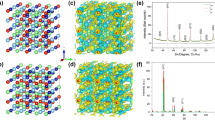
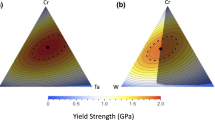
Abstract
Predicting the synthetic accessibility of multi-principal element alloys (MPEAs) across the global chemical space remains a challenge. In this study, we show that the synthesizability of MPEAs across broad compositional and structural spaces can be predicted using a physical model that expresses the total energy of any MPEA as a linear combination of energies from lower-dimensional subsystems. The model is validated with a large computational dataset and supported by the experimental synthesis of multiple MPEAs, achieving mean absolute errors near or below 7 meV/atom on a density functional theory dataset of 135,791 MPEAs spanning 28 metals and up to ten components. Its accuracy is comparable to state-of-the-art deep learning models while maintaining interpretability through cluster-expansion theory. Moreover, we show that the stability of high-entropy alloys can be predicted using a linear combination of energies from lower-dimensional systems with low errors, indicating a flatter energy landscape at high compositional complexity.
Similar content being viewed by others
Data availability
All data for reproducing this work have been included in manuscript and supplementary information. Source data is provided with this paper. All data generated or analyzed in this study are provided in the Source Data file included with this manuscript. There are no restrictions on data access. Source data are provided with this paper.
References
George, E. P., Raabe, D. & Ritchie, R. O. High-entropy alloys. Nat. Rev. Mater. 4, 515–534 (2019).
Senkov, O. N., Miller, J. D., Miracle, D. B. & Woodward, C. Accelerated exploration of multi-principal element alloys with solid solution phases. Nat. Commun. 6, 6529 (2015).
Wang, L. & Ouyang, B. Phase selection rules of multi-principal element alloys. Adv. Mater. 36, 2307860 (2024).
Miracle, D. B. & Senkov, O. N. A critical review of high entropy alloys and related concepts. Acta Mater. 122, 448–511 (2017).
Hsu, W.-L., Tsai, C.-W., Yeh, A.-C. & Yeh, J.-W. Clarifying the four core effects of high-entropy materials. Nat. Rev. Chem. 8, 471–485 (2024).
Ye, Z. et al. Using surface composition and energy to control the formation of either tetrahexahedral or hexoctahedral high-index facet nanostructures. J. Am. Chem. Soc. 146, 13519–13526 (2024).
Ye, Z. et al. A data-driven approach for the guided regulation of exposed facets in nanoparticles. Nat. Synth. 3, 922–929 (2024).
Dey, G. R. et al. Colloidal nanoparticles of high entropy materials: Capabilities, challenges, and opportunities in synthesis and characterization. ACS Nanosci. Au 4, 3–20 (2024).
Veglak, J. M., Tsai, A., Soliman, S. S., Dey, G. R. & Schaak, R. E. Disentangling competitive and synergistic chemical reactivities during the seeded growth of high-entropy alloys on high-entropy metal sulfide nanoparticles. J. Am. Chem. Soc. 146, 19521–19536 (2024).
Yang, C. et al. Overcoming immiscibility toward bimetallic catalyst library. Sci. Adv. 6, eaaz6844 (2020).
Yao, Y. et al. High-entropy nanoparticles: Synthesis-structure-property relationships and data-driven discovery. Science 376, eabn3103 (2022).
Cui, M. et al. Multi-principal elemental intermetallic nanoparticles synthesized via a disorder-to-order transition. Sci. Adv. 8, eabm4322 (2022).
Birbilis, N., Choudhary, S., Scully, J. R. & Taheri, M. L. A perspective on corrosion of multi-principal element alloys. npj Mater. Degrad. 5, 14 (2021).
Senkov, O. N., Miller, J. D., Miracle, D. B. & Woodward, C. Accelerated exploration of multi-principal element alloys for structural applications. Calphad 50, 32–48 (2015).
Chen, Y. et al. A focused review on engineering application of multi-principal element alloy. Front. Mater. 8, 816309 (2022).
