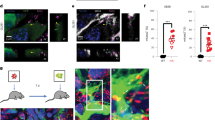
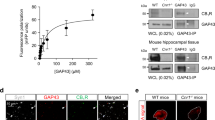
Abstract
Transcription regulates axon outgrowth and regeneration. However, to date, no transcription complexes have been shown to control axon outgrowth and regeneration by regulating axon growth genes. Here, we report that the tumor suppressor p53 and its acetyltransferases CBP/p300 form a transcriptional complex that regulates the axonal growth-associated protein 43, a well-characterized pro-axon outgrowth and regeneration protein. Acetylated p53 at K372-3-82 drives axon outgrowth, GAP-43 expression, and binds specific elements on the neuronal GAP-43 promoter in a chromatin environment through CBP/p300 signaling. Importantly, in an axon regeneration model, both CBP and p53 K372-3-82 are induced following axotomy in facial motor neurons, where p53 K372-3-82 occupancy of GAP-43 promoter is enhanced as shown by in vivo chromatin immunoprecipitation. Finally, by comparing wild-type and p53 null mice, we demonstrate that the p53/GAP-43 transcriptional module is specifically switched on during axon regeneration in vivo. These data contribute to the understanding of gene regulation in axon outgrowth and may suggest new molecular targets for axon regeneration.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Abbreviations
- C/EBP:
-
CCAAT-enhancer-binding proteins
- CBP:
-
CREB-binding protein
- CREB:
-
cAMP-responsive element-binding protein
- JNKs:
-
c-Jun N-terminal kinases
- MAPK:
-
mitogen-activated protein kinases
- NFAT:
-
nuclear factor of activated T-cells
- P/CAF:
-
p300/CBP-associated factor
- STAT3:
-
signal transducer and activator of transcription 3
References
Makwana M, Raivich G . Molecular mechanisms in successful peripheral regeneration. FEBS J 2005; 272: 2628–2638.
Teng FY, Tang BL . Axonal regeneration in adult CNS neurons--signaling molecules and pathways. J Neurochem 2006; 96: 1501–1508.
Laux T, Fukami K, Thelen M, Golub T, Frey D, Caroni P . GAP43, MARCKS, and CAP23 modulate PI(4,5)P(2) at plasmalemmal rafts, and regulate cell cortex actin dynamics through a common mechanism. J Cell Biol 2000; 149: 1455–1472.
Perrone-Bizzozero NI, Weiner D, Hauser G, Benowitz LI . Extraction of major acidic Ca2+ dependent phosphoproteins from synaptic membranes. J Neurosci Res 1988; 20: 346–350.
De la Monte SM, Federoff HJ, Ng SC, Grabczyk E, Fishman MC . GAP-43 gene expression during development: persistence in a distinctive set of neurons in the mature central nervous system. Brain Res Dev Brain Res 1989; 46: 161–168.
Jacobson RD, Virag I, Skene JH . A protein associated with axon growth, GAP-43, is widely distributed and developmentally regulated in rat CNS. J Neurosci 1986; 6: 1843–1855.
Benowitz LI, Perrone-Bizzozero NI, Neve RL, Rodriguez W . GAP-43 as a marker for structural plasticity in the mature CNS. Prog Brain Res 1990; 86: 309–320.
Tetzlaff W, Alexander SW, Miller FD, Bisby MA . Response of facial and rubrospinal neurons to axotomy: changes in mRNA expression for cytoskeletal proteins and GAP-43. J Neurosci 1991; 11: 2528–2544.
Tetzlaff W, Zwiers H, Lederis K, Cassar L, Bisby MA . Axonal transport and localization of B-50/GAP-43-like immunoreactivity in regenerating sciatic and facial nerves of the rat. J Neurosci 1989; 9: 1303–1313.
Hulsebosch CE, DeWitt DS, Jenkins LW, Prough DS . Traumatic brain injury in rats results in increased expression of Gap-43 that correlates with behavioral recovery. Neurosci Lett 1998; 255: 83–86.
Carmichael ST . Plasticity of cortical projections after stroke. Neuroscientist 2003; 9: 64–75.
Gianola S, Rossi F . GAP-43 overexpression in adult mouse Purkinje cells overrides myelin-derived inhibition of neurite growth. Eur J Neurosci 2004; 19: 819–830.
Aigner L, Arber S, Kapfhammer JP, Laux T, Schneider C, Botteri F et al. Overexpression of the neural growth-associated protein GAP-43 induces nerve sprouting in the adult nervous system of transgenic mice. Cell 1995; 83: 269–278.
