonuclease. Proc. Natl. Acad. Sci. USA 102, 14392–14397 (2005).
Bacman, S. R., Williams, S. L., Duan, D. & Moraes, C. T. Manipulation of mtDNA heteroplasmy in all striated muscles of newborn mice by AAV9-mediated delivery of a mitochondria-targeted restriction endonuclease. Gene Ther. 19, 1101–1106 (2012).
Tanaka, M. et al. Gene therapy for mitochondrial disease by delivering restriction endonuclease smaI into mitochondria. J. Biomed. Sci. 9, 534–541 (2002).
Alexeyev, M. F. et al. Selective elimination of mutant mitochondrial genomes as therapeutic strategy for the treatment of NARP and MILS syndromes. Gene Ther. 15, 516–523 (2008).
Gammage, P. A. et al. Near-complete elimination of mutant mtDNA by iterative or dynamic dose-controlled treatment with mtZFNs. Nucleic Acids Res. 44, 7804–7816 (2016).
Gammage, P. A. et al. Genome editing in mitochondria corrects a pathogenic mtDNA mutation in vivo. Nat. Med. 24, 1691–1695 (2018).
Hashimoto, M. et al. MitoTALEN: a general approach to reduce mutant mtDNA loads and restore oxidative phosphorylation function in mitochondrial diseases. Mol. Ther. 23, 1592–1599 (2015).
Qasim, W. et al. Molecular remission of infant B-ALL after infusion of universal TALEN gene-edited CAR T cells. Sci. Transl. Med. 9, eaaj2013 (2017).
Miller, J. C. et al. A TALE nuclease architecture for efficient genome editing. Nat. Biotechnol. 29, 143–148 (2011).
Gammage, P. A. et al. Mitochondrially targeted ZFNs for selective degradation of pathogenic mitochondrial genomes bearing large-scale deletions or point mutations. EMBO Mol. Med. 6, 458–466 (2014).
Persson, Ö. et al. Copy-choice recombination during mitochondrial L-strand synthesis causes DNA deletions. Nat. Commun. 10, 759 (2019).
Falabella, M. et al. Gene therapy for primary mitochondrial diseases: experimental advances and clinical challenges. Nat. Rev. Neurol. 18, 689–698 (2022).
Pulicherla, N. et al. Engineering liver-detargeted AAV9 vectors for cardiac and musculoskeletal gene transfer. Mol. Ther. 19, 1070–1078 (2011).
Minczuk, M. et al. Sequence-specific modification of mitochondrial DNA using a chimeric zinc finger methylase. Proc. Natl Acad. Sci. USA 103, 19689–19694 (2006).
Reddy, P. et al. Selective elimination of mitochondrial mutations in the germline by genome editing. Cell 161, 459–469 (2015).
Wai, T. et al. The role of mitochondrial DNA copy number in mammalian fertility. Biol. Reprod. 83, 52–62 (2010).
Wang, T. et al. Polar body genome transfer for preventing the transmission of inherited mitochondrial diseases. Cell 157, 1591–1604 (2014).
Arnould, S. et al. The I-CreI meganuclease and its engineered derivatives: applications from cell modification to gene therapy. Protein Eng. Des. Sel. 24, 27–31 (2011).
Zincarelli, C., Soltys, S., Rengo, G. & Rabinowitz, J. E. Analysis of AAV serotypes 1-9 mediated gene expression and tropism in mice after systemic injection. Mol. Ther. 16, 1073–1080, (2008).
Barrera-Paez, J. D. & Moraes, C. T. Mitochondrial genome engineering coming-of-age. Trends Genet. 38, 869–880 (2022).
Aushev, M. & Herbert, M. Mitochondrial genome editing gets precise. Nature 583, 521–522 (2020).
Qi, X. et al. Precision modeling of mitochondrial disease in rats via DdCBE-mediated mtDNA editing. Cell Discov. 7, 95 (2021).
Silva-Pinheiro, P. et al. In vivo mitochondrial base editing via adeno-associated viral delivery to mouse post-mitotic tissue. Nat. Commun. 13, 750 (2022).
Tan, L. et al. A conditional knockout rat resource of mitochondrial protein-coding genes via a DdCBE-induced premature stop codon. Sci. Adv. 9, eadf2695 (2023).
Chen, X. et al. DdCBE-mediated mitochondrial base editing in human 3PN embryos. Cell Discov. 8, 8 (2022).
Guo, J. et al. Precision modeling of mitochondrial diseases in zebrafish via DdCBE-mediated mtDNA base editing. Cell Discov. 7, 78 (2021).
Guo, J. et al. A DddA ortholog-based and transactivator-assisted nuclear and mitochondrial cytosine base editors with expanded target compatibility. Mol. Cell 83, 1710–1724.e1717 (2023).
Guo, J. et al. DdCBE mediates efficient and inheritable modifications in mouse mitochondrial genome. Mol. Ther. Nucleic Acids 27, 73–80 (2022).
Nilsen, H. et al. Nuclear and mitochondrial uracil-DNA glycosylases are generated by alternative splicing and transcription from different positions in the UNG gene. Nucleic Acids Res. 25, 750–755 (1997).
Lee, H. et al. Mitochondrial DNA editing in mice with DddA-TALE fusion deaminases. Nat. Commun. 12, 1190 (2021).
Richter, M. F. et al. Phage-assisted evolution of an adenine base editor with improved Cas domain compatibility and activity. Nat. Biotechnol. 38, 883–891 (2020).
Lim, K., Cho, S. I. & Kim, J. S. Nuclear and mitochondrial DNA editing in human cells with zinc finger deaminases. Nat. Commun. 13, 366 (2022).
Willis, J. C. W. et al. Compact zinc finger base editors that edit mitochondrial or nuclear DNA in vitro and in vivo. Nat. Commun. 13, 7204 (2022).
Berglund, A.-K. et al. Nucleotide pools dictate the identity and frequency of ribonucleotide incorporation in mitochondrial DNA. PLoS Genet. 13, e1006628 (2017).
Fernández-Silva, P., Enriquez, J. A. & Montoya, J. Replication and transcription of mammalian mitochondrial DNA. Exp. Physiol. 88, 41–56 (2003).
Fusté, J. M. et al. Mitochondrial RNA polymerase is needed for activation of the origin of light-strand DNA replication. Mol. Cell 37, 67–78 (2010).
Gustafsson, C. M., Falkenberg, M. & Larsson, N.-G. Maintenance and expression of mammalian mitochondrial DNA. Annu. Rev. Biochem. 85, 133–160, (2016).
Jedynak-Slyvka, M., Jabczynska, A. & Szczesny, R. J. Human mitochondrial RNA processing and modifications: overview. Int. J. Mol. Sci. 22, 7999 (2021).
Huang, T. et al. Iron oxide nanoparticles augment the intercellular mitochondrial transfer-mediated therapy. Sci. Adv. 7, eabj0534 (2021).
Bandiera, S. et al. Nuclear outsourcing of RNA interference components to human mitochondria. PLoS One 6, e20746 (2011).
Mustafa, M. F., Fakurazi, S., Abdullah, M. A. & Maniam, S. Pathogenic mitochondria DNA mutations: current detection tools and interventions. Genes 11, 192 (2020).
Liu, S. & Chong, W. Roles of LncRNAs in regulating mitochondrial dysfunction in septic cardiomyopathy. Front. Immunol. 12, 802085 (2021).
Zhu, L.-R. et al. Advances in RNA epigenetic modifications in hepatocellular carcinoma and potential targeted intervention strategies. Front. Cell Dev. Biol. 9, 777007 (2021).
Sahel, J. A. et al. Gene therapies for the treatment of leber hereditary optic neuropathy. Int. Ophthalmol. Clin. 61, 195–208 (2021).
Castanotto, D. & Rossi, J. J. The promises and pitfalls of RNA-interference-based therapeutics. Nature 457, 426–433, (2009).
Yu, A.-M., Choi, Y. H. & Tu, M.-J. RNA drugs and RNA targets for small molecules: principles, progress, and challenges. Pharm. Rev. 72, 862–898 (2020).
Zhou, L.-Y. et al. Current RNA-based therapeutics in clinical trials. Curr. Gene Ther. 19, 172–196 (2019).
Sahin, U., Karikó, K. & Türeci, Ö. mRNA-based therapeutics—developing a new class of drugs. Nat. Rev. Drug Discov. 13, 759–780, (2014).
Traber, G. M. & Yu, A.-M. RNAi-based therapeutics and novel RNA bioengineering technologies. J. Pharm. Exp. Ther. 384, 133–154 (2023).
Chernega, T., Choi, J., Salmena, L. & Andreazza, A. C. Mitochondrion-targeted RNA therapies as a potential treatment strategy for mitochondrial diseases. Mol. Ther. Nucleic Acids 30, 359–377 (2022).
Maghsoudnia, N. et al. Mitochondrial delivery of microRNA mimic let-7b to NSCLC cells by PAMAM-based nanoparticles. J. Drug Target 28, 818–830 (2020).
