Abstract
Vitamin D deficiency is an emerging public health problem globally, with devastating health consequences. Some studies suggest that exogenous sex hormones, found in hormonal contraceptives, may enhance vitamin D levels. However, the reasons for this association are not fully understood, as women using hormonal contraception may have different lifestyle habits affecting their vitamin D status. Therefore, this study seeks to explore the relationship between hormonal contraceptive use and vitamin D levels. A Facility based comparative cross-sectional study was conducted in Gondar town from February to April 2023, involving a total of 162 women using three types of hormonal contraceptives (Norplant, DMPA, and COC) and 162 age and BMI-matched non-users as controls in a 1:1 ratio. Participants were selected using systematic random sampling. A semi-structured questionnaire was used to collected data regarding the socio-demographic, economic, obstetric, lifestyle, and clinical information. 5 milliliters of blood samples were collected from each participant for Laboratory analysis of serum vitamin D, calcium, and alkaline phosphates using a Beckman Coulter chemistry analyzer. Independent t-tests, ANOVA with post hoc Bonferroni test was used to compare statistics between the two groups, and logistic regression models to identify factors associated with Vitamin D deficiency. The mean serum Vitamin D levels of Norplant, DMPA, and COC users were 24.08 (± 5.17), 24.83 (± 5.52), and 31.90 (± 6.94) respectively; whereas control group has mean Vitamin D level of 22.00 (± 7.97). On the current study the prevalence of Vitamin D deficiency (< 20 ng/ml) among hormonal contraceptive users was found to be 21.6% (35/162), whereas 48.14% (78/162) of non-user controls had vitamin D deficiency. The odds of having Vitamin D deficiency was higher among participants who attained higher education, who never eat fish and have never been used vitamin D Supplements. However, the use of combined oral contraceptives (COC) shown to reduce the odd of having vitamin D deficiency by 90%. Similarly, individuals with normal and hypercalcemia state shown to have lower odd of having Vitamin D deficiency. Users of combined oral contraceptives (COC) had significantly higher mean serum Vitamin D levels compared to users of Norplant and DMPA, as well as non-users. The prevalence of Vitamin D deficiency was lower among COC users compared to non-users, highlighting a potential protective effect of COC use against Vitamin D deficiency.
Similar content being viewed by others
Introduction
Vitamin D (calciferol) is a fat-soluble vitamin that is important for normal functions of the body1,2. It is present in the body in to forms Vitamin D3 (Cholecalciferol) and Vitamin D2 (Ergocalciferol). Cholecalciferol secreted in the skin in response to active sunlight exposure whereas Ergocalciferol usually obtained from diet1. Vitamin D regulate calcium and phosphorus level by regulating their metabolism and absorption3. In addition, Vitamin D also demonstrated to plays a role in immune cells function, cell growth, and inflammatory responses4,5.
Even though vitamin D is crucial for normal function of the body, several epidemiological studies have shown that a significant percentage of the population globally have insufficient levels of this important nutrient1,6,7. This is very concerning since vitamin D deficiency and insufficiency have been linked to a higher risk of several health issues, including osteoporosis, adverse cardiovascular outcomes, autoimmune disease, and cancers7,8,9. Some reports have also indicated that vitamin D plays a role in various forms of adverse reproductive outcomes on the offspring10. Blood 25-hydroxycholecalciferol (25(OH) D) level believed to be the best indicator of person vitamin D status is, as it reflects both cutaneous synthesis and dietary consumption of the nutrient7.
Vitamin D deficiency is currently a public health problem, affecting individuals of all classes and populations with varying epidemiology1,11. The prevalence of vitamin D deficiency varies across different regions, with prevalence of serum 25(OH)D deficiency (< 30 nmol/L) varied from 5.5% in the Region of the Americas to 35.2% in the Eastern Mediterranean Region. It is also reported that females were more vulnerable to vitamin D deficiency than males11. It has been gaining much attention globally due to its health effects on the reduction of mortality and morbidity1,11.
Several human factors reported to affect vitamin D levels in the body. The most significant factor is sun light exposure, since skin produces vitamin D when exposed to sun. Moreover, individuals age, skin color, Body mass index (BMI) and dietary intake of vitamin D-rich food can also influence vitamin D levels1,12. It is also believed that Hormonal contraceptives (HCs) containing estrogens or estrogens coupled with progestins may affect vitamin D serum concentrations13. Some studies suggest exogenous estrogen may improve vitamin D status, but the etiology is unclear because women who use hormones may make lifestyle choices that differentially affect vitamin D status10,13.
Hormonal Contraceptives (HCs) are birth control methods containing artificial Estrogen (Ethinylestradiol (EE)) and/or progestin14. These contraceptives work by shifting hormone levels in body14,15. Ethinylestradiol (EE2) is a analogous of 17β-estradiol16, the major endogenous human estrogen. EE2 is used in many forms of hormonal contraception and is one of the most commonly used medications for this purpose. EE2 exert its function by acting as a ling and activating the estrogen receptor and exerts its hormonal effect16,17. Another component of HC which administrated solely or in combination with EE is progestin18,19,20,21. The molecularly modified progestin has strong progestogenic effects. Molecular modifications, such as the adding of a 17α-alkyl group and alteration to the steroid backbone, enhance the efficiency of progestins in HC preparations19.
Epidemiological studies reported that HCs are the most commonly used methods of birth control worldwide22,23. World Fertility and Family Planning 2020 reported that in 2019, 49% of all women of reproductive age were using contraceptives globally24 and half of them were using HCs containing progestin and/or Estrogen25. The hormonal contraceptive prevalence varies globally, with higher rates observed in developed countries compared to developing nations23. In Ethiopia, the contraceptive prevalence rate (CPR) is currently 22.2%12.
Serum level of alkaline phosphate (ALP) frequently analyzed along with Vitamin D, which is a crucial indicator of bone health26,27,28. ALP is an enzyme present in liver, bones, kidneys, and digestive system; and that is involved in the process of bone mineralization, and elevated levels of ALP may indicate bone turnover13. Calcium is another essential nutrient frequently analyzed along with ALP and Vitamin D; that plays a role in bone growth and regeneration, muscle function, and nerve transmission26,29.
