Abstract
Kaempferol (KMF) possesses notable anti-tumor bioactivity, which indicates its promising action in the therapy of gynecologic cancers. Here, we examined the therapeutic potential of the naturally occurring flavonoid kaempferol coated on niosome nanoparticles (NPs) and its impact on the breast cancer cell line MCF-7. Niosome NPs containing KMF were prepared by thin-layer hydration. The generated niosome/KMF NPs cytotoxicity on MCF-7 and MCF-10 cell lines were assessed by MTT assay. The physicochemical properties of the niosome/KMF NPs were characterized by SEM, DLS zeta potential, and FTIR. Flow cytometry was used to quantify primary and secondary apoptosis, necrosis and cell cycle arrest. Finally, the expression of apoptosis (Bax and caspase 3) and metastasis genes (ITGA5 and MMP2) was analyzed by Real-time PCR. A Scratch test was performed to investigate the anti-metastatic effect of synthesized nanoparticles. The results showed that the synthesized niosome/KMF NPs have a diameter of 500 nm, a zeta potential of 33.9 mV and a PDI of 0.169. The FTIR spectrum of niosome NPs containing KMF showed distinct peaks in the range of 600–3400 cm− 1 belonging to different components. The results of the MTT assay showed that treatment of the MCF-7 cell line with a concentration of 0.0873 µMol of niosome NPs containing KMF resulted in the death of 50% of the cells. Niosome/KMF NPs caused 64% apoptosis in MCF-7 cells. Real-time PCR results showed a 2.95- and 2.75-fold increase in Bax and caspase 3 gene expression compared to the control group (p < 0.001). After 72 h of treatment with niosome NPs containing KMF, ITGA5 and MMP2 gene expression decreased by 0.58- and 0.53-fold, respectively (p < 0.001). In summary, KMF-loaded niosome NPs efficiently induced apoptosis and inhibited metastasis-related gene expression in MCF-7 cells, exhibiting notable anti-cancer activity.
Similar content being viewed by others
Introduction
Cancer is one of the most common causes of death worldwide. In 2020 alone, 19.3 million new cases and 10 million deaths are attributed to cancer1. Breast cancer (BC) is the most commonly diagnosed malignancy in women worldwide. In 2020, an estimated 2.3 million new cases of BC were detected, resulting in 685 thousand deaths; this corresponds to one in six cancer-related deaths2. In addition, BC is a heterogeneous cancer subgroup categorized into four main types: Basal-like BC, human epidermal growth factor receptor 2 (HER2)-enriched BC, luminal A BA and luminal B3. Conventional cancer therapies such as chemotherapy, radiotherapy and surgery have several disadvantages, such as widespread toxicity, ineffective distribution of drugs in the body and the development of drug resistance. The treatment of BC usually involves multidisciplinary procedures, such as systemic therapy (chemotherapy, endocrine therapy, targeted HER2 therapy, etc.) and local therapy (surgical treatment and radiation)4. The choice of treatment approach depends on the type and stage of the disease in breast cancer. While improvements in these treatment strategies have increased the chances of cure for approximately 70–80% of patients with early breast cancer, metastatic breast cancer is still considered incurable5. In addition, standard anticancer drugs have many drawbacks, including their inability to specifically target tumors, low solubility, high toxicity, susceptibility to multidrug resistance, short half-life, and poor chemical stability. These limitations generally lead to unsatisfactory therapeutic efficacy. Therefore, it is necessary to conduct more comprehensive research to explore more efficient and less harmful therapeutic approaches to provide customized therapy for BC6.
Natural chemicals are biologically active molecules derived from various sources such as plants, fungi and marine animals. Due to their potential therapeutic effects against cancer, these chemicals have attracted increasing attention in recent years. These compounds have been shown to have antioxidant, anti-inflammatory and anti-cancer properties, making them attractive options for cancer therapy7,8. In addition, natural chemicals have been shown to exhibit selectivity towards cancer cells, minimizing the negative effects on healthy cells. Although natural substances are very promising for therapy, they also have some limitations that can impair their efficacy. One of the main disadvantages of these compounds is that they are poorly soluble in water, which makes their transport to the tumor cells difficult. Another disadvantage is their low bioavailability, which means that they are rapidly metabolized and excreted from the bloodstream before they can reach their target9. Kaempferol (KMF) with a molecular weight of 286.24 g/mol, also known as 3,4′,5,7-tetrahydroxyflavone, is a yellowish chemical that belongs to the flavonoid class. It is characterized by the presence of four hydroxy groups located at positions − 3, -4′, -5 and − 710. It can be found in many plant components such as leaves, flowers, seeds, vegetables and fruits. KMF and its glycosides have a number of beneficial properties, including antidiabetic, anticancer, neuroprotective, antioxidant, antitumor, anti-inflammatory, antimicrobial and cardioprotective effects11. The anti-cancer properties of KMF and its numerous applications have attracted a great deal of attention from researchers in the field of cancer therapy. The development of cancer cells is often inhibited by promoting apoptosis, thereby preventing their proliferation12. In fact, KMF inhibits the proliferation of malignant cells by arresting the cell cycle at the G2-M stage, promoting apoptosis and suppressing signaling pathways such as phosphatidylinositol 3-kinase-protein kinase B (PI3K-PKB)13. However, the low solubility and poor stability of this compound in water and body fluids have limited the use of this potent compound as a therapeutic strategy. The combined use of natural products and nanomaterials has significant potential for the treatment of cancer. By embedding natural compounds in nanoparticles, their bioavailability and pharmacokinetics can be improved, leading to higher therapeutic efficacy. In addition, nano-delivery techniques have the potential to reduce the negative effects of natural chemicals on normal cells while increasing their ability to target cancer cells14.
Therefore, there is a growing interest in the development of novel cancer therapies that are more effective, less toxic and easier to accept15. Nanotechnology has become a promising topic in recent years, as it has enormous potential in the development of new methods of drug delivery in cancer therapy. Nanotechnology in medicine has many advantages, such as improved treatment efficacy, precise drug distribution, minimization of adverse effects and better patient outcomes16. Nanobiotechnology, an emerging and specialized field of study, has successfully produced various nanostructures, including nanoparticles, through the application of protocols, methods and techniques from other scientific disciplines17. Due to their different attributes and physiological properties, these nanostructures or nanocarriers have facilitated the development of extensive therapeutic strategies and techniques, including drug delivery systems for gene therapies, tissue regeneration, tissue engineering and immunotherapies, as well as against microbial infections and cancers18. In addition to advances in nanomaterials, scientists are now focusing on the development of novel nanosystems that regulate the release of various molecules with biological properties. Nanocarriers and novel pharmacological formulations have a significant impact on improving the bioavailability of drugs or natural substances by concentrating them at the intended site19. Vesicular nanoparticles (NPs), including liposomes, transferosomes, ethosomes, niosomes, and polymeric micelles, are considered to be one of the most exciting methods for the delivery of nanodrugs. These NPs have the ability to transport both hydrophobic and hydrophilic therapeutic substances20. They have the ability to protect the drug from degradation, improve efficacy and prevent adverse effects on non-targeted organs. Niosomes are a type of vesicular system that has a variety of uses21. Niosomes are lipid-based NPs formed by the self-assembly of nonionic surfactants and other lipid components in a spherical bilayer form22. The bilayer structure of these particles allows for versatility, as hydrophilic drugs can be entrapped in the central aqueous compartment, while hydrophobic drugs can be entrapped in the lipid membrane. The confirmation of this structure, makes niosomes a very effective drug delivery mechanism23. Various non-ionic surfactants can be used for the preparation of niosomes, including Span, Tween and Brij surfactants. To increase the rigidity and reduce the ability of substances to penetrate the bilayer of the prepared niosomes, cholesterol can be added24. Niosome NPs exhibit greater stability compared to liposomes and can encapsulate both hydrophilic and lipophilic compounds with biological properties. These carriers are not capable of triggering an immune response, are compatible with living organisms, can degrade naturally, are inexpensive and have a long shelf life24. The aim of producing niosomes for drug delivery is to achieve consistent and controllable particle size distribution and controlled release of the loaded drug over extended periods of time. Due to their numerous advantages over conventional nano systems, niosomes have considerable potential as nanocarriers25.