Yao, Y. et al. Computationally aided, entropy-driven synthesis of highly efficient and durable multi-elemental alloy catalysts. Sci. Adv. 6, eaaz0510 (2020).
Sun, Y. & Dai, S. High-entropy materials for catalysis: A new frontier. Sci. Adv. 7, eabg1600 (2021).
Bolar, S., Ito, Y. & Fujita, T. Future prospects of high-entropy alloys as next-generation industrial electrode materials. Chem. Sci. 15, 8664–8722 (2024).
Mao, X., Chen, Q. & Granick, S. Entropy favours open colloidal lattices. Nat. Mater. 12, 217–222 (2013).
Troparevsky, M. C., Morris, J. R., Kent, P. R. C., Lupini, A. R. & Stocks, G. M. Criteria for predicting the formation of single-phase high-entropy alloys. Phys. Rev. X 5, 011041 (2015).
Chen, Z. & Yang, Y. Data-driven design of eutectic high entropy alloys. J. Mater. Inform. 3, 10 (2023).
Liu, X. et al. Machine learning-based glass formation prediction in multicomponent alloys. Acta Mater. 201, 182–190 (2020).
He, Q. F., Ding, Z. Y., Ye, Y. F. & Yang, Y. Design of high-entropy alloy: A perspective from nonideal mixing. JOM 69, 2092–2098 (2017).
Yin, J., Pei, Z. & Gao, M. C. Neural network-based order parameter for phase transitions and its applications in high-entropy alloys. Nat. Comput. Sci. 1, 686–693 (2021).
Pei, Z., Yin, J., Hawk, J. A., Alman, D. E. & Gao, M. C. Machine-learning informed prediction of high-entropy solid solution formation: Beyond the Hume-Rothery rules. npj Comput. Mater. 6, 50 (2020).
Feng, R. et al. Phase stability and transformation in a light-weight high-entropy alloy. Acta Mater. 146, 280–293 (2018).
Brown, P. & Zhuang, H. Quantum machine-learning phase prediction of high-entropy alloys. Mater. Today 63, 18–31 (2023).
Rittiruam, M. et al. High-throughput materials screening algorithm based on first-principles density functional theory and artificial neural network for high-entropy alloys. Sci. Rep. 12, 16653 (2022).
Sarker, P. et al. High-entropy high-hardness metal carbides discovered by entropy descriptors. Nat. Commun. 9, 4980 (2018).
Eckert, H. & Curtarolo, S. A formula to predict the synthesizability of high-entropy materials. Nature Research Briefing (2024).
Xie, T. & Grossman, J. C. Crystal graph convolutional neural networks for an accurate and interpretable prediction of material properties. Phys. Rev. Lett. 120, 145301 (2018).
Wang, L., He, T. & Ouyang, B. The Impact of Domain Knowledge on Universal Machine Learning Models https://doi.org/10.26434/chemrxiv-2024-fmq8p. (2024)
Ong, S. P., Wang, L., Kang, B. & Ceder, G. Li−Fe−P−O2 phase diagram from first principles calculations. Chem. Mater. 20, 1798–1807 (2008).
Lun, Z. et al. Cation-disordered rocksalt-type high-entropy cathodes for Li-ion batteries. Nat. Mater. 20, 214–221 (2021).
Ouyang, B. et al. Synthetic accessibility and stability rules of NASICONs. Nat. Commun. 12, 5752 (2021).
Jain, A. et al. Commentary: The Materials Project: A materials genome approach to accelerating materials innovation. APL Mater. 1, 011002 (2013).
Saal, J. E., Kirklin, S., Aykol, M., Meredig, B. & Wolverton, C. Materials design and discovery with high-throughput density functional theory: The open quantum materials database (OQMD). JOM 65, 1501–1509 (2013).
Kirklin, S. et al. The Open Quantum Materials Database (OQMD): Assessing the accuracy of DFT formation energies. npj Comput. Mater. 1, 15010 (2015).