Hormonal contraceptives are generally utilized by women for birth control and family planning purpose. However, there are some evidences suggesting they may affect some biomolecules, including their vitamin D status10,30,31. Few studies have investigated the relationship between HC and serum vitamin D levels, with conflicting results32,33,34,35. Some studies reported combined HC shown to increase blood levels of vitamin D, although the exact molecular mechanism is unclear10,13,34,35,36. There is also report affirming the use of combined oral contraceptives (COC) may decrease Vitamin D levels37. On the other hand, other epidemiological studies investigating the association between hormonal contraceptive usage and serum vitamin D levels have reported contradictory findings. Some studies have found no significant association between hormonal contraceptive use and serum vitamin D levels or even lower vitamin D level among hormonal contraceptive users37,38.
Additional investigation about the association between HC and vitamin D is needed to briefly explain their association, because of the potential effects of HC preparations on blood vitamin D levels. Moreover, Understanding the variables that influence vitamin D levels among those on HC may help healthcare professionals to identify those who are at risk of vitamin D deficiency and develop targeted medicines to address this issue. The purpose of this study is to analyze blood vitamin D levels and identify the prevalence of Vitamin D deficiency and associated factors among hormonal contraceptive users and non-user controls in Gondar town in 2023, with a focus on serum ALP and calcium levels.
Method and material
Study area, design, and period
After receiving ethical clearance letter from “School of Biomedical and Laboratory Sciences Institutional Review Board by reference number SBLS/184/14; A facility-based comparative cross-sectional study was conducted. The study was carried out at three healthcare institutions in Gondar town (The University of Gondar Comprehensive Specialized Hospital, Ethiopian Family Guidance Association Gondar branch, and Gondar Poly Clinic). The study data was collected from February 6 to April 28, 2023.
Population
Source population
All eligible reproductive age group women who visited the two institutes for family planning service in 2023 (hormonal contraceptives user group). And a control group of age and BMI matched women who were refrain from using hormonal contraceptives.
Study population
The study populations were HC user who visited the selected health institutes seeking routine family planning services and consultations, with selection based on meeting the defined inclusion criteria at the time of data collection from February 6 to April 28 2023, while the second cohort comprised age and BMI-matched women who were not using hormonal contraceptives.
Eligibility criteria
Inclusion criteria for hormonal contraceptives user group
Volunteer women aged 18–45 years with regular use of hormonal contraceptives (Norplant, DMPA and COC) for at least 6 months. And able to provide information related to demography, life and clinical variables; and willing to provide 5 ml of blood.
Inclusion criteria for non-user controls
Volunteer women aged 18–45 years with abstinence of hormonal contraceptives usage in the past 6 months. And able to provide information related to demography, life and clinical variables; and willing to provide 5 ml of blood.
Exclusion criteria for both groups
Women with poor HC compliance, mental and hearing problem or other health issues limiting their ability to provide information. women on anti-Tuberculosis and antiepileptic medications. Women with acute or chronic liver disease. Women with acute or chronic kidney disease. Pregnant or lactating women or history of pregnancy or miscarriage in the past 12 months History of vitamin D deficiency or calcium metabolism disorders.
Variables
Dependent variable
-
Serum vitamin D level.
Independent variables
-
Socio-demographic factors (Age, Educational Status, Monthly Income, Marital Status, Residency, Housing Condition and Occupation).
-
Anthropometric (BMI, WHR and Skin color).
-
Clinical and obstetric conditions (Blood pressure, hormonal contraceptive class, duration of hormonal contraceptive usage, gravidity, and parity).
-
Behavioral characteristics (Coffee drinking habit, physical activity, smoking and alcohol consumption).
-
Sun light exposure modulators (Sunlight exposure time in a day, utilization of sun screen lotions, type of clothing).
-
Usual food content and Dietary diversity.
Sample size and sampling technique
A total of 324 Women (162 for HC user group and 162 for Non-user Controls) was enrolled on the study. Systematic random sampling technique was applied to recruit participants. The sample size was allocated proportionally. We obtained data from the three health institutes in the previous year (2022) to determine the skip interval for our systematic random sample during the duration of data collection. Every 5th of study participants arriving to the clinics were asked for participation and included in the study.
Operational definition
Hormonal contraceptive user women aged 18 years and older who have consistently used hormonal contraceptives (such as combined oral contraceptives (COC), Depo-medroxyprogesterone acetate (DMPA), or Norplant) for a minimum duration of 6 months39.
Hormonal contraceptive non-user (Controls) Women of reproductive age group abstains of HC use, individuals who use non-hormonal contraceptives (Condoms, Cervical Cap and diaphragm).
Vitamin D deficiencySerum levels of 25-hydroxyvitamin D (25(OH)D) below < 20 ng/mL or < 50 nmol/L13,40.
Vitamin D insufficiency serum levels of 25-hydroxyvitamin D (25(OH)D) between 20 and 30 ng/mL or 50–75 nmol/L13,40.
Skin Color - Skin color classification is based on the Fitzpatrick scale to describe skin pigmentation as (“Type IV” light brown, “Type V” dark brown Type and “VI- very dark brown”).
Physical exercise or activity refers to any physiological movement produced by skeletal muscles that needs energy expenditure and raises heart rate at least once every day for 20–30 min in a continuous manner. Walking, running, cycling, swimming, weight lifting, and participating in other sports or fitness classes are all examples of this. For the objectives of this study, physical exercise or activity will be measured in terms of frequency (number of sessions per week) and length (minutes per session). “Active” individuals engage in regular physical exercise or activity for at least 150 min per week, while “inactive” individuals report no engagement in physical exercise or activity for at least 150 min per week41.