Wang, G. et al. Correcting human mitochondrial mutations with targeted RNA import. Proc. Natl Acad. Sci. USA 109, 4840–4845 (2012).
Mukherjee, S., Bhattacharyya, S. N. & Adhya, S. Stepwise transfer of tRNA through the double membrane of leishmania mitochondria. J. Biol. Chem. 274, 31249–31255 (1999).
Mukherjee, S., Mahata, B., Mahato, B. & Adhya, S. Targeted mRNA degradation by complex-mediated delivery of antisense RNAs to intracellular human mitochondria. Hum. Mol. Genet. 17, 1292–1298, (2008).
Kawamura, E., Hibino, M., Harashima, H. & Yamada, Y. Targeted mitochondrial delivery of antisense RNA-containing nanoparticles by a MITO-Porter for safe and efficient mitochondrial gene silencing. Mitochondrion 49, 178–188 (2019).
Cruz-Zaragoza, L. D. et al. An in vitro system to silence mitochondrial gene expression. Cell 184, 5824–5837.e5815 (2021).
Ciesielski, S. J. et al. Interaction of J-protein co-chaperone Jac1 with Fe-S scaffold Isu is indispensable in vivo and conserved in evolution. J. Mol. Biol. 417, 1–12 (2012).
Zhang, X. et al. MicroRNA directly enhances mitochondrial translation during muscle differentiation. Cell 158, 607–619 (2014).
Bobbin, M. L. & Rossi, J. J. RNA interference (RNAi)-based therapeutics: delivering on the promise? Annu. Rev. Pharm. Toxicol. 56, 103–122, (2016).
Aigner, A. Gene silencing through RNA interference (RNAi) in vivo: strategies based on the direct application of siRNAs. J. Biotechnol. 124, 12–25 (2006).
Agrawal, N. et al. RNA interference: biology, mechanism, and applications. Microbiol. Mol. Biol. Rev. 67, 657–685 (2003).
Hoye, A. T. et al. Targeting mitochondria. Acc. Chem. Res. 41, 87–97 (2008).
Yang, S. et al. Mitochondria: a novel therapeutic target in diabetic nephropathy. Curr. Med. Chem. 24, 3185–3202 (2017).
Ishikawa, T. et al. Mitochondrial transgene expression via an artificial mitochondrial DNA vector in cells from a patient with a mitochondrial disease. J. Control Release 274, 109–117 (2018).
Kren, B. T. et al. MicroRNAs identified in highly purified liver-derived mitochondria may play a role in apoptosis. RNA Biol. 6, 65–72 (2009).
Erturk, E. et al. Mitochondrial miRNAs (MitomiRs): their potential roles in breast and other cancers. Mitochondrion 66, 74–81 (2022).
Gao, K. et al. Active RNA interference in mitochondria. Cell Res. 31, 219–228 (2021).
Yamada, Y. et al. Mitochondrial delivery of an anticancer drug via systemic administration using a mitochondrial delivery system that inhibits the growth of drug-resistant cancer engrafted on mice. J. Pharm. Sci. 109, 2493–2500 (2020).
Kawamura, E. et al. Validation of gene therapy for mutant mitochondria by delivering mitochondrial RNA Using a MITO-Porter. Mol. Ther. Nucleic Acids 20, 687–698 (2020).
Mahato, B., Jash, S. & Adhya, S. RNA-mediated restoration of mitochondrial function in cells harboring a Kearns Sayre Syndrome mutation. Mitochondrion 11, 564–574 (2011).
Yamada, Y. et al. Validation of a mitochondrial RNA therapeutic strategy using fibroblasts from a Leigh syndrome patient with a mutation in the mitochondrial ND3 gene. Sci. Rep. 10, 7511 (2020).
Jiang, L. et al. Dual mRNA therapy restores metabolic function in long-term studies in mice with propionic acidemia. Nat. Commun. 11, 5339 (2020).
Choudhury, R. et al. Engineering RNA endonucleases with customized sequence specificities. Nat. Commun. 3, 1147 (2012).
Takeshita, D. et al. Crystal structure of the PIN domain of human telomerase-associated protein EST1A. Proteins 68, 980–989 (2007).
Sieber, F., Duchêne, A. M. & Maréchal-Drouard, L. Mitochondrial RNA import: from diversity of natural mechanisms to potential applications. Int. Rev. Cell Mol. Biol. 287, 145–190, (2011).
Kim, K. M., Noh, J. H., Abdelmohsen, K. & Gorospe, M. Mitochondrial noncoding RNA transport. BMB Rep. 50, 164–174 (2017).
Rubio, M. A. et al. Mammalian mitochondria have the innate ability to import tRNAs by a mechanism distinct from protein import. Proc. Natl. Acad. Sci. USA 105, 9186–9191 (2008).
Rubio, M. A. & Hopper, A. K. Transfer RNA travels from the cytoplasm to organelles. Wiley Interdiscip. Rev. RNA 2, 802–817, (2011).
Smirnov, A., Entelis, N., Martin, R. P. & Tarassov, I. Biological significance of 5S rRNA import into human mitochondria: role of ribosomal protein MRP-L18. Genes Dev. 25, 1289–1305 (2011).
Salinas-Giegé, T., Giegé, R. & Giegé, P. tRNA biology in mitochondria. Int. J. Mol. Sci. 16, 4518–4559 (2015).
Schneider, A. Mitochondrial tRNA import and its consequences for mitochondrial translation. Annu. Rev. Biochem 80, 1033–1053 (2011).
Steitz, J. A. et al. A 5S rRNA/L5 complex is a precursor to ribosome assembly in mammalian cells. J. Cell Biol. 106, 545–556 (1988).
Smirnov, A. et al. Two distinct structural elements of 5S rRNA are needed for its import into human mitochondria. RNA 14, 749–759 (2008).
Autour, A. et al. Fluorogenic RNA Mango aptamers for imaging small non-coding RNAs in mammalian cells. Nat. Commun. 9, 656 (2018).
Smirnov, A. et al. Mitochondrial enzyme rhodanese is essential for 5S ribosomal RNA import into human mitochondria. J. Biol. Chem. 285, 30792–30803 (2010).
Hiller, S. et al. The 3D structures of VDAC represent a native conformation. Trends Biochem. Sci. 35, 514–521 (2010).
Campo, M. L., Peixoto, P. M. & Martínez-Caballero, S. Revisiting trends on mitochondrial mega-channels for the import of proteins and nucleic acids. J. Bioenerg. Biomembr. 49, 75–99 (2017).
Becker, T. & Wagner, R. Mitochondrial outer membrane channels: emerging diversity in transport processes. Bioessays 40, e1800013 (2018).
Raja, M. A. G., Katas, H. & Amjad, M. W. Design, mechanism, delivery and therapeutics of canonical and dicer-substrate siRNA. Asian J. Pharm. Sci. 14, 497–510 (2019).
Kara, G., Calin, G. A. & Ozpolat, B. RNAi-based therapeutics and tumor-targeted delivery in cancer. Adv. Drug Deliv. Rev. 182, 114113 (2022).
Bryson, T. E., Anglin, C. M., Bridges, P. H. & Cottle, R. N. Nuclease-mediated gene therapies for inherited metabolic diseases of the liver. Yale J. Biol. Med. 90, 553–566 (2017).
Nayerossadat, N., Maedeh, T. & Ali, P. A. Viral and nonviral delivery systems for gene delivery. Adv. Biomed. Res. 1, 27 (2012).
Wu, Z., Yang, H. & Colosi, P. Effect of genome size on AAV vector packaging. Mol. Ther. 18, 80–86 (2010).
Ramasamy, T., Munusamy, S., Ruttala, H. B. & Kim, J. O. Smart nanocarriers for the delivery of nucleic acid‐based therapeutics: a comprehensive review. Biotechnol. J. 16, 1900408 (2021).
Nakamura, T. et al. Innovative nanotechnologies for enhancing nucleic acids/gene therapy: controlling intracellular trafficking to targeted biodistribution. Biomaterials 218, 119329 (2019).
Yamada, Y. et al. Power of mitochondrial drug delivery systems to produce innovative nanomedicines. Adv. Drug Deliv. Rev. 154-155, 187–209 (2020).
Liew, S. S. et al. Smart design of nanomaterials for mitochondria‐targeted nanotherapeutics. Angew. Chem. Int. Ed. 60, 2232–2256 (2021).
Jhaveri, A. & Torchilin, V. Intracellular delivery of nanocarriers and targeting to subcellular organelles. Expert Opin. Drug Deliv. 13, 49–70 (2016).
Davis, M. E., Chen, Z. G. & Shin, D. M. Nanoparticle therapeutics: an emerging treatment modality for cancer. Nat. Rev. Drug Discov. 7, 771–782 (2008).
Petros, R. A. & DeSimone, J. M. Strategies in the design of nanoparticles for therapeutic applications. Nat. Rev. Drug Discov. 9, 615–627 (2010).