Therefore, the aim of this research is to encapsulate kaempferol into niosome nanostructures to improve its solubility, achieve controlled and gradual release, and investigate its anti-cancer and anti-metastatic properties against MCF-7 cell line. MCF-7 cells are useful for in vitro breast studies because they retained several ideal characteristics particular to mammary epithelium.
Materials and methods
Chemicals and reagents
Cholesterol (CAS 57-88-5), and ethanol (CAS 64-17-5) were acquired from Merck Co. in Germany. Span 80, manufactured by Samchun Pure Chemical Co., Ltd. in Korea (CAS 00S1172), was employed in addition to tween 80 and dichloromethane, supplied by Sharlua and Sharlab S.L (CAS: 9005-65-6). in Spain, respectively. The substances such as agarose, ethidium bromide, ethylene diamine tetra acetic acid (EDTA), polyvinyl alcohol, MTT kit, annexin V FITC, and Trypan blue were acquired from Sigma-Aldrich, USA. In the present study, kaempferol with a purity of more than 90%, melting point of 277 °C, and molecular weight of 286.24 g/mol was purchased from Sigma-Aldrich, USA with catalog number K0133-50MG.
MCF-7 and MCF-10 cell lines were obtained from the Pasteur Institute of Iran (Iran, Tehran) with accession codes ATCC HTB-22 and CRL-10,317-ATCC, respectively. Also, Dulbecco’s Modified Eagle Medium (DMEM), fetal bovine serum (FBS), horse serum, and streptomycin were obtained from Gibco, USA. Trypsin-EDTA was purchased from DACell, Iran. All the other compounds utilized during the investigation were procured from local sources and met the standards of analytical grade. All tests in this study were repeated 3 times.
Preparation of niosomes/kmf NPs
Niosome NPs were prepared by the thin-layer hydration method. To prepare niosome NPs, 2 ml of Tween 80, 1 ml of Span 80 (Merck, Germany) and 1 mg of cholesterol were dissolved in 20 ml of chloroform solution. Then the resulting solution was thoroughly stirred until the components were completely dissolved. The ratio of chloroform to methanol was 2:1 to obtain the required lipid content. The resulting solution was then stirred vigorously until all components were completely dissolved. A rotary device evaporated the solvent at a temperature of 60 °C and a speed of 150 rpm under vacuum conditions. The process of solvent evaporation took 30 min. After this process, a lipid layer forms as a thin coating on the surface of the balloon. During the hydration phase, a solution was prepared by first dissolving 40 mg of KMF in a minimal volume of ethanol (1–2 ml) to ensure complete dissolution. This ethanolic solution of KMF was then added dropwise to 10 ml of phosphate buffer with a pH of 7.4 under stirring. This final hydro-alcoholic solution, containing 40 mg/100 ml of KMF, was then used to hydrate the previously formed lipid film. Film was rotated at 60 °C for thirty minutes to ensure sufficient hydration. After hydration, the particles were sonicated (Model 150 UPS, Iranian Technology Company Research Nasir, Iran) for 15 min to reduce the particle size21.
Characterization of KMF-NPs
Evaluation of morphology and polydispersity index
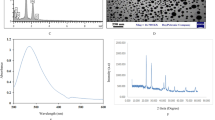
The surface morphology of the KMF-loaded niosomes was observed using a scanning electron microscope (SEM, Hitachi Model 3400 N, Japan). 15 µl sample of the niosomal suspension was placed on a glass slide and air dried at ambient temperature. Before scanning the samples in the SEM, they were fixed on a die and coated with a layer of gold to exclude charging effects. The SEM was operated with an accelerating voltage of 20 KV. The KMF-loaded niosomes were diluted with distilled water at a ratio of 1:100. The droplet size and polydispersity index were measured using the Zeta Sizer instrument (Malvern Instruments, U.K). The measurements were performed at a light scattering angle of 90 degrees26. Standard deviation against particle size and Zeta potential by Zetasizer was Zeta: 33.9 ± 0.0002 and Sem: 4.438 ± 500 nm.
Fourier transform infrared spectrum (FTIR)
FTIR (ALPHA II, Bruker, Germany) was performed to determine the interaction between drug and excipient and to investigate possible interactions between the substances used in the formulation. FTIR was used to measure the absorption peaks of both free niosomes and KMF-loaded niosome NPs. The samples were formed in a KBr disk with a ratio of 2 mg KMF NPs to 200 mg KBr. The preparation was carried out using a hydrostatic press with a force of 275790.292 Pa for 5 min. The resulting spectra were obtained over the entire wavelength range from 4000 to 400 cm− 127.
Cell culture and in vitro cytotoxicity investigation
MCF-7 cells were grown in DMEM-F12 complete medium containing 10% FBS, 100 units/ml penicillin and 100 µg/ml streptomycin and incubated at 37 °C and 5% CO2. In addition, MCF-10 cultured under the following conditions. The culture medium was changed twice a week until the cells had reached 70–80% confluence. The cells were harvested by placing them in a 0.25% trypsin-EDTA solution. After centrifugation, the cells were counted and immersed in PBS for the subsequent experiment. The inhibitory effect of KMF-loaded niosome NPs on BC cells was determined using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) method, and the half-maximal inhibitory concentration (IC50) was calculated. IC50 refers to the concentration of a drug that inhibits the growth of cancer cells by 50%30. It is often used to evaluate the therapeutic potential of a treatment in the laboratory. In this study, the effect of three factors was examined: free niosomes, free KMF and KMF-loaded niosome NPs. “Free niosomes” indicate empty niosomal nanoparticles without any encapsulated kaempferol was used as the negative control. “Free KMF” indicates kaempferol in its unencapsulated form, dissolved in the cell culture medium at the same concentrations used in the kaempferol-loaded niosomes. The “kaempferol-loaded NPs” are the niosomal nanoparticles containing kaempferol. A total of 104 cells were grown in each well of 96-well plates and exposed to free niosomes, free KMF and KMF-loaded niosome NPs at concentrations of 40, 20, 10, 5, 2.5, 1.25 and 0.625 mg/100 ml for 24, 48 and 72 h. The molar concentration used was equal to 1.39, 0.69, 0.34, 0.17, 0.0873, 0.0436 and 0.0218 µMol (Supplementary Table 1). At the end of the incubation period, the culture medium was removed. Subsequently, 100 µl of MTT (Sigma- Aldrich, USA) solution (0.5 mg/ml in PBS) was added to each well and the plate was incubated at a temperature of 37 °C for 3 h. The MTT reagent was extracted, and 100 µl of DMSO (Sigma- Aldrich, USA) was added to disperse the formazan crystals. The absorbance of each sample was measured at an optical density (OD) of 570 nm using an ELISA reader (ChroMate, The Netherlands)28. Cell proliferation was quantified using the following formula.