Sun light exposure time in a day: Common sunlight exposure time in a day refers to the duration of time spent outdoors in direct sunlight, which can contribute to the synthesis of vitamin D in the skin. For the purpose of this study, sunlight exposure time will be categorized into three time periods: “Sun raise/morning” The time period between 7:00 and 9:00 AM, “Mid-day” The time period between 10:00 and 3:00 PM, and Sunset/evening “The time period between 4:00 and 6:00 PM”.
Utilization of Sunscreen Lotion refers to the frequency of applying sunscreen on the skin to protect it from the harmful effects of UV radiation from the sun. For the purpose of this study among study participants, sunscreen usage will be categorized into three levels “Sunscreen users” Participants who regularly use sunscreen or lotion when exposed to sunlight, regardless of their eating habits. “Sunscreen non-users” Participants who do not use sunscreen or lotion when exposed to sunlight, regardless of their eating habits. “Variable sunscreen or lotion users” Participants who inconsistently use sunscreen or lotion when exposed to sunlight, depending on factors such as the weather or activities they are engaged in.
Utilization of Umbrella refers to the frequency of using Umbrella to protect skin from the harmful effects of UV radiation from the sun. For the purpose of this study umbrella use will be categorized in to three usage level “Regular Umbrella Users” Participants who use an umbrella for sun protection daily, or on a consistent basis when outdoors. “Occasional Umbrella Users” Participants who use an umbrella for sun protection on a weekly or monthly basis, depending on weather conditions or activities. “Umbrella Non-Users” Participants who never use an umbrella for sun protection, regardless of weather or outdoor activities.
Dietary Diversityrefers to the variety of different foods and food groups consumed by an individual within a specified period, typically a day or a week. For the purpose of this study dietary diversity will be categorized “High Dietary Diversity” typically consume foods from fruits, vegetables, grains, proteins, dairy, and fats/oils. “Moderate Dietary Diversity” individual consumes foods from a variety of food groups but may not include all major food groups or may lack variety within specific food groups. “Low Dietary Diversity” Low dietary diversity signifies limited variation in food choices, with a focus on a narrow range of foods or heavy reliance on a few food groups. “Very low diet diversity” consume the same types of foods or neglect certain food groups.
Dietary Vitamin D intake quantification To be considered a consumer of Vitamin D through dietary intake, participants must meet the following criteria: “Liver Consumer”: Having consumed a serving of beef liver greater than 150 g in one meal per day. “Fish Consumer”: Having a meal consisting of medium 85–170 g of fatty fish, such as salmon and sardines, per day “Fish Oil Consumer”: Having consumed fish oil products containing 0.25–0.5 g (1–2 teaspoons) of combined EPA (eicosapentaenoic acid) and DHA (docosahexaenoic acid) per day. “Fortified Cereals Consumer”: Having consumed at least 250 g of fortified cereal products per day. “Egg Consumer”: Having a meal consisting of at least 3 eggs per day. “Fortified Dairy Products Consumer”: Having consumed serving sizes of fortified dairy products that provide about 100 IU of Vitamin D, such as: 1 cup of fortified milk (about 250 ml) or 1 cup of fortified yogurt (about 250 ml) or 1.5 ounces of fortified cheese.
Serum calcium level “Hypocalcemia” serum calcium < 8.5 mg/dL, “Hypercalcemia” serum calcium > 10.5 mg/dL.
Data collection tool and procedure
Individuals who met the inclusion criteria for each study group were invited to participate after being fully briefed about the study’s objectives. Participants who agreed to participate were asked to give written signed informed consent. A standard questionnaire was prepared and pre-tested to collected variables about sociodemographic, economical, behavioral and clinical characteristics. The questionnaire was first prepared in English, and then it was translated into local Amharic language. the prepared questions were pre-tested at “Azezzo” health center to adapt to the local context based on the study objectives through face-to-face interview. Information on hormonal contraceptive use, including type, dosage, and duration, was also obtained from medical records.
Weight in kilograms (kg) was measured to the closest tenth kg with a digital balance attached to a Seca digital weighted scale (Germany), with subjects barefoot and dressed in light clothing. Height (cm) was measured twice and reported to the nearest centimeter using a Seca (Germany) calibrated Stadiometer. BMI (kg/m2) was calculated using measured height and weight. The waist circumference was measured with a meter halfway between the lowest rib border and the iliac crest. The hip circumference was measured with a meter over the widest region of the hips, crossing behind the back. Blood pressure (BP) was measured using an analog sphygmomanometer (Omron blood pressure monitor Japan) and stethoscope (Omron Japan).
Following the interview, the upper arm region was thoroughly cleansed with alcohol wipes, and subsequently allowed to completely dry. The needle was then swiftly inserted at a 30–45-degree angle to obtain venous blood sample of 5 ml from the medial cubital vein of the left arm using a syringe, which was then transferred into a gel-coated serum separator tube (SST) with a specific ID label. The blood sample was left to coagulate at room temperature for half an hour before being centrifuged at 5000 rotations per minute (RPM) for 3 min. The resulting serum was then moved to a sterile “Nunc tube” and transported to the university of Gondar comprehensive specialized hospital clinical chemistry section for analysis.
Serum vitamin D levels were analyzed utilizing a standardized laboratory immunoassay technique on the Unicel DxI 800 Beckman Coulter Clinical Chemistry Analyzer, with Beckman Coulter reagents. The Vitamin D Total assay employed a two-step competitive binding immunoenzymatic procedure, wherein the initial incubation involves the addition of a sample to a reaction vessel with a DBP releasing agent and paramagnetic particles coated with sheep monoclonal anti-25(OH) vitamin D antibody. This process allows for the release of 25(OH) vitamin D from DBP and binding to the immobilized monoclonal anti-25(OH) vitamin D on the solid phase. Subsequently, a 25(OH) vitamin D analogue-alkaline phosphatase conjugate is introduced to compete for binding to the immobilized monoclonal anti-25(OH) vitamin D. Following a subsequent incubation, materials bound to the solid phase are separated in a magnetic field while unbound materials are washed away. The chemiluminescent substrate is then added to the vessel, and the light generated by the reaction is measured using a luminometer. The intensity of light production is inversely related to the concentration of 25(OH) vitamin D in the sample. The result was reported as ng/ml.