Cabral, H., Miyata, K., Osada, K. & Kataoka, K. Block copolymer micelles in nanomedicine applications. Chem. Rev. 118, 6844–6892 (2018).
Mitchell, M. J. et al. Engineering precision nanoparticles for drug delivery. Nat. Rev. Drug Discov. 20, 101–124 (2021).
Torchilin, V. P. Recent approaches to intracellular delivery of drugs and DNA and organelle targeting. Annu. Rev. Biomed. Eng. 8, 343–375 (2006).
Sakhrani, N. M. & Padh, H. Organelle targeting: third level of drug targeting. Drug Des. Dev. Ther. 7, 585–599 (2013).
Parodi, A. et al. Enabling cytoplasmic delivery and organelle targeting by surface modification of nanocarriers. Nanomedicine 10, 1923–1940 (2015).
Ma, X. et al. Future of nanotherapeutics: targeting the cellular sub-organelles. Biomaterials 97, 10–21 (2016).
Owens, D. E. 3rd & Peppas, N. A. Opsonization, biodistribution, and pharmacokinetics of polymeric nanoparticles. Int. J. Pharm. 307, 93–102 (2006).
White, P. J., Anastasopoulos, F., Pouton, C. W. & Boyd, B. J. Overcoming biological barriers to in vivo efficacy of antisense oligonucleotides. Expert Rev. Mol. Med. 11, e10 (2009).
Kalyanaraman, B. et al. A review of the basics of mitochondrial bioenergetics, metabolism, and related signaling pathways in cancer cells: therapeutic targeting of tumor mitochondria with lipophilic cationic compounds. Redox Biol. 14, 316–327 (2018).
Jeena, M. T., Kim, S., Jin, S. & Ryu, J. H. Recent progress in mitochondria-targeted drug and drug-free agents for cancer therapy. Cancers 12, (2019).
Slone, J. & Huang, T. The special considerations of gene therapy for mitochondrial diseases. NPJ Genom. Med. 5, 7 (2020).
Jackson, C. B., Turnbull, D. M., Minczuk, M. & Gammage, P. A. Therapeutic manipulation of mtDNA heteroplasmy: a shifting perspective. Trends Mol. Med. 26, 698–709 (2020).
Yasuzaki, Y., Yamada, Y., Kanefuji, T. & Harashima, H. Localization of exogenous DNA to mitochondria in skeletal muscle following hydrodynamic limb vein injection. J. Control Release 172, 805–811 (2013).
Bonnefoy, N. & Fox, T. D. Directed alteration of Saccharomyces cerevisiae mitochondrial DNA by biolistic transformation and homologous recombination. Methods Mol. Biol. 372, 153–166, (2007).
Jiang, Y. et al. Recent advances in nanotechnology approaches for non-viral gene therapy. Biomater. Sci. 10, 6862–6892 (2022).
Hamimed, S., Jabberi, M. & Chatti, A. Nanotechnology in drug and gene delivery. Naunyn-Schmiedeberg’s Arch. Pharmacol. 395, 769–787 (2022).
Mirón-Barroso, S., Domènech, E. B. & Trigueros, S. Nanotechnology-based strategies to overcome current barriers in gene delivery. Int. J. Mol. Sci. 22, 8537 (2021).
Kinnear, C. et al. Form follows function: nanoparticle shape and its implications for nanomedicine. Chem. Rev. 117, 11476–11521 (2017).
Kamenski, P. A., Krasheninnikov, I. A. & Tarassov, I. 40 years of studying RNA import into mitochondria: from basic mechanisms to gene therapy strategies. Mol. Biol. 53, 924–932 (2019).
Faria, R. et al. Physicochemical characterization and targeting performance of triphenylphosphonium nano-polyplexes. J. Mol. Liq. 316, 113873 (2020).
Weissig, V. & Torchilin, V. P. Towards mitochondrial gene therapy: DQAsomes as a strategy. J. Drug Target. 9, 1–13 (2001).
Weissig, V. & Torchilin, V. P. Cationic bolasomes with delocalized charge centers as mitochondria-specific DNA delivery systems. Adv. Drug Deliv. Rev. 49, 127–149 (2001).
Weissig, V. et al. DQAsomes: a novel potential drug and gene delivery system made from Dequalinium. Pharm. Res. 15, 334–337 (1998).
Lyrawati, D., Trounson, A. & Cram, D. Expression of GFP in the mitochondrial compartment using DQAsome-mediated delivery of an artificial mini-mitochondrial genome. Pharm. Res. 28, 2848–2862 (2011).
Bae, Y. et al. Functional nanosome for enhanced mitochondria-targeted gene delivery and expression. Mitochondrion 37, 27–40 (2017).
Yamada, Y. & Harashima, H. MITO-porter for mitochondrial delivery and mitochondrial functional analysis. Handb. Exp. Pharm. 240, 457–472 (2017).
Yasuzaki, Y., Yamada, Y. & Harashima, H. Mitochondrial matrix delivery using MITO-Porter, a liposome-based carrier that specifies fusion with mitochondrial membranes. Biochem. Biophys. Res. Commun. 397, 181–186 (2010).
Yamada, Y. et al. The use of a MITO-Porter to deliver exogenous therapeutic RNA to a mitochondrial disease’s cell with a A1555G mutation in the mitochondrial 12S rRNA gene results in an increase in mitochondrial respiratory activity. Mitochondrion 55, 134–144 (2020).
Khalil, I. A., Kogure, K., Futaki, S. & Harashima, H. High density of octaarginine stimulates macropinocytosis leading to efficient intracellular trafficking for gene expression. J. Biol. Chem. 281, 3544–3551 (2006).
Nikzamir, M., Hanifehpour, Y., Akbarzadeh, A. & Panahi, Y. Applications of dendrimers in nanomedicine and drug delivery: a review. J. Inorg. Organomet. Polym. Mater. 31, 1–16 (2021).
Boas, U. & Heegaard, P. M. Dendrimers in drug research. Chem. Soc. Rev. 33, 43–63 (2004).
Vögtle, F. et al. Functional dendrimers. Prog. Polym. Sci. 25, 987–1041 (2000).
Svenson, S. & Tomalia, D. A. Dendrimers in biomedical applications—reflections on the field. Adv. Drug Deliv. Rev. 64, 102–115 (2012).
Surekha, B. et al. PAMAM dendrimer as a talented multifunctional biomimetic nanocarrier for cancer diagnosis and therapy. Colloids Surf. B Biointerfaces 204, 111837 (2021).
Oddone, N. et al. In vitro and in vivo uptake studies of PAMAM G4.5 dendrimers in breast cancer. J. Nanobiotechnol. 14, 45 (2016).
Biswas, S., Dodwadkar, N. S., Piroyan, A. & Torchilin, V. P. Surface conjugation of triphenylphosphonium to target poly(amidoamine) dendrimers to mitochondria. Biomaterials 33, 4773–4782 (2012).
Wolfram, R. K., Heller, L. & Csuk, R. Targeting mitochondria: esters of rhodamine B with triterpenoids are mitocanic triggers of apoptosis. Eur. J. Med. Chem. 152, 21–30 (2018).
Santos, J., Sousa, F., Queiroz, J. & Costa, D. Rhodamine-based plasmid DNA nanoparticles for mitochondrial gene therapy. Colloids Surf. B Biointerfaces 121, 129–140 (2014).
Salvado, R., Sousa, F., Queiroz, J. & Costa, D. Development of mitochondrial targeting plasmid DNA nanoparticles: characterization and in vitro studies. Colloids Surf. A Physicochem. Eng. Asp. 480, 287–295 (2015).
Costa, D. et al. Targeting of cellular organelles by fluorescent plasmid DNA nanoparticles. Biomacromolecules 18, 2928–2936 (2017).
Chacinska, A. et al. Importing mitochondrial proteins: machineries and mechanisms. Cell 138, 628–644 (2009).
Abe, Y. et al. Structural basis of presequence recognition by the mitochondrial protein import receptor Tom20. Cell 100, 551–560 (2000).
van der Laan, M. et al. Motor-free mitochondrial presequence translocase drives membrane integration of preproteins. Nat. Cell Biol. 9, 1152–1159 (2007).
Shiota, T. et al. Molecular architecture of the active mitochondrial protein gate. Science 349, 1544–1548 (2015).
Garg, S. et al. Conservation of transit peptide-independent protein import into the mitochondrial and hydrogenosomal matrix. Genome Biol. Evol. 7, 2716–2726 (2015).
Gyimesi, G. & Hediger, M. A. Sequence features of mitochondrial transporter protein families. Biomolecules. 10 (2020).
Khan, S. M. & Bennett, J. P. Jr. Development of mitochondrial gene replacement therapy. J. Bioenerg. Biomembr. 36, 387–393 (2004).
Farge, G. et al. Protein sliding and DNA denaturation are essential for DNA organization by human mitochondrial transcription factor A. Nat. Commun. 3, 1013 (2012).
Keeney, P. M. et al. Mitochondrial gene therapy augments mitochondrial physiology in a Parkinson’s disease cell model. Hum. Gene Ther. 20, 897–907 (2009).