Aigner L, Caroni P . Depletion of 43-kD growth-associated protein in primary sensory neurons leads to diminished formation and spreading of growth cones. J Cell Biol 1993; 123: 417–429.
Alonso G, Ridet JL, Oestreicher AB, Gispen WH, Privat A . B-50 (GAP-43) immunoreactivity is rarely detected within intact catecholaminergic and serotonergic axons innervating the brain and spinal cord of the adult rat, but is associated with these axons following lesion. Exp neurol 1995; 134: 35–48.
Bomze HM, Bulsara KR, Iskandar BJ, Caroni P, Skene JH . Spinal axon regeneration evoked by replacing two growth cone proteins in adult neurons. Nat Neurosci 2001; 4: 38–43.
Beckel-Mitchener AC, Miera A, Keller R, Perrone-Bizzozero NI . Poly(A) tail length-dependent stabilization of GAP-43 mRNA by the RNA-binding protein HuD. J Biol Chem 2002; 277: 27996–28002.
Chung S, Eckrich M, Perrone-Bizzozero N, Kohn DT, Furneaux H . The Elav-like proteins bind to a conserved regulatory element in the 3′-untranslated region of GAP-43 mRNA. J Biol Chem 1997; 272: 6593–6598.
Bolognani F, Tanner DC, Merhege M, Deschenes-Furry J, Jasmin B, Perrone-Bizzozero NI . In vivo post-transcriptional regulation of GAP-43 mRNA by overexpression of the RNA-binding protein HuD. J Neurochem 2006; 96: 790–801.
Bolognani F, Tanner DC, Nixon S, Okano HJ, Okano H, Perrone-Bizzozero NI . Coordinated expression of HuD and GAP-43 in hippocampal dentate granule cells during developmental and adult plasticity. Neurochem Res 2007; 32: 2142–2151.
Smith CL, Afroz R, Bassell GJ, Furneaux HM, Perrone-Bizzozero NI, Burry RW . GAP-43 mRNA in growth cones is associated with HuD and ribosomes. J Neurobiol 2004; 61: 222–235.
Nedivi E, Basi GS, Akey IV, Skene JH . A neural-specific GAP-43 core promoter located between unusual DNA elements that interact to regulate its activity. J Neurosci 1992; 12: 691–704.
Starr RG, Lu B, Federoff HJ . Functional characterization of the rat GAP-43 promoter. Brain Res 1994; 638: 211–220.
Weber JR, Skene JH . The activity of a highly promiscuous AP-1 element can be confined to neurons by a tissue-selective repressive element. J Neurosci 1998; 18: 5264–5274.
Uittenbogaard M, Martinka DL, Chiaramello A . The basic helix-loop-helix differentiation factor Nex1/MATH-2 functions as a key activator of the GAP-43 gene. J Neurochem 2003; 84: 678–688.
Di Giovanni S, Knights CD, Rao M, Yakovlev A, Beers J, Catania J et al. The tumor suppressor protein p53 is required for neurite outgrowth and axon regeneration. EMBO J 2006; 25: 4084–4096.
Di Giovanni S, De Biase A, Yakovlev A, Finn T, Beers J, Hoffman EP et al. In vivo and in vitro characterization of novel neuronal plasticity factors identified following spinal cord injury. J Biol Chem 2005; 280: 2084–2091.
Yakovlev AG, Di Giovanni S, Wang G, Liu W, Stoica B, Faden AI . BOK and NOXA are essential mediators of p53-dependent apoptosis. J Biol Chem 2004; 279: 28367–28374.
Saha RN, Pahan K . HATs and HDACs in neurodegeneration: a tale of disconcerted acetylation homeostasis. Cell Death Differ 2006; 13: 539–550.
Liu L, Scolnick DM, Trievel RC, Zhang HB, Marmorstein R, Halazonetis TD et al. p53 sites acetylated in vitro by PCAF and p300 are acetylated in vivo in response to DNA damage. Mol Cell Biol 1999; 19: 1202–1209.
Avantaggiati ML, Ogryzko V, Gardner K, Giordano A, Levine AS, Kelly K . Recruitment of p300/CBP in p53-dependent signal pathways. Cell 1997; 89: 1175–1184.
Lill NL, Grossman SR, Ginsberg D, DeCaprio J, Livingston DM . Binding and modulation of p53 by p300/CBP coactivators. Nature 1997; 387: 823–827.
Knights CD, Catania J, Di Giovanni S, Muratoglu S, Perez R, Swartzbeck A et al. Distinct p53 acetylation cassettes differentially influence gene-expression patterns and cell fate. J Cell Biol 2006; 173: 533–544.