ODC refers to the optical density of untreated cells, while ODT refers to the optical density of treated cells. Cell survival results were analyzed and IC50 values were measured based on the dose-response pattern29.
Flow cytometry and cell apoptosis measurements
Apoptosis analysis of MCF-7 cells was performed using an Annexin V-FITC apoptosis detection kit from BD Biosciences (Franklin Lakes, NJ, USA). MCF-7 cells were plated in 6-well plates at a concentration of 104 cells per well. They were then treated for 72 h with the IC50 concentration of free KMF, free niosome and KMF-loaded NPs. After rinsing twice with cold, sterile PBS (pH 7.4), a suspension of 104 MCF-7 cells per well was prepared in a 6-well plate using the 1X binding buffer provided by the reagent. Cells were processed according to the manufacturer’s instructions by adding precise amounts of Annexin V-FITC (green fluorescence) and propidium iodide (red fluorescence). The incubation lasted 10 min under normal room temperature conditions. The cell samples were transferred to a flow cytometric tube and analyzed by flow cytometry (Spectrum Two, Perkin Elmer, USA) in the final phase30.
Investigation of cell cycle arrest
MCF-7 cell proliferation was assessed by propidium iodide (PI) staining. The DNA content is used to determine the phase of the cell cycle, as the amount of DNA present is directly proportional to the binding of PI to the DNA. Cells were cultured in complete medium in 6-well plates at a density of 1 × 106 cells per well. After overnight incubation and washing three times with PBS, cells were treated with IC50 concentration of free KMF, free niosomes and KMF-loaded NPs in complete medium for 72 h. Cells were then harvested and stored in 70% chilled ethanol at 4 °C for one night. Cells were then exposed to 450 µl PI solution (including RNase) without light for 20 min at room temperature. Finally, the cells were analyzed by flow cytometry. The experiments were reproduced three times31.
Assessment of cell migration (scratch test)
In order to assess the anti-metastatic activity of the treatments administered, a scratch assay was performed according to a standard protocol. In brief, MCF-7 breast cancer cells were seeded at 100,000 cells/well of 6-well plates, a density that would ensure confluent growth after overnight incubation at 37 °C and 5% CO2. Once complete confluence was reached, a homogeneous linear scratch was made in each well with the help of a 10 µl pipette tip. The culture medium was removed, and the cell layers were washed two times with phosphate-buffered saline to remove any non-adherent cells and debris. The cells were then exposed to IC50 concentration of kaempferol-loaded NPs, free niosome and free KMP (at 0.0873 µMol concentration). Migration of cells into the scratched area was seen and noted after 24 h of incubation using an inverted microscope (KERN, Germany) at 10x magnification. Photographs at this time point were then compared to ascertain the extent of wound closure, reflective of the migratory capacity of the treated cells32.
Real-time PCR
In order to examine the effect of kaempferol-loaded niosomes on gene expression, MCF-7 and MCF-10 cell lines were treated with a dose of 0.0873 µMol of the NPs, which was found to be the IC50 dose on MCF-7 cells. After incubation times of 24, 48, and 72 h, quantitative real-time PCR (qRT-PCR) was performed to assess the mRNA expression levels of MMP2, Caspase 3, Bax, and ITGA5. This analysis was performed under different IC50 concentrations of free KMF, free niosome and KMF-loaded NPs. GAPDH was used as a reference gene. Total RNA was extracted using TRIzol reagent (Thermo Fisher Scientific, Waltham, MA, USA), and cDNA synthesis was performed using a cDNA synthesis kit (Parstous, Iran). The thermal protocol for Real-time PCR (Ausdiagnostic, Australia) consisted of an initial step at 95 °C for 5 min, followed by 35 cycles of 95 °C for 30 s, 54 °C for 30 s, and 72 °C for 30 s. PCR was performed using 12.5 µl of SYBR-PCR master mix (Amplicon, Denmark) with each primer at a concentration of 100 nM and 1 µl of RT product brought to a final volume of 25 µl with water. The primer sequences for the indicated genes are listed separately for the forward and reverse orientations in Table 1. The 2–ΔΔCT method was used to determine the fold changes in the control group. The studies were performed on three separate occasions33.
Statistical analysis
GraphPad Prism 5.0 was used for statistical analysis, with results provided as mean ± standard deviation and mean ± SD. The data were examined using a one-way analysis of variance (ANOVA). A T-test with a p-value of 0.05 was employed to compare group pairings.
Results
Investigating the physicochemical properties of synthesized NPs
In this research, SEM was used to evaluate the morphological characteristics and size of the niosome NPs containing KMF. Figure 1 shows the KMF-containing NPs, which have a uniform morphology and smooth surface. SEM analysis showed that the average diameter of the synthesized NPs was less than 500 nm. The hydrodynamic diameter of KMF-loaded niosome NPs determined by DLS is 100 to 500 nm (Table 2). The diameter of the KMF-loaded niosome NPs determined by SEM was smaller than that determined by dynamic light scattering (DLS). This change may be due to drying of the samples prior to SEM imaging. SEM images usually provide morphological information about nanoparticles in dry form (measuring the exact diameter of each NP), while DLS indicates the hydrodynamic diameter, which includes the size of the core and all adsorbed molecules (water and ions) on the surface. Niosome NPs containing KMF have a zeta potential of 33.9 mV (Table 2) and a PDI of 0.165.
SEM-based morphological characterization of niosome/KMF nanoparticles. SEM imaging findings demonstrate that niosome/KMF nanoparticles have favorable properties for the cellular transport of KMF. The small size, spherical shape, and smooth surface indicate a well-designed nanoparticle delivery system.
FTIR analysis
FTIR analysis was used to confirm the presence of KMF in niosome NPs. Table 3 presents the FTIR spectra of the free niosome and niosome nanoparticles with KMF. The FTIR spectrum showed distinct peaks in the range of 400–3500 cm− 1. The FTIR spectrum of niosome NPs containing KMF shows distinct peaks in the 600–3400 cm− 1 range belonging to different components of the niosome system. The detected peak at 3400.39 cm− 1 was attributed to OH stretching of cholesterol and span 80. The peak at 1569.68 cm− 1 is related to the symmetric and asymmetric stretching of carbon in the CH3 group, which can be seen in the FTIR pattern of the niosome after KMF loading. In the spectrum of the niosome/KMF NPs, C = C stretching of the cholesterol structure and C = O and C-O stretching of the KMF ester groups were observed at 1414.01, 1059.44 and 1059.51 cm− 1, respectively.