Iyer, S. et al. Recombinant mitochondrial transcription factor A with N-terminal mitochondrial transduction domain increases respiration and mitochondrial gene expression. Mitochondrion 9, 196–203 (2009).
Iyer, S. et al. Mitochondrial gene therapy improves respiration, biogenesis, and transcription in G11778A Leber’s hereditary optic neuropathy and T8993G Leigh’s syndrome cells. Hum. Gene Ther. 23, 647–657 (2012).
Kang, D., Kim, S. H. & Hamasaki, N. Mitochondrial transcription factor A (TFAM): roles in maintenance of mtDNA and cellular functions. Mitochondrion 7, 39–44 (2007).
Iyer, S. et al. Mitochondrial gene replacement in human pluripotent stem cell-derived neural progenitors. Gene Ther. 19, 469–475 (2012).
Shih, W. M., Quispe, J. D. & Joyce, G. F. A 1.7-kilobase single-stranded DNA that folds into a nanoscale octahedron. Nature 427, 618–621 (2004).
Rothemund, P. W. Folding DNA to create nanoscale shapes and patterns. Nature 440, 297–302 (2006).
Goodman, R. P. et al. Rapid chiral assembly of rigid DNA building blocks for molecular nanofabrication. Science 310, 1661–1665 (2005).
Choi, C. H. et al. Mechanism for the endocytosis of spherical nucleic acid nanoparticle conjugates. Proc. Natl. Acad. Sci. USA 110, 7625–7630 (2013).
Liang, L. et al. Single-particle tracking and modulation of cell entry pathways of a tetrahedral DNA nanostructure in live cells. Angew. Chem. Int. Ed. Engl. 53, 7745–7750 (2014).
Chan, M. S. & Lo, P. K. Nanoneedle-assisted delivery of site-selective peptide-functionalized DNA nanocages for targeting mitochondria and nuclei. Small 10, 1255–1260 (2014).
Qin, X. et al. Tetrahedral framework nucleic acids-based delivery of microRNA-155 inhibits choroidal neovascularization by regulating the polarization of macrophages. Bioact. Mater. 14, 134–144 (2022).
Li, S. et al. Bioswitchable delivery of microRNA by framework nucleic acids: application to bone regeneration. Small 17, e2104359 (2021).
Chai, X. et al. A redox-activatable DNA nanodevice for spatially-selective, AND-gated imaging of ATP and glutathione in mitochondria. Nano Lett. 21, 10047–10053 (2021).
Luo, L. et al. Ratiometric fluorescent DNA nanostructure for mitochondrial ATP imaging in living cells based on hybridization chain reaction. Anal. Chem. 93, 6715–6722 (2021).
Liu, J. et al. Reductase and light programmatical gated DNA nanodevice for spatiotemporally controlled imaging of biomolecules in subcellular organelles under hypoxic conditions. ACS Appl. Mater. Interfaces 13, 33894–33904 (2021).
Wold, W. S. & Toth, K. Adenovirus vectors for gene therapy, vaccination and cancer gene therapy. Curr. Gene Ther. 13, 421–433 (2013).
Naso, M. F., Tomkowicz, B., Perry, W. L. 3rd & Strohl, W. R. Adeno-associated virus (AAV) as a vector for gene therapy. Biodrugs 31, 317–334 (2017).
Büning, H. & Srivastava, A. Capsid modifications for targeting and improving the efficacy of AAV vectors. Mol. Ther. Methods Clin. Dev. 12, 248–265 (2019).
Penaud-Budloo, M. et al. Adeno-associated virus vector genomes persist as episomal chromatin in primate muscle. J. Virol. 82, 7875–7885 (2008).
Bouchard, S. et al. Long-term transgene expression in cardiac and skeletal muscle following fetal administration of adenoviral or adeno-associated viral vectors in mice. J. Gene Med. 5, 941–950 (2003).
Newman, N. J. et al. Randomized trial of bilateral gene therapy injection for m.11778G > A MT-ND4 Leber optic neuropathy. Brain 146, 1328–1341 (2023).
Koilkonda, R. D. et al. Safety and effects of the vector for the Leber hereditary optic neuropathy gene therapy clinical trial. JAMA Ophthalmol. 132, 409–420 (2014).
Vignal-Clermont, C. et al. Safety of lenadogene nolparvovec gene therapy over 5 years in 189 patients with leber hereditary optic neuropathy. Am. J. Ophthalmol. 249, 108–125 (2023).
Hocquemiller, M. et al. Adeno-associated virus-based gene therapy for CNS diseases. Hum. Gene Ther. 27, 478–496 (2016).
Benkhelifa-Ziyyat, S. et al. Intramuscular scAAV9-SMN injection mediates widespread gene delivery to the spinal cord and decreases disease severity in SMA mice. Mol. Ther. 21, 282–290 (2013).
Wang, D. et al. A rationally engineered capsid variant of AAV9 for systemic CNS-directed and peripheral tissue-detargeted gene delivery in neonates. Mol. Ther. Methods Clin. Dev. 9, 234–246 (2018).
Cabrera-Pérez, R. et al. Alpha-1-antitrypsin promoter improves the efficacy of an adeno-associated virus vector for the treatment of mitochondrial neurogastrointestinal encephalomyopathy. Hum. Gene Ther. 30, 985–998 (2019).
Hioki, H. et al. Efficient gene transduction of neurons by lentivirus with enhanced neuron-specific promoters. Gene Ther. 14, 872–882 (2007).
Xu, R. et al. Quantitative comparison of expression with adeno-associated virus (AAV-2) brain-specific gene cassettes. Gene Ther. 8, 1323–1332 (2001).
Talbot, G. E. et al. Desmin-regulated lentiviral vectors for skeletal muscle gene transfer. Mol. Ther. 18, 601–608 (2010).
Wang, D., Tai, P. W. L. & Gao, G. Adeno-associated virus vector as a platform for gene therapy delivery. Nat. Rev. Drug Discov. 18, 358–378 (2019).
Yu, H. et al. Gene delivery to mitochondria by targeting modified adenoassociated virus suppresses Leber’s hereditary optic neuropathy in a mouse model. Proc. Natl Acad. Sci. USA 109, E1238–1247 (2012).
Lyu, P. et al. Adenine base editor ribonucleoproteins delivered by lentivirus-like particles show high on-target base editing and undetectable RNA off-target activities. CRISPR J. 4, 69–81 (2021).
Yu-Wai-Man, P. et al. Bilateral visual improvement with unilateral gene therapy injection for Leber hereditary optic neuropathy. Sci. Transl. Med. 12 (2020).
Xie, Q. et al. Mitochondrial transplantation attenuates cerebral ischemia-reperfusion injury: possible involvement of mitochondrial component separation. Oxid. Med. Cell Longev. 2021, 1006636 (2021).
Lin, H.-Y. et al. Mitochondrial transfer from Wharton’s jelly-derived mesenchymal stem cells to mitochondria-defective cells recaptures impaired mitochondrial function. Mitochondrion 22, 31–44 (2015).
Liu, D. et al. Intercellular mitochondrial transfer as a means of tissue revitalization. Signal Transduct. Target. Ther. 6, 65 (2021).
Borcherding, N. & Brestoff, J. R. The power and potential of mitochondria transfer. Nature 623, 283–291 (2023).
Gurke, S. et al. Tunneling nanotube (TNT)-like structures facilitate a constitutive, actomyosin-dependent exchange of endocytic organelles between normal rat kidney cells. Exp. Cell Res. 314, 3669–3683 (2008).
He, K. et al. Long-distance intercellular connectivity between cardiomyocytes and cardiofibroblasts mediated by membrane nanotubes. Cardiovasc. Res. 92, 39–47 (2011).
Koyanagi, M. et al. Cell-to-cell connection of endothelial progenitor cells with cardiac myocytes by nanotubes: a novel mechanism for cell fate changes? Circ. Res. 96, 1039–1041 (2005).
Liao, P. et al. Osteocyte mitochondria regulate angiogenesis of transcortical vessels. Nat. Commun. 15, 2529 (2024).
Yip, H. K. et al. Melatonin rescues cerebral ischemic events through upregulated tunneling nanotube-mediated mitochondrial transfer and downregulated mitochondrial oxidative stress in rat brain. Biomed. Pharmacother. 139, 111593 (2021).
Davis, C. H. et al. Transcellular degradation of axonal mitochondria. Proc. Natl. Acad. Sci. USA 111, 9633–9638 (2014).
English, K. et al. Astrocytes rescue neuronal health after cisplatin treatment through mitochondrial transfer. Acta Neuropathol. Commun. 8, 36 (2020).
Hayakawa, K. et al. Transfer of mitochondria from astrocytes to neurons after stroke. Nature 535, 551–555 (2016).
van der Vlist, M. et al. Macrophages transfer mitochondria to sensory neurons to resolve inflammatory pain. Neuron 110, 613–626.e619 (2022).