Serum Calcium and ALP analyses were conducted using the Beckman Coulter DxC700 Automated Chemistry Analyzer, with test results reported in mg/dl for calcium and International Units per liter (IU/L) for enzymatic ALP levels. The DxC 700 AU clinical chemistry analyzer represents the latest technological advancement produced by Beckman Coulter Inc., a company under the Danaher Corporation based in Brea, California, United States.
Data analysis and interpretation
The data collected underwent manual verification, categorization, and coding procedures. It was then entered into EPI-DATA software for organization. Subsequently, The data underwent rigorous cleaning procedures, including dealing with missing values, correcting inaccuracies, removing duplicates and handling missing values, ensuring data consistency and transferred to STATA VERSION 17 for detailed descriptive statistical analysis. The normal distribution of continuous variables was scrutinized utilizing the Kolmogorov-Smirnov test. Various statistical methods such as Independent t-test, ANOVA, and logistic regression were employed to assess and compare values across different groups, while also identifying factors associated with Vitamin D deficiency.
A one-way ANOVA with Bonferroni post-hoc test was executed to compare the mean serum levels of Vitamin D, Total Calcium, and Alkaline phosphate between Hormonal contraceptive users and the control group. Furthermore, binary and multivariate logistic regression models were utilized to ascertain the factors linked to Vitamin D deficiency. Variables demonstrating a significance level of ≤ 0.25 in the bivariate analysis were reevaluated in the multivariate logistic regression model to control for potential confounding variables and to pinpoint the independent factors related to Vitamin D deficiency. Both crude and adjusted odds ratios (COR and AOR) were computed, along with their corresponding 95% confidence intervals (CI), to determine the strength of associations between variables. A statistical significance of < 0.05 was assumed for the multivariate regression model to assess statistical significance.
Ethical considerations
The study protocol was approved by the Institutional Review Board of School of Biomedical and Laboratory Sciences, college of medicine and health sciences University of Gondar; by reference number SBLS/184/14. The study was conducted in accordance with the Declaration of Helsinki guidelines and regulations. Permission was also gained from the medical director of each three of the health institute the data was collected. “Informed consent” was obtained from all participants (Supplementary material 1), and confidentiality of data was maintained throughout the study. In addition, to ensure confidentiality, codes were used as identifier of participants on the questionnaire and laboratory requests. The collected data was not used for another purpose other than the present study.
Results
Socio-demographic characteristics
In the current study a total of 324 women were included in two study groups. There were 162 women who were using hormone-based contraceptives (Norplant, DMPA, and COC) in one group, while the other group consisted of 162 women who did not use hormonal contraceptives, matched by age and BMI. The mean age for women using hormonal contraceptives was 26.88 years (± 5.50), and for the control group, it was 26.80 years (± 5.60). Over half of the study participants, 178 (54.92%) had attained at least a secondary education level or higher (Table 1).
Anthropometric, clinical and obstetric characteristics
The mean body mass index (BMI) for those using hormonal contraceptives was 22.76 (± 3.85), and 22.52 (± 3.85) for the non-user control group. Out of the participants, 155 (47.84%) had dark brown (type V) skin, while only 74 (22.84%) had light skin (type IV). Additionally, 154 (47.53%) of the study subjects were in a primigravida state, with only 27 (8.33%) being nulliparous (Table 2).
Hormonal contraceptive status of participants
The current study enrolls users of three contraceptive 54 in each group and age and BMI matched non-user controls (Fig. 1).
Dietary intake of vitamin D rich foods
About 183 (56.48%) of the women participated on this study never consumed fish and 312 (96.29%) never consumed Fish oil. 283 (87.34%) of participants reported on never been used vitamin D supplements ever. On the other hand, 100 (30.86%) consumed fortified milk routinely (Table 3).
Sunlight exposure and lifestyle characteristics
About 93 (28.70%) of participants reported on that they never use Sun screen lotion and 194 (59.88%) of the participants usually use umbrella whenever they go outside of home. 126 (38.89%) of the participants usually exposed to sun light on the mid-day. 220 (67.90%) of the participants reported vegetables as their usual food content (Table 4).
Serum vitamin D, calcium and alkaline phosphate level of participants
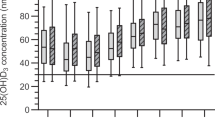
The mean serum Vitamin D levels were found to be 26.94 (± 6.87) for hormonal contraceptive users and 22.00 (± 7.97) for control group participants (Table 5). It was observed that different types of hormonal contraceptives had varying effects on the serum Vitamin D levels of the study participants, as indicated in the ANOVA table below. Specifically, the mean Vitamin D levels for Norplant, DMPA, and COC were 24.08 (± 5.17), 24.83 (± 5.52), and 31.90 (± 6.94) respectively (Table 6). Furthermore, the Bonferroni post hoc analysis revealed that the mean serum Vitamin D levels differed significantly between each contraceptive class and were found to be statistically significant (p < 0.001) (Table 6).
Prevalence of vitamin D deficiency and insufficiency
On the current study the prevalence of Vitamin D deficiency (< 20 ng/ml) among hormonal contraceptive users was found to be 21.6% (35/162), whereas 48.14% (78/162) of non-user controls had vitamin D deficiency. On the other hand, the prevalence of Vitamin D insufficiency (20.01–29.99 ng/ml) was found to be 68.51% (111/162) among hormonal contraceptive users and 83.33% (135/162) among non-user controls. The overall prevalence of Vitamin D deficiency and insufficiency among the study participants found to be 113 (34.87%) and 246 (75.92%) respectively (Table 7).