Results of MTT assay
Cytotoxicity of KMF-loaded niosome NPs on MCF-7 breast cancer cells was evaluated using the MTT assay. The IC50 concentration, i.e., the dose at which 50% of the cancer cells were inhibited, was determined by treating MCF-7 cells with various concentrations (1.39, 0.69, 0.34, 0.17, 0.0873, 0.0436, and 0.0218 µMol) of free niosomes, free KMF, and KMF-loaded niosomes for 24 h. The results indicated that KMF-loaded niosomes exerted a concentration-dependent cytotoxic effect, with 0.0873 µMol reducing MCF-7 cell viability by 50%. Therefore, the IC50 value was established at 0.0873 µMol (Fig. 2A). Interestingly, neither free KMF nor unencapsulated niosomes exhibited significant cytotoxic effects on MCF-7 cells, confirming the importance of KMF encapsulation for anticancer activity.
Following IC50 determination, further experiments assessed the cytotoxicity of free KMF, free niosomes, and KMF-loaded niosomes on MCF-7 and MCF-10 cells over 24, 48, and 72 h (Fig. 2B and C). Free KMF and free niosomes did not show significant cytotoxic effects on either cell line (p > 0.05). However, the IC50 concentration of KMF-loaded niosomes caused a time-dependent increase in cytotoxicity in MCF-7 cells, with viability declining from 50% at 24 h to 43% at 72 h (p < 0.001).
Importantly, no significant cytotoxicity was observed in normal MCF-10 cells at the IC50 concentration effective for MCF-7. Cell viability in MCF-10 remained above 80% after 72 h, suggesting low toxicity to healthy cells. These results emphasize the selective effect of KMF-loaded niosomes and their potential for targeted therapy.
Based on the ISO 10993-5 standard, the results show non-cytotoxicity at 24 and 48 h, with weak cytotoxicity at 72 h. The finding implies that the niosomal delivery system has the potential to facilitate the targeted delivery of KMF to neoplastic cells while at the same time reducing notable cytotoxic effects on normal tissues, even though a weak effect was detected at the longest exposure time.
(A) Evaluation of cell viability and determination of the IC50 value after administration of different concentrations of KMF-loaded niosome NPs to MCF-7 cells. (B and C) The cytotoxic effects of free KMF, free niosome and KMF-loaded niosome NPs on MCF-10 and MCF-7 cell lines at 0.0873 µMol concentration. Data are presented as percentage of cell viability with standard deviation (SD, n = 3). *p < 0.05, **p < 0.01, ***p < 0.001, ns: Results of the viability of MCF-10 cells treated with nanoparticles containing KMF compared to niosomes and free KMF.
Evaluation of apoptosis using flow cytometry
The Annexin-V-FITC assay was used to evaluate the induction of apoptosis in the MCF-7 cell line after 72 h of treatment with PBS (as negative control), free niosome, free KMF and KMF-loaded niosome NPs. The Annexin-V preparations contain Annexin-V to assess membrane asymmetry and a viability dye to measure membrane integrity. Phosphatidylserine (PS), a negatively charged phospholipid located on the inner surface of the plasma membrane, is present on the outer cell surface at an early stage of apoptosis. The result of the experiment indicates that the Q1 region includes necrotic cells, the Q2 region has cells in the late phase of apoptosis, the Q3 region contains cells in the early phase of apoptosis, and the Q4 region contains viable cells. Figure 3 illustrates the immediate induction of apoptotic effects (Q2 + Q3) in MCF-7 cells upon exposure to niosome nanoparticles containing KMF. The most notable difference is the markedly reduced proportion of viable cells in the niosome/KMF group (35%) compared to the free KMF (83%). This indicates that the KMF-loaded NPs have a much greater impact on diminishing cell viability. The investigation revealed that niosome and free KMF, at the IC50 concentration, did not substantially influence the survival rate of MCF-7 cells in comparison to the niosome loaded with KMF. A survival rate of 90% was detected in MCF-7 cells exposed to free niosome. The maximum primary and secondary apoptotic rates of 53% and 11%, respectively, were determined in MCF-7 cells treated with niosome nanoparticles containing KMF, demonstrating a significant difference compared to free KMF and free niosomes (p < 0.01). In the free KMF group, primary and secondary apoptosis was reported to be 4% and 10%, respectively, which was not significant compared to the empty niosomes group. Also, the percentage of viable cells in the free niosomes, free KMF, and KMF-loaded nanoparticles groups was reported to be 90%, 83%, and 35%, respectively. The niosome/KMF nanoparticles exhibit enhanced effectiveness as a delivery system for KMF, promoting both early and late-stage apoptosis, hence reducing cell viability (p < 0.01). The group treated with niosome/KMF NPs had the lowest survival rate (35%) and the greatest apoptotic rate (64%) of MCF-7 cells, demonstrating a statistically significant difference compared to other groups (p < 0.01).
Investigation of cell cycle alterations
Flow cytometry was used to evaluate cell cycle modifications in MCF-7 cells after treatment with free nisome, free KMF and niosome NPs encapsulated with KMF (Fig. 4). Niosome NPs loaded with KMF, a recognized anti-cancer agent, induced cell cycle arrest in the G0/G1 phase (67%) while reducing the proportion of cells in the G2/M phase (23%) (p < 0.01). Prolonged arrest in G0/G1 may trigger apoptosis, or programmed cell death, in MCF-7 cells.
Assessment of cell migration
The effects of free niosome, free KMF and niosomes/KMF NPs on the migration of MCF-7 cells were investigated using a scratch/wound healing assay. The results are shown in Fig. 5. The scratch assay results showed that administration of the IC50 dose of niosomes/KMF prevented the remigration of the cell population to the scratched area within 24 h. This indicates suppression of cell migration and or proliferation. Cells treated with free niosomes and free KMF had little effect on the inhibition of cell migration in the wound area. Cells treated with free niosome and KMF eventually migrated and filled the wounded area within 24 h of treatment. As shown in Fig. 5, the length of the scratched area in the free niosomes and free KMF groups was 98 and 100 μm, respectively. After 24 h, the scratched area was filled with cell migration and the length of the scratched area decreased to 35 and 55 μm, respectively (p < 0.05). The administration of niosome NPs containing KMF to the MCF-7 cell line led to a significant reduction in metastasis, as indicated by the fact that the scratch area had not changed significantly after 24 h compared to the first measurement. The results showed that KMF-loaded niosome NPs were able to inhibit cell migration.