Jung, J. E. et al. The mitochondria-derived peptide human in improves recovery from intracerebral hemorrhage: implication of mitochondria transfer and microglia phenotype change. J. Neurosci. 40, 2154–2165 (2020).
Joshi, A. U. et al. Fragmented mitochondria released from microglia trigger A1 astrocytic response and propagate inflammatory neurodegeneration. Nat. Neurosci. 22, 1635–1648 (2019).
Liu, K. et al. Mesenchymal stem cells rescue injured endothelial cells in an in vitro ischemia-reperfusion model via tunneling nanotube like structure-mediated mitochondrial transfer. Microvasc. Res. 92, 10–18 (2014).
Sinclair, K. A., Yerkovich, S. T., Hopkins, P. M. & Chambers, D. C. Characterization of intercellular communication and mitochondrial donation by mesenchymal stromal cells derived from the human lung. Stem Cell Res. Ther. 7, 91 (2016).
Acquistapace, A. et al. Human mesenchymal stem cells reprogram adult cardiomyocytes toward a progenitor-like state through partial cell fusion and mitochondria transfer. Stem Cells 29, 812–824 (2011).
Vallabhaneni, K. C., Haller, H. & Dumler, I. Vascular smooth muscle cells initiate proliferation of mesenchymal stem cells by mitochondrial transfer via tunneling nanotubes. Stem Cells Dev. 21, 3104–3113 (2012).
Plotnikov, E. Y. et al. Cytoplasm and organelle transfer between mesenchymal multipotent stromal cells and renal tubular cells in co-culture. Exp. Cell Res. 316, 2447–2455 (2010).
Zampieri, L. X., Silva-Almeida, C., Rondeau, J. D. & Sonveaux, P. Mitochondrial transfer in cancer: a comprehensive review. Int. J. Mol. Sci. 22 (2021).
Cho, Y. M. et al. Mesenchymal stem cells transfer mitochondria to the cells with virtually no mitochondrial function but not with pathogenic mtDNA mutations. PLoS One 7, e32778 (2012).
Tan, A. S. et al. Mitochondrial genome acquisition restores respiratory function and tumorigenic potential of cancer cells without mitochondrial DNA. Cell Metab. 21, 81–94 (2015).
Arneth, B. Tumor microenvironment. Medicina 56, 15 (2019).
Antanavičiūtė, I. et al. Long-distance communication between laryngeal carcinoma cells. PLoS One 9, e99196 (2014).
Lou, E. et al. Tunneling nanotubes provide a unique conduit for intercellular transfer of cellular contents in human malignant pleural mesothelioma. PLoS One 7, e33093 (2012).
Pinto, G. et al. Patient-derived glioblastoma stem cells transfer mitochondria through tunneling nanotubes in tumor organoids. Biochem. J. 478, 21–39 (2021).
Ippolito, L. et al. Cancer-associated fibroblasts promote prostate cancer malignancy via metabolic rewiring and mitochondrial transfer. Oncogene 38, 5339–5355 (2019).
Lu, J. et al. Tunneling nanotubes promote intercellular mitochondria transfer followed by increased invasiveness in bladder cancer cells. Oncotarget 8, 15539–15552 (2017).
Moschoi, R. et al. Protective mitochondrial transfer from bone marrow stromal cells to acute myeloid leukemic cells during chemotherapy. Blood 128, 253–264 (2016).
Burt, R. et al. Activated stromal cells transfer mitochondria to rescue acute lymphoblastic leukemia cells from oxidative stress. Blood 134, 1415–1429 (2019).
Salaud, C. et al. Mitochondria transfer from tumor-activated stromal cells (TASC) to primary Glioblastoma cells. Biochem. Biophys. Res. Commun. 533, 139–147 (2020).
Pasquier, J. et al. Preferential transfer of mitochondria from endothelial to cancer cells through tunneling nanotubes modulates chemoresistance. J. Transl. Med. 11, 94 (2013).
Wang, X. & Gerdes, H. H. Transfer of mitochondria via tunneling nanotubes rescues apoptotic PC12 cells. Cell Death Differ. 22, 1181–1191 (2015).
Wang, J. et al. Cell adhesion-mediated mitochondria transfer contributes to mesenchymal stem cell-induced chemoresistance on T cell acute lymphoblastic leukemia cells. J. Hematol. Oncol. 11, 11 (2018).
Elliott, R. L., Jiang, X. P. & Head, J. F. Mitochondria organelle transplantation: introduction of normal epithelial mitochondria into human cancer cells inhibits proliferation and increases drug sensitivity. Breast Cancer Res. Treat. 136, 347–354 (2012).
Skildum, A., Dornfeld, K. & Wallace, K. Mitochondrial amplification selectively increases doxorubicin sensitivity in breast cancer cells with acquired antiestrogen resistance. Breast Cancer Res. Treat. 129, 785–797 (2011).
Suganuma, K. et al. Energy metabolism of leukemia cells: glycolysis versus oxidative phosphorylation. Leuk. Lymphoma 51, 2112–2119 (2010).
Marlein, C. R. et al. NADPH oxidase-2 derived superoxide drives mitochondrial transfer from bone marrow stromal cells to leukemic blasts. Blood 130, 1649–1660 (2017).
Marlein, C. R. et al. CD38-driven mitochondrial trafficking promotes bioenergetic plasticity in multiple myeloma. Cancer Res. 79, 2285–2297 (2019).
Hall, A. M. et al. In vivo multiphoton imaging of mitochondrial structure and function during acute kidney injury. Kidney Int. 83, 72–83 (2013).
Islam, M. N. et al. Mitochondrial transfer from bone-marrow-derived stromal cells to pulmonary alveoli protects against acute lung injury. Nat. Med. 18, 759–765 (2012).
Wang, W. et al. Mitochondria dysfunction in the pathogenesis of Alzheimer’s disease: recent advances. Mol. Neurodegener. 15, 30 (2020).
Marti Gutierrez, N. et al. Horizontal mtDNA transfer between cells is common during mouse development. iScience 25, 103901 (2022).
Zhang, H. et al. Systematic investigation of mitochondrial transfer between cancer cells and T cells at single-cell resolution. Cancer Cell 41, 1788–1802.e1710 (2023).
Mulcahy, L. A., Pink, R. C. & Carter, D. R. Routes and mechanisms of extracellular vesicle uptake. J. Extracell Vesicles 3 (2014).
Smeets, H. J. Preventing the transmission of mitochondrial DNA disorders: selecting the good guys or kicking out the bad guys. Reprod. Biomed. Online 27, 599–610 (2013).
Bae, S. K. et al. A ratiometric two-photon fluorescent probe reveals reduction in mitochondrial H2S production in Parkinson’s disease gene knockout astrocytes. J. Am. Chem. Soc. 135, 9915–9923 (2013).
Li, H. et al. Mitochondrial transfer from bone marrow mesenchymal stem cells to motor neurons in spinal cord injury rats via gap junction. Theranostics 9, 2017–2035 (2019).
Wan, M. et al. Oxidized but not native cardiolipin has pro-inflammatory effects, which are inhibited by Annexin A5. Atherosclerosis 235, 592–598 (2014).
King, M. P. & Attardi, G. Injection of mitochondria into human cells leads to a rapid replacement of the endogenous mitochondrial DNA. Cell 52, 811–819 (1988).
Pinkert, C. A., Irwin, M. H., Johnson, L. W. & Moffatt, R. J. Mitochondria transfer into mouse ova by microinjection. Transgenic Res. 6, 379–383 (1997).
Macheiner, T. et al. Magnetomitotransfer: An efficient way for direct mitochondria transfer into cultured human cells. Sci. Rep. 6, 35571 (2016).
Nzigou Mombo, B. et al. MitoCeption: transferring isolated human MSC mitochondria to glioblastoma stem cells. J. Vis. Exp. (2017).
Caicedo, A. et al. MitoCeption as a new tool to assess the effects of mesenchymal stem/stromal cell mitochondria on cancer cell metabolism and function. Sci. Rep. 5, 9073 (2015).
Wu, T. H. et al. Mitochondrial transfer by photothermal nanoblade restores metabolite profile in mammalian cells. Cell Metab. 23, 921–929 (2016).
Sercel, A. J. et al. Stable transplantation of human mitochondrial DNA by high-throughput, pressurized isolated mitochondrial delivery. Elife 10 (2021).
Sercel, A. J. et al. Generating stable isolated mitochondrial recipient clones in mammalian cells using MitoPunch mitochondrial transfer. STAR Protoc. 2, 100850 (2021).
Shakoor, A. et al. Automated optical tweezers manipulation to transfer mitochondria from fetal to adult MSCs to improve antiaging gene expressions. Small 17, e2103086 (2021).
Sun, J. et al. High-efficiency quantitative control of mitochondrial transfer based on droplet microfluidics and its application on muscle regeneration. Sci. Adv. 8, eabp9245 (2022).