Chen, P.-C. et al. Interface and heterostructure design in polyelemental nanoparticles. Science 363, 959–964 (2019).
Chen, P.-C. et al. Polyelemental nanoparticle libraries. Science 352, 1565–1569 (2016).
Tsao, T.-K., Sheikh, S. & Murakami, H. Development of platinum-group-metals-containing high entropy alloys with outstanding thermal capability and hot hardness. Appl. Phys. Lett. 119, 191901 (2021).
Shen, B. et al. Crystal structure engineering in multimetallic high-index facet nanocatalysts. Proc. Natl. Acad. Sci. 118, e2105722118 (2021).
Sreeramagiri, P. & Balasubramanian, G. Directed energy deposition of multi-principal element alloys. Front. Mater. 9, 825276 (2022).
Borg, C. K. H. et al. Expanded dataset of mechanical properties and observed phases of multi-principal element alloys. Sci. Data 7, 430 (2020).
Coury, F. G., Zepon, G. & Bolfarini, C. Multi-principal element alloys from the CrCoNi family: outlook and perspectives. J. Mater. Res. Technol. 15, 3461–3480 (2021).
Huang, L. et al. Multimetallic high-index faceted heterostructured nanoparticles. J. Am. Chem. Soc. 142, 4570–4575 (2020).
Qi, J., Cheung, A. M. & Poon, S. J. High entropy alloys mined from binary phase diagrams. Sci. Rep. 9, 15501 (2019).
Li, M. et al. High-entropy alloy electrocatalysts go to (sub-)nanoscale. Sci. Adv. 10, eadn2877 (2024).
Sun, W., Kitchaev, D. A., Kramer, D. & Ceder, G. Non-equilibrium crystallization pathways of manganese oxides in aqueous solution. Nat. Commun. 10, 573 (2019).
Chen, B.-R. et al. Understanding crystallization pathways leading to manganese oxide polymorph formation. Nat. Commun. 9, 2553 (2018).
Piner, R. D., Zhu, J., Xu, F., Hong, S. & Mirkin, C. A. “Dip-Pen” Nanolithography. Science 283, 661–663 (1999).
Huo, F. et al. Polymer pen lithography. Science 321, 1658–1660 (2008).
Chai, J. et al. Scanning probe block copolymer lithography. Proc. Natl. Acad. Sci. 107, 20202–20206 (2010).
Shen, B. et al. Morphology engineering in multicomponent hollow metal chalcogenide nanoparticles. ACS Nano 17, 4642–4649 (2023).
Shen, B. et al. Synthesis of metal-capped semiconductor nanowires from heterodimer nanoparticle catalysts. J. Am. Chem. Soc. 142, 18324–18329 (2020).
Smith, P. T., Wahl, C. B., Orbeck, J. K. H. & Mirkin, C. A. Megalibraries: Supercharged acceleration of materials discovery. MRS Bull. 48, 1172–1183 (2023).
Lai, M., Shin, D., Jibril, L. & Mirkin, C. A. Combinatorial synthesis and screening of mixed halide perovskite megalibraries. J. Am. Chem. Soc. 144, 13823–13830 (2022).
Hume-Rothery, W. Researches on the nature, properties, and conditions of formation of intermetallic compounds, with special reference to certain compounds of tin. PhD thesis, University of London (1926).
Pickering, E. J., Muñoz-Moreno, R., Stone, H. J. & Jones, N. G. Precipitation in the equiatomic high-entropy alloy CrMnFeCoNi. Scr. Mater. 113, 106–109 (2016).
Chen, W. et al. A map of single-phase high-entropy alloys. Nat. Commun. 14, 2856 (2023).
Ye, Y. F., Wang, Q., Lu, J., Liu, C. T. & Yang, Y. High-entropy alloy: Challenges and prospects. Mater. Today 19, 349–362 (2016).