Factors associated with vitamin D deficiency among participants
Variable screening was carried out to choose independent variables for the regression model. The independence and exclusiveness of each predictor variable were confirmed during the design phase, while variables exhibiting multicollinearity were excluded from the regression model in the second analysis stage. A chi-squared (chi2) test was conducted for each categorical independent variable. Variables that met the assumption were then analyzed in a bivariate logistic regression. The Hosmer–Lemeshow goodness of fit test was performed for the logistic regression model, yielding a p-value of 0.70.
In the bivariable analysis, the following predictors were found to have a significant association (p < 0.05) with Vitamin D deficiency: hormonal contraceptive status, type of hormonal contraceptive, educational status, systolic blood pressure (SBP), food diversity score, use of sunscreen lotion, use of an umbrella, habit of consuming fish, and serum calcium level. All variables with a p-value < 0.25 in binary regression were included in the multivariable logistic analysis. In the multivariable analysis, it was observed that participants with higher education (p = 0.011), those who never consume fish (p = 0.018), and individuals who do not use Vitamin D supplements (p = 0.016) had higher odds of experiencing vitamin D deficiency. On the other hand, users of combined oral contraceptives (COC) (p = 0.000) and those with normal or high levels of calcium in the blood had lower odds of developing vitamin D deficiency, with p-values of (0.041) and (0.049) respectively (Table 8).
The odds of having Vitamin D deficiency was 2.99 times higher among participants who attained higher education. Conversely, Participant who never eat fish shown to have increased odds of having Vitamin D deficiency by 12.36 times. Participants who reported to have never been used vitamin D Supplements was observed to have 4.1 times higher odds of having Vitamin D deficiency. However, user of combined oral contraceptives (COC) revealed to be 90% less likely to for occurrence of Vitamin D deficiency with AOR of 0.10. Similarly, individuals with normal serum calcium level or hypercalcemia state shown to have lower odd of having Vitamin D deficiency (Table 8).
Discussion
In the present study the users of Hormonal contraceptives (Norplant, DMPA and COC) had higher mean value of serum Vitamin D (25(OH)D) level than controls. These findings were consistent with previous findings from Italy42, Norway43, Germany34, United Kingdom32,44, Netherlands45, United State10,33,36,46,47, Canada31,48 and Uruguay49. The observed higher mean serum vitamin D levels among hormonal contraceptive users compared to non-users could potentially be explained by the role of hormonal contraceptives in increasing vitamin D binding proteins (VDBP)50,51. Estrogen components in Hormonal contraceptives has been shown to increase the production of VDBP in the liver50,52. VDBP is responsible for transporting vitamin D in the bloodstream, and higher levels of VDBP can lead to increased binding and circulation of vitamin D in the body. As a result, individuals with higher estrogen levels, such as those using hormonal contraceptives, may have higher levels of VDBP and therefore higher serum vitamin D levels. Additionally, estrogen has been shown to have a direct effect on the activity of enzymes involved in vitamin D metabolism. Estrogen can enhance the conversion of vitamin D into its active form, which may also contribute to higher serum vitamin D levels in hormonal contraceptive users10. Similarly, it is also hypothesized that hormonal contraceptives might increase absorption of vitamin D in the intestine, and leading to higher levels of vitamin D in the bloodstream35.
Opposing results were found in the current study regarding the association between the use of hormonal contraceptives and serum Vitamin D (25(OH)D) levels when compared to previous studies conducted in Iraq38 and Turkey37. These studies suggested that the use of hormonal contraceptives may have no effect on serum Vitamin D levels, or even lead to a decrease in the mean level of Vitamin D among users. The conflicting results between our study and the studies from Iraq and Turkey may be attributed to factors such as differences in study populations and methodologies. For instance, the study from Turkey focused on women with polycystic ovary syndrome (PCOS), a condition known to impact Vitamin D levels53. The variation in findings between these studies and ours could be explained by the difference in study participants, as our study included individuals without PCOS.
The present study also revealed that Users of combined oral contraceptives (COC) had significantly higher mean serum Vitamin D levels compared to users of Norplant and DMPA, as well as non-users. This finding may partly be explained by the fact that COCs consist of a combination of estrogen and progestin, whereas other classes of hormonal contraceptives are primarily composed of progestin only. Estrogen has been shown to have a potential role in enhancing Vitamin D synthesis and metabolism, which could account for the higher levels observed in COC users. Conversely, progestin-only contraceptives may not have the same effect on Vitamin D levels. This differentiation in hormonal composition may help explain the variability in serum Vitamin D levels among users of different types of hormonal contraceptives. This hypothesis is supported by several previous studies10,13,36,46,47,49,54. However, further research is needed to fully elucidate the underlying mechanisms responsible for these differences.
On the present study the prevalence of Vitamin D deficiency (< 20 ng/ml) among hormonal contraceptive users was found to be 21.6% (15.90–28.65), whereas 48.14% (40.50–55.87) of non-user controls had vitamin D deficiency. This finding is consistent with previous studies from Norway (17%)43 and the United States (19.1%)46 which reports hormonal contraceptive users have lower vitamin D prevalence. This may be explained by the action of Estrogen to have a potential role in enhancing Vitamin D synthesis and metabolism, which could account for the higher levels observed in among HC users10. It can also further elucidate that better compliance with healthcare recommendations among women on hormonal contraceptives; this is because women who use hormonal contraceptives may have more consistent interaction with the healthcare system, including regular visits to their healthcare providers for prescription refills and check-ups. During these visits, healthcare providers may discuss the importance of maintaining adequate vitamin D levels and provide recommendations for achieving this, such as dietary changes or supplementation. This increased engagement with healthcare services may help women using hormonal contraceptives better manage their health, including their vitamin D status15,54.
In contrast, the reported prevalence of Vitamin D deficiency among hormonal contraceptive users on the current study 21.6% (15.90–28.65), is lower than previous reports from Italy (35%)42 and Canada (50%)31, but higher than result from Uruguay49 and United state10; The differences in prevalence rates across these studies may be attributed to several factors, including variations in study design, participant and other environmental factors. These variations may influence the prevalence of vitamin D deficiency among hormonal contraceptive users in different populations. Our study was also conducted in dry season considering high exposure to sunlight unlike other studies which did not consider these factors.