Evaluation of gene expression by real-time PCR
The expression of the Bax, caspase3, MMP2 and ITGA5 genes was examined using the q-PCR method in the MCF-7 and MCF-10 cell lines after treatment with free niosome, free KMF and KMF-loaded niosome NPs. The results showed that Bax gene expression increased significantly in the niosome NPs containing KMF-treated group at 24 and 48 h, with a fold change of 1.98 and 2.15 (***p˂0.001), respectively. Bax gene expression was significantly increased up to 2.95-fold (***p˂0.001) in the 72-hour treatment with KMF-loaded niosomes (Fig. 6). Likewise, the level of caspase-3, an executioner caspase in the apoptosis pathway, was significantly elevated by 1.45-fold, 2.25-fold, and 2.80-fold at 24, 48, and 72 h (**p < 0.01, ***p < 0.001), respectively, when treated with KMF-loaded niosomes. This indicates that the KMF-loaded nanoparticles are successful in inducing the apoptotic machinery within the breast cancer cells, and the impact is augmented with time. On the other hand, the metastasis-related gene expressions MMP2 and ITGA5 were significantly decreased in the MCF-7 cells treated with KMF-loaded niosomes, especially at the time point of 72 h. The MMP2 and ITGA5 expression levels were decreased to around 0.53 and 0.58 folds of their controls (***p˂0.001), respectively, suggesting the suppressive action against the metastatic ability of these cancer cells. One of the most important conclusions of this study is the specific targeting of cancer cells by the KMF-loaded niosome nanoparticles (Fig. 6). In the MCF-10 cell line of normal breast epithelial cells, neither free niosomes nor free KMF nor KMF-loaded niosome nanoparticles produced any significant change in the expression of the studied genes (Bax, caspase-3, MMP2, and ITGA5) at any of the examined time points (24, 48, and 72 h). This lack of significant gene expression change in the normal cell line suggests that the therapeutic effect of the KMF-loaded niosomes is likely targeted against the cancer cells, minimizing potential off-target effects against normal breast tissue.
Discussion
Flavonoids are currently being investigated as antioxidant compounds for the prevention of various malignancies, including gastric, breast, colon, pancreatic, skin, liver, lung and prostate cancers34. Therefore, research has been conducted to improve the stability and bioavailability of these substances using nanoparticle-based methods to increase their efficacy in biological systems at lower doses35. Kaempferol, which is found in a variety of plants and foods, has attracted interest as a valuable flavonoid due to its potential biological properties, such as its anti-cancer properties. Recent research suggests that kaempferol is effective in the inhibition and treatment of various cancers, such as lung, ovarian and breast cancer36. The disadvantages of free kaempferol include insufficient distribution in the body, limited absorption by the body and poor solubility in water. Therefore, the use of nanotechnology is a viable method to improve the anti-cancer and anti-metastatic properties of this flavonoid37.
Numerous studies have examined niosomes in detail, focusing on their physicochemical properties and potential use as drug carriers. Nano-sized niosomes serve as drug carriers and expand the possibilities of pharmacokinetics by reducing toxicity, improving drug solubility and increasing bioavailability38. In this study, niosomal nanoparticles were used to increase the bioavailability and solubility of kaempferol. Tween 80, Span 80 and cholesterol were used to synthesize KMF-loaded niosome NPs. Various analyses were performed to evaluate their chemical and physical properties, including DLS, FTIR, zeta potential and SEM. The SEM results show that the diameter of the synthesized nanoparticles is less than 500 nm, while the average diameter of the synthesized nanoparticles was determined to be between 100 and 500 nm using the DLS technique. The PDI of the niosome NPs containing kaempferol loaded was 0.165, indicating that the loaded niosomes exhibited reasonable size uniformity with acceptable PDI values. The zeta potential may be an indication of the stability of the formulation. The zeta potential of the developed formulations is in a range that ensures good emulsion stability due to electrostatic repulsion. Nanoparticles with a zeta potential between − 10 and + 10 mV are considered approximately neutral, while nanoparticles with zeta potentials of more than + 30 mV or less than − 30 mV are considered strongly cationic or strongly anionic39. The zeta potential in this study was given as 33.9 mV. The PDI is a numerical measure that evaluates the degree of uniformity of particle size and quantifies the size range in a sample. The PDI is a number between 0 and 140. The results of our study show that probe sonication effectively reduces the particle size of niosomes, leading to an improvement in the effectiveness of drug entrapment, and that the FTIR spectra, characterized by clear absorption peaks, confirm the effective incorporation of kaempferol into niosome NPs.
Several studies have demonstrated the anticancer properties of KMF, and various mechanisms have been proposed, but little research has been conducted in the field of nano formulation of kaempferol for drug delivery. Experiments have shown that kaempferol nanoparticles can inhibit the development of cancer in ovarian cancer cells (A2780/CP70 and OVCAR-3)41, rat glioma cells (C6)39 and lung cancer cells (A549)42. In a 2023 study by He et al., to overcome the solubility constraints of kaempferol and augment its antitumor efficacy, they synthesized a kaempferol nanosuspension (KMF-NSps) utilizing D-α-tocopherol polyethylene glycol 1000 succinate (TPGS) as a stabilizing agent and assessed its antitumor effects. Their findings indicated that TPGS-KAE-NSps demonstrated enhanced cytotoxicity and inhibition of cell migration, along with elevated intracellular ROS levels and increased death rates relative to in vitro cell tests with KMF43. The MTT analysis demonstrated that KMF-loaded niosomes had a significant, concentration- and time-dependent lethal impact on MCF-7 cancer cells, with an IC50 of 0.0873 µMol at 24 h. Significantly, free KMF and empty niosomes exhibited less cytotoxicity, highlighting the essential function of niosomal encapsulation in KMF’s anticancer efficacy. The KMF-loaded niosomes exhibited notable selectivity, inflicting less damage on normal MCF-10 cells, indicating a viable tailored drug delivery strategy for cancer treatment with reduced risk of adverse effects.
Previous research showed that kaempferol has the ability to inhibit cell growth and cause cell cycle arrest in HT-29 human colon cancer cells44. Kaempferol has been shown to reduce angiogenesis by decreasing the extracellular signal-regulated kinase (ERK)-NFκB-cMyc-p21-VEGF network in a cancer cell line45. Kaempferol has demonstrated the ability to hinder the growth of cells in a manner that is influenced by the dosage. This is achieved by controlling the activity of cyclin-dependent kinase 1 (CDK1) and cyclin B, which are responsible for the transition from the G2 phase to the M phase. Additionally, kaempferol regulates the expression of either p53 or PLK-1, both of which are tumor suppressor genes that play a crucial role in halting the cell cycle. These effects have been observed in MCF-7 breast cancer cells and HeLa cervical cancer cells. Additionally, kaempferol regulates the expression of either p53 or PLK-1, both of which are tumor suppressor genes that play a crucial role in halting the cell cycle. These effects have been observed in MCF-7 BC cells and HeLa cervical cancer cells46.