Chang, J. C. et al. Peptide-mediated delivery of donor mitochondria improves mitochondrial function and cell viability in human cybrid cells with the MELAS A3243G mutation. Sci. Rep. 7, 10710 (2017).
Wu, S. et al. Polymer functionalization of isolated mitochondria for cellular transplantation and metabolic phenotype alteration. Adv. Sci. 5, 1700530 (2018).
Nakano, T. et al. Mitochondrial surface coating with artificial lipid membrane improves the transfer efficacy. Commun. Biol. 5, 745 (2022).
Patananan, A. N. et al. Pressure-driven mitochondrial transfer pipeline generates mammalian cells of desired genetic combinations and fates. Cell Rep. 33, 108562 (2020).
Gäbelein, C. G. et al. Mitochondria transplantation between living cells. PLoS Biol. 20, e3001576 (2022).
Meister, A. et al. FluidFM: combining atomic force microscopy and nanofluidics in a universal liquid delivery system for single cell applications and beyond. Nano Lett. 9, 2501–2507 (2009).
Wu, Y. C. et al. Massively parallel delivery of large cargo into mammalian cells with light pulses. Nat. Methods 12, 439–444 (2015).
Ashkin, A., Dziedzic, J. M. & Yamane, T. Optical trapping and manipulation of single cells using infrared laser beams. Nature 330, 769–771 (1987).
Al Amir Dache, Z. et al. Blood contains circulating cell-free respiratory competent mitochondria. FASEB J. 34, 3616–3630 (2020).
Silva, A. K., Letourneur, D. & Chauvierre, C. Polysaccharide nanosystems for future progress in cardiovascular pathologies. Theranostics 4, 579–591 (2014).
Dave, K. M. et al. Mitochondria-containing extracellular vesicles (EV) reduce mouse brain infarct sizes and EV/HSP27 protect ischemic brain endothelial cultures. J. Control Release 354, 368–393 (2023).
Morrison, T. J. et al. Mesenchymal stromal cells modulate macrophages in clinically relevant lung injury models by extracellular vesicle mitochondrial transfer. Am. J. Respir. Crit. Care Med. 196, 1275–1286 (2017).
Crewe, C. et al. Extracellular vesicle-based interorgan transport of mitochondria from energetically stressed adipocytes. Cell Metab. 33, 1853–1868.e1811 (2021).
Rosina, M. et al. Ejection of damaged mitochondria and their removal by macrophages ensure efficient thermogenesis in brown adipose tissue. Cell Metab. 34, 533–548.e512 (2022).
Heit, B., Yeung, T. & Grinstein, S. Changes in mitochondrial surface charge mediate recruitment of signaling molecules during apoptosis. Am. J. Physiol. Cell Physiol. 300, C33–C41 (2011).
Kates, M., Syz, J. Y., Gosser, D. & Haines, T. H. pH-dissociation characteristics of cardiolipin and its 2’-deoxy analogue. Lipids 28, 877–882 (1993).
Yang, W., Abe, S. & Tabata, Y. Association with cationized gelatin nanospheres enhances cell internalization of mitochondria efficiency. Regen. Ther. 24, 190–200 (2023).
Baudo, G. et al. Polymer-functionalized mitochondrial transplantation to fibroblasts counteracts a pro-fibrotic phenotype. Int. J. Mol. Sci. 24, (2023).
Hammond, P. T. Building biomedical materials layer-by-layer. Mater. Today 5, 196–206 (2012).
Chen, W. et al. Mitochondrial surface engineering for multidrug resistance reversal. Nano Lett. 19, 2905–2913 (2019).
Maeda, H. et al. TAT-dextran–mediated mitochondrial transfer enhances recovery from models of reperfusion injury in cultured cardiomyocytes. J. Cell Mol. Med. 24, 5007–5020 (2020).
Chang, J. C. et al. Mitochondrial transplantation regulates antitumour activity, chemoresistance and mitochondrial dynamics in breast cancer. J. Exp. Clin. Cancer Res 38, 30 (2019).
Chang, J. C. et al. Allogeneic/xenogeneic transplantation of peptide-labeled mitochondria in Parkinson’s disease: restoration of mitochondria functions and attenuation of 6-hydroxydopamine-induced neurotoxicity. Transl. Res. 170, 40–56.e43 (2016).
Chen, W. et al. Chemotaxis-based self-accumulation of surface-engineered mitochondria for cancer therapeutic improvement. Nano Today 35, 100966 (2020).
Liu, J. et al. Engineering living mitochondria with AIE photosensitizer for synergistic cancer cell ablation. Nano Lett. 20, 7438–7445 (2020).
Berry, B. J. et al. Optogenetic rejuvenation of mitochondrial membrane potential extends C. elegans lifespan. Nat. Aging 3, 157–161 (2023).
Xu, C., Martin, N., Li, M. & Mann, S. Living material assembly of bacteriogenic protocells. Nature 609, 1029–1037 (2022).
Park, H. et al. Artificial organelles for sustainable chemical energy conversion and production in artificial cells: artificial mitochondrion and chloroplasts. Biophys. Rev. 4, (2023).
Discher, D. E. & Eisenberg, A. Polymer vesicles. Science 297, 967–973 (2002).
Carlsen, A., Glaser, N., Le Meins, J.-F. O. & Lecommandoux, S. Block copolymer vesicle permeability measured by osmotic swelling and shrinking. Langmuir 27, 4884–4890 (2011).
Lin, Z. et al. Cryo transmission electron microscopy study of vesicles and micelles in siloxane surfactant aqueous solutions. Langmuir 10, 1008–1011 (1994).
Marušič, N. et al. Constructing artificial respiratory chain in polymer compartments: Insights into the interplay between bo3 oxidase and the membrane. Proc. Natl. Acad. Sci. USA 117, 15006–15017 (2020).
Otrin, L. et al. En route to dynamic life processes by SNARE-mediated fusion of polymer and hybrid membranes. Nat. Commun. 12, 4972 (2021).
Nordlund, G., Brzezinski, P. & von Ballmoos, C. SNARE-fusion mediated insertion of membrane proteins into native and artificial membranes. Nat. Commun. 5, 4303 (2014).
Biner, O., Schick, T., Müller, Y. & von Ballmoos, C. Delivery of membrane proteins into small and giant unilamellar vesicles by charge-mediated fusion. FEBS Lett. 590, 2051–2062 (2016).
Weber, T. et al. SNAREpins: minimal machinery for membrane fusion. Cell 92, 759–772 (1998).
Jahn, R. & Scheller, R. H. SNAREs–engines for membrane fusion. Nat. Rev. Mol. Cell Biol. 7, 631–643 (2006).
Stamatatos, L., Leventis, R., Zuckermann, M. J. & Silvius, J. R. Interactions of cationic lipid vesicles with negatively charged phospholipid vesicles and biological membranes. Biochemistry 27, 3917–3925 (1988).
Biner, O., Schick, T., Ganguin, A. A. & von Ballmoos, C. Towards a synthetic mitochondrion. Chim. 72, 291–296 (2018).
Krenn, B. E. et al. Co-reconstitution of the H+-ATP synthase and cytochrome b-563c-554 complex from a thermophilic cyanobacterium: High ATP yield and mutual effects on the enzymatic activities. Biochim Biophys. Acta Bioenerg. 1140, 271–281 (1993).
Gemperli, A. C., Dimroth, P. & Steuber, J. Sodium ion cycling mediates energy coupling between complex I and ATP synthase. Proc. Natl. Acad. Sci. USA 100, 839–844 (2003).
Otrin, L. et al. Toward artificial mitochondrion: mimicking oxidative phosphorylation in polymer and hybrid membranes. Nano Lett. 17, 6816–6821 (2017).
von Ballmoos, C., Biner, O., Nilsson, T. & Brzezinski, P. Mimicking respiratory phosphorylation using purified enzymes. Biochim. Biophys. Acta Bioenerg. 1857, 321–331 (2016).
Nilsson, T. et al. Lipid-mediated protein-protein interactions modulate respiration-driven ATP synthesis. Sci. Rep. 6, 24113 (2016).
Li, J.-H. et al. Cyclodextrin-based microcapsules as bioreactors for ATP biosynthesis. Biomacromolecules 14, 2984–2988 (2013).
Duan, L. et al. Proton gradients produced by glucose oxidase microcapsules containing motor F0F1-ATPase for continuous ATP biosynthesis. J. Phys. Chem. B 113, 395–399 (2009).
Qi, W. et al. Motor protein CF0F1 reconstituted in lipid-coated hemoglobin microcapsules for ATP synthesis. Adv. Mater. 20, 601–605 (2008).
Di Meo, I., Lamperti, C. & Tiranti, V. Mitochondrial diseases caused by toxic compound accumulation: from etiopathology to therapeutic approaches. EMBO Mol. Med 7, 1257–1266 (2015).
Di Meo, I. et al. Chronic exposure to sulfide causes accelerated degradation of cytochrome c oxidase in ethylmalonic encephalopathy. Antioxid. Redox Signal 15, 353–362 (2011).