On the current study the the odds of having Vitamin D deficiency were 2.99 times higher among participants who attained higher education. This may be due to the reason that Individuals with higher education levels may have sedentary lifestyles or spend more time indoors due to their occupations, which can limit their exposure to sunlight, the primary source of Vitamin D. Lack of sunlight exposure can contribute to Vitamin D deficiency55. Conversely, Participant who never eat fish shown to have increased odds of having Vitamin D deficiency by 12.36 times. This may elucidate by the fact that Fish, especially fatty fish like salmon, tuna, and mackerel, are rich sources of Vitamin D. By never consuming fish, individuals are missing out on a significant dietary source of Vitamin D, which can lead to deficiency56. On the other hand, Participants who reported to have never been used vitamin D Supplements was observed to have 4.1 times higher odds of having Vitamin D deficiency. This finding is in line with previous report from United state10 which state consumption of vitamin D containing supplements was positively associated found a positive association between consumption of vitamin D-containing supplements and serum total Vitamin D levels supports the current finding. Using vitamin D supplements can increase the amount of Vitamin D in the bloodstream, thereby improving total serum Vitamin D levels and reducing the risk of deficiency57.
On the present study, the utilization of combined oral contraceptives (COC) shown to reduce the risk of vitamin D deficiency by 90% (AOR 0.10 95% CI (0.02–0.36, p 0.000); this is agreement with previous reports from studies conducted among COC users31,33,38,43,46,47,49. This finding may be explained the presence of higher concentration of estrogen in a combination of progestin in COC. Although the exact molecular mechanism is still unclear Estrogen, has been shown to potentially play a role in enhancing the synthesis and metabolism of Vitamin D in the body and thus leading to higher levels of Vitamin D in individuals who use COCs that contain estrogen34.
The odds of having Vitamin D deficiency shown to be lower among participants with normal serum calcium concentration (AOR 0.50 (0.26–0.97)) and Hypercalcemic state with AOR 0.31 (0.10–0.99); this is in line with previous reports38,49. Vitamin D plays a crucial role in regulating calcium levels in the body. It helps in the absorption of calcium from the intestines and maintains calcium levels in the blood within a narrow range. If there is a deficiency of vitamin D, it can lead to decreased calcium absorption and levels in the blood, potentially causing weak and brittle bones49,58.
Conclusion
On the current study we found the use of hormonal contraceptive use may affect Vitamin D levels. The mean serum Vitamin D levels were higher among combined oral contraceptive (COC) users compared to Norplant and DMPA users and non-users. The prevalence of Vitamin D deficiency was lower among hormonal contraceptive users, particularly those using COCs, compared to non-users. Factors such as higher education, lack of fish consumption, and not using vitamin D supplements were associated with higher odds of Vitamin D deficiency. Additionally, individuals with normal and hypercalcemia state had lower odds of Vitamin D deficiency.
Recommendations
Based on the findings of this study, it is recommended that healthcare providers consider monitoring Vitamin D levels in individuals using hormonal contraceptives, especially those using Norplant and DMPA. Education and counseling on the importance of consuming fish and taking vitamin D supplements should be provided to individuals at risk of Vitamin D deficiency. Further research is needed to explore the mechanisms underlying the relationship between hormonal contraceptive use and Vitamin D levels, as well as the potential benefits of COCs in reducing the risk of Vitamin D deficiency. Additionally, public health interventions should be developed to improve awareness and access to Vitamin D supplementation for individuals at risk of deficiency.
Strength and limitation of the study
Strength of the study
It is the first study in Ethiopia to assess serum vitamin D level of Hormonal contraceptive users and among a few in worldwide. The Comparative study design nature of the current study and by comparing the Vitamin D levels and prevalence of deficiency among hormonal contraceptive users and non-users, we were able to identify some potential relationship. Large sample size: The study included 324 participants, which increases the generalizability of the findings. The current study also considered factors such as sociodemographic, behavioral and clinical as well as diet characteristics and calcium levels in the analysis, which strengthens the validity of the findings. All the laboratory analysis is performed in referral hospital laboratory which increase the quality of the data obtained.
Limitation of the study
Even though we incorporate a reference control group on the current study; the study design limits the ability to establish causality between hormonal contraceptive use and Vitamin D levels. We also use a self-reported data and the reliance on self-reported data for factors such as fish consumption and supplement use may introduce recall bias. The current study also conducted purposefully in a limited geographic scope. Due to unavailability of these assays serum ALP was analyzed without identifying bones specific ALP. The study was conducted in a specific region (Gondar Town), which may limit the generalizability of the findings to other populations.
Data availability
Most of data generated or analyzed during this study are included in this published article; and Addition research data set associated with a paper is available, can be accessed in contact with corresponding author.
Abbreviations
- 25(OH) D):
-
25-hydroxycholecalciferol
- ALP:
-
Alkaline phosphate
- ANOVA:
-
Analysis of variance
- BMI:
-
Body Mass Index
- BP:
-
Blood pressure
- CI:
-
Confidence interval
- COC:
-
Combined oral contraceptives
- CPR:
-
Contraceptive prevalence rate
- DBP:
-
Diastolic blood pressure
- DMPA:
-
Depo-medroxyprogesterone acetate
- EE2:
-
Ethinylestradiol
- ETB:
-
Ethiopian Birr
- HCs:
-
Hormonal contraceptives
- PCOS:
-
Polycystic ovary syndrome
- SBP:
-
Systolic blood pressure
- UOGCSH:
-
University of Gondar Comprehensive Specialized Hospital
- VDBP:
-
Vitamin D binding proteins
- WHR:
-
Waist to Hip ratio
References
Cashman, K. D. Vitamin D deficiency: Defining, prevalence, causes, and strategies of addressing. Calcif. Tissue Int.106(1), 14–29 (2020).