Apoptosis is a significant occurrence that results in programmed cell death, which is crucial for proper physiological processes, including organism growth and maintenance47. Cancer cells use many mechanisms to circumvent apoptosis. Hence, to efficiently combat or cure cancer, it is preferable to discover substances that possess the capacity to specifically eliminate cancerous cells while also safeguarding healthy cells48. The Objective of this research was to examine the potential of niosome NPs containing kaempferol to cause apoptosis in MCF-7 cells. To do this, MCF-7 cancer cells were exposed to a concentration of 0.0873 µMol of niosome/kaempferol NPs for 72 h. Annexin V staining demonstrated that the administration of KMF-loaded niosome nanoparticles at a concentration of 0.0873 µMol substantially induced apoptosis (p < 0.05) in MCF-7 cells compared to other groups (free niosome, free KMF, and untreated cells). Annexin V staining results showed that the presence of niosome NPs containing kaempferol significantly (p < 0.05) induced apoptosis in MCF-7 cells when administered at a dose of 0.0873 µMol. However, the addition of the same concentration of niosome NPs containing kaempferol to MCF-10 cell culture, a normal cell of the breast epithelium, did not result in a significant change in cell viability. In addition, the Real-time PCR method showed upregulation of mRNA expression of apoptosis-related genes, namely Bax and caspase 3, in the groups treated with niosome NP containing kaempferol at the IC50 concentration. The decrease in membrane potential and the release of cytochrome C are the result of the coordination of outer mitochondrial membrane permeability by Bcl-2 family proteins. These events lead to the formation of the apoptosome complex, which subsequently activates caspase-9. Caspase 3 was activated by cleaved caspase 9, which subsequently initiated the apoptotic process49. The results that we obtained indicated that the treatment with KMF/niosome NPs after 72 h also resulted in a 2.95 and 2.75-fold increase in the expression of the Bax and caspase-3 genes in MCF-7 cells (p˂0.001). Caspase 3 is an effector that induces DNA degradation and the formation of apoptotic bodies. In 2023, Afzal et al. conducted a study where they examined the levels of mitochondrial apoptosis proteins Bax and cytochrome c in MDA-MB-231 cells treated with a concentration of 40 µMol kaempferol using a confocal microscope50. The microscopy results revealed an increase in the expression of both proteins. The duration of cytochrome c release from mitochondria was closely related to caspase activation. As a result, the levels of caspase-3 and − 9 were measured using ELISA, and it was observed that they were highly expressed after treatment with a kaempferol50. Consistent with the results of the present study, Zho et al. discovered that kaempferol effectively inhibited the growth of MDA-MB-231 TNBC cells. Interestingly, the growth inhibitory effect of kaempferol was more pronounced in MDA-MB-231 cells than in the estrogen receptor-positive BT474 cell line51. In addition, 48-hour treatment with kaempferol resulted in a significant decrease in the percentage of cells in the G1 phase from 85.48 to 51.35% and a significant increase in the percentage of cells in the G2 phase from 9.27 to 37.5%. These results indicate that kaempferol contributes to the induction of G2/M arrest51. In another study, Nandi et al. demonstrated the strong efficacy of kaempferol in inhibiting the growth of breast tumors grown outside the body (ex-vivo) and of patients with TNBC who underwent neoadjuvant chemotherapy. This efficacy is achieved by decreasing the nuclear levels of p53, CD44, ALDH1, NANOG, MDR1, Ki67, and BCL2 and increasing the level of caspase 3. The results of this study confirmed our research and showed that treatment with the IC50 dose of kaempferol leads to an increase in the expression of caspase 352.
G2/M cell cycle arrest is a common cellular reaction to DNA damage induced by radiation-induced agents. This response is linked to genomic instability, the development of tumors, and the treatment process. Recent research has shown that kaempferol frequently leads to cell cycle arrest at the G2/M phase when used to treat gynecological malignancies53. Kaempferol hinders the growth of MDA-MB-453 cells, a type of human breast cancer cell, by disrupting the cell cycle. It causes cell cycle arrest at the G2/M checkpoint and is thought to promote apoptosis through p53 phosphorylation54. In agreement with the results of previous studies, the cell cycle results in this study showed that cells treated with niosome NPs containing kaempferol had the lowest rate of entry into G2/M phase and the highest rate of entry into G0/G1 phase (p < 0.05*).
Tumor cell invasion and metastasis are key features of malignant tumors; the most important biomarkers are MMP-2, MMP-9, N-cadherin and E-cadherin55. Li et al. found that kaempferol impeded the invasion of MDA-MB-231 breast cancer cell, by blocking the PKC/MAPK/AP-1 signaling pathway and reducing the production of MMP-956. Our studies confirmed that treatment with the IC50 dose of kaempferol, inhibited the migration and metastasis of MCF-7 cells by reducing the expression of MMP2 and ITGA5 by 0.53- and 0.58-fold, respectively (***p˂0.001). Lee et al. (2017) found that kaempferol impedes the epithelial-mesenchymal transition (EMT) process as well as the movement and infiltration of MCF-7 breast cancer cells by acting on the estrogen receptor (ER). This is accomplished via controlling the protein production of genes associated with EMT and the spread of cancer cells to other parts of the body, such as N-cadherin, Snail, Slug, and Cathepsin B57. Zhang et al. (2021) found that Kaempferol 3-O-gentiobioside effectively hindered the movement and infiltration of cancer cells. Additionally, it reversed the expression of regulatory factors associated with EMT and decreased tumor development in living organisms by obstructing the conventional TGF-β/ALK5/Smad pathway58. Yang et al. (2019) found that kaempferol induces a decrease in the growth of OVCAR-3 human ovarian cancer cells by increasing the levels of proteins involved in cell death, specifically caspase 3, caspase 8, caspase 9, and Bax. These proteins promote apoptosis, block the cell cycle at the G0/G1 phase, and affect the MEK/ERK and STAT3 pathways59. Kim et al. (2016) found that kaempferol can effectively inhibit the growth of breast cancer cells induced by TCS (the antiproliferative effects of kaempferol in triclosan) and E2 (17β-estradiol). It acts as an antagonist of ER and IGF-1R signaling, while reducing the levels of cyclin D1, cyclin E and cathepsin D proteins. In addition, it increases the levels of p21, Bax, pIRS-1, pAkt and pMEK1/2 proteins60. The collective results of this study and previous research suggest that kaempferol has significant antitumor properties, especially against aggressive malignant tumors such as breast, lung, liver and pancreatic cancer. Numerous studies have shown that kaempferol can induce programmed cell death in tumor cells, inhibit their growth, prevent metastasis and invasion, and induce self-destruction of tumor cells in female malignancies such as breast, ovarian and endometrial cancer.
Conclusion
In summary, this study was successful in designing and characterizing kaempferol-loaded niosome nanoparticles as a promising strategy to enhance the therapeutic potential of kaempferol against breast cancer. In vitro investigation in MCF-7 cells confirmed that this nanoformulation significantly augmented the cytotoxicity of kaempferol, induced apoptosis via the upregulation of Bax and caspase-3, and inhibited metastasis via the downregulation of MMP2 and ITGA5 with negligible cytotoxicity against normal MCF-10 cells. These findings indicate the potential of niosomal encapsulation to augment kaempferol’s anti-cancer activity and as a target drug delivery system in the treatment of cancer, with suggestions for future in vivo research.
Limitations
Because the research costs were paid personally, it was not possible to use multiple cell lines.