Tiranti, V. et al. Loss of ETHE1, a mitochondrial dioxygenase, causes fatal sulfide toxicity in ethylmalonic encephalopathy. Nat. Med. 15, 200–205 (2009).
Yang, G., Sun, X. & Wang, R. Hydrogen sulfide-induced apoptosis of human aorta smooth muscle cells via the activation of mitogen-activated protein kinases and caspase-3. FASEB J. 18, 1782–1784 (2004).
Di Meo, I. et al. Effective AAV-mediated gene therapy in a mouse model of ethylmalonic encephalopathy. EMBO Mol. Med. 4, 1008–1014 (2012).
Lake, N. J., Bird, M. J., Isohanni, P. & Paetau, A. Leigh syndrome: neuropathology and pathogenesis. J. Neuropathol. Exp. Neurol. 74, 482–492 (2015).
Reynaud-Dulaurier, R. et al. Gene replacement therapy provides benefit in an adult mouse model of Leigh syndrome. Brain 143, 1686–1696 (2020).
Parés, M. et al. Preclinical assessment of a gene-editing approach in a mouse model of mitochondrial neurogastrointestinal encephalomyopathy. Hum. Gene Ther. 32, 1210–1223 (2021).
Bottani, E. et al. AAV-mediated liver-specific MPV17 expression restores mtDNA levels and prevents diet-induced liver failure. Mol. Ther. 22, 10–17 (2014).
Kaukonen, J. et al. Role of adenine nucleotide translocator 1 in mtDNA maintenance. Science 289, 782–785 (2000).
Flierl, A. et al. Adeno-associated virus-mediated gene transfer of the heart/muscle adenine nucleotide translocator (ANT) in mouse. Gene Ther. 12, 570–578 (2005).
Wang, S. et al. AAV gene therapy prevents and reverses heart failure in a murine knockout model of Barth syndrome. Circ. Res. 126, 1024–1039 (2020).
Cook, A. & Giunti, P. Friedreich’s ataxia: clinical features, pathogenesis and management. Br. Med. Bull. 124, 19–30 (2017).
Perdomini, M. et al. Prevention and reversal of severe mitochondrial cardiomyopathy by gene therapy in a mouse model of Friedreich’s ataxia. Nat. Med. 20, 542–547 (2014).
Peverelli, L. et al. Leber’s hereditary optic neuropathy: a report on novel mtDNA pathogenic variants. Front. Neurol. 12, 657317 (2021).
Newman, N. J. et al. Efficacy and safety of intravitreal gene therapy for Leber hereditary optic neuropathy treated within 6 months of disease onset. Ophthalmology 128, 649–660 (2021).
Newman, N. J. et al. Intravitreal gene therapy vs. natural history in patients with leber hereditary optic neuropathy carrying the m.11778G > A ND4 mutation: systematic review and indirect comparison. Front. Neurol. 12, 662838 (2021).
Bonnet, C. et al. The optimized allotopic expression of ND1 or ND4 genes restores respiratory chain complex I activity in fibroblasts harboring mutations in these genes. Biochim. Biophys. Acta 1783, 1707–1717 (2008).
Koilkonda, R. et al. LHON gene therapy vector prevents visual loss and optic neuropathy induced by G11778A mutant mitochondrial DNA: biodistribution and toxicology profile. Investig. Ophthalmol. Vis. Sci. 55, 7739–7753 (2014).
Calkins, D. J. et al. Biodistribution of intravitreal (lenadogene) nolparvovec gene therapy in nonhuman primates. Mol. Ther. Methods Clin. Dev. 23, 307–318 (2021).
Sabharwal, A. et al. The FusX TALE Base Editor (FusXTBE) for rapid mitochondrial DNA programming of human cells in vitro and zebrafish disease models in vivo. CRISPR J. 4, 799–821 (2021).
Lee, S. et al. Enhanced mitochondrial DNA editing in mice using nuclear-exported TALE-linked deaminases and nucleases. Genome Biol. 23, 211 (2022).
Kim, M. J., Lee, J. M., Min, K. & Choi, Y. S. Xenogeneic transplantation of mitochondria induces muscle regeneration in an in vivo rat model of dexamethasone-induced atrophy. J. Muscle Res. Cell Motil. 45, 53–68 (2024).
Zeng, J. et al. Mitochondrial transplantation reduces lower limb ischemia-reperfusion injury by increasing skeletal muscle energy and adipocyte browning. Mol. Ther. Methods Clin. Dev. 31, 101152 (2023).
Liang, X. et al. Direct administration of mesenchymal stem cell-derived mitochondria improves cardiac function after infarction via ameliorating endothelial senescence. Bioeng. Transl. Med. 8, e10365 (2023).
Masuzawa, A. et al. Transplantation of autologously derived mitochondria protects the heart from ischemia-reperfusion injury. Am. J. Physiol. Heart Circ. Physiol. 304, H966–982 (2013).
Huang, C. C. et al. Mitochondrial transplantation attenuates traumatic neuropathic pain, neuroinflammation, and apoptosis in rats with nerve root ligation. Mol. Pain 19, 17448069231210423 (2023).
Blitzer, D. et al. Delayed transplantation of autologous mitochondria for cardioprotection in a porcine model. Ann. Thorac. Surg. 109, 711–719 (2020).
Jiang, D. et al. Donation of mitochondria by iPSC-derived mesenchymal stem cells protects retinal ganglion cells against mitochondrial complex I defect-induced degeneration. Theranostics 9, 2395–2410 (2019).
Konari, N., Nagaishi, K., Kikuchi, S. & Fujimiya, M. Mitochondria transfer from mesenchymal stem cells structurally and functionally repairs renal proximal tubular epithelial cells in diabetic nephropathy in vivo. Sci. Rep. 9, 5184 (2019).
Zhang, Z. et al. Muscle-derived autologous mitochondrial transplantation: a novel strategy for treating cerebral ischemic injury. Behav. Brain Res. 356, 322–331 (2019).
Pourmohammadi-Bejarpasi, Z. et al. Mesenchymal stem cells-derived mitochondria transplantation mitigates I/R-induced injury, abolishes I/R-induced apoptosis, and restores motor function in acute ischemia stroke rat model. Brain Res Bull. 165, 70–80 (2020).
Huang, P. J. et al. Transferring xenogenic mitochondria provides neural protection against ischemic stress in ischemic rat brains. Cell Transpl. 25, 913–927 (2016).
Cloer, C. M. et al. Mitochondrial transplant after ischemia reperfusion promotes cellular salvage and improves lung function during ex-vivo lung perfusion. J. Heart Lung Transpl. 42, 575–584 (2023).
Doulamis, I. P. et al. Mitochondrial transplantation by intra-arterial injection for acute kidney injury. Am. J. Physiol. Ren. Physiol. 319, F403–f413 (2020).
Alway, S. E. et al. Mitochondria transplant therapy improves regeneration and restoration of injured skeletal muscle. J. Cachexia Sarcopenia Muscle 14, 493–507 (2023).
Xu, J. et al. Targeted transplantation of engineered mitochondrial compound promotes functional recovery after spinal cord injury by enhancing macrophage phagocytosis. Bioact. Mater. 32, 427–444 (2024).
Nitzan, K. et al. Mitochondrial transfer ameliorates cognitive deficits, neuronal loss, and gliosis in Alzheimer’s disease mice. J. Alzheimers Dis. 72, 587–604 (2019).
Nakamura, Y., Lo, E. H. & Hayakawa, K. Placental mitochondria therapy for cerebral ischemia-reperfusion injury in mice. Stroke 51, 3142–3146 (2020).
Shi, X., Zhao, M., Fu, C. & Fu, A. Intravenous administration of mitochondria for treating experimental Parkinson’s disease. Mitochondrion 34, 91–100 (2017).
Guo, Y. et al. Mitochondria transfer enhances proliferation, migration, and osteogenic differentiation of bone marrow mesenchymal stem cell and promotes bone defect healing. Stem Cell Res. Ther. 11, 245 (2020).
Court, A. C. et al. Mitochondrial transfer from MSCs to T cells induces Treg differentiation and restricts inflammatory response. EMBO Rep. 21, e48052 (2020).
Ikeda, G. et al. Mitochondria-rich extracellular vesicles from autologous stem cell-derived cardiomyocytes restore energetics of ischemic myocardium. J. Am. Coll. Cardiol. 77, 1073–1088 (2021).
McCully, J. D. et al. Injection of isolated mitochondria during early reperfusion for cardioprotection. Am. J. Physiol. Heart Circ. Physiol. 296, H94–h105 (2009).
Kaza, A. K. et al. Myocardial rescue with autologous mitochondrial transplantation in a porcine model of ischemia/reperfusion. J. Thorac. Cardiovasc. Surg. 153, 934–943 (2017).
Babenko, V. A. et al. Improving the post-stroke therapeutic potency of mesenchymal multipotent stromal cells by cocultivation with cortical neurons: the role of crosstalk between cells. Stem Cells Transl. Med. 4, 1011–1020 (2015).