Lips, P. Vitamin D physiology. Prog. Biophys. Mol. Biol.92(1), 4–8 (2006).
Wacker, M., Holick, M. F. & Sunlight, D. A global perspective for health. Dermato-endocrinology. 5(1), 51–108 (2013).
Schwalfenberg, G. K. A review of the critical role of vitamin D in the functioning of the immune system and the clinical implications of vitamin D deficiency. Mol. Nutr. Food Res.55(1), 96–108 (2011).
Condoleo, V. et al. Role of vitamin D in cardiovascular diseases. Endocrines. 2(4), 417–426 (2021).
Forrest, K. Y. & Stuhldreher, W. L. Prevalence and correlates of vitamin D deficiency in US adults. Nutr. Res.31(1), 48–54 (2011).
Roth, D. E. et al. Global Prevalence and Disease Burden of Vitamin D Deficiency: A Roadmap for Action in low-and middle-income Countries (Wiley Online Library, 2018). Report No 0077–8923.
de Haan, K., Groeneveld, A. J., de Geus, H. R., Egal, M. & Struijs, A. Vitamin D deficiency as a risk factor for infection, sepsis and mortality in the critically ill: Systematic review and meta-analysis. Crit. Care18, 1–8 (2014).
Grandi, N. C., Breitling, L. P. & Brenner, H. Vitamin D and cardiovascular disease: Systematic review and meta-analysis of prospective studies. Prev. Med.51(3–4), 228–233 (2010).
Harmon, Q. E., Umbach, D. M. & Baird, D. D. Use of estrogen-containing contraception is associated with increased concentrations of 25-hydroxy vitamin D. J. Clin. Endocrinol. Metabolism. 101(9), 3370–3377 (2016).
Cui, A. et al. Global and regional prevalence of vitamin D deficiency in population-based studies from 2000 to 2022: a pooled analysis of 7.9 million participants. Front. Nutr.10, 1070808 (2023).
Bekele, D. et al. Contraceptive prevalence rate and associated factors among reproductive age women in four emerging regions of Ethiopia: A mixed method study. Contracept. Reproduct. Med.6(1), 18 (2021).
Ciebiera, M. et al. Vitamin D serum levels in women using contraception containing drospirenone–a preliminary study. Arch. Med. Sci.15(2), 554–557 (2019).
Wiegratz, I. & Thaler, C. J. Hormonal contraception—what kind, when, and for whom? Deutsches Ärzteblatt Int.108(28–29), 495 (2011).
Doran, A. Self-Confidence and Hormonal Contraceptive Use. (2023).
Stanczyk, F. Z., Archer, D. F. & Bhavnani, B. R. Ethinyl estradiol and 17β-estradiol in combined oral contraceptives: Pharmacokinetics, pharmacodynamics and risk assessment. Contraception87(6), 706–727 (2013).
Keam, S. J. & Wagstaff, A. J. Ethinylestradiol/drospirenone: A review of its use as an oral contraceptive. Treat. Endocrinol.2(1), 49–70 (2003).
Amiri, M. et al. Effects of oral contraceptives on metabolic profile in women with polycystic ovary syndrome: A meta-analysis comparing products containing cyproterone acetate with third generation progestins. Metab. Clin. Exp.73, 22–35 (2017).
Edwards, M. & Can, A. S. Progestin. StatPearls [Internet]. (2021).
Graham, S. & Fraser, I. S. The progestogen-only mini-pill. Contraception. 26(4), 373–388 (1982).
Jacobstein, R. & Polis, C. B. Progestin-only contraception: Injectables and implants. Best Pract. Res. Clin. Obstet. Gynecol.28(6), 795–806 (2014).
Haakenstad, A. et al. Measuring contraceptive method mix, prevalence, and demand satisfied by age and marital status in 204 countries and territories, 1970–2019: A systematic analysis for the global burden of Disease Study 2019. Lancet400(10348), 295–327 (2022).
Wang, M. et al. Contraceptive and reproductive health practices of unmarried women globally, 1999 to 2018: Systematic review and meta-analysis. Medicine99(49), e23368 (2020).
UN. World Fertility and Family Planning 2020: Highlights. United Nations, Department of Economic and Social Affairs, Population… (2020).
Nations, U. Contraceptive use by method (United Nations, Department of Economic and Social Affairs New York, 2019).
Need, A. G. et al. Vitamin D metabolites and calcium absorption in severe vitamin D deficiency. J. Bone Miner. Res.23(11), 1859–1863 (2008).
Kalantar-Zadeh, K. et al. Kidney bone disease and mortality in CKD: Revisiting the role of vitamin D, calcimimetics, alkaline phosphatase, and minerals. Kidney Int.78, S10–S21 (2010).
Need, A. G. Bone resorption markers in vitamin D insufficiency. Clin. Chim. Acta. 368(1–2), 48–52 (2006).
Ladhani, S., Srinivasan, L., Buchanan, C. & Allgrove, J. Presentation of vitamin D deficiency. Arch. Dis. Child.89(8), 781–784 (2004).
García-Bailo, B. et al. Plasma 25-hydroxyvitamin D, hormonal contraceptive use, and the plasma proteome in caucasian, east Asian, and south Asian young adults. J. Proteome Res.12(4), 1797–1807 (2013).
García-Bailo, B., Josse, A. R., Jamnik, J., Badawi, A. & El-Sohemy, A. Positive association between 25-hydroxyvitamin D and C-reactive protein is confounded by hormonal contraceptive use. J. Women’s Health. 22(5), 417–425 (2013).
Wyon, M. A. et al. The influence of hormonal contraception on vitamin D supplementation on serum 25 (OH) D 3 status in premenopausal women: A prospective double-blind placebo random controlled trial. J. Endocrinol. Metabolism7(4), 117–121 (2017).
Stanczyk, F. Z. et al. Effect of oral contraceptives on total and bioavailable 25-hydroxyvitamin D. J. Steroid Biochem. Mol. Biol.211, 105879 (2021).