Data availability
The materials used in this study are available from the corresponding author upon request.
Abbreviations
- BC:
-
Breast cancer
- HER2:
-
Human epidermal growth factor receptor 2
- KMF:
-
Kaempferol
- PI3K-PKB:
-
Phosphatidylinositol 3-kinase-protein kinase B
- NPs:
-
Nanoparticles
- EDTA:
-
Ethylene diamine tetra acetic acid
- SEM:
-
Scanning electron microscope
- FTIR:
-
Fourier transform infrared spectrum
- IC50 :
-
Half-maximal inhibitory concentration
- OD:
-
Optical density
- PI:
-
Propidium iodide
- q-PCR:
-
Real-time polymerase chain reaction
- DLS:
-
Dynamic Light Scattering
- MTT:
-
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- PS:
-
Phosphatidylserine
- PDI:
-
Polydispersity index
- ERK:
-
Extracellular signal-regulated kinase
- CDK1:
-
Cyclin-dependent kinase 1
- TNBC:
-
Triple-negative breast cancer
- EMT:
-
Epithelial-mesenchymal transition
- ER:
-
Estrogen receptor
- DMEM:
-
Dulbecco’s Modified Eagle Medium
- FBS:
-
Fetal bovine serum
References
Sung, H. et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Cancer J. Clin. 71 (3), 209–249 (2021).
Kumar, G. et al. Codelivery of phytochemicals with conventional anticancer drugs in form of nanocarriers. Pharmaceutics 15 (3), 889 (2023).
Rakha, E. A., Tse, G. M. & Quinn, C. M. An update on the pathological classification of breast cancer .Histopathology 82(1), 5–16 (2023).
Kaur, R., Bhardwaj, A. & Gupta, S. Cancer treatment therapies: traditional to modern approaches to combat cancers. Mol. Biol. Rep. 50 (11), 9663–9676 (2023).
Burguin, A., Diorio, C. & Durocher, F. Breast cancer treatments: updates and new challenges. J. Personalized Med. 11 (8), 808 (2021).
Asadipour, E. et al. Nano-biotechnology and challenges of drug delivery system in cancer treatment pathway. Chem. Biodivers. 20 (6), e202201072 (2023).
Silva-Pinto, P. A. et al. Phytochemical Insights into Flavonoids in Cancer: Mechanisms, Therapeutic Potential, and the Case of Quercetin. (Heliyon, 2025).
Parisi, O. I. et al. Polyphenols and their formulations: different strategies to overcome the drawbacks associated with their poor stability and bioavailability. In Polyphenols in Human Health and Disease. 29–45. (Elsevier, 2014).
Khan, A. Q. & Uddin, S. Anticancer activity of natural compounds. Asian Pac. J. Cancer Prev. 22 (S1), 1–2 (2021).
Qattan, M. Y. et al. Therapeutic importance of Kaempferol in the treatment of cancer through the modulation of cell signalling pathways. Molecules 27 (24), 8864 (2022).
Shahbaz, M. et al. Anticancer, antioxidant, ameliorative and therapeutic properties of Kaempferol. Int. J. Food Prop. 26 (1), 1140–1166 (2023).
Imran, M. et al. Chemo-preventive and therapeutic effect of the dietary flavonoid kaempferol: A comprehensive review. Phytother. Res. 33 (2), 263–275 (2019).
Bangar, S. P. et al. Kaempferol: A flavonoid with wider biological activities and its applications. Crit. Rev. Food Sci. Nutr. 63 (28), 9580–9604 (2023).
Zare, M. et al. Improving the cancer prevention/treatment role of carotenoids through various nano-delivery systems. Crit. Rev. Food Sci. Nutr. 61 (3), 522–534 (2021).
Mirzaei-Parsa, M. J. et al. Preparation, characterization, and evaluation of the anticancer activity of artemether-loaded nano-niosomes against breast cancer. Breast Cancer. 27, 243–251 (2020).
Alrushaid, N. et al. Nanotechnology in cancer diagnosis and treatment. Pharmaceutics 15 (3), 1025 (2023).
Dessale, M., Mengistu, G. & Mengist, H. M. Nanotechnology: a promising approach for cancer diagnosis, therapeutics and theragnosis. Int. J. Nanomed. 17, 3735 (2022).
Fan, D. et al. Nanomedicine in cancer therapy. Signal. Transduct. Target. Therapy. 8 (1), 293 (2023).
Mosleh-Shirazi, S. et al. Nanotechnology advances in the detection and treatment of cancer: an overview. Nanotheranostics 6 (4), 400 (2022).
Piri-Gharaghie T, Zarinnezhad A, Naghian B, Babaei R. Molecular detection of fungal APR1 gene in serum of multiple sclerosis patients: a personalized medicine research. PM Journal. 7(25),15–24 (2022).
Witwer, K. W. & Wolfram, J. Extracellular vesicles versus synthetic nanoparticles for drug delivery. Nat. Reviews Mater. 6 (2), 103–106 (2021).
Zargarani, N. et al. A potential new strategy for BC treatment: NPs containing solanine and evaluation of its anticancer and antimetastatic properties. BMC Cancer. 25, 860 (2025).
Rad, M. E. et al. Optimization of Curcumin loaded niosomes for drug delivery applications. Colloids Surf., A. 654, 129921 (2022).
Tariq, F. et al. Design, optimization & characterization of Niosomal & polymeric nanoparticles. Int. J. Polym. Mater. Polym. Biomaterials. 73 (15), 1353–1366 (2024).
Moghtaderi, M. et al. Niosomes: a novel targeted drug delivery system for cancer. Med. Oncol. 39 (12), 240 (2022).
Homaeii, S. et al. Investigating the apoptotic and antimetastatic effect of daphnetin-containing nano niosomes on MCF-7 cells. Adv. Cancer Biology – Metastasis. 14, 100139 (2025).
Bozorgi, A. et al. The anti-cancer effect of chitosan/resveratrol polymeric nanocomplex against triple‐negative breast cancer; an in vitro assessment. IET Nanobiotechnol. 17 (2), 91–102 (2023).
Kavousi, M. et al. Solanum pseudo-capsicum effects on Bax and Bcl-2 gene expression and apoptosis in MCF-7 cell line. Genetika-Belgrade 55 (2), 523–536 (2023).
Ebadi, M. et al. Investigation of the apoptotic and antimetastatic effects of nano-niosomes containing the plant extract Anabasis setifera on HeLa: In vitro cervical cancer study. Chem. Biodivers. e202402599.
Fong, S. S. et al. Chitosan-coated-PLGA nanoparticles enhance the antitumor and antimigration activity of stattic–a STAT3 dimerization blocker. Int. J. Nanomed.. 137–150 (2022).
Kavousi, M. et al. Effect of Coated Carbon Nanotubes with Chitosan and Cover of Flaxseed in the Induction of MDA-MB-231 Apoptosis by Analyzing the Expression of Bax and Bcl-2. Vol. 26 (Human Gene (Meta Gene), 2020).