Abdullahi, W., Tripathi, D. & Ronaldson, P. T. Blood-brain barrier dysfunction in ischemic stroke: targeting tight junctions and transporters for vascular protection. Am. J. Physiol. Cell Physiol. 315, C343–c356 (2018).
Zhao, J. et al. Mitochondria transplantation protects traumatic brain injury via promoting neuronal survival and astrocytic BDNF. Transl. Res. 235, 102–114 (2021).
Johnatas Dutra, S. et al. Mesenchymal stromal cell extracellular vesicles rescue mitochondrial dysfunction and improve barrier integrity in clinically relevant models of ARDS. Eur. Respir. J. 58, 2002978 (2021).
Ahmad, T. et al. Miro1 regulates intercellular mitochondrial transport & enhances mesenchymal stem cell rescue efficacy. EMBO J. 33, 994–1010 (2014).
Chang, C. H. et al. Posttranscriptional control of T cell effector function by aerobic glycolysis. Cell 153, 1239–1251 (2013).
Yuan, Y. et al. Mitochondrial transfer from mesenchymal stem cells to macrophages restricts inflammation and alleviates kidney injury in diabetic nephropathy mice via PGC-1α activation. Stem Cells 39, 913–928 (2021).
Jabbari, H. et al. Mitochondrial transplantation ameliorates ischemia/reperfusion-induced kidney injury in rat. Biochim. Biophys. Acta Mol. Basis Dis. 1866, 165809 (2020).
Emani, S. M. et al. Autologous mitochondrial transplantation for dysfunction after ischemia-reperfusion injury. J. Thorac. Cardiovasc. Surg. 154, 286–289 (2017).
Guariento, A. et al. Autologous mitochondrial transplantation for cardiogenic shock in pediatric patients following ischemia-reperfusion injury. J. Thorac. Cardiovasc. Surg. 162, 992–1001 (2021).
Jacoby, E. et al. Mitochondrial augmentation of hematopoietic stem cells in children with single large-scale mitochondrial DNA deletion syndromes. Sci. Transl. Med. 14, eabo3724 (2022).
Elliott, H. R. et al. Pathogenic mitochondrial DNA mutations are common in the general population. Am. J. Hum. Genet. 83, 254–260 (2008).
Schaefer, A. M. et al. Prevalence of mitochondrial DNA disease in adults. Ann. Neurol. 63, 35–39 (2008).
Wang, L.-y, Wang, D.-h, Zou, X.-y & Xu, C.-m Mitochondrial functions on oocytes and preimplantation embryos. J. Zhejiang Univ. Sci. B 10, 483–492 (2009).
Rodríguez-Varela, C. & Labarta, E. Role of mitochondria transfer in infertility: a commentary. Cells 11 (2022).
Muggleton-Harris, A., Whittingham, D. G. & Wilson, L. Cytoplasmic control of preimplantation development in vitro in the mouse. Nature 299, 460–462 (1982).
Cohen, J. et al. Ooplasmic transfer in mature human oocytes. Mol. Hum. Reprod. 4, 269–280 (1998).
Zhang, J. et al. In vitro maturation of human preovulatory oocytes reconstructed by germinal vesicle transfer. Fertil. Steril. 71, 726–731 (1999).
Tachibana, M. et al. Mitochondrial gene replacement in primate offspring and embryonic stem cells. Nature 461, 367–372 (2009).
Zhang, J. et al. Live birth derived from oocyte spindle transfer to prevent mitochondrial disease. Reprod. Biomed. Online 34, 361–368 (2017).
Craven, L. et al. Pronuclear transfer in human embryos to prevent transmission of mitochondrial DNA disease. Nature 465, 82–85 (2010).
Zhang, S. P. et al. Polar body transfer restores the developmental potential of oocytes to blastocyst stage in a case of repeated embryo fragmentation. J. Assist. Reprod. Genet. 34, 563–571 (2017).
Ma, H. et al. Functional human oocytes generated by transfer of polar body genomes. Cell Stem Cell 20, 112–119 (2017).
Ferreira, C. R. et al. The kinetics of donor cell mtDNA in embryonic and somatic donor cell-derived bovine embryos. Cloning Stem Cells 9, 618–629 (2007).
Rodriguez-Varela, C., Herraiz, S. & Labarta, E. Mitochondrial enrichment in infertile patients: a review of different mitochondrial replacement therapies. Ther. Adv. Reprod. Health 15, 26334941211023544 (2021).
Tzeng, C. et al. Mitochondria transfer (MIT) into oocyte from autologous cumulus granulosa cells (cGCs). Fertil. Steril. 82, S53 (2004).
Woods, D. C. & Tilly, J. L. Autologous Germline Mitochondrial Energy Transfer (AUGMENT) in human assisted reproduction. Semin Reprod. Med. 33, 410–421 (2015).
Wang, Z. B. et al. Transfer of autologous mitochondria from adipose tissue-derived stem cells rescues oocyte quality and infertility in aged mice. Aging 9, 2480–2488 (2017).
Mobarak, H. et al. Autologous mitochondrial microinjection; a strategy to improve the oocyte quality and subsequent reproductive outcome during aging. Cell Biosci. 9, 95 (2019).
Farnezi, H. C. M. et al. Three-parent babies: Mitochondrial replacement therapies. JBRA Assist. Reprod. 24, 189–196 (2020).
Miller, T. E. et al. Mitochondrial variant enrichment from high-throughput single-cell RNA sequencing resolves clonal populations. Nat. Biotechnol. 40, 1030–1034 (2022).
Keraite, I. et al. A method for multiplexed full-length single-molecule sequencing of the human mitochondrial genome. Nat. Commun. 13, 5902 (2022).
Mahrouf-Yorgov, M. et al. Mesenchymal stem cells sense mitochondria released from damaged cells as danger signals to activate their rescue properties. Cell Death Differ. 24, 1224–1238 (2017).
Sun, X. et al. Increased mtDNA copy number promotes cancer progression by enhancing mitochondrial oxidative phosphorylation in microsatellite-stable colorectal cancer. Signal Transduct. Target Ther. 3, 8 (2018).
Fan, S. et al. Mitochondrial miRNA determines chemoresistance by reprogramming metabolism and regulating mitochondrial transcription. Cancer Res. 79, 1069–1084 (2019).
Kumar, J., Mohammad, G., Alka, K. & Kowluru, R. A. Mitochondrial genome-encoded long noncoding RNA and mitochondrial stability in diabetic retinopathy. Diabetes 72, 520–531 (2023).
Lu, T. et al. Extracellular vesicles derived from mesenchymal stromal cells as nanotherapeutics for liver ischaemia-reperfusion injury by transferring mitochondria to modulate the formation of neutrophil extracellular traps. Biomaterials 284, 121486 (2022).
Liu, H. L. et al. Factors associated with rapid improvement in visual acuity in patients with Leber’s hereditary optic neuropathy after gene therapy. Acta Ophthalmol. 98, e730–e733 (2020).
Yang, S. et al. Long-term outcomes of gene therapy for the treatment of Leber’s hereditary optic neuropathy. EBioMedicine 10, 258–268 (2016).
Yuan, J. et al. Seven-year follow-up of gene therapy for leber’s hereditary optic neuropathy. Ophthalmology 127, 1125–1127 (2020).
Guy, J. et al. Gene therapy for leber hsereditary optic neuropathy: low- and medium-dose visual results. Ophthalmology 124, 1621–1634 (2017).
Acknowledgements
This work was supported by the National Key R&D Program of China (2023YFB4606700/03), the National Natural Science Foundation of China (82272561), and 135 Project of Center for High Altitude Medicine, West China Hospital, Sichuan University (GYYX24013).
Author information
Authors and Affiliations
Contributions
Z.Y. and S.B. conceptualized the initial manuscript. L.M.Y. and W.L.M. designed and organized the figures and tables. Supervision and strategic direction were provided by Z.Y. and S.B., who also contributed key ideas that shaped the manuscript. L.M.Y., W.L.M. draft, and refined the manuscript. L.M.Y., S.H.B., W.Y.G., and L.Y. revised and refined the manuscript. All authors have read and approved the review article.
Corresponding authors
Ethics declarations
Competing interests
One of the authors (S.B.) has received, during the study period, funding from the National Natural Science Foundation of China (82272561), National Key R&D Program of China (2023YFB4606700/03) and 135 Project of Center for High Altitude Medicine, West China Hospital, Sichuan University (GYYX24013). Each other author certifies that he has no commercial associations (consultancies, stock ownership, equity interest, patent/licensing arrangements, etc.) that might pose a conflict of interest in connection with the submitted article.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, M., Wu, L., Si, H. et al. Engineered mitochondria in diseases: mechanisms, strategies, and applications. Sig Transduct Target Ther 10, 71 (2025). https://doi.org/10.1038/s41392-024-02081-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41392-024-02081-y
This article is cited by
-
Strategic Targeting of Mitochondria: Bridging Biology and Therapy for Health Benefits
Cell Biochemistry and Biophysics (2025)