Pilz, S. et al. Hormonal contraceptive use is associated with higher total but unaltered free 25-hydroxyvitamin D serum concentrations. J. Clin. Endocrinol. Metabolism. 103(6), 2385–2391 (2018).
Møller, U. K. et al. Increased plasma concentrations of vitamin D metabolites and vitamin D binding protein in women using hormonal contraceptives: A cross-sectional study. Nutrients5(9), 3470–3480 (2013).
Harris, S. S. & Dawson-Hughes, B. The association of oral contraceptive use with plasma 25-hydroxyvitamin D levels. J. Am. Coll. Nutr.17(3), 282–284 (1998).
Namli Kalem, M. et al. Effect of combined oral contraceptive use on serum 25-hydroxy vitamin D levels and ultrasound parameters in patients with polycystic ovary syndrome. Gynecol. Endocrinol.32(4), 281–284 (2016).
Abd Kadim, A. & Mohammed, S. H. Effect of oral contraceptive pills on levels of calcium and Vitamin D in women in Al-Najaf Province. Indian J. Forensic Med. Toxicol.14(4), 739–743 (2020).
Teferra, A. S. & Wondifraw, A. A. Determinants of long acting contraceptive use among reproductive age women in Ethiopia: Evidence from EDHS 2011. (2015).
Bouillon, R., Norman, A. W. & Lips, P. Vitamin D deficiency. N. Engl. J. Med.357(19), 1980–1981 (2007).
WHO, Physical activity: & WHO. (2020). https://www.who.int/news-room/fact-sheets/detail/physical-activity
Adami, S. et al. 25-hydroxy vitamin D levels in healthy premenopausal women: Association with bone turnover markers and bone mineral density. Bone45(3), 423–426 (2009).
Öberg, J. et al. 100 YEARS OF VITAMIN D: Combined hormonal contraceptives and vitamin D metabolism in adolescent girls. Endocr. Connect.11(3), (2022).
Wyon, M. A. et al. Estrogen contraception pill doubled the response to 1,000 IU of vitamin D–RCT Sept 2017. RCT (2017).
Schreurs, W. & van Rijn, H. Serum 25-hydroxycholecalciferol levels in women using oral contraceptives. Contraception23(4), 399–406 (1981).
Brinker, K. A. Oral contraceptive use and vitamin D status among women ages 15–44 in the US: A cross-sectional study (Emory University, 2012).
Huff, L. L. et al. Oral contraceptive pills increase circulating 25-Hydroxy-vitamin D concentrations in women who are Lactating. Am. J. Perinatol.41(S 01), e2759–e66 (2024).
García-Bailo, B., Karmali, M., Badawi, A. & El-Sohemy, A. Plasma 25-hydroxyvitamin D, hormonal contraceptive use, and cardiometabolic disease risk in an ethnically diverse population of young adults. J. Am. Coll. Nutr.32(5), 296–306 (2013).
Donangelo, C. M., Cornes, R., Sintes, C. & Bezerra, F. F. Combined oral contraceptives: Association with Serum 25-Hydroxyvitamin D and calcium and bone homeostasis (Journal of Women’s Health, 2024).
Xie, Z., Santora, A. C., Shapses, S. A. & Wang, X. Vitamin D binding protein and vitamin D levels 638263 (Hindawi Publishing Corporation, 2014).
Oleröd, G., Hultén, L. M., Hammarsten, O. & Klingberg, E. The variation in free 25-hydroxy vitamin D and vitamin D-binding protein with season and vitamin D status. Endocr. Connect.6(2), 111–120 (2017).
Huff, L. L. et al.Oral contraceptive pills increase circulating 25-Hydroxy-Vitamin D concentrations in women who are lactating. Am. J. Perinatol. (2023).
Morgante, G. et al. PCOS physiopathology and vitamin D deficiency: Biological insights and perspectives for treatment. J. Clin. Med.11(15), 4509 (2022).
Haider, Z. & D’Souza, R. Non–contraceptive benefits and risks of contraception. Best Pract. Res. Clin. Obstet. Gynecol.23(2), 249–262 (2009).
Arias-Palencia, N. M. et al. Levels and patterns of objectively assessed physical activity and compliance with different public health guidelines in university students. PLoS ONE10(11), e0141977 (2015).
Macdonald, H. M. Contributions of sunlight and diet to vitamin D status. Calcif. Tissue Int.92, 163–176 (2013).
Bouillon, R. et al. The health effects of vitamin D supplementation: Evidence from human studies. Nat. Rev. Endocrinol.18(2), 96–110 (2022).
Ceglia, L. & Harris, S. S. Vitamin D and its role in skeletal muscle. Calcif. Tissue Int.92, 151–162 (2013).
Acknowledgements
We would also like to express our gratitude to the participants for being voluntary to participate in the study and generously shared their time and experiences for this study. We would like to acknowledge UOGCSH Clinical chemistry laboratory staffs for their support and laboratory analysis that made this research possible. Special thanks to data collectors for their assistance with data collection.
Author information
Authors and Affiliations
Contributions
E.C. conceptualized and designed the study, including the research question, study design, and data collection methods. E.C., B.B.T. and D.M.B. was responsible for overseeing the data collection process, ensuring adherence to study protocols, and verifying data accuracy. E.C., B.B.T., D.M. and A.M. conducted the statistical analysis, interpreted the results, and contributed to the writing of the methodology and results sections of the manuscript; and assisted in the interpretation of the results, provided critical feedback on the manuscript drafts, and revised the final version of the manuscript. E.C., M.T., Z.Y., A.A.A. contributed to the literature review, study background, and discussion sections of the manuscript. E.C., B.B.T., D.M. and N.C. was involved in the study conception and design, data analysis, and interpretation of the findings, as well as providing overall supervision of the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chane, E., Teketlew, B.B., Berta, D.M. et al. A comparative study of hormonal contraceptive use and vitamin D levels at Gondar Town 2023. Sci Rep 14, 22162 (2024). https://doi.org/10.1038/s41598-024-73014-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-73014-6