Dabbagh Moghaddam, F. et al. Delivery of melittin-loaded niosomes for breast cancer treatment: an in vitro and in vivo evaluation of anti-cancer effect. Cancer Nanotechnol. 12 (1), 14 (2021).
Bojar Doulaby, D. et al. Effect of Dioscorea extract on Bax and Bcl-2 gene expression in MCF- 7 and HFF cell lines. Egypt. J. Med. Hum. Genet. 24, 70 (2023).
Slika, H. et al. Therapeutic potential of flavonoids in cancer: ROS-mediated mechanisms. Biomed. Pharmacother. 146, 112442 (2022).
Farhan, M. et al. Current Understanding of flavonoids in cancer therapy and prevention. Metabolites 13 (4), 481 (2023).
Almatroudi, A. et al. Effects and mechanisms of Kaempferol in the management of cancers through modulation of inflammation and signal transduction pathways. Int. J. Mol. Sci. 24 (10), 8630 (2023).
Chandekar, L., Katgeri, R. & Takke, A. The potential clinical uses and nanoformulation strategies of kaempferol, a dietary flavonoid. Revista Brasileira De Farmacognosia. 32 (5), 693–707 (2022).
Aldawsari, M. F. et al. Formulation optimization, in vitro and in vivo evaluation of Niosomal nanocarriers for enhanced topical delivery of Cetirizine. Saudi Pharm. J. 31 (9), 101734 (2023).
Colombo, M. et al. Kaempferol-loaded mucoadhesive nanoemulsion for intranasal administration reduces glioma growth in vitro. Int. J. Pharm. 543 (1–2), 214–223 (2018).
Iqbal, M. S., Abbas, K. & Qadir, M. I. Synthesis, characterization and evaluation of biological properties of selenium nanoparticles from Solanum lycopersicum. Arab. J. Chem. 15 (7), 103901 (2022).
Piri-Gharaghie T, Ebrahimi M, Dehganzad B, Seraj Z. The development of a novel multiepitope vaccine targeting the secretion systems of Pseudomonas aeruginosa using a reverse vaccinology and immunoinformatic strategy. Discover Applied Sciences. 7(5):1-29 (2025).
Govindaraju, S. et al. Kaempferol conjugated gold nanoclusters enabled efficient for anticancer therapeutics to A549 lung cancer cells. Int. J. Nanomed.. 5147–5157 (2019).
He, W. et al. Preparation, characterization, and evaluation of the antitumor effect of Kaempferol nanosuspensions. Drug Delivery Translational Res. 13 (11), 2885–2902 (2023).
Cho, H. J. & Park, J. H. Y. Kaempferol induces cell cycle arrest in HT-29 human colon cancer cells. J. cancer Prev. 18 (3), 257 (2013).
Luo, H. et al. Kaempferol inhibits angiogenesis and VEGF expression through both HIF dependent and independent pathways in human ovarian cancer cells. Nutr. Cancer. 61 (4), 554–563 (2009).
Kang, G. Y. et al. Downregulation of PLK-1 expression in kaempferol-induced apoptosis of MCF-7 cells. Eur. J. Pharmacol. 611 (1–3), 17–21 (2009).
Das, S. et al. Mechanism of interaction between autophagy and apoptosis in cancer. Apoptosis. 1–22 (2021).
Cetraro, P. et al. A review of the current impact of inhibitors of apoptosis proteins and their repression in cancer. Cancers 14 (7), 1671 (2022).
Wolf, P., Schoeniger, A. & Edlich, F. Pro-apoptotic complexes of BAX and BAK on the outer mitochondrial membrane. Biochim. Et Biophys. Acta (BBA)-Molecular Cell. Res. 1869 (10), 119317 (2022).
Afzal, M. et al. Antiproliferative mechanisms of a polyphenolic combination of Kaempferol and Fisetin in triple-negative breast cancer cells. Int. J. Mol. Sci. 24 (7), 6393 (2023).
Zhu, L. & Xue, L. Kaempferol suppresses proliferation and induces cell cycle arrest, apoptosis, and DNA damage in breast cancer cells. Oncol. Res. 27 (6), 629 (2019).
Nandi, S. K. et al. Kaempferol attenuates viability of ex-vivo cultured post-NACT breast tumor explants through downregulation of p53 induced stemness, inflammation and apoptosis evasion pathways. Pathology-Research Pract. 237, 154029 (2022).
Ma, X. et al. The role of Kaempferol in gynaecological malignancies: progress and perspectives. Front. Pharmacol. 14, 1310416 (2023).
Choi, E. J. & Ahn, W. S. Kaempferol induced the apoptosis via cell cycle arrest in human breast cancer MDA-MB-453 cells. Nutr. Res. Pract. 2 (4), 322–325 (2008).
Li, H. et al. The relationship between MMP-2 and MMP-9 expression levels with breast cancer incidence and prognosis. Oncol. Lett. 14 (5), 5865–5870 (2017).
Li, C. et al. Inhibitory effects of Kaempferol on the invasion of human breast carcinoma cells by downregulating the expression and activity of matrix metalloproteinase-9. Biochem. Cell Biol. 93 (1), 16–27 (2015).
Lee, G. A., Choi, K. C. & Hwang, K. A. Kaempferol, a phytoestrogen, suppressed triclosan-induced epithelial-mesenchymal transition and metastatic-related behaviors of MCF-7 breast cancer cells. Environ. Toxicol. Pharmacol. 49, 48–57 (2017).
Zhang, Z. et al. Kaempferol 3-O‐gentiobioside, an ALK5 inhibitor, affects the proliferation, migration, and invasion of tumor cells via Blockade of the TGF‐β/ALK5/Smad signaling pathway. Phytother. Res. 35 (11), 6310–6323 (2021).
Yang, S. et al. Kaempferol exerts anti-proliferative effects on human ovarian cancer cells by inducing apoptosis, G0/G1 cell cycle arrest and modulation of MEK/ERK and STAT3 pathways. J. Buon. 24 (3), 975–981 (2019).
Kim, S. H., Hwang, K. A. & Choi, K. C. Treatment with Kaempferol suppresses breast cancer cell growth caused by Estrogen and triclosan in cellular and xenograft breast cancer models. J. Nutr. Biochem. 28, 70–82 (2016).
Author information
Authors and Affiliations
Contributions
SH.M conceptualized and wrote the manuscript. MK wrote the manuscript and did the necessary editing, supervised and performed the investigations and statistical analysis. FJ helped in the editing of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics declaration
The study was approved by the Ethics Committee of Islamic Azad University (East Tehran Branch, Tehran, Iran) and Iran National Committee for Ethics in Biomedical Research (IR.IAU.ET.REC.1403.003).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Minaei, S., Kavousi, M. & Jamshidian, F. The apoptotic and anti-metastatic effects of niosome kaempferol in MCF-7 breast cancer cells. Sci Rep 15, 20741 (2025). https://doi.org/10.1038/s41598-025-07221-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-07221-0
Keywords
This article is cited by
-
Phytocompounds as sustainable therapeutics for breast cancer treatment: a comprehensive review on isolation and delivery strategies
Discover Applied Sciences (2025)