Cantor, B. Multicomponent high-entropy Cantor alloys. Prog. Mater. Sci. 120, 100754 (2021).
Magyari-Köpe, B., Vitos, L. & Grimvall, G. Anomalous behavior of lattice parameters and elastic constants in hcp Ag− Zn alloys. Phys. Rev. B 70, 052102 (2004).
Liu, S., Liu, B., Gong, C. & Li, Z. A nanoporous Cu-Ag thin film at the Cu-Ag-Zn alloy surface by spontaneous dissolution of Zn and Cu in different degrees as a highly sensitive non-enzymatic glucose sensor. Electrochim. Acta 320, 134599 (2019).
Barroso-Luque, L. et al. Cluster expansions of multicomponent ionic materials: Formalism and methodology. Phys. Rev. B 106, 144202 (2022).
Sanchez, J. M., Ducastelle, F. & Gratias, D. Generalized cluster description of multicomponent systems. Phys. A: Stat. Mech. Appl. 128, 334–350 (1984).
Sanchez, J. M. Cluster expansions and the configurational energy of alloys. Phys. Rev. B 48, 14013–14015 (1993).
Tepesch, P. D., Garbulsky, G. D. & Ceder, G. Model for configurational thermodynamics in ionic systems. Phys. Rev. Lett. 74, 2272–2275 (1995).
Asta, M., Foiles, S. M. & Quong, A. A. First-principles calculations of bulk and interfacial thermodynamic properties for fcc-based Al-Sc alloys. Phys. Rev. B 57, 11265–11275 (1998).
Nguyen, A. H., Rosenbrock, C. W., Reese, C. S. & Hart, G. L. W. Robustness of the cluster expansion: Assessing the roles of relaxation and numerical error. Phys. Rev. B 96, 014107 (2017).
Levy, O., Hart, G. L. W. & Curtarolo, S. Uncovering compounds by synergy of cluster expansion and high-throughput methods. J. Am. Chem. Soc. 132, 4830–4833 (2010).
Zunger, A., Wei, S. H., Ferreira, L. G. & Bernard, J. E. Special quasirandom structures. Phys. Rev. Lett. 65, 353–356 (1990).
van de Walle, A. et al. Efficient stochastic generation of special quasirandom structures. Calphad 42, 13–18 (2013).
Esters, M. et al. Settling the matter of the role of vibrations in the stability of high-entropy carbides. Nat. Commun. 12, 5747 (2021).
Zhang, Z., Li, M., Cavin, J., Flores, K. & Mishra, R. A fast and robust method for predicting the phase stability of refractory complex concentrated alloys using pairwise mixing enthalpy. Acta Mater. 241, 118389 (2022).
Sun, W. et al. The thermodynamic scale of inorganic crystalline metastability. Sci. Adv. 2, e1600225 (2016).
Wang, L. et al. Elemental stability rules for high entropy disordered rocksalt type Li-ion battery positive electrodes. Adv. Energy Mater. 15, 2404982 (2025).
Walsh, F., Zhang, M., Ritchie, R. O., Minor, A. M. & Asta, M. Extra electron reflections in concentrated alloys do not necessitate short-range order. Nat. Mater. 22, 926–929 (2023).
Zhang, R. et al. Short-range order and its impact on the CrCoNi medium-entropy alloy. Nature 581, 283–287 (2020).
Yin, B., Yoshida, S., Tsuji, N. & Curtin, W. A. Yield strength and misfit volumes of NiCoCr and implications for short-range-order. Nat. Commun. 11, 2507 (2020).
Sanchez, J. M. & de Fontaine, D. The fcc Ising model in the cluster variation approximation. Phys. Rev. B 17, 2926–2936 (1978).
Sanchez, J. M. & de Fontaine, D. in Industrial Chemistry Library Vol. 2 (eds O’Keeffe M & Navrotsky A.) 117-132 (Elsevier, 1981).
Zhang, R. et al. Direct imaging of short-range order and its impact on deformation in Ti-6Al. Sci. Adv. 5, eaax2799 (2019).