Raivich G, Bohatschek M, Da Costa C, Iwata O, Galiano M, Hristova M et al. The AP-1 transcription factor c-Jun is required for efficient axonal regeneration. Neuron 2004; 43: 57–67.
Namgung U, Routtenberg A . Transcriptional and post-transcriptional regulation of a brain growth protein: regional differentiation and regeneration induction of GAP-43. Eur J Neurosci 2000; 12: 3124–3136.
Takahashi M, Sato Y, Nakagami Y, Miyake K, Iijima S . Identification of cis-acting regions that contribute to neuron-specific expression of the GAP-43 gene. Biosci Biotechnol Biochem 2006; 70: 1492–1495.
Hoh J, Jin S, Parrado T, Edington J, Levine AJ, Ott J . The p53MH algorithm and its application in detecting p53-responsive genes. Proc Natl Acad Sci USA 2002; 99: 8467–8472.
Xu Y . Regulation of p53 responses by post-translational modifications. Cell Death Differ 2003; 10: 400–403.
Song J, Chao C, Xu Y . Ser18 and Ser23 phosphorylation plays synergistic roles in activating p53-dependent neuronal apoptosis. Cell Cycle 2007; 6: 1412–1414.
Zhu Y, Mao XO, Sun Y, Xia Z, Greenberg DA . p38 Mitogen-activated protein kinase mediates hypoxic regulation of Mdm2 and p53 in neurons. J Biol Chem 2002; 277: 22909–22914.
Chen RW, Qin ZH, Ren M, Kanai H, Chalecka-Franaszek E, Leeds P et al. Regulation of c-Jun N-terminal kinase, p38 kinase and AP-1 DNA binding in cultured brain neurons: roles in glutamate excitotoxicity and lithium neuroprotection. J Neurochem 2003; 84: 566–575.
Glozak MA, Sengupta N, Zhang X, Seto E . Acetylation and deacetylation of non-histone proteins. Gene 2005; 363: 15–23.
Luo J, Li M, Tang Y, Laszkowska M, Roeder RG, Gu W . Acetylation of p53 augments its site-specific DNA binding both in vitro and in vivo. Proc Natl Acad Sci USA 2004; 101: 2259–2264.
Gu W, Roeder RG . Activation of p53 sequence-specific DNA binding by acetylation of the p53 C-terminal domain. Cell 1997; 90: 595–606.
Gao Y, Deng K, Hou J, Bryson JB, Barco A, Nikulina E et al. Activated CREB is sufficient to overcome inhibitors in myelin and promote spinal axon regeneration in vivo. Neuron 2004; 44: 609–621.
Acknowledgements
We thank Jorge Garay for conducting part of the facial nerve transection experiments, Sergio Schinelli for critically reviewing the paper, and Sanam Vakil for her help editing the paper. In addition, we are grateful to Dr. Maria Laura Avantaggiati for providing the p53 acetylation mutant plasmid DNAs, to Dr. Ulrike Neumann for the p53 R273H and R248W mutant plasmid DNAs, and to Dr. Katsuhide Miyake for providing the CBP/p300 RNAi. This work was supported by the Hertie Foundation; the Fortune Grant, University of Tuebingen, the NIH R21 NS052640 and the DFG DI 1497/1-1 grants (all granted to Simone Di Giovanni).
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by D Kaplan
Supplementary Information accompanies the paper on Cell Death and Differentiation website (http://www.nature.com/cdd)
Rights and permissions
About this article
Cite this article
Tedeschi, A., Nguyen, T., Puttagunta, R. et al. A p53-CBP/p300 transcription module is required for GAP-43 expression, axon outgrowth, and regeneration. Cell Death Differ 16, 543–554 (2009). https://doi.org/10.1038/cdd.2008.175
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/cdd.2008.175
Keywords
This article is cited by
-
Transcriptional Control of Peripheral Nerve Regeneration
Molecular Neurobiology (2023)
-
Macrophage-specific RhoA knockout delays Wallerian degeneration after peripheral nerve injury in mice
Journal of Neuroinflammation (2021)
-
Cancer and Alzheimer’s disease inverse relationship: an age-associated diverging derailment of shared pathways
Molecular Psychiatry (2021)
-
SIRT2 Inhibition Improves Functional Motor Recovery After Peripheral Nerve Injury
Neurotherapeutics (2020)
-
Intrinsic mechanisms of neuronal axon regeneration
Nature Reviews Neuroscience (2018)