Seol, J. B. et al. Mechanically derived short-range order and its impact on the multi-principal-element alloys. Nat. Commun. 13, 6766 (2022).
Ferrari, A., Körmann, F., Asta, M. & Neugebauer, J. Simulating short-range order in compositionally complex materials. Nat. Comput. Sci. 3, 221–229 (2023).
Ouyang, B. et al. Effect of fluorination on lithium transport and short-range order in disordered-rocksalt-type lithium-ion battery cathodes. Adv. Energy Mater. 10, 1903240 (2020).
Riebesell, J. et al. A framework to evaluate machine learning crystal stability predictions. Nat. Mach. Intell. 7, 836–847 (2025).
Ong, S. P. et al. Python Materials Genomics (pymatgen): A robust, open-source python library for materials analysis. Comput. Mater. Sci. 68, 314–319 (2013).
van de Walle, A., Asta, M. & Ceder, G. The alloy theoretic automated toolkit: A user guide. Calphad 26, 539–553 (2002).
Ong, S. P., Jain, A., Hautier, G., Kang, B. & Ceder, G. Thermal stabilities of delithiated olivine MPO4 (M=Fe, Mn) cathodes investigated using first principles calculations. Electrochem. Commun. 12, 427–430 (2010).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Kresse, G. & Hafner, J. Ab initio molecular-dynamics simulation of the liquid-metal–amorphous-semiconductor transition in germanium. Phys. Rev. B 49, 14251–14269 (1994).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for liquid metals. Phys. Rev. B 47, 558–561 (1993).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Monkhorst, H. J. & Pack, J. D. Special points for Brillouin-zone integrations. Phys. Rev. B 13, 5188–5192 (1976).
Methfessel, M. & Paxton, A. T. High-precision sampling for Brillouin-zone integration in metals. Phys. Rev. B 40, 3616–3621 (1989).
Acknowledgements
This work is supported by startup funding from Florida State University (B.O.). Additional support was provided by the American Chemical Society Petroleum Research Fund (ACS-PRF # 68184-DNI10) (L.W. and B.O.). Computational resources were provided by ACCESS (B.O.), the National Energy Research Scientific Computing Center (NERSC), a U.S. DOE Office of Science User Facility supported under Contract No. DE-AC02-05CH11231 (B.O.), and the Research Computing Center at Florida State University (B.O.). The Department of Energy’s Office of Energy Efficiency and Renewable Energy at the National Renewable Energy Laboratory also supported computation and data processing (B.O.). The experimental work of this paper was supported by the Toyota Research Institute, Inc. (B.S., Z.Y., and C.A.M.) and the U.S. Army DEVCOM ARL Army Research Office (ARO) Energy Sciences Competency (Electrochemistry) Program award W911NF-23-1-0285 (Z.Y. and C.A.M.). The views and conclusions contained in this document are those of the authors and should not be interpreted as representing the official policies, either expressed or implied, of the U.S. Army or the U.S. Government.
Author information
Authors and Affiliations
Contributions
B.O. supervised and planned all aspects of the research. B.O. and L.W. designed the computations and generated all computational results; B.O., L.W., and Z.H. analyzed the data and generated all figures; B.S. and Z.Y. did chemical synthesis and analysis; L.W., B. S., Z.Y., Y.Z., C.A.M., and B.O. contributed to the writing.
Corresponding author
Ethics declarations
Competing interests
C.A.M. has financial interests in Mattiq Inc. which could potentially benefit from the outcomes of this research. Northwestern University has financial interests relative to intellectual property related to this research. As a result of these interests, Northwestern University could ultimately potentially benefit financially from the outcomes of this research.
Peer review
Peer review information
Nature Communications thanks the anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, L., Shen, B., He, ZD. et al. Universal framework for efficient estimation of stability in multi-principal element alloys. Nat Commun (2026). https://doi.org/10.1038/s41467-026-69585-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-